3.1 and 3.2 Redox and standard electrode potential and Redox reactions
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Define oxidation
Loss of electrons
Define reduction
Gain of electrons
Oxidation number of group 1 metals
Always +1
Oxidation number of group 2 metals
Always +2
Oxidation number of oxygen
Usually -2, except perioxides where it is -1
Oxidation number of hydrogen
Usually +1, except metal hydrides where it is -1
Oxidation number of Florine
-1
Oxidation number of chloride
-1, unless bonded to F or O
Oxidation number of ions
Charge of ion.
If a substance is reduced, does the oxidation number increase or decrease?
decreases
What is a disproportionation reaction
A single substance is simultaneously oxidised and reduced
5 steps to construct a redox equation?
Balance atoms
Add water to balance oxygens
Add H+ to balance hydrogens
Add electrons to balance hydrogens
Combine to make a redox equation
What is a half cell?
Comprises of an element in 2 different oxidation states.
Structure of a simple half cell?
Strip of metal placed in an aqueous solution of its ions.
How to tell If a half cell is likely to loose or gain electrons?
Use a standard electrode as a reference.
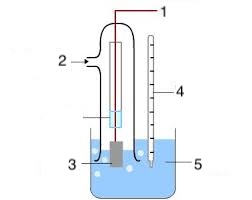
Label the 5 parts of the standard hydrogen electrode.
Copper wire
Hydrogen gas at 1atm
Platinum electrode
298K
H+ (aq) at 1moldm-3 (e.g. HCl)
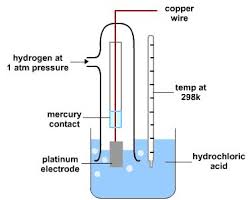
Why is a platinum electrode used?
It is inert.
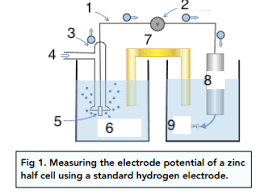
Label the 9 parts of the copper half cell connected to a SHE (electrolysis)
Copper wire
Voltmeter
Standard hydrogen electrode
H2 gas (1atm)
platinum electrode
H+ at 1moldm-3
Salt bridge
Copper
Cu2+ (aq) (1moldm-3)
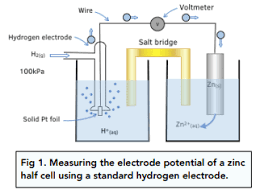
What happens in the system when two half cells are connected? Equation for this.
Copper electrode will get bigger and solution will become paler, as electrons flow from the hydrogen half cell to the copper half cell.
Cu2+ + 2e- → Cu
Why is a salt bridge needed? Give an example.
KNO3, needed to connect and complete the circuit. KNO3 suitable as it is soluble, so no precipitate formed.
Rule for direction of electron flow in half cells?
The most positive E∘ goes forward (right) and the other backwards (left)
How to calculate E∘
Most positive - most negative
What indicates a feasible reaction?
Positive E∘
What indicates a spontaneous reaction?
A voltage reading above 0.4V
How to allow 2 half cells reaction to run to completion
Remove voltmeter, but no reading will be available.
2 limitations of half cells
Rate could be slow due to high activation energy
Conditions may be different in actual situations
How to read mixture of different oxidation states?
Use a platinum electrode (e.g. iron with a mix of Fe2+ and Fe3+)
Find the forward reaction, backwards and overall reaction, feasibility and whether this reaction is spontaneous?
Cu2+ (aq) + 2e- ⇌ Cu (s); E0 = +0.34V
Sn+ (aq) + 2e- ⇌ Sn (s); E0 = -0.14V
Cu2+ (aq) + 2e- → Cu (s): more positive, so forwards)
Sn → Sn2+ 2e-
Overall reaction: Cu2+ + Sn → Cu + Sn2+
E0 = 0.34 - (-0.14) =0.48V
Reaction feasible and spontaneous
How to create a spontaneous reaction if voltage isn’t above 0.4V
Change conditions.