metal prenstation
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Only __ nonmetals exist, but they are biologically essential (O, C, H, N, P, S)
Noble gases = most unreactive elements
General Properties
more electronegative then metals
Form:
__ compounds with metals
__ compounds with other nonmetals
Physical states:
__: C, S, P, I
__: Br
__: H₂, N₂, O₂, F₂, Cl₂, noble gases
25
ionic
covalent
solids
liquid
gases
Most abundant element in the universe
colorless, odorless, nonpoisonous, formed after big bang
Exists as:
¹H (protium) – most common
²H (deuterium)
³H (tritium, radioactive)
Types of Hydrides
__ (saline) – Group 1 & 2 metals except Be
white, high melting point solids, s block
___(molecular) – nonmetals
p block, volatile, low melting point
__ (metallic) – transition metals
formed by heating transitions metals in H2, black , powdery
Production of Hydrogen
mixture of co2 and h2= water gas
production form oil refineries
Electrolysis of water
Metal + acid reactions
hydrogen
ionic
covalent
interstitial
group 1
Main Points
Most __ metals
Never found free in nature
Obtained by electrolysis
Key Reactions
React violently with __:
Produce H₂ gas + metal hydroxide
Reactivity increases __ the group
Only __ reacts with N₂
Other Notes
Strong __ agents
Form __ compounds
Form __ (Li₂O, Na₂O₂, KO₂)
can even reduce the hydrogen in __
alkali metals
reactive
nature
electrolysis
water
down
Li
reducing
ionic
oxides
water
____ (Group 2) Main Points
Reactive, but less than Group 1
Form basic oxides and hyroxides, from CA, SR, and BA
Found in minerals (limestone, dolomite)
Important Concepts
__ of calcium is stored in your bones and teeth
__ is in your blood and soft tissue-helps contraction
__ used for producing glass for tv
__=machinable metal-electronic, bleaches, dyes, fireworks, well drilling
__- radioactive, natural in rocks solids, ground water, 224, 226, 228
reaction
__ does not react with water
__ burns in air
__ reacts with
__ may ignite in air
__ compounds are very toxic
alkaline earth metals
less
basic hyroxides
minerals
99%
1%
barium
radium
Be
Mg
Ca
Ba
beryillium
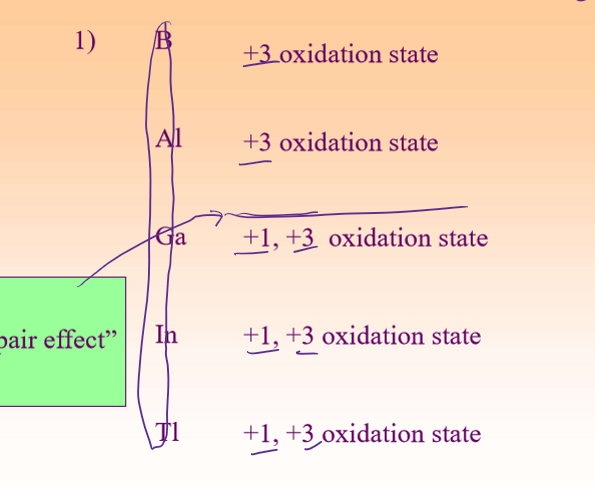
Group 13 (___) Oxidation States
Usually __
Heavier elements show __
__ the tendency to from cations two unit lower in charge then expected due to different energies of valance p and s electrons
Key Elements
___
Metalloid
__ oxides
__ retards the spread of a flame
made domestically in __
Aluminum
Most __ metal in crust
Light, strong, corrosion-resistant, ___-acid or base
becomes __ when exposed to air= wont rust
obtained through __: reverse rather than from rocks
US has largest production
boron family
+3
+1
inert pair effect
boron
acid
boric acid
California
aluminum
abundant
amphoteric
passivated
hall process
Carbon & Group 14 Carbon
Exists as ___ (diamond, graphite)
Forms __ bonds (unique ability)
Important Compounds
CO – toxic
not an acidic oxide
burns readily in o2 to from co2
CO₂ – non-toxic, used in beverages
when solid __
Special Structures
Fullerenes (C₆₀)
Trend
Metallic character __ down group (Si → Pb)
lead based paint hazard in houses- mainly found in older homes
primary pathway of lead posining in young children
allotrope
multiple
dry ice
increases
Carbides & Cyanides
Carbides CaC2:
Very hard (e.g., SiC)
strong Bronsted __
__ carbides
__ carbides
interstitial carbides
Cyanides CN:
Extremely __ (bind to iron in enzymes)
used in extraction of __
noble prize for buckminsterfullerene
__ C60 and named for American architect
base
toxic
gold
bucky ball
(Group 15)
78% of atmosphere
Important compounds:
NO- paramagnetic
NO₂ (toxic gases)-laughing gas
highly toxic __ gas
__: mixture of 1: 3 ratio of HNO3 to HCL
will oxidize gold
Ammonia (NH₃)- most important compound of group 15
Weak Bronsted __
strong __ base
Very __ in water
Important in __
Nitric Acid (HNO₃)
Used in —— and —
fertilizer bomb parked in truck
— liquid that boils ay 83 Celsius
__ produced by reduction of nitrates with hot metal
soluble in water, mildly toxic, used to cure meat, ___
nitrogen family
78%
yellow brown
aqua regia
base
Lewis
soluble
fertilizers
fertilizer and explosives
colorless
nitrites
carcinogen
Phosphorus
greek- light bringer
__ most abundant element in the body
obtained from __, mineral from of calcium phsopahte
white from the condensed vapor and red from striking a match
Found in __ rocks
Exists as allotropes:
__ phosphorus (reactive) (P4)
__ phosphorus (stable)(P8)
Environmental Impact
Causes ___:
__ blooms
Low __
__ of aquatic life
contains two metalloids: __
phosphate
second
phosphate
white
red
As and Sb
eutrophication
algal
oxygen
death
B, Si, Ge, As, Sb, Te, Po
have the appearance and some physical properties of meatal but behave chemically like a non metal
metalloids
(Group 16)
___
also know as ___
Ozone (O₃) is reactive- blue gas
found in __ ores and copper
liquid=pale blue
gas = colorless
Sulfur
Found in __ & gases
Forms:
SO₂, SO₃
H₂SO₄ (sulfuric acid)
Sulfuric Acid
Most important __ chemical
Properties:
Strong __
__ agent
__ agent
colorless, corrosive, oily liquid
oxygen family
paramagnetic
chalcogens
copper
minerals
industrial
acid
dehydrating
oxidizing
(Group 17) Key Traits
fluorine, chlorine, bromine, and iodine
Highly reactive ___
Very __- decrease from fl to I
“Salt formers”
HF has a high Boling point -hydrogen bonding
Trends
Reactivity __ down group
oxidation state -1 to +7
Acid strength:
HF < HCl < HBr < HI
Uses
HF → etching __
HCl → __ processes
__: iodine astaine
__: bromine
__: fluorine chlorine
halogens
non metal
toxic
decreases
glass
industrial
solid
liquid
gas
glass
(Group 18) Main Points
Very ____ (full electron shells)
Some compounds exist mainly __ with fl and o2
powerful fluorinating agent and xenon oxides are powerful oxidizing agents
Special Property
Liquid helium → __ at very low temps
__: the ability to flow without viscosity
noble gases
unreactive
xenon
superfluidity

CaCo3xMgCO3
dolamite

CaCo3
limestone, calcite, chalk

3BeOxAl2O3×6SiO2
beryl
Ca5(PO4)3OH
hydroxyapatite
tooth enamel
Ca5(PO4)3F
fluorapatite
floride to protect teeth
N2O
laughing gas
NH3
ammonia
HNO3
nitric acid
Sb2S3
stibnite
As2S4
orpiment

As4S4
realgar

PbS
Galena

HgS
Cinnabar

FeS2
pyrite

ZnS
sphalerite
CaSO42H2O
gypsum