f Block Chemistry
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
Why are they called the rare earths if they are more abundant than some other elements?
Because they are difficult to extract and purify.
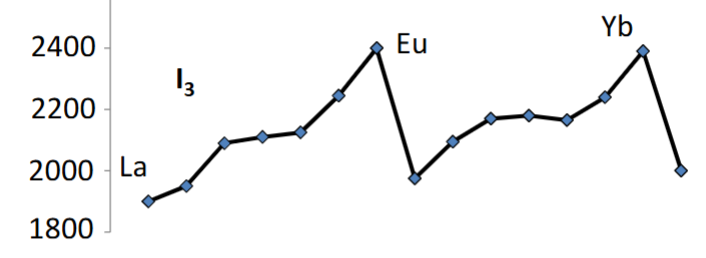
Explain the trends depicted in this graph.
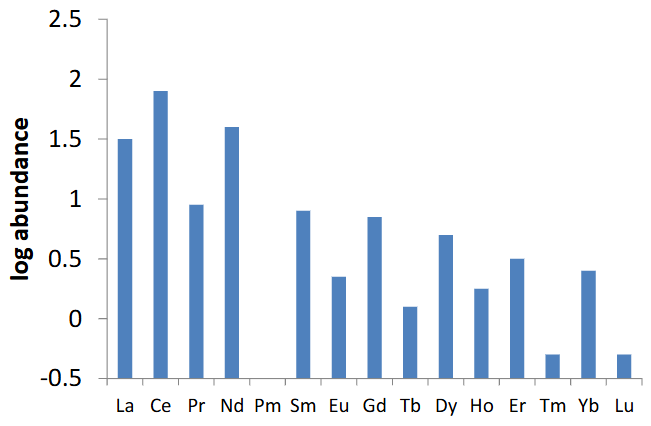
Lanthanides generally get rarer as they get heavier due to decreasing nuclear stability.
This is because of the increase in coulombic repulsion in nuclei with more protons.
The odd/even pattern is due to certain nuclei numbers being more stable.
Pm is so radioactive that it has a very short half life and hence a very low abundance.
How are the different lanthanides separated?
On an ion exchange column.
Different ligands which coordinate to different lanthanides are fixed to a column.
What is HEX?
HEX is UF6.
HEX is used in 235U separation (from 238U) by diffusion.
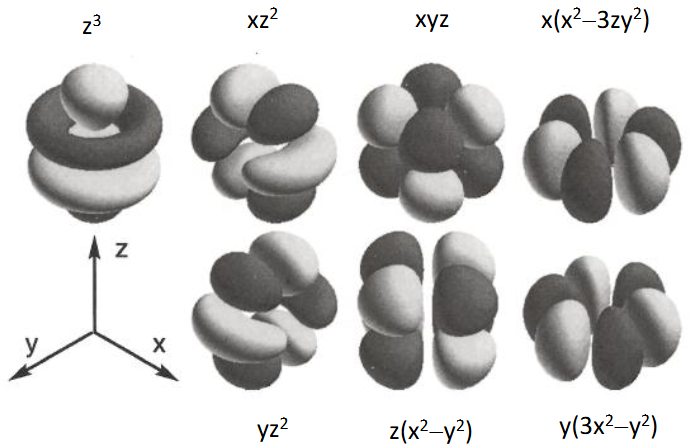
What does Paul mean by ‘lanthanides are lumpy’?
They have high angular nodality
Explain the Relativistic Effect in relation to lanthanides.
A trend of decreasing Ln3+ size across the row is observed.
This was previously thought to be just due to the increase in effective nuclear charge (Zeff).
However, it is now thought to be partially due to relativistic effects:
as an electron approaches the nucleus, it speeds up considerably
at higher speeds, the mass of the electron increases to around 1.35x its usual mass
therefore there is higher electron density near the nucleus, resulting in the observed nuclear contraction
What is the consequence of f block elements having high angular nodality?
They have high corrdination numbers.
They also behave like spheres when it comes to bonding.
Are f orbitals u or g?

They are ungerade.
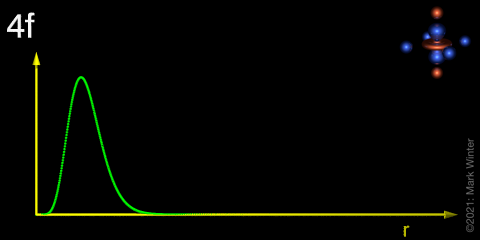
Draw a radial distribution plot to demonstrate the differences between the 4f, 5f and 6s orbitals and describe the key features.
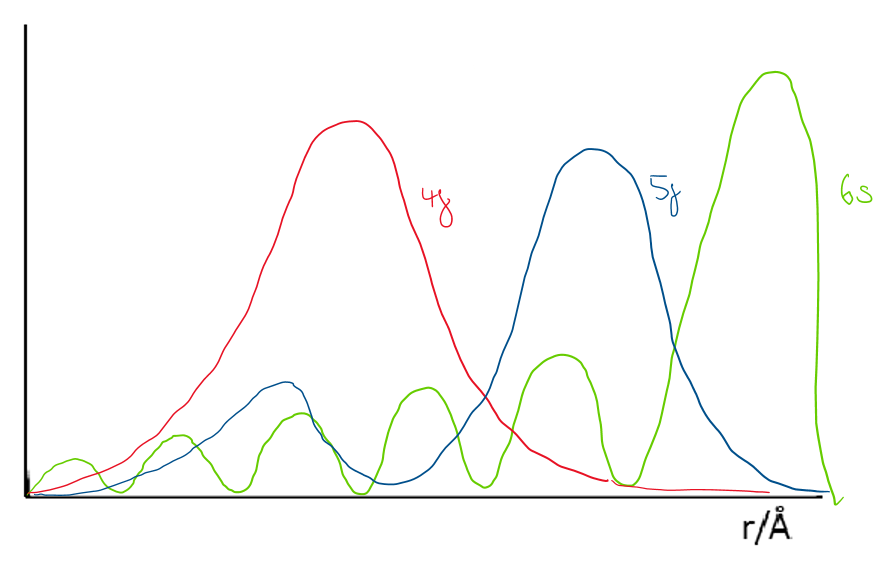
4f orbital has no radial nodes and electron density is closer to the nucleus than 6s so is “core-like”
5f has one node and is less core-like - 5f electrons can therefore interact with things like ligands (“the outside world”)
Approximately how far from the nucleus do ligands sit? How does this explain the bonding of lanthanides?
Ligands sit ~2.5 Å away from the nucleus.
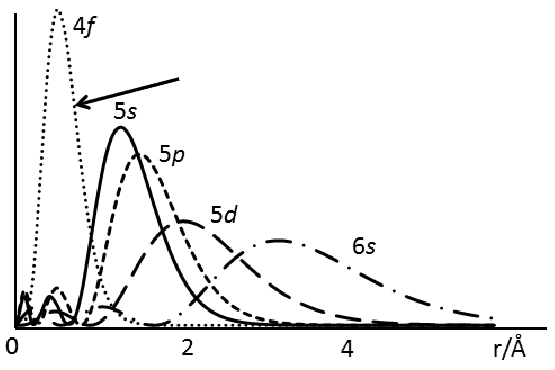
The 4f orbital is far too contracted to interact with the ligands in any covalent way.
Therefore, all of the bonding in lanthanides is ionic.
(There is a little bit of covalency in actinides because the 5f orbital is less core-like)
Explain the different magnitudes of significance of the relativistic effect on 4f and 6s orbitals.
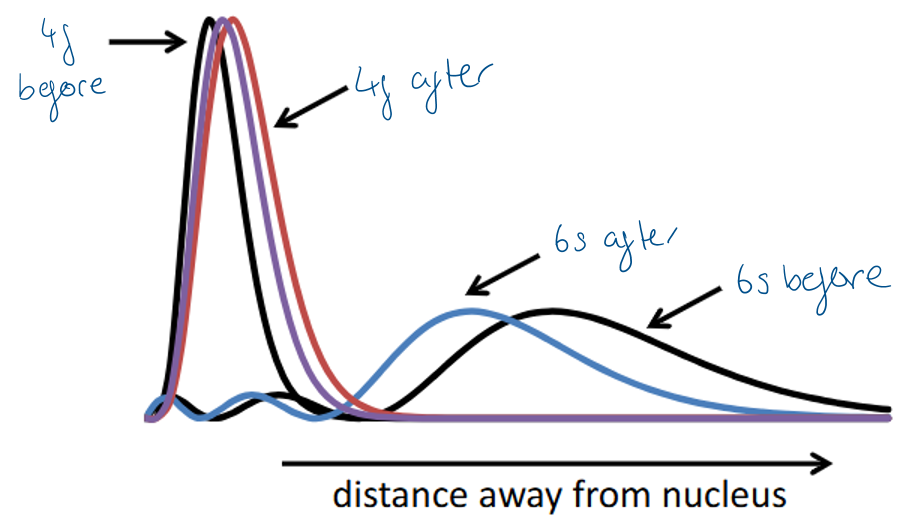
The 6f orbital is more significantly impacted by relativistic effects than 4f orbitals.This is because the 6s electrons can pass right through the nucleus, so reach higher speeds, hence increasing electron density closer to the nucleus.
Why are Ln-ligand bond lengths longer than Ac-ligand bonds?
Actanides are larger than lanthanides (due to occupitation of 5f orbitals).
(f-block - ligand bonding is mostly ionic)
What is Lanthanide Contraction?
The trend of decreasing size across the f block rows caused by relativistic effects (which impact the 6s orbital significantly).
Explain why the 5d elements are smaller than expected?
5d transition metals are contracted due to the relativistic effect shrinking orbitals, such as the 5s orbital.
This is why Ti → Zr → Hf doesn’t really increase in size much, even though theres a significant increase in atomic number. Hence, 4d and 5d elements have similar chemistry (and are therefore difficult to separate).
What does the Aufbau Principle tell us about the ionisation of lanthanides and actanides?
That f electrons are expected to be the first to be removed because they sit at higher energies.
Why is 3+ the most common oxidation state for f-block elements?
The 4f/5f orbitals are the highest in energy so are the first to be ionised (Aufbau Principle).
However, once an f electron is removed, the f orbitals drop in energy and the 6s or 5d orbitals become the highest in energy.
Therefore, the lanthanide loses one f electron, then 6s or 5d electrons until it gets to 3+.
It is difficult to ionise beyond 3+.
Valence electrons in a 3+ lanthanide ion are in f orbitals, which explains their chemistry.
What is the general trend of metallic radius in lanthanides? Explain why it is observed.
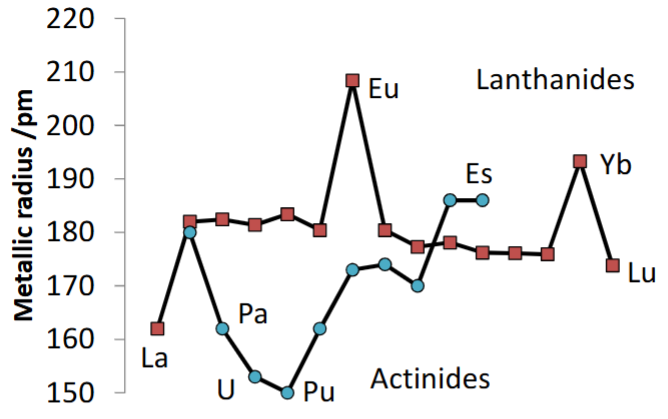
General trend of a very slight decrease in size.
This similarity in size is observed because of the nature of metallic bonding - positive cations in a sea of electrons. Because all lanthanide ions are 3+, the strength of metallic bonding is the same for each so little change is observed.
Eu and Yb are outliers - they have some stability in a +2 state so can exist in a weaker bonding metal system.
What makes Eu and Yb different from the other lanthanides?
Eu2+: f7
Yb2+: f14
While other lanthanides are stable at 3+ oxidation states, Eu and Yb can also exist as 2+ ions because of their electron configurations (↑). Their half filled (f7) and fully filled (f14) sublevels in the +2 state make this state more stable and disfavours the third ionisation:
What notable oxidation state is known for Ce and why is it important?
Ce can exist as Ce4+, which is used as a strong oxidising agent.
How do actinide oxidation states differ from lanthanides?
Actinide oxidation states are much more complex than lanthanides, which generally just occupy +3.
Actinides occupy a variety of oxidation states due to their 5f orbitals being less core-like:
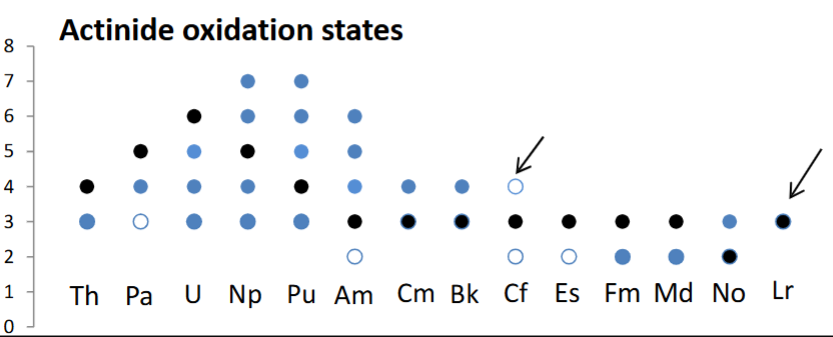
These oxidation states exhibit different solubilities (filled circle is aqueous, unfilled is solid) which can cause issues for radioactive waste handling.
Explain the trend exhibited in the Ligand Field column, specifying the difference between the two circle values

Larger metal ions experience greater splitting by ligands because the valence electrons are held closer to the ligand electrons.
The 4f orbital is core-like and its electron density is held close to the nucleus, so ligands have very little impact on lanthanide energy levels. Molecules which bind lanthanides behave as if the lanthanide is barely bonded at all and it acts like a gas phase ion.
The 5f orbital is less core-like, and holds some electron density further out from the nucleus. Therefore, a larger (but still fairly small) impact is felt on the actinide electrons by the ligand electrons.
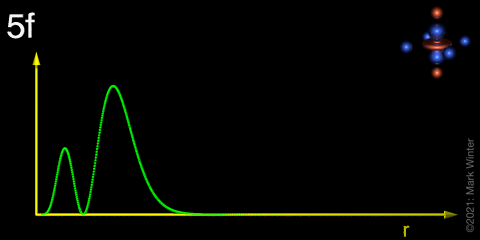
Explain the trend exhibited in the e-e repulsion column, specifying the difference between the two circle values
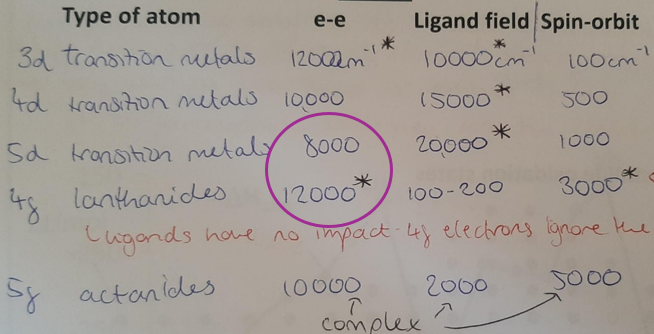
3d → 4d → 5d: orbitals are getting bigger so there is less electron-electron repulsion
4f orbital is core-like so electrons are held close together near the nucleus, hence significant increase in repulsion.
5f orbital is larger and has a radial node so less electron-electron repulsion, but still fairly significant due to the contracted nature of f orbitals.
Explain the trend exhibited in the spin-orbit coupling column.

3d → 4d → 5d → 4f → 5f: electrons have higher angular momentum, hence experience more spin-orbit coupling.
This is denoted by the electrons attraction to the nucleus (distance and size).
What is the Russell Saunders coupling scheme?
The simplification of spin-orbit coupling by summing all of the spin (s) and angular momentum (l) components:
s1 + s2 + s3 … = S
l1 + l2 + l3 … = L
J = S + L
What is the formula for making a term symbol?
What are the rules for J values?
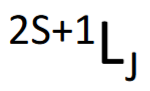
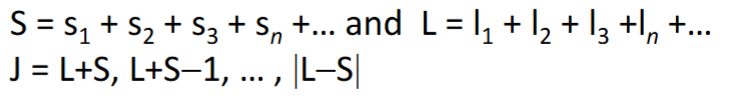
What are Hund’s Rules which define the ground state term symbol?
1) Largest S (maximum spin multiplicity) is the ground state
2) If there are two states with the same S, the ground (/lower) state is the one with the largest L
3) For >half filled states, the maximum value of J is the ground state.
Determine the ground state term symbol for Ce3+
Ce3+ = 4f1
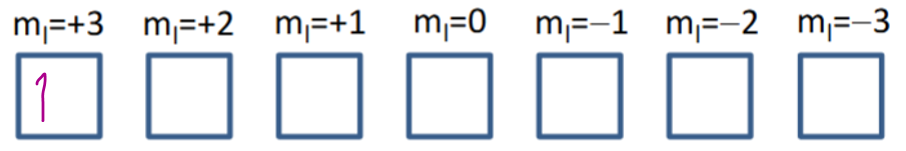
S = (+) ½
(2S+1 = 2)
L = + 3
(L = 0 (S), L = 1 (P), L = 2 (D), L = 3 (F), L = 4 (G), …)
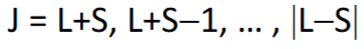
Subshell is < half full so minimum J value is ground state.
J = 3.5, 2.5
Term symbol = 2F2.5
Determine the ground state term symbol for Dy3+
Dy3+ = 4f9
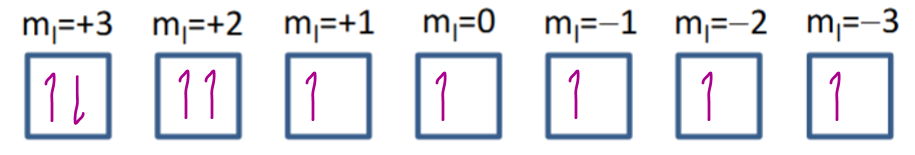
S = (+) 5/2
(2S+1 = 6)
L = + 5
(L = 0 (S), L = 1 (P), L = 2 (D), L = 3 (F), L = 4 (G), L = 5 (H), … )
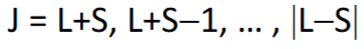
Subshell is > half full so maximum J value is ground state.
J = 7.5, 6.5, 5.5, 4.5, 3.5, 2.5
Term symbol = 6H7.5
How does calculating the magnetic moment (μeff) differ between transition metals and lanthanides?
For transition metals, we can approximate using the spin-only formula because the ligands quench the angular momentum (L):
μeff = 2√(S(S+1)
For lanthanides, L isn’t quenched so we use the Landé formula:
μeff = g√(J(J+1)
where g =
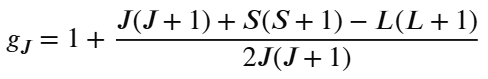
(note that equations will be given in the exam*)
Which two lanthanides must you be careful to spot in an exam question?
Eu and Yb - these are ‘evil’ and will not follow the expected lanthanide trends.
Eu and Yb can be stable in the 2+ oxidation state.
What is the assumption made in using the Landé formula to calculate the magnetic moment of a lanthanide?
That the lanthanide (and its electrons) are acting like that of a gas phase ion.
Explain why Eu have matching calculated and experimental values for magnetic moment.
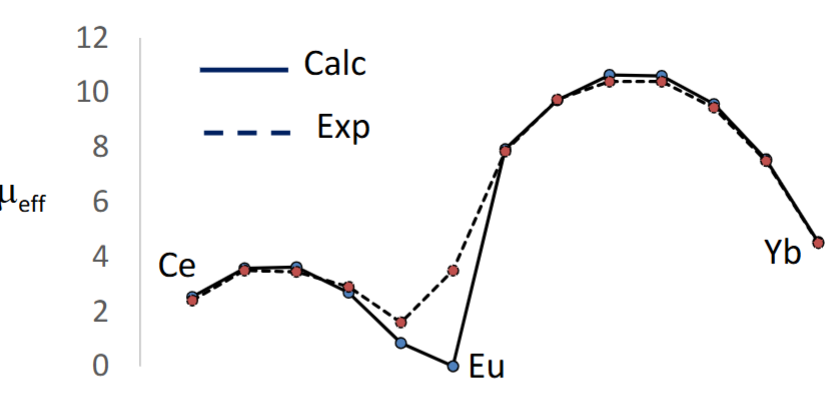
Europium’s magnetic ground state is very close to some low excited states, meaning that it occupies states other than the ground state at room temp.
Hund’s rules and the Landé formula assume occupation of the ground state, making the calculated value different to the experimental value.
Describe the general magnitude of lanthanide magnetic moments.
Generally fairly enormous

Describe magnetism in actinides.
Due to their size, spin-orbit coupling begins to dominate.
This means that the actinide magnetic moments are much more difficult to predict.
Describe the difference between 3d transition metal and lanthanide electronic absorption spectrum.
Transition metal electronic absorption spectra contain broad peaks due to vibrating ligand distances. Lanthanide spectra contain very sharp peaks because the f orbitals do not interact with the ligands.
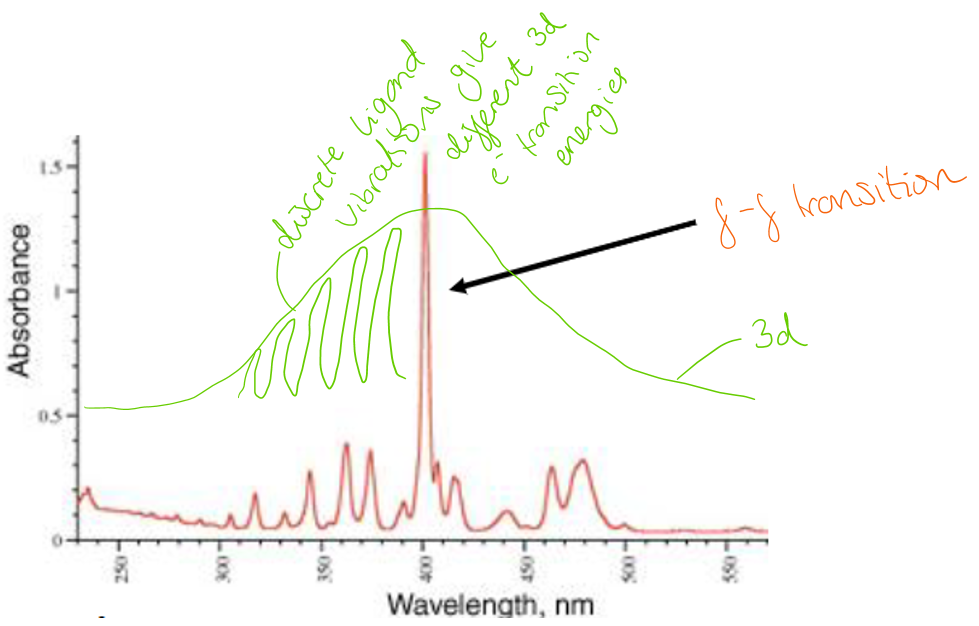
Describe the expected and observed colours of lanthanides.
Expected: f-f transitions give sharp peaks so we expect lanthanides to give ‘brilliant’ colours
Reality: in reality, f-f transitions don’t obey the symmetry selection rule (u → u: disallowed). This means that lanthanides are essentially colourless.
Note: the spin selection rule is obeyed by lanthanides (f-f) because they contains many (Russell-Saunders) states of the same spin multiplicity, so this is not limiting the electronic transitions, just the symmetry selection rule.
Describe the expected and observed colours of actinides. Describe how this differs from lanthanides.
Lanthanides are essentially colourless due to f-f transitions disobeying the symmetry selection rule.
Actinides can have some colour because the 5f orbital is a little more extended and can slightly interact with the ligands (symmetry allowed → peaks more intense → ε value is larger (similar magnitude to 3d TM transitions)). This makes their electronic absorption peaks slightly broader than lanthanides, causing the colours to be muddy.
Draw and describe the energy levels and transitions involved in a Nd YAG laser.
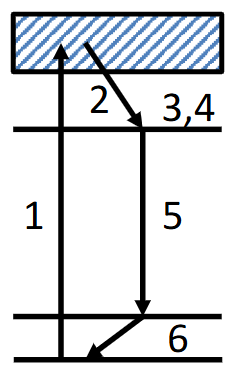
1) UV promoted, symmetry allowed excitation (eg. charge transfer)
2) Relaxation to an excited f-state by intersystem crossing
3,4) Build up of population in excited f state (population inversion)
5) Intense transition between f states, giving an almost perfectly monochromatic emission
6) Relaxation back to ground state
Why do lanthanides produce such strong magnets?
They contain lots of unpaired electrons which allows for some high, static magnetic fields.
Draw and describe the energy levels and transitions involved in lanthanide luminescence.
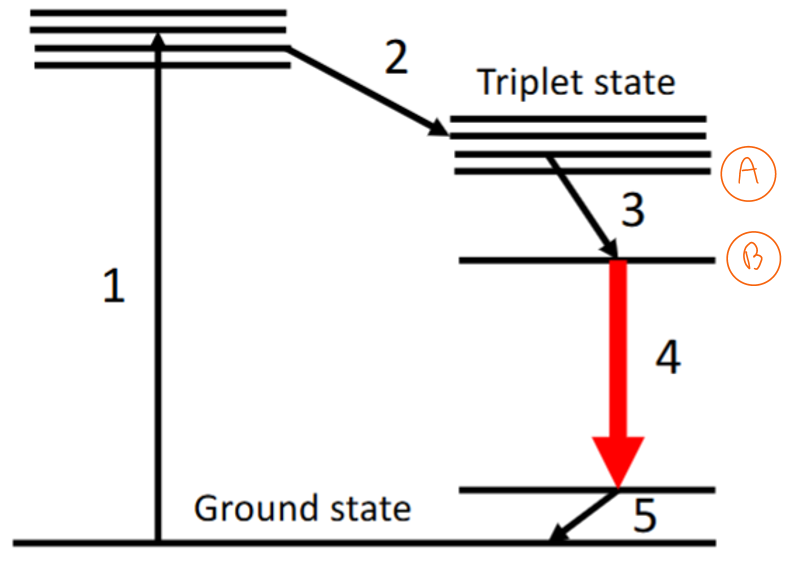
1) Absorption, typically based on the ligand (eg. MLCT), giving an excited state. Usually done with UV and is a fully allowed transition.
2) Intersystem crossing (within the ligand)
3) Excitation goes to an f state on the lanthanide. Rate of this transfer is dependent on the energy difference between states A and B
4) Emission
5) Relaxation back to ground state
Describe how lanthanides can be used as a sensor and why tuning is important.

Lanthanides can be attached to a ligand which absorbs at a specific wavelength (1). The excitation passes to the lanthanide in step 3, then luminescence is observed (4).
Observation of luminescence indicates the excitation of the ligand
Tuning the energy gap between A and B allows you to maximise transfer and minimise back-transfer, allowing for a more intense luminsecence for observation.
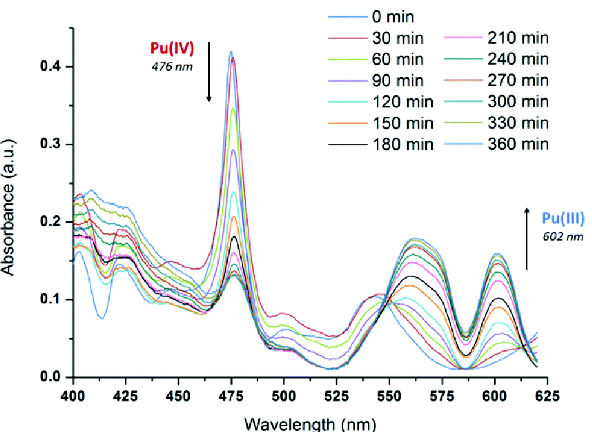
Describe how this graph demonstrates how the Eu complex acts as a senor.
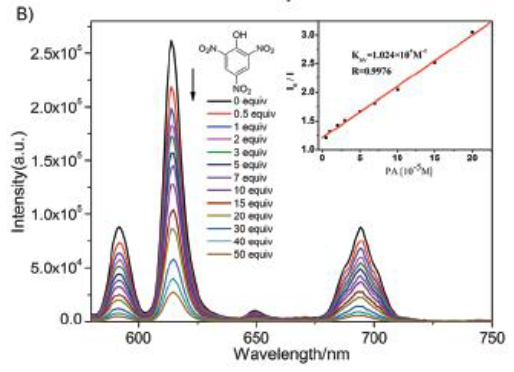
A trinitrophenol molecule can sit in the midde of the tetrahedral Eu complex.

This changes the energy level of the ligand excited state (A).
This quenches the fluorescence and indicates the presence of trinitrophenol (explosive like TNT).
What is one application of the monochromatic emission of lanthanides?
Monochromatic emission = bright colours
Lanthanide systems are used in phone screens and other displays as ‘screen phosphors’.
Examples:
Red: Eu3+ embedded in Y2O2S
Green: Tb3+ embedded in La2O2S
Notice how often mutiple lanthanides are present in these structures.
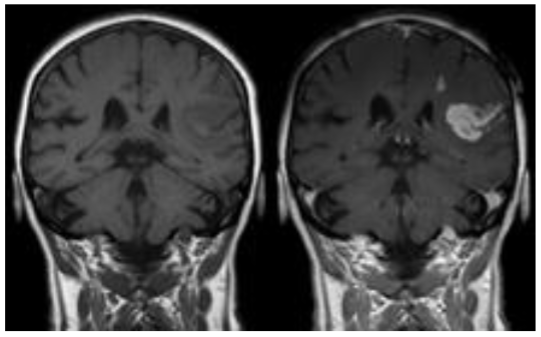
What is an application of lanthanide magnetism?
Lanthanides have high inherent spin magnetism.
We can therefore use them as ‘relaxation agents’.
Their magnetic field can cause relaxation the spins of nuclei in nearby molecules.
This is used in MRI imaging to visualise the position of a stroke site.
Gd3+ is typically used for this because it has the maximum number of unpaired f electrons (7) and therefore has a huge magnetic field and can relax other nuclei very fast. (Gd is unfortunately very toxic)
Describe the bonding of lanthanides to ligands.
The 4f orbital is too core-like to participate in any significant amount of covalent bonding to ligands.
Therefore, lanthanide ligands must be charged to give an ionic interaction.
Lanthanide ions, therefore, bond well to oxygen based ligands (they are oxophilic).
Lanthanide ions can have very high coordination numbers due to their large size.
What is the typical coordination number range for lanthanides?
6-12 is possible.
Larger end of the scale is generally only accessible with small bite angle/ non-bulky ligands.
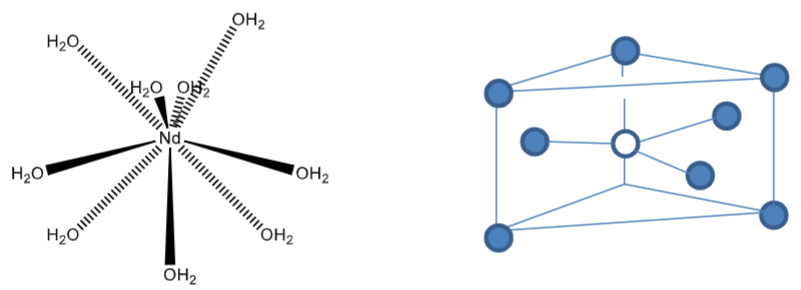
Draw a tricapped trigonal prism (9 coordinate) geometry.
Draw the bonding of NO3- as a ligand to a lanthanide centre. Describe the key features.
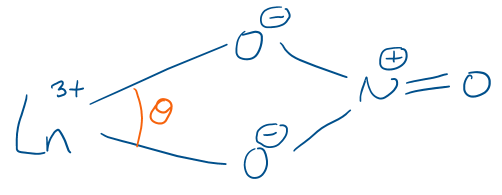
Small bite angle (θ) so up to 6 can coordinate to one centre (12 coordinate)
Lanthanides are oxophilic so bond well to the negatively charged oxygens in NO3-
Valence lanthanide orbitals are 4f and 6s so there is little directional symmetry, so small bite angle ligands can bond
What is the chelate effect and how does it relate to lanthanide complexes?
Chelate effect: significantly strong bonding to polydentate ligands than monodentate.
Because lanthanide-ligand bonding is ionic and weak, these complexes can be thermodynamically unstable.
Often, polydentate ligands are required to strongly bind lanthanide ions.
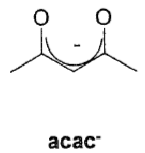
Draw an acetonyl acetate (acac) ligand binding to a lanthanide ligand.
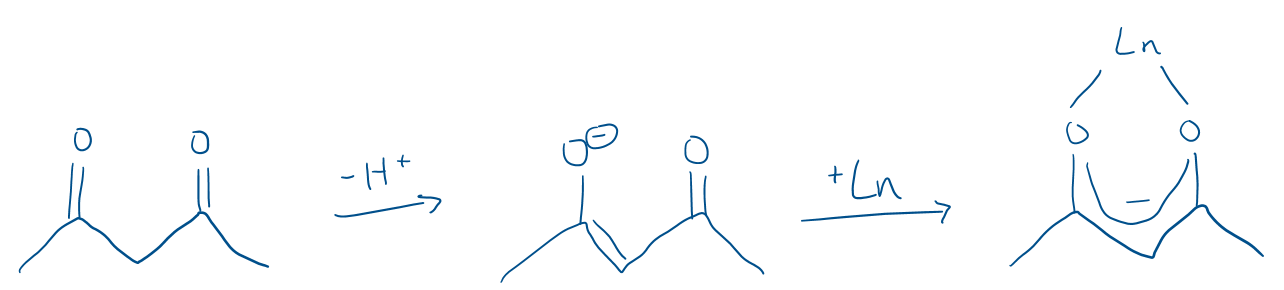
This makes use of the chelate effect and the oxophilic nature of lanthanides.
Draw an oxalate ligand binding to a lanthanide ligand.
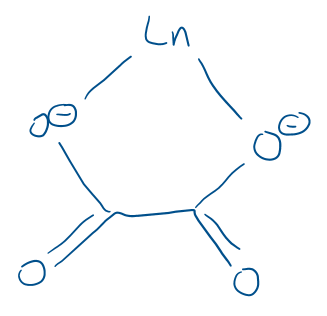
This makes use of the chelate effect and the oxophilic nature of lanthanides.
Describe how a DOTA molecule coordinates to a lanthanide ion.
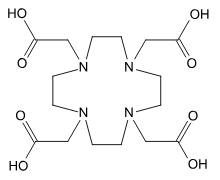
Chelates to 8 different points (idk why it bonds to the nitrogen it just sorta seems to?)
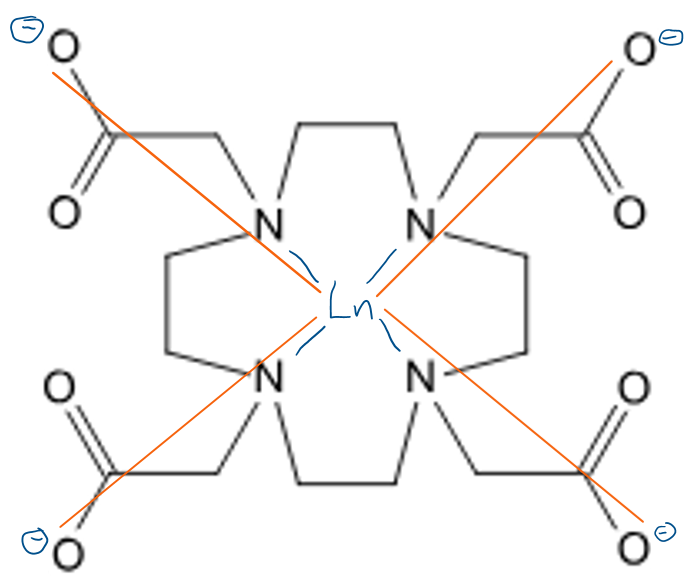
This makes use of the chelate effect and the oxophilic nature of lanthanides.
Give the function of tributyl phosphate (TBP) bonding to lanthanides.
TBP uses the oxophilic nature of lanthanides to bind to lanthanides.
It is used to extract uranium in nuclear reprocessing.
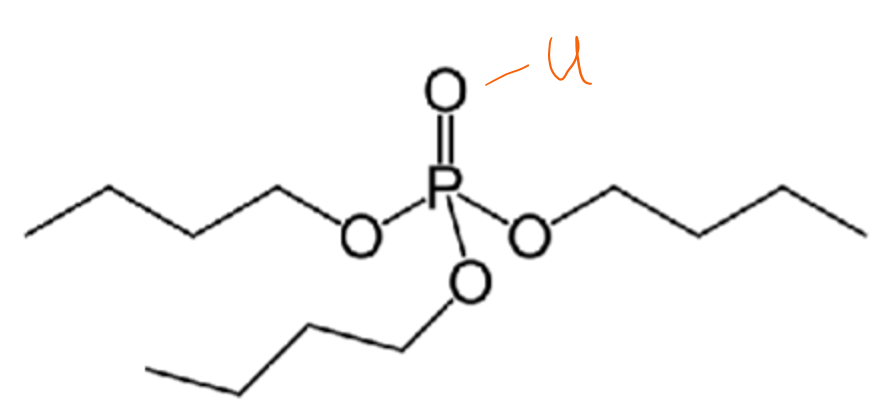
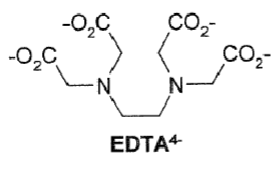
Draw EDTA ligand binding to a lanthanide ligand.
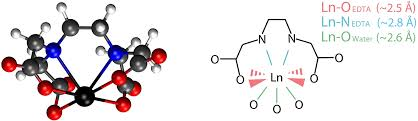
Draw a graph depicting the trend of stability constants of lanthanides with acac and EDTA across the period.

Across the period, Zeff increases so ionic bonding to ligands gets stronger:
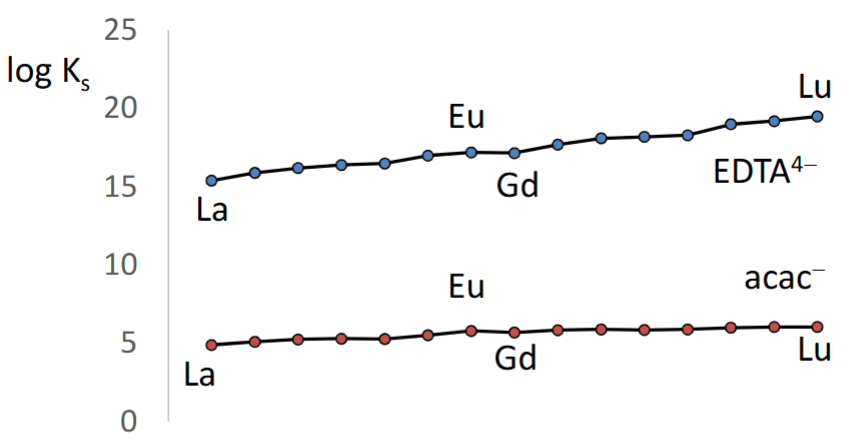
What does this graph tell us about lanthanide separation.

The chelate effect is critical for lanthanide binding.
Minimal differences in binding across the period means that it is hard to separate lanthanides by binding.
We can immobilise a ligand on a resin and separate using these slightly different KS values.
This is called ion exchange chromatography.
How do we separate lanthanides?
Ion exchange chromatography:
We can immobilise a ligand on a resin which binds different lanthanides differently.
We then flow aqueous solution over the resin and separate using the lanthanides’ slightly different KS values.
KS values only vary a little across the period and the trend is due to increasing Zeff.

Describe how lanthanide complexes act as a catalyst.
They are mostly redox inactive and act as Lewis acids (electron acceptors).
This is because they are typically 3+
Lanthanide catalysts also have high ligand exchange rates (so high rates of reaction).
Describe the action of SmI2.
The Sm in SmI2 is in its 2+ oxidation state. We know that lanthanides are stable in 3+ states so we expect (and observe) the Sm to be able to lose an electron.
Therefore, SmI2 acts as a reducing agent.
It can facilitate Pinacol coupling:
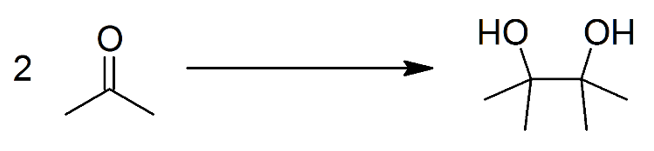
Give the simple equation for Pinacol coupling.

Give an example of a lanthanide that acts as an oxidising agent and briefly explain why.
Ce(IV)
We know that lanthanides are stable as 3+ ions, so Ce4+ readily gains an electron, thus acts as an oxidising agent.
Draw the general structure of lanthanide complexes which exhibit some covalent bonding. What is notable about their application?
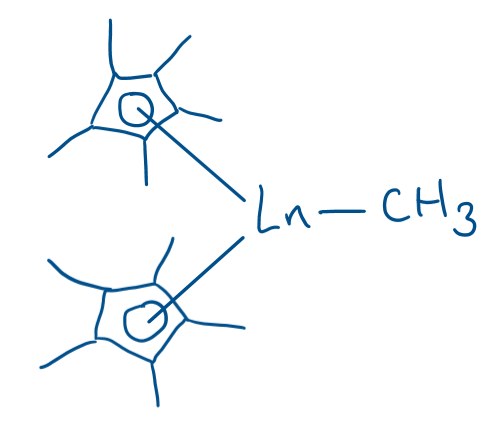
(Two Cp* ligands)
These catalysts can be used to activate methane, which is ordinarily very difficult.
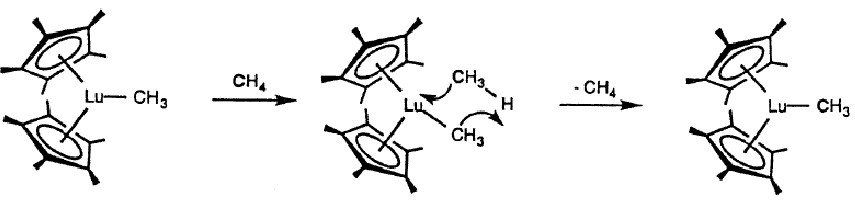
This can be observed using labelled methane (eg. isotopes)
Give the mechanism for methane exchange on a lutetium complex.

Draw the mechanism for polymerisation of methylmethacrylate using a lanthanide complex.
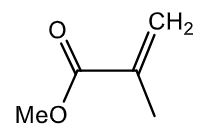
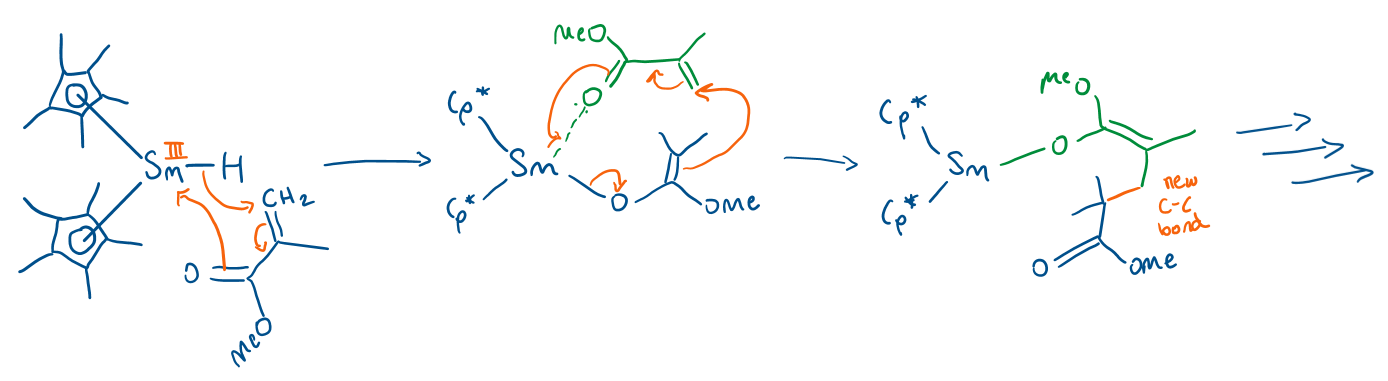
Gives polymethylmethacrylate (perspex).
Sm acts as a Lewis acid (accepts electrons from methylmethacrylate), doesn’t do oxidative addition/reductive elimination.
Give the trend of half life for the actinides. Why is this trend observed?

Half life (generally) decreases with the heavier actinides.
There is also an up/down pattern → this is because nuclei have stable numbers (similar to electronic configurations).
Draw graph and explain the Liquid Drop Model.
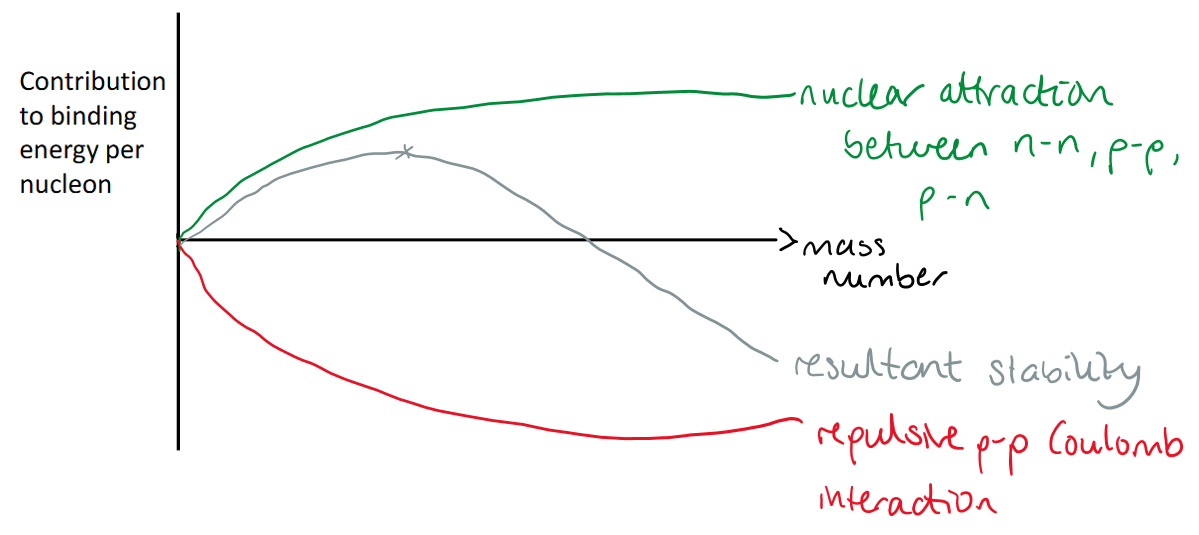
At higher mass numbers (heavier elements), the repulsive force wins out and the element has a higher tendency to decay.
(The marked position on the greyline is the maximum stabilty, which is around iron.)
This explains the decrease in half life across the actinides.
Describe the interaction displayed in this plot:
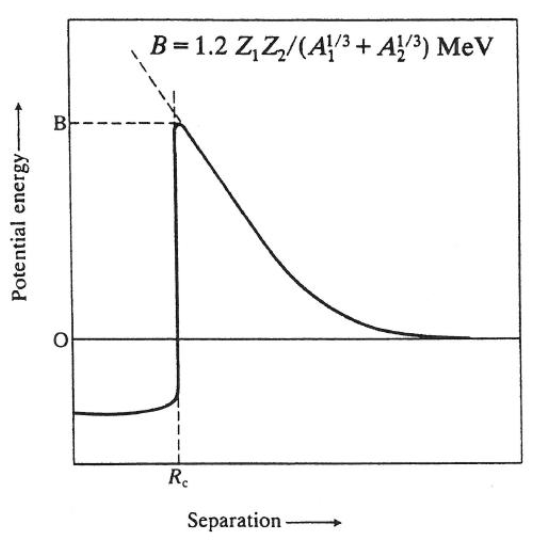
The plot shows how coulombic repulsion of a proton increases as it approaches the nucleus.
(equation not required knowledge)
The height of the Coulomb barrier is a proportional to Z (nuclear charge) and inversely proportional to A (nuclear mass). Ie. larger mass = lower barrier.
However, the barrier height is in mega eV, so even with large A, crossing this barrier should not be possible.
(does happen because of proton tunnelling)
If the Coulomb barrier (shown below) is mega eVs high, how can nuclear reactions still occur?

Protons can tunnel the energy barrier.
This means that the limiting factor isn’t the height of the barrier, but the width.
The equation for the probability of tunnelling is:
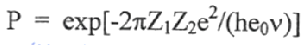
(equation not required knowledge)
This demonstrates that the probability of successful tunnelling is dependent on the nuclear charge (higher charge = higher velocity of emitted radiation particle = higher chance of tunnelling = increased radioactivity)
Therefore, almost all f block elements are α-emitters because of their high nuclear charge.
What does this plot demonstrates about nuclear stability of heavier elements?
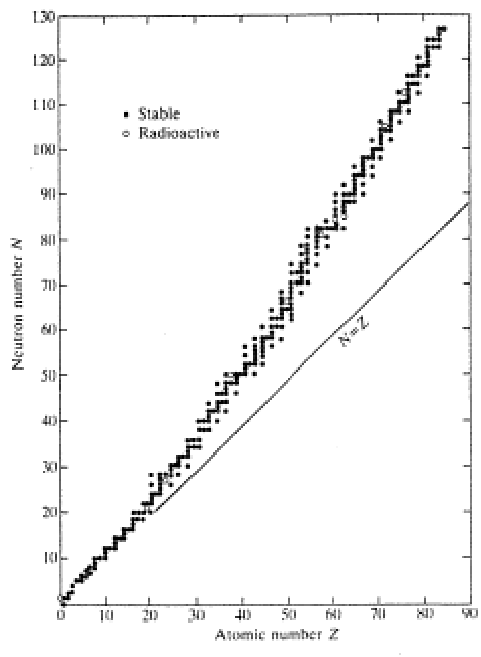
This plot shows that at around atomic number 20, the number of neutrons begins to outweigh the number of protons.
This is because coulombic repulsion between protons begins to get too strong and more neutrons are needed to reduce it.
In unstable nuclei (when there aren’t enough nuclei to balance the protons), the protons decay (β+ decay) as below:
proton → neutron + γ ray + positron (e+)
What happens when a proton decays (β+ decay) and what is the application of this?
proton → neutron + γ ray + positron (e+)
When a positron interacts with an electron:
positron + electron → 2 γ rays
This is used in tumor imaging.