Chemistry OCR - Module 3: Section 2 Physical Chemistry
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
What is enthalpy change?
Enthalpy change, ΔH, is the heat energy transferred in a reaction at constant pressure. The units for ΔH are kJ mol-1.
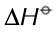
What does the image mean?
The measurements were made under standard conditions and that the elements were in their standard states (i.e. their physical states under standard conditions). Standard conditions are 100 kPa (about 1 atm) pressure and a temperature of 298 K (25°C)
What is an exothermic reaction?
Exothermic reactions give out energy. ΔH is negative.
Oxidation is usually exothermic.
The temperature often goes up.
The oxidation of carbohydrates is exothermic.
Combustion is exothermic.
What is an endothermic reaction?
Endothermic reactions absorb energy. ΔH is positive.
Temperature often falls.
The thermal decomposition of calcium carbonate is endothermic.
The main reactions of photosynthesis are also endothermic.
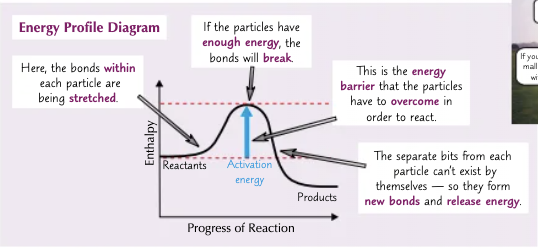
What are enthalpy profile diagrams?
They show you how the enthalpy changes during reactions.
What is the activation energy Ea?
The minimum amount of energy needed to begin breaking reactant bonds and start a chemical reaction.
What does the enthalpy profile diagram look like for an endothermic and exothermic reaction?
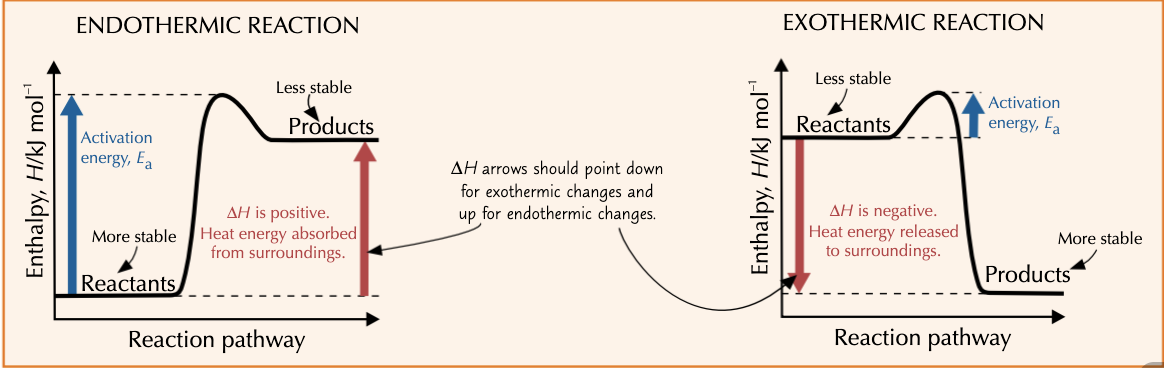
In terms of enthalpy, when is a substance more stable?
The less enthalpy a substance has, the more stable it is.
What are the different ΔH? (° will be used for the weird symbol on top right that looks like an underground tube sign)
Standard enthalpy change of reaction - ΔrH°, is the enthalpy change when the reaction occurs in the molar quantities shown in the chemical equation, under standard conditions.
Standard enthalpy change of formation - ΔfH°, is the enthalpy change when 1 mole of a compound is formed from its elements in their standard states, under standard conditions.
Standard enthalpy change of combustion - ΔcH°, is the enthalpy change when 1 mole of a substance is completely burned in oxygen, under standard conditions.
Standard enthalpy change of neutralisation - ΔneutH°, is the enthalpy change when an acid and an alkali react together, under standard conditions, to form 1 mole of water.
What is the enthalpy change for breaking bonds?
You need energy to break bonds, so bond breaking is endothermic ΔH is positive
What is the enthalpy change of making bonds?
Energy is released when bonds are formed, so this is exothermic (ΔH is negative).
What is the enthalpy change for a reaction?
It is the overall effect of breaking and making bonds. If you need more energy to break bonds than is released when bonds are made, ΔH is positive. If you need less, ΔH is negative.
How is energy needed to break the attraction between atoms and ions?
In ionic bonding, positive and negative ions are attracted to each other. In covalent molecules, the positive nuclei are attracted to the negative charge of the shared electrons in a covalent bond.
You need energy to break this attraction, stronger bonds take more energy to break. The amount of energy you need per mole is called the bond dissociation enthalpy.
Bond dissociation enthalpies always involve bond breaking in gaseous compounds.
Why are average bond enthalpies not exact? What is average bond enthalpy?
Water has two O-H bonds. It does not take the same amount of energy to break both. (OH- is a bit easier to break apart because of the extra electron repulsion.)
The data book says the bond enthalpy for O-H is +463kJmol-1. It is a bit different because it is the average for a much bigger range of molecules not just water.
Average bond enthalpy:
The energy needed to break one mole of bonds in the gas phase, averaged over many different compounds.
What do you need to measure enthalpy change in a lab?
The number of moles of the stuff that is reacting.
The change in temperature.
What equation can be used to calculate enthalpy change?
q = mcΔT
q = heat lost or gained in joules. This is the same as the enthalpy change if the pressure is constant.
m = mass of water in the calorimeter, or solution in the insulated container in grams.
c = specific heat capacity of water 4.18 J g-1 K-1
ΔT = the change in temperature of the water or solution in K.
What is Hess’ Law?
The total enthalpy change of a reaction is always the same, no matter which route is taken.
How can enthalpy changes be worked out from enthalpies of formation?
Enthalpy changes of formation are useful for calculating enthalpy changes you cannot find directly. You need to know enthalpy change of formation for all the reactants and products that are compounds.
Enthalpy change of reaction = the sum of enthalpy change of formation of products - the sum of enthalpy change of formation of the reactants.
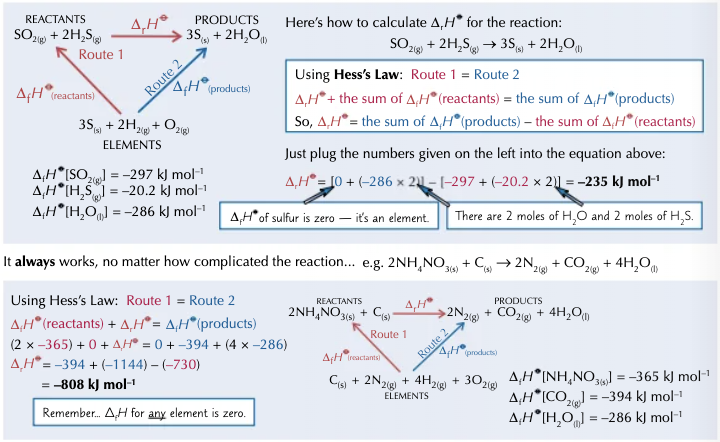
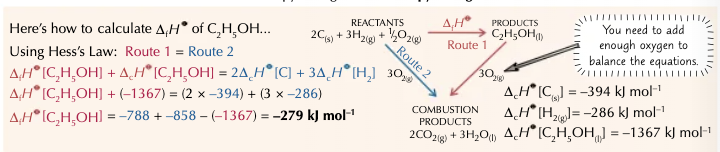
How can enthalpy changes be worked out from enthalpies of combustion?
A similar method to that of enthalpy change of formation.
How can enthalpy changes be calculated from average bond enthalpies?
In any chemical reaction, energy is absorbed to break bonds and given out during bond formation. The difference between the energy absorbed and released is the overall enthalpy change of reaction.
Enthalpy Change of Reaction = Total Energy Absorbed - Total Energy Released
When will two particles react?
They collide in the right direction. They need to be facing each other the right way.
They collide with at least a certain minimum amount of kinetic energy.
What is the activation energy? Ea
The minimum amount of kinetic energy particles need to react.
The particles need this energy to break the bonds to start the reaction.
How can activation impact how a easily a reaction might occur.
Reactions with low activation energies often happen pretty easily.
Reactions with high activation energies do not happen pretty easily and the particles need more energy by heating them.
What does an energy profile diagram look like?
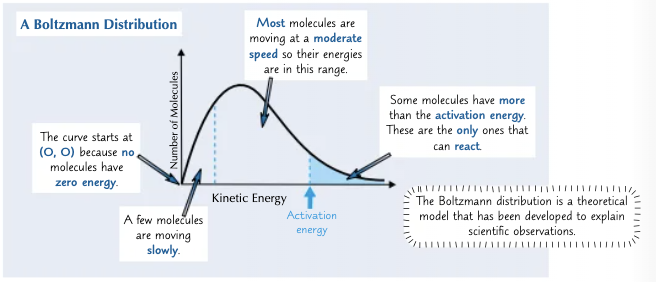
What is a Boltzmann Distribution and what does it look like?
If you plot a graph of the number of molecules in a substance with different kinetic energies you get a Boltzmann distribution.
The particles beyond the activation energy under the Boltzmann distribution are the particles that can react.
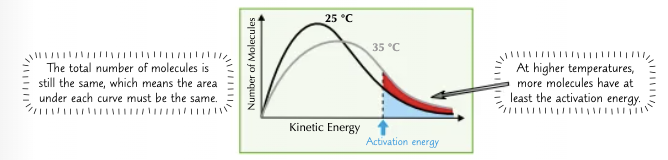
How does increasing temperature make reactions faster?
If you increase the temperature, the particles will, on average, have more kinetic energy and will move faster.
So, a greater proportion of molecules will have at least the activation energy and be able to react. This changes the shape of the Boltzmann distribution curve, it pushes it over to the right.
Because the molecules are flying about faster, they will collide more often. This is another reason why increasing the temperature makes a reaction faster.
How does increasing concentration speed up reactions?
If you increase the concentration of reactants in a solution, the particles will be closer together, on average. If they are closer, they will collide more frequently. If there are more collisions, they will have more chances to react.
How deos increasing pressure speed up reactions?
If any of your reactants are gases, increasing the pressure will increase the rate of reaction. It is pretty much the same as increasing the concentration of a solution, at higher pressures, the particles will be closer together, increasing the chance of successful collisions.
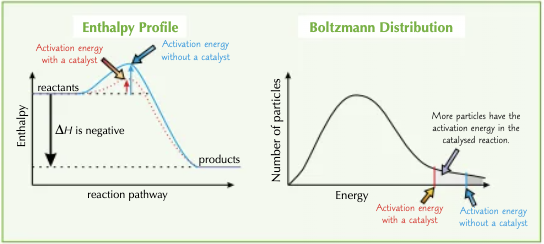
How can catalysts speed up reactions?
A catalyst increases the rate of a reaction by providing an alternative reaction pathway with a lower activation energy. The catalyst is chemically unchanged at the end of the reaction.
How do the enthalpy profiles and Boltzmann distributions show why catalysts work?
The catalyst lowers the activation energy, meaning there are more particles with enough energy to react when they collide. So, in a certain amount of time, more particles react.
What are the different types of catalyst?
Heterogeneous - A catalyst that is in a different phase from the reactants i.e. in a different physical state. The reaction happens on the surface of the heterogeneous catalyst. So, increasing the surface area of the catalyst increases the number of molecules that can react at the same time, increasing the rate of the reaction.
Homogeneous - Catalysts that are in the same physical state as the reactants. Usually a homogeneous catalyst is an aqueous catalyst for a reaction between two aqueous solutions. A homogeneous catalyst works by forming an intermediate species. The reactants combine with the catalyst to make an intermediate species, which then reacts to form the products and reform the catalyst.
Why are catalysts good for industries?
Loads of industries rely on catalysts. They can dramatically lower production costs, give you more product in a shorter time and help make better products.
Why are catalysts good for environmental sustainability?
Using catalysts means that lower temperatures and pressures can be used. So energy is saved, meaning less CO2 is released, and fossil fuel reserves are preserved. Catalysts can also reduce waste by allowing a different reaction to be used with a better atom economy.
Catalytic converters on cars are made from alloys of platinum, palladium and rhodium. They reduce the pollution released into the atmosphere by speeding up the reaction.