CSB520 Disease Process Mid semester
1/103
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
104 Terms
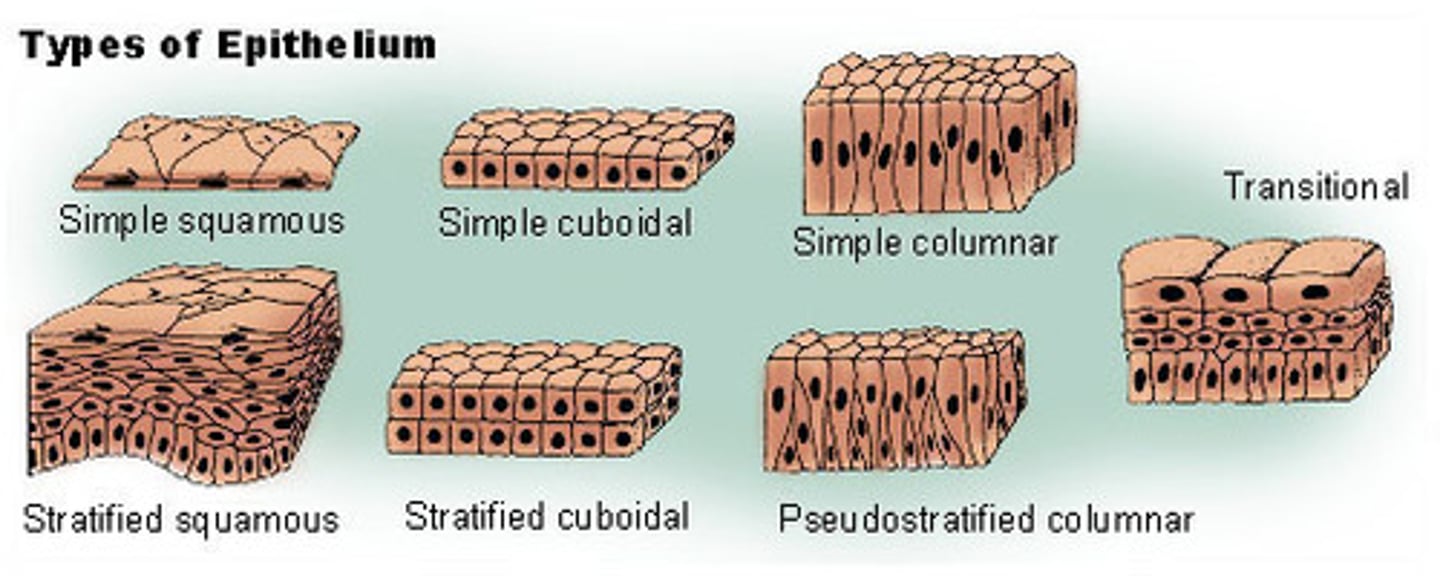
Give some examples of cell types that are considered epithelial (outside lining)(6)
Simple (one layer) - squamous (alveoli) , cuboidal (exocrine glands), columnar (secretion like mucus)
Stratified (multiple layers) - squamous (skin, oesophagus)
Pseudostratified - upper respiratory tract
Transitional Epithelium - urinary system
Give some examples of cell types that are considered connective
- Endothelium (lining of blood vessels)
- Cartilage (chondrocytes)
- bone (osteoblasts, osteocytes)
- Fibroblasts (collagen, used in scaring)
- Fat
Give some examples of cell types that are considered 'others'
Mesothelial cells (layer around body cavities)
Melanocytes (produces melanin)
Germ cells -sperm
Lymphoid tissue - (behind epithelium tissue)
Bone marrow
What is meant by labile, stable and permanent cells, give some examples (types of cells)
Labile - continuously dividing. eg Skin, GIT, reproductive. Haemopoetic stem cells
Stable - quiescent / can divide when stimulated. eg liver, kidney, lung, pancreas. Smooth muscle, fibroblasts, endothelial
Permanent - non-dividing. eg Cardiac and skeletal myocytes, neurons. Can't get cancers but can in children due to embrogensis (cell death apoptosis)
Describe the main differences between apoptosis and necrosis
Apoptosis- cell suicide / programmed cell death (slow bleed). Separates itself from other cells then dies (active dismantling). No inflammation or scarring. Active form of cell death
Necrosis - accidental / catastrophic death (stroke). Kills multiple cells with its death. Passive form of cell death
What is an infarct and how does it occur
Area of necrotic tissue = infarct (caused by sudden severe stress)
Define Autophagy
Cell reduces in size by eating itself (still alive)
What determines whether a stimulus causes atrophy or infarction (3)
Severity, duration and types
Infarct happens with disruption in blood which leads to ischeamia
What effect does ageing have on cells and organs
causes cells to become less able to divide and function, tissues to lose mass and become more rigid, and organs to slowly lose function (due to replacement of functional tissue with collagen, increasing the risk of age-related diseases
Define Atrophy
Tissue/organ shrinkage through cellular apoptosis and/or autophagy
Define Hyperplasia
Increase in cell number through cell division
Define Metaplasia
a reversible adaptation where one normal, differentiated cell type transforms into another normal, differentiated cell type in response to chronic stress.
Define types of cellular adaptation (5) and understand when these process are not possible/reversible
- atrophy: decrease in cell size and/or no. resulting in a reduction in organ size
- hypertrophy: increase in all size and increase in tissue/organ size
-hyperplasia: increase in cell no. and increase in tissue/organ size
- metaplasia: reversible change in which one normal, well-differentiated cell type is replaced by another
- dysplasia: abnormal growth and maturation of cells w/ disordered structure and loss of uniformity
Not reversible in the heart
Is metaplasia an active or passive cellular change and is it reversible
Active, it requires the use of energy/ATP & in some cases there will now be increased cell turn-over e.g.stratified squamous cells constantly divide at the basal layer. The only passive process covered isnecrosis.
Yes if the environment changes back or stress removed
Hypoxia
Lack of oxygen, inadequate oxygen tension at a cellular level
Hypozaemia / Hypoxemia
Lack of oxygen (reduced concentration) in the arterial blood
A- (prefix)
Without / away from
Hyper- / Hypo -
Hyper - 'excessive', 'above' or 'beyond'
Hypo - 'under', 'below', beneath'
Aemia / emia
Suffix means 'blood' or relates to 'blood condition'. The prefix haemameans blood
Epi, Endo, Myo -
Epi - On, upon
Endo - inward, within
Myo- muscles
What type of epithelium is the best at withstanding a physical stress?
Stratified squamous epithelium.
Define neoplasia
new abnormal growth (other words tumour)
Define Tumour
An abnormal mass of tissue that forms when cells grow and divide more than they should or do not die when they should
Define Malignancy
The ability of abnormal cells to grow uncontrollably to invade nearby tissues and spread /Potentially fatal
Difference between Benign and Malignant tumours (4)
1. Never Metastasizes / Can potentially metastasize
2. Encapsulated / Infiltrative growth pattern
3. Homogenous (uniformity of cells)/ Heterogenous (pleomorphic - cells lack uniformity)
4. Well differentiated / Well differentiated or poorly or undifferentiated (anaplastic)
Define Dysplasia
Presence of cells of an abnormal architecture, shape and size
Most dysplasia is destroyed by the immune system, if not, leads to cancer
Define Angiogenesis
formation of new blood vessels
Difference between Homogenous vs Heterogenous
Well differentiated vs varying degrees of differentiation
Looks the same everywhere vs looks different
Less variation in tumours vs different mutations, cell types and metabolic needs
Define Encapsulated tumour
A tumour (benign) that is surrounded by well-defined fibrous capsule, separating it from surrounding tissues
How is a undifferentiated cell differentiated (6)
Appearance, function, growth rate, tissue organisation, hormones
Define Carcinoma and Carcinoma-in-situ
Malignant tumour, arising from epithelial cells, outside skin and lining of organs, 90% of cancers
A type where cells have not yet invaded anywhere else (found in the place they were first formed)
Define Metastasize
When the cancer moves from its original location to another site
This can be via blood, lymphatics and direct seeding
Nomenclature of tumours
Malignant tumours- 'Sarc' = malignant and connnective tissue cell of origin
Benign tumours - 'oma' benign cell of origin
See week 1 half way down for cell types
What does the term mean, break down the word:
Chondrosarcoma
Adenocarcinoma
Leiomyosarcoma
Cystadenoma
Osteosarcoma
Chondrosarcoma: Chondro = cartilage, Sar = malignant & connective tissue origin
Adenocarcinoma: Adeno = glandular (secretory) epithelial, Car =malignant & epithelial origin
Leiomyosarcoma: Leio = smooth, myo = muscle, Sar = malignant &connective tissue origin
Cystadenoma: Cyst = fluid-filled sack (describing pattern of growth),Adeno = glandular (secretory) epithelial, NO Car so benign
Osteosarcoma: Osteo = bone, Sar = malignant & connective tissue origin
What does the term mean, break down the word:
Liposarcoma
Haemangioma
Angiogenesis
Liposarcoma: Lipo = fat, Sar = malignant & connective tissue origin• Haemangioma:
Haem = blood, angio = vessel, NO SAR so benign•
Angiogenesis: Angio = blood vessel, genesis = generate
Why are carcinomas the most common cancer diagnosed in adults
Cells that are dividing have the greatest chance of sustaining a mutation (exposed to outside environment). Epithelial cells are capable of dividing and some continuously divide.
Epithelial cells are on the front line
List the 3 main routes of metastasis and common sites affected
bloodstream (liver , lungs, brain, bone)
lymphatic spread (axillary nodes for breast, cervical nodes for heart and neck)
direct seeding (Ovarian, breast)
What are 2 cellular adaptations that increase the risk of tumour development?
Hyperplasia (increase cell division) & metaplasia (change from one normal/well-differentiated cell type to another normal/well-differentiated cell type).Dysplasia is NOT an adaptation
Define Hyperaemia
Increase of blood in vessels. Active response. Inflammatory response
Define Oedema (exudate vs transudate)
Excess fluid in interstitial fluid/body cavities (fluid between cells).
Exudate - Inflammatory extravascular fluid with high protein concentration
Transudate - Extravascular fluid with low protein concentration
Define Effusion
abnormal accumulation of fluid in the body / fluid outside blood vessels
Define resolution
restoration of normal architecture and function (healing without scarring)
Define organization
replacement with scar tissue (healing with scarring)
What is an ulcer, and how do acute and chronic ulcers differ?
An ulcer is a localised lesion involving necrosis (death) of tissue on a body surface such as the skin, stomach, or gastrointestinal tract, where there is loss of the surface epithelium and underlying tissue — often described as a “sore.”
Acute ulcer:
The necrotic core is removed, collagen fills the defect, and epithelial cells proliferate to cover the surface → resulting in healing.Chronic ulcer:
The necrotic core persists and fails to heal properly, leading to ongoing inflammation, fibrosis, and an increased risk of carcinoma (malignant transformation).
Define Abscess
occur in a solid tissue organ, body walls off inflammation. Normally containing pus
What form of cell death causes an inflammatory response
Necrosis
What are the 3 features of acute inflammation-
1. Hyperemia,
2. Oedema in the form of an exudate,
3. Neutrophil - white blood cells (help recruitment and activation of other immune cells)
What are the 3 possible outcomes of acute inflammation
Resolution, organization, chronic inflammation (wall off / containment inflammation)
What are the 3 main components of granulation tissue and their role/purpose in repair
Granulation tissue (immature scar tissue) forms during the repair phase and has three main components:
Macrophages – Remove debris and secrete growth factors that promote healing.
Fibroblasts / Myofibroblasts – Secrete collagen to fill the tissue defect and provide structural strength.
Angiogenesis (new capillaries) – Form new blood vessels to deliver oxygen and nutrients to the healing tissue.
Once repair is complete, fibroblasts and macrophages leave, and the new vessels regress by apoptosis, leaving a mature collagen scar.
In chronic inflammation, granulation tissue is continuously formed due to persistent injury.
What are the consequences of healing through organization
During acute, lost some functional tissue which has been replaced with collagen. Overtime collagen can contract which does bring further problems to some organ systems
What are the 3 features/components of acute inflammation
Hyperaemia, oedema (exudate) & neutrophils
Describe the possible negative consequences of chronic inflammation (2)
lose functional tissue and if epithelial cells are present, increase risk of carcinoma (cancer)
What are the 3 features/components of chronic inflammation?
Ongoing injury
Repeated attempts at repair (through the formation of granulation tissue & proliferation of surrounding cells if occurring in labile/stable tissues)
Lymphocytes.
What are the 3 ways in which we get chronicinflammation?
Stimulus causing acute inflammation persists or remains unresolved.
Acute inflammation is repeated over time.
It occurs in special cases such as autoimmune diseases (e.g., rheumatoid arthritis) and persistent infections (e.g., tuberculosis).
What is meant by sterile and non-sterile sites in the human body
sterile - no microorganisms present(blood, brain and CSF, Bone and marrow, lower respiratory, upper urogenital, stomach).
Non sterile- microorganisms presents (skin, GIT, upper respiratory, lower urogenital (lower urethra))
Understand the main differences between the innate and adaptive systems
Innate- response doesn't change, same acute response, fast response.
Adaptive - able to differentiate between targets (including your own immune systems), slow, learn and remembers
What is the difference between autoimmune and hypersensitivity responses, and what are the main types of hypersensitivity reactions?
Autoimmune response:
The immune system targets the body’s own normal tissues, mistaking them for foreign.
Hypersensitivity response:
The immune system overreacts to a harmless or exaggerated stimulus, causing tissue injury or disease.
Types of hypersensitivity:
Type I – Immediate (IgE-mediated):
Prototypic: Allergies, anaphylaxis
Pathologic: Tissue damage/inflammation
Type II – Antibody-mediated (IgG or IgM):
Prototypic: Autoimmune reactions
Pathologic: Functional impairment without direct cell injury (e.g., myasthenia gravis)
Type III – Immune complex-mediated:
Antigen–antibody complexes deposit in tissues → inflammation and tissue injury (e.g., lupus, glomerulonephritis)
Type IV – Cell-mediated (T-cell):
Sensitised T lymphocytes cause tissue damage
TH1/TH17 cells cause chronic inflammation
TH2 cells can contribute to lesions seen in some immediate hypersensitivity reactions but are not considered Type IV
Have a basic understanding of what is meant by the term immune-compromised and the patients at risk (3)
immune systems is depleted, makes person susceptible to opportunistic infections. Can be from lost innate responses (macrophages, neutrophils, epithelium, cough reflex), genetic, from drugs /treatment
Following a myocardial infarction, what type of inflammation and repair occur?
Inflammation: Acute inflammation occurs
Repair: Organisation (a type of repair) follows
Why does chronic inflammation increase the risk of carcinoma formation?
Chronic inflammation increases cancer risk because surrounding epithelial cells proliferate (undergo hyperplasia) in a futile attempt to repair ongoing tissue injury.
This constant cell division, combined with the presence of free radicals and inflammatory mediators, increases the likelihood of DNA mutations and malignant transformation.
➡ It’s important to note that granulation tissue itself does not cause cancer — it simply forms as part of the healing process and is present at all sites of chronic inflammation.
What is the mature scar tissue made out of (apart fromin the brain)?
Collagen protein (in the brain it is glial cells)
Define Congestion
passive accumulation of blood
Define haemorrhage and describe what determines its consequences.
The ongoing loss of blood from a damaged blood vessel.
The consequences depend on:
Site: e.g. in the brain → can cause death, in tissue → causes a bruise (haematoma).
Amount of blood lost: large volumes can lead to shock or death.
Speed of loss: rapid loss is more dangerous than slow loss, even if the total volume is the same
Define haematoma
Accumulation of blood, outside the blood vessels usually clotted
Define Thrombus
Blood clot attached to the blood vessel wall
Define Embolus and examples
An embolus is an undissolved mass that travels through the bloodstream until it lodges in a vessel and blocks blood flow (embolism).
Examples include:
Thromboembolism: a fragment of a blood clot.
Tumour emboli: clumps of cancer cells.
Septic emboli: clumps of microbes.
Gas emboli: air or nitrogen bubbles.
Fat emboli: droplets of fat (e.g. after long bone fractures).
Amniotic fluid emboli: entry of amniotic fluid into maternal circulation.
Foreign material: e.g. injected particles.
Define Aneurysm
an abnormal, localised, dilatation of an artery or ventricle. Occurs in high pressure. Prone to rupture
Define atheroma and explain the meaning of its word parts.
Sclerotic plaque in an artery
Athero = arterial
Sclero= hardening
Osis = process of
Define Atherosclerosis
Chronic inflammatory process within the wall of an artery (ongoing injury, attempts at repair, inflammatory cells plus oxidised lipids and calcium crystals) (crystals feel like glass when feeling the artery)
the buildup of fatty plaques in artery walls causing narrowing and hardening of arteries.
Define Ischemia
blood flow to organ is reduced
Define Hypertension
higher than normal blood pressure
Define stasis
blood not moving likely due to being stationary
Define Hypercoagulability
blood more likely to clot
Identify blood flow and pressure differences in the systemic, pulmonary and (systemic) venous circuits
high pressure systemic arterial ( so blood can be pushed to capillaries for gas exchange), low pressure in pulmonary circuit (lungs), low pressure in systematic venous circuit
Identify Constituents of blood and the organs that are relevant for its synthesis / maintenance (5)
Plasma 55% and Formed elements (platelets, leukocytes, erythrocytes) 45%
Bone marrow, liver, spleen, kidneys, lymphatic system
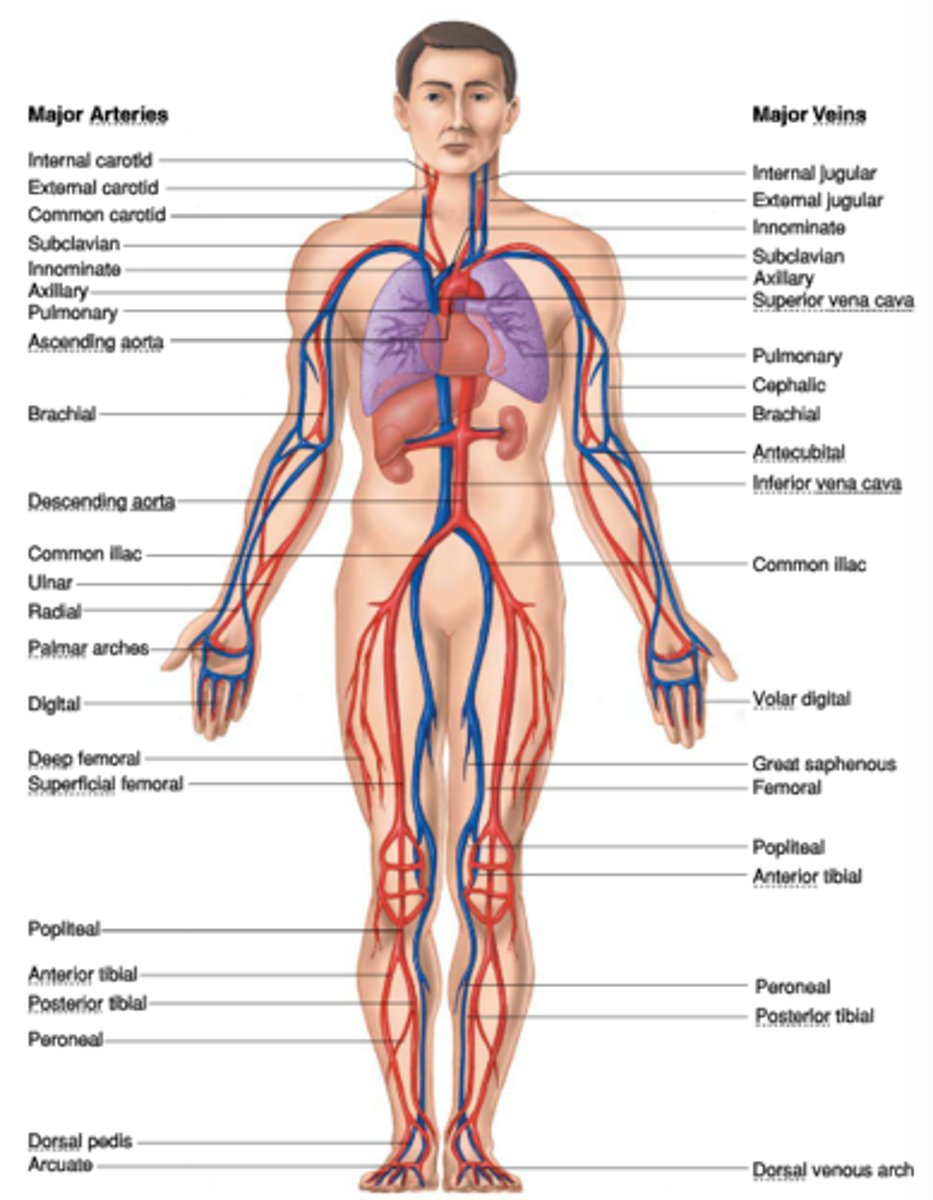
Location of aorta (abdominal), carotid and cerebral arteries, coronary arteries, inferior and superior vena cava, femoral and iliac veins
Compare superficial with deep vein thrombi (DVT)
Superficial (close to surface of skin, varicose veins (spider veins), Pain, ulceration of overlying skin, oedema 'downstream' of distal to clot) vs Deep Veins (often silent, may cause swelling of the limb, deep vein thrombi often embolise as a large mass)
How do the kidneys contribute to systemic hypertension
RAAS (Renin Angiotensin Aldosterone System)- System is activated when the body has low blood pressure or if there is an injury
Renin (released by kidneys) --> Angiotensin 2 (causes vasoconstriction (constriction on blood vessels, leads to vascular resistance)) --> Aldosterone (from adrenal gland (causes kidney to uptake sodium (Na+) which takes up water which increases blood volume)
List the risk factors of DVT and track their path if they embolise
Stasis & hypercoagulability and end of path lungs
List the main risk factors for the development of atherosclerosis (7)
Increasing age, being male, smoking, systemic hypertension, hyper or dyslipidaemia, diabetes, being of Aboriginal & Torres Strait Islander descent
How could atherosclerosis in the abdominal aorta lead to our sudden death
Predisposes to the formation of an aneurysm which if it ruptures kills us quickly via internal haemorrhage & hypovolemic shock. The abdominal aorta is carrying oxygenated blood downwards towards the lower limbs, it feeds into smaller arteries, arterioles &capillaries where gas exchange occurs
What is the difference between angina and a myocardial infarction?
Angina is chest pain that occurs because the heart muscle does not receive enough oxygenated blood due to narrowed / partial blockage coronary arteries. Myocardial infarction is a heart attack occurring from prolonged blockage of a coronary artery
Describe the inflammation and repair that occurs following an MI
The infarcted tissue triggers an acute inflammatory response, causing hyperaemia, oedema, and neutrophil infiltration.
Hyperaemia increases hydrostatic pressure, pushing fluid into the damaged tissue.
Increased vascular permeability allows plasma proteins to leak out, forming an exudate.
Since cardiac muscle is a permanent tissue, repair occurs via granulation tissue formation rather than regeneration.
Macrophages remove necrotic debris and apoptotic neutrophils.
Fibroblasts migrate in and secrete collagen fibres to replace the lost myocytes.
New capillaries (angiogenesis) grow in to supply oxygen, nutrients, and growth factors.
As healing progresses, the macrophages and fibroblasts leave, the new capillaries regress, and the granulation tissue matures into a collagen scar.
This scar contracts over time, pulling the edges of viable myocardium together — a process known as organisation
Briefly describe what complications can occur following an MI
heart failure, loss of some function and electrical impulse
Lift side heart failure causes, forward effects, backward effects
Causes - Ischaemic heart disease, Systemic hypertension, Aortic / mitral Bicuspid valve disease
Forward effects (same as right side)- Renal response to reduced CO, Compensation via RAAS (Angiotensin 2 causes increased vascular resistance, Aldosterone causes kidneys to retain salt (Na+) and water to increase blood volume)
Backward effects- Pulmonary congestion, Increased hydrostatic pressure, Pulmonary oedema
What are two signs of left sided heart failure
Dyspnea and cough with blood tinged phlegm
Right side causes, forward effects, backward effects
Causes - LHF or pulmonary hypertension due to congestion in pulmonary circulation
Forward effects (same as left side) Lack of output to lungs and blood retrun to left side of heart --> decreased CO --> decreased perfusion to tissues of the body --> fatigue
Decreased CO --> kidney compensation activation of the RAAS
Backward effects- Systemic VENOUS congestion, Peripheral oedema, increased hydrostatic pressure
What is the sign of right sided heart failure
Severe oedema (pitting oedema) in the limbs, distended jugular vein
List some of the common causes and possible consequences of endocarditis
is the inflammation or infection of the inner lining of the heart (endocardium), typically involving the heart valves.
Common causes:
Bacteria entering the bloodstream (eg. from surgery)
Damaged or abnormal heart valves that provide a surface for bacteria to adhere to.
Intravenous drug use, which introduces bacteria directly into the circulation.
Possible consequences:
Loss of functional heart tissue due to inflammation and destruction of valve structures.
Heart failure from valvular damage or impaired pumping efficiency.
Emboli formed from infected material breaking off and spreading to other organs (e.g. brain, kidneys, lungs).
Systemic infection or sepsis if the bacteria spread beyond the heart.
List some of the common causes and possible consequences of pericarditis-
bacterial infections, autoimmune disorders, complications after a heart attack or surgery, injuries to the chest, kidney failure, and certain medications or cancer treatments. Consequences - fluid build up (pericardial effusion), cardiac tamponade (life-threatening pressure on the heart), and constrictive pericarditis (scarring and thickening of the pericardium), potentially causing heart failure.
List the main impacts of ageing on the cardiovascular system
increase BP (hypertension), As cells age, they atrophy and die. The remaining cells have to pick up the slack of the dead cells. This means the cells are working harder. They create collagen between the cells instead of elastic fibres. This is inevitable with age
What is endocarditis/pericarditis/valvestenosis/valve insufficiency or incompetence?
Peri = around, referring to the sac around the heart
Endo = inner, referring to inner layer of the heart including the valves
Card = heart
-itis = inflammation of
Valve stenosis: valve no longer fully opens
Valve insufficiency/incompetence: valve fails tofully close thus allowing regurgitation
Which organ(s) are at risk of damage in a patient with right-sided heart failure?
Organs with a large outflow into the inferior vena cava typically the liver & kidneys.
Which organ(s) are at risk of damage in a patient with left-sided heart failure?
Lungs, from the chronic congestion and hemorrhaging; right side of the heart due to pulmonary hypertension
Describe the innate defences in the conductive & respiratory portions of the lungs.
Conductive portion (nose, trachea, bronchi):
Nasal hairs and turbinates filter and trap large particles from inspired air.
Goblet cells secrete mucus, which traps dust, microbes, and debris.
Ciliated epithelium sweeps the mucus upward toward the pharynx (mucociliary escalator).
Airway reflexes such as coughing and sneezing expel trapped material.
Respiratory portion (bronchioles → alveoli):
Cellular defences include macrophages, neutrophils, and lymphocytes, which destroy inhaled pathogens and debris.
👉 Together, these innate mechanisms keep the airways clear and protect the lungs from infection and injury.
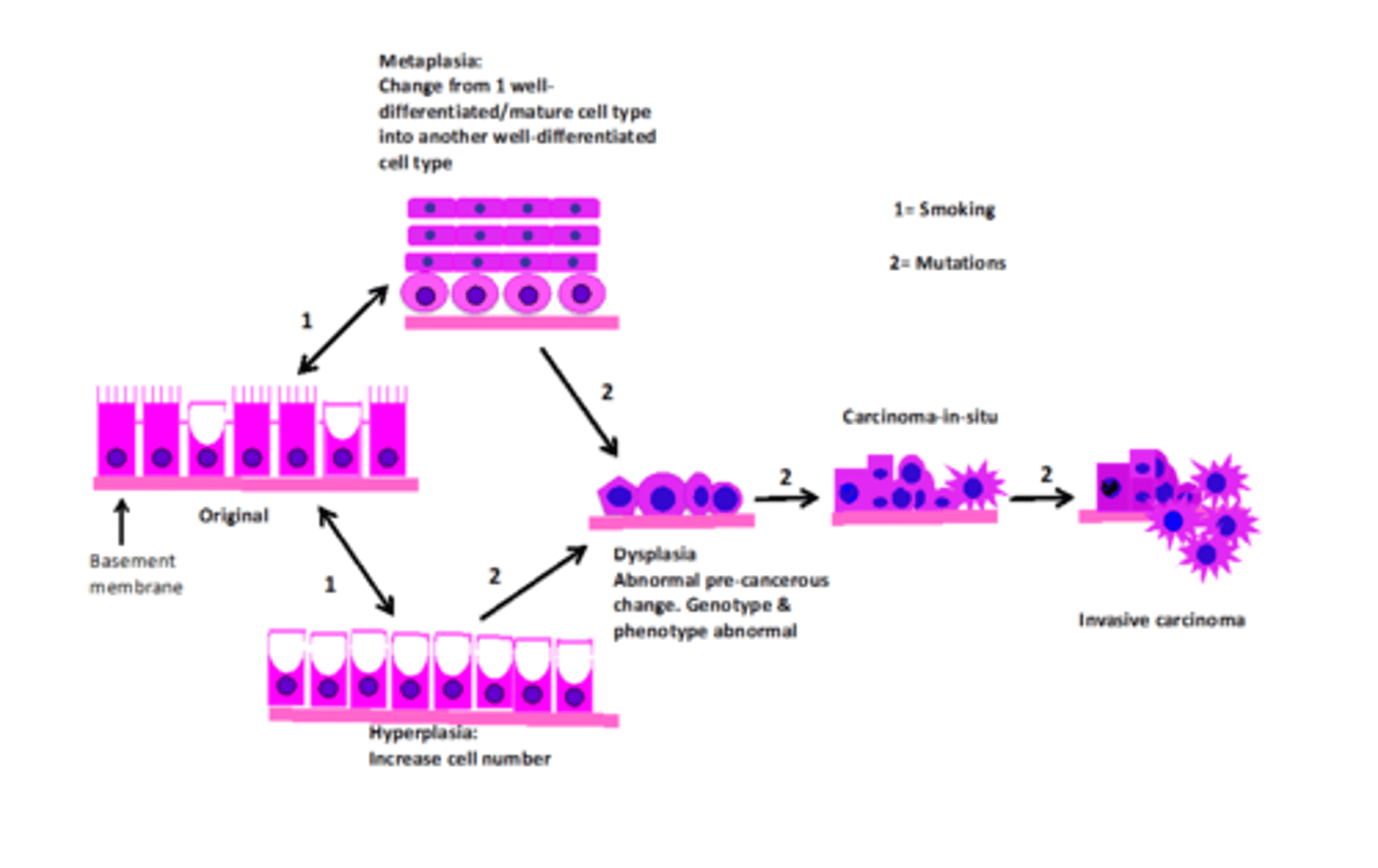
Using a diagram, describe the pathogenesis of the 2 most common primary lung cancers.
Cilia --> Metaplasia --> Stratified squamous --> Dysplasia --> Squamous cell carcinoma
Goblet cell --> Hyperplasia (produce more mucus) --> Secretory Epithelium --> Dysplasia --> Adenocarcinoma
Why are the lungs a common site for secondary cancers?
Lungs are the most common site because all fluid travels to the lungs (lymph, blood etc)
How are the lungs affected by left ventricular failure?
Left ventricular failure (LVF) leads to pulmonary congestion and edema due to increased pressure in the pulmonary circulation. This occurs because the failing left ventricle cannot effectively pump blood forward, causing backward pressure buildup into the pulmonary veins and capillaries.
How do lung diseases contribute to right ventricular failure?
Lung diseases can lead to right ventricular (RV) failure, primarily through pulmonary hypertension, which increases the workload on the right ventricle. This condition is called cor pulmonale, referring to right heart failure due to chronic lung disease.
Where do most pulmonary emboli (PE) travel from?
The majority of pulmonary emboli (PE) originate from deep vein thrombosis (DVT) in the lower limbs.