Radiotherapy & Radiation physics
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
Ionising radiation properties
Contains enough energy to remove electrons from atoms creating ions.
Interacts strongly with tissues.
Damages cells/DNA
Detected for imaging
Main types of ionizing radiation & their properties
Alpha particles: heavy, positively charged, don’t travel far & are stopped by paper / skin.
Not commonly used in imaging as they don’t penetrate far enough.
Beta particles: high speed electrons or positrons (positively charged electrons), lighter & travel further than alpha, stopped by aluminium.
X-rays & Gamma rays: high energy electromagnetic waves with no mass & no charge, are attenuated by bone/lead.
Important in imaging as they can be picked up by detectors outside the body.
Neutrons: Indirectly ionizing via collision / activation & are stopped by low-z elements.
Not commonly used in imaging
Ionizing radiation sources
Radioactive decay of radionuclides
X-ray tube (is a form of particle accelerator)
Nuclear reactors
Particle accelerators
Nuclear fusion
Cosmic rays
Process & effect of radioactive decay of radionuclides
Ionising radiation produced when unstable radionuclide release energy/particles to achieve a more stable state.
Particles emitted knock electrons off atoms in surrounding matter causing ionisation.
Process of creating ionizing radiation with an X-ray tube & nuclear reactors
X-ray tube:
Electrons accelerated towards a metal target.
Sudden deceleration when they hit target releases X-rays.
Nuclear reactors:
Nuclear fission causes heavy nuclei to split & release energy, neutrons, gamma radiation.
Particle accelerator uses
Charged particle beams (used in radiotherapy) - directly treat cancer by firing high energy particles at tumour, which gets damaged by energy deposited.
Form radionuclides via nuclear reaction - produce radioactive isotopes used in imaging/therapy.
Fire high-energy particles at target nucleus causing nuclear reaction & target becomes radionuclide.
Produces radioactive isotopes used in imaging/therapy.
Process of creating ionizing radiation with Nuclear fusion & Cosmic rays
Nuclear fusion - light nuclei combine to form heavier nucleus releasing energy.
Cosmic rays - high energy particles from space interact with atmosphere producing ionising radiation.
Characteristic X-rays production & its difference to gamma
Gamma ray emission comes from nucleus & X ray comes from electron cloud.
Atomic electrons transitions produce characteristic X-rays.
Each element has it own electron energy levels & its corresponding characteristic X-ray energy.
Binding energy definition & X-ray production process
Electrons in atoms sit in shells (K, L, M) each with a specific binding energy.
If inner shell electron is removed the outer electron can fall down to fill the gap.
Atom emits X-ray with energy equal to difference between energy levels.
Binding energy (negative wrt unbound state) - energy required to remove electron from atom.
X-ray fluorescence (XRF)
Analytical technique where a material gets bombarded with high energy X-rays/ Gamma rays to become excited.
A characteristic x-ray gets emitted & used to identify the material.
Nuclear structure components definitions (Z, A, N, Isotopes, Isobars, Isotones)
Z: number of protons = atomic number.
A: total number of nucleons = mass number.
A (protons + neutrons) ≈ atomic weight.
N: number of neutrons = A-Z.
Isotopes: nuclides with same Z.
Isobars: nuclides with same A.
Isotones: nuclides with same N.
Forces in nucleus
Coulomb force (repulsive) – protons push each other apart.
Nuclear force (attractive) – holds nucleons together.
Stability – balance between coulomb & nuclear force.
Nuclear shell model definitions & energy state types
Nuclear shell model – nucleus has energy levels & nucleons occupy specific energy states.
Ground state – lowest nucleus energy state.
Excited state (doesn’t last long) – nucleus has extra energy.
Metastable state – excited state that last unusually long.
What occurs to unstable nuclei?
Radioactive decay – particles/photons emitted to achieve more stable state (higher binding energy per nucleon).
Higher binding energy ≈ lower value on total energy level diagram.
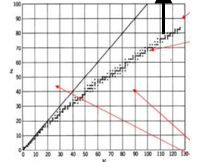
Define light & heavy nuclei
Light nuclei (N=Z): equal number of protons & neutrons.
Heavy nuclei: more neutrons than protons.
Protons repel each other (coulomb force) & extra neutrons help stabilise the nucleus.
Unstable nucleus (not in stability band) decays to move back to stability.
Decay types
Beta plus decay: neutron deficient nuclides.
Beta minus decay: neutron has excess nuclides.
Beta minus emission properties
Beta minus emission (too many neutrons) - neutron in nucleus changes into a proton, electron & a neutrino,
Neutron in nucleus changes into a proton, an electron & a neutrino.
Z increases by 1 & A stays the same.
Beta minus decay
Continuous spectrum of electron energies (up to Emax) - energy shared between electron & neutrino.
Average energy E = 1/3*Emax
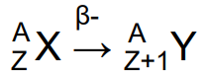
Beta minus & gamma decay properties and define isometric transition
Daughter nucleus is still excited after beta minus decay & emits a gamma ray immediately after.
Can emit multiple gamma - have well defined energies characteristic of radionuclide.
Example: 131-Iodine
Isometric transition - delayed gamma emission when daughter left in a long-lived metastable state
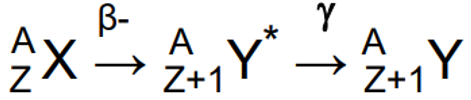
Electron capture
Proton in nucleus captures an orbital electron from K or L shell & becomes a neutron.
Z decreases by 1 & A stays the same.
Captured inner-shell electron means atom has an electron vacancy.
Outer electron that fill the gap cause the atom to emit: characteristic X-rays/Auger electron/ Gamma emission (if nucleus is left excited).
Positron (Beta plus) decay properties
Proton changes into a neutron, a positron & a neutrino.
Z decreases by 1 & A stays the same
Positron annihilates with atomic electron.
Mass is converted into energy: 2 gamma photons (each with 511keV energy) that travel in opposite directions.
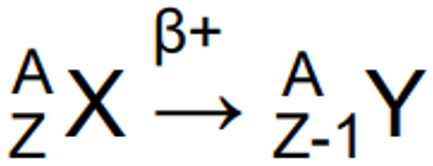
Annihilation coincidence detection process & line of response
Radionuclide decays.
Positron is emitted.
Positron annihilates with an electron.
2 551keV photons are detected by a ring of detectors around the patient.
Line of response - allows PET to localise where tracer is in body.
2 detectors detect photons at similar times.
System assumes annihilation occurred along line between them.
Beta plus vs Electron capture decay
Beta plus & EC decays are alternate decay routes to same nucleus.
Can reduce proton in proton rich nucleus by either: emitting a positron/ capturing an electron.
Low-Z : mostly beta-plus.
High-Z : mostly EC.
Define Becquerels, Branching ratio and the equation for radioactivity amount & half life.
Radioactivity measure in Becquerels
1decay per second = 1 Becquerel.
Each decay can lead to several/ no emission.
Branching ratio (%) - fraction of decays that can lead to a particular emission.
Radioactivity amount: A(t) = A0e-𝜆t
Half-life (time for activity to fall to ½ its original value): T1/2=0.693 / λ
Radiation types & interaction mechanism determine
Charged particle: alpha & beta particles.
Electromagnetic radiation: x-rays & gamma rays.
Interaction mechanisms determine: radiation detection, shielding, dosimetry
Collisional losses definition & causes
Charged particles interact with electrons & nuclei & slow down (lose energy) as they travel.
Causes:
Ionization
Excitation
Bremsstrahlung
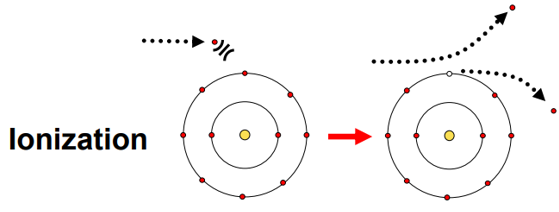
Ionization process and results & effects.
Incident charged particle gives enough energy to knock electron completely out of atom from outer shell.
Can create secondary electrons: delta (𝛿) – rays.
Result: atom is ionized, free electron is produced that can go on to cause more ionization.
Effects:
Damage to tissue
Produces electrical signals in detectors
Deposits dose in matter
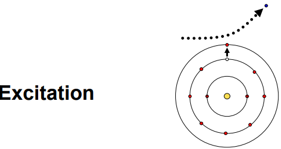
Excitation process & effect
Charged particle gives energy to atom/molecule to raise electron to higher energy state.
Atom is disturbed not ionised – no electron fully removed.
Energy stored temporarily (can be released later).
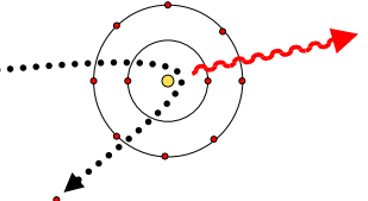
Bremsstrahlung process & what its good for
Radiative energy loss mechanism - incident charged particles interact with nucleus, changes direction & emits a photon.
Produces a continuous spectrum - energy can vary continuously from 0 up to max energy.
Good for: High Z absorber materials, high incident energy, small particle mass.
Charged particle tracks
Gradually slows down & loses energy – passes through material, leaving a trail of ionized atoms.
Heavy particles (proton alpha) – straight tracks, densely ionizing.
Electrons (𝑒-,𝑒+) – convoluted path, less dense ionization.
Ionization volume (measured by radiation detectors) - detect particles in radiation detector.
Path length & range definition
Path length - total distance travelled.
Range - net penetration distance.
Path length > range for beta particles.
Twisted electrons path travel long distances but not deep.
Indirect ionisation properties and interaction types
High energy photon interaction are indirectly ionising: X-rays, Gamma rays, annihilation photons, bremsstrahlung.
Photons have no charge.
Produce secondary charged particles (ejected electrons, electron-positron pairs) during interaction.
Charged particles then cause ionisation.
Interaction types: photoelectric effect, Compton scattering, pair production, Rayleigh scattering.
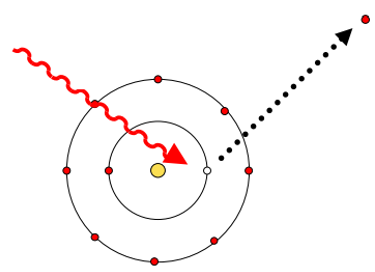
Photoelectric effect process
Incident gamma is absorbed by atom
Photoelectron is ejected (usually from inner shell if enough energy is available).
Epe= Eincident - Ebinding
Inner shell vacancy leads to X-rays/Augers being emitted when electrons from other shells fall down to fill the gap.
Photoelectron causes an ionization train.
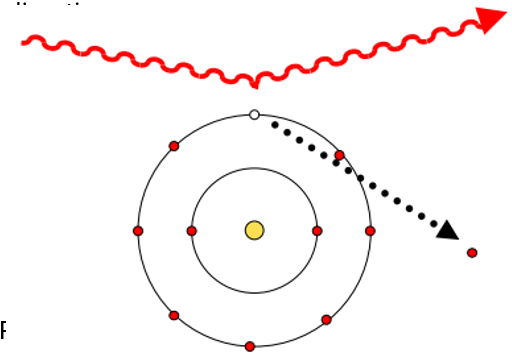
Compton scattering properties
Incident gamma is scattered
Outer-shell electron is ejected as recoil electron: Erecoil = Eincident - Escattered
Scattered photon has lower energy than the incident photon: Emin_scattered < Escattered < Eincident
Forward peaked - scattering probability determined by Klein Nishina distribution
Problems of scattered photons in Compton scattering
Go in wrong direction
Still reach detector sometimes
Reduce image contrast
Add noise & mispositioned count
Pair Production properties
Photon disappear & β+, β- pair created.
Minimum photon energy for pair production: 2×511keV = 1.022MeV
Beta particles go on to cause ionization
β+ creates 511keV annihilation photons.
Pair production process
High energy photon comes near nucleus.
Photon disappears.
Energy becomes mass + kinetic energy of: 1 electron, 1 positron.
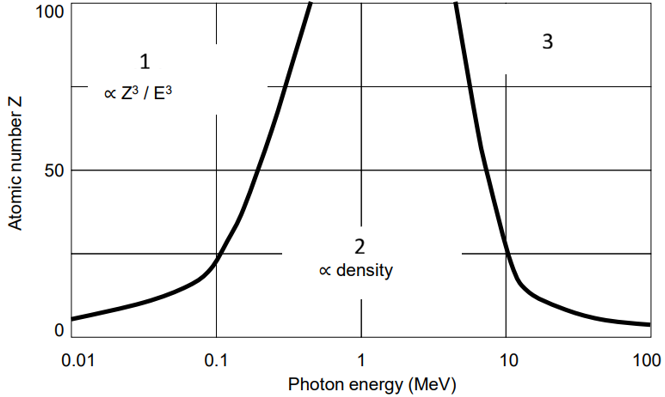
Label the photon interaction chart
Photoelectric effect - used when Z is high (bione, iodine) & energy is low.
Compton scattering
Pair production - only occurs at high energies.
What are definitions and equations for attenuation, linear attenuation, mass attenuation, transmitted beam intensity & half value layer
Attenuation - photons are absorbed/ scattered out of beam as they pass through a material.
Linear attenuation coefficient 𝜇l (units cm-1).
Mass attenuation coefficient: 𝜇m(Z,E) = 𝜇l /𝜌.
Transmitted beam intensity: e-𝜇lx
Half value layer (50% intensity decrease) - large HVL (weaker attenuation, need more material for shielding), small HVL (strong attenuation, need less material for shielding).
Radiation detectors purpose and types
Radiation detectors - convert incident radiation to detectible form (light/ charge).
Convert radiation to measurable electrical signal: directly ( charge created in detector volume), indirectly (use light signal).
Main detector types:
Scintillation detectors (indirect).
Gas-filled detectors (direct)
Semi-conductor detectors (direct)
Quantum efficiency definition & properties that affect it
Quantum efficiency (%) - probability that radiation (usually gamma) reaching detector is absorbed & detected.
Varies with radiation & energy type.
Depends on attenuation coefficient (𝜇 cm-1) or radiation length (1/𝜇).
Affected by: detector material thickness & radiation energy.
Sensitivity definition & properties
Sensitivity (%) - how accurate detector can tell where radiation came from.
Depends on efficiency & detector size (solid angle).
Is a property of whole system.
High sensitivity: large area (compensates for low QE), better image quality (less noise).
Energy resolution definition
Energy resolution (ΔE %) - detector’s ability to distinguish between different photon energies.
Characterised by full width at half maximum (FWHM).
Smaller FWHM has better energy resolution.
ΔE = f (no. electrons/ photon collected).
More charge carriers means better statistical accuracy & resolution
Requirements of good energy resolution
Radiation energy identification.
Energy discrimination/ scatter rejection - removes low energy scattered photons.
Positioning schemes - some detectors use energy information.
Enables advances/ novel imaging techniques.
Spatial resolution definition
Spatial resolution (essential for imaging) - detector’s ability to determines analogue/ digital position of incoming radiation.
Measure by FWHM (mm) - better spatial resolution & sharper image when smaller.
Use many small detectors.
Minimises/ simplifies amount of electronic readout needed.
Ideally robust, cheap.
Temporal resolution definition
Temporal resolution - detector’s ability to measure time of radiation event accurately.
Determines when particle/ photon is detected.
Need fast & accurate (high statistics signal).
Essential for coincidence detection accuracy.
Important in PET (time of flight: 10ps).
Count rate capability/ dead time definition
Count rate capability/ dead time - detector’s ability to accurately record events at high count rates:
Dead time - period after detection when detector can’t record another event.
Lose data at high count rates.
Readout of each detected pulse takes too long - pulses overlap.
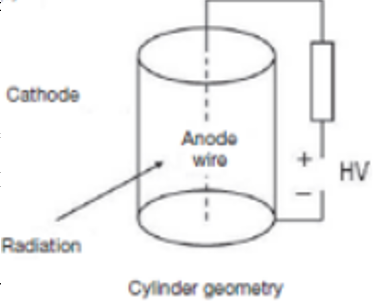
Gas filled detectors set up and situations where they are ineffective
Gas contained between 2 electrodes.
Ionisation collected & small current flows.
Low QE & low sensitivity for X-rays & gamma-rays - gasses aren’t dense so photons are less likely to interact with it.
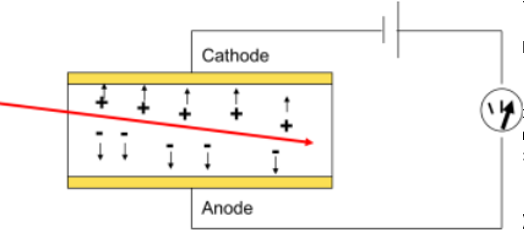
Process of how gas filled detectors function
Radiation enters gas & ionises the gas atoms.
Free electron & positive ions are produced.
Electric field moves them to electrodes.
Produces a small current/pulse.
Different types of gas filled detectors
Survey Meter (radiation safety monitor)
Calibrated in mSv/hr
Protective cap removed for alpha, beta & very-low energy (<10 keV) photons.
Dose calibrator - measures amount of radioactive tracer in syringe/vial before patient administration.
Calibrated in Bq.
Response depends on energy so calibration factors required.
Properties of gas filled detectors in terms of quantum efficiency, sensitivity, energy resolution, temporal resolution, count rate capability, spatial resolution.
Survey meter:
Quantum efficiency: low (gas)
Sensitivity: very low (doesn’t matter as count rate is high).
Energy/ temporal/spatial resolution: none
Count rate capability: moderate
Dose calibrator:
Quantum efficiency: low (gas)
Sensitivity: low (improved by large volume, large activities measured so doesn’t matter).
Energy/ temporal/spatial resolution: none
Count rate capability: moderate
Scintillation detectors
Scintillation detector - convert energy from ionisation & excitation into visible light.
Use inorganic crystals / organic substances dissolved in liquid solution.
Amount of light is proportional to energy deposited.
Original experiments counted scintillation on zinc sulphide screen - now use highly sensitive photodetectors.
Process of scintillation detector
Radiation enters scintillator & deposits energy.
Scintillator emits visible light
Photodetector converts light to electric signal.
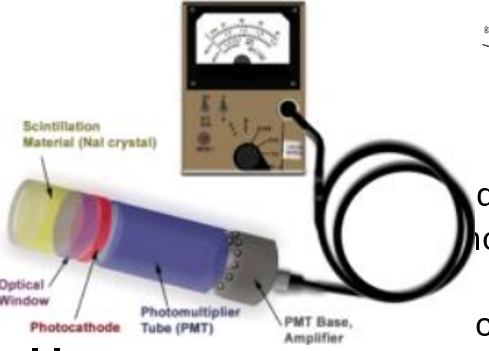
Scintillator components
Most are impurity activated.
Nal(Ti) crystal - detector of choice for most routine apps.
Dense, high energy (Z), cheap, high light yield.
Hygroscopic (need Aluminium jacket/ optical window) - absorbs moisture from air so must be sealed.
Some with thin aluminium or Be entrance window for low energy radiations.
Photocathode (QE: 10-30%) - converts light to electrons.
Photomultiplier tube: gain 106 at each dynode.
Voltage must be very stable - multiplication factor very sensitive to voltage changes.
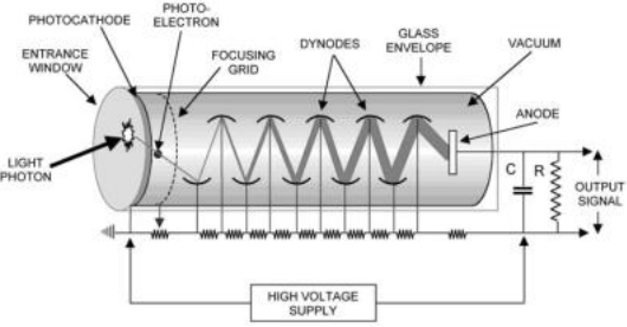
Process for photomultiplier tube
Receive flash from scintillator.
Convert light to electrons at photocathode
Multiply electrons through dynodes & output strong electric pulse.
Scintillation detector examples & their properties in terms of quantum efficiency, sensitivity, energy resolution, temporal resolution, count rate capability & spatial resolution.
Well counter - quantifies small activities (e.g. blood samples).
Very high (>50%): quantum efficiency (very large Nal detector >5cm diameter), sensitivity.
Good energy resolution.
No temporal / spatial resolution.
Poor count rate capability (not required as only measuring small activities).
Contamination monitor:
High : quantum efficiency
Low: sensitivity (small detector volume)
Good: Energy resolution
Moderate: count rate capability
No temporal/ spatial resolution
Gamma Camera:
Very high (thick Nal crystal): quantum efficiency
High: sensitivity (large area detectors).
Good: energy resolution (~50% must reject scattered radiation), Count rate capability, temporal resolution (not needed for SPECT)
3mm FWHM spatial resolution.
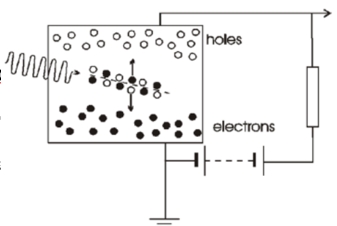
Semi conductor detectors properties
Semi-conductor detectors provide direct radiation detection.
Solid state version of gas-filled detector.
High density (>x2000) & high sensitivity.
Need cooling using liquid nitrogen to remain at 77 kelvin: high purity germanium HPGe, Ge(Li) & Si(Li) operated & stored at low temperatures.
Room temp SCs (CdZnT) are becoming available.
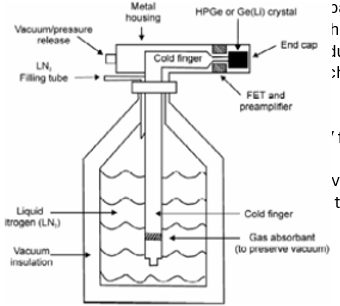
HPGe Detector properties
Standard choice for gamma spectroscopy.
Operate at 77 kelvin.
Energy resolution 1keV for 122 keV gamma ray.
10cm*10cm cylinders available.
Drift (charge collection) times up to 100nS.
CdTe & CZT properties
Starting to be used in imaging system.
Can be operated at room temperature without excessive electronic noise.
Worse energy resolution than HPGe.
High atomic number (>Ge) - relatively thin detectors have good stopping efficiency for detecting gamma rays.
Gamma ray spectroscopy properties in terms of quantum efficiency, sensitivity, energy resolution, temporal resolution, count rate capability & spatial resolution.
Best for energy accuracy but not fast timing.
Good: quantum efficiency, sensitivity (depends on configuration).
Excellent: energy resolution
No temporal resolution (not usually needed but poor for PET) & poor count rate capability.
Spatial resolution (can be used for imaging).
Film spectroscopy properties in terms of quantum efficiency, sensitivity, energy resolution, temporal resolution, count rate capability & spatial resolution.
Very low: quantum efficiency (used in combination with phosphor/scintillator).
Moderate sensitivity (used in with phosphor/scintillator).
No energy/temporal resolution
Poor count rate capability (film is easily overexposed).
Excellent spatial resolution