1 intermolecular interactions
1/61
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms



























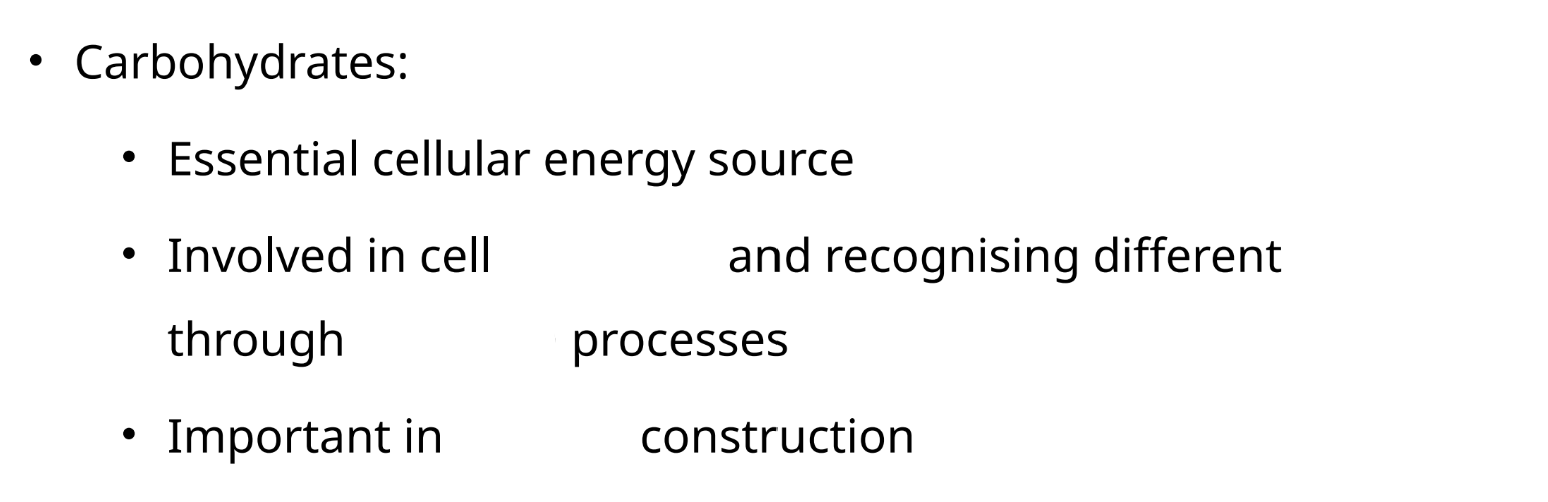




monomer of proteins?
amino acids
monomer of nucleic acids?
nucleotides
monomer of carbohydrates?
monosaccharides
equation linking enthalpy and entropy
ΔG = ΔH - TΔS
what do charge-charge interactions require
the presence of two ionisable groups
pH of living systems
approx 7.4
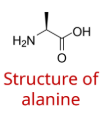
structure of amino acids in living systems at acidic, neutral and basic pH
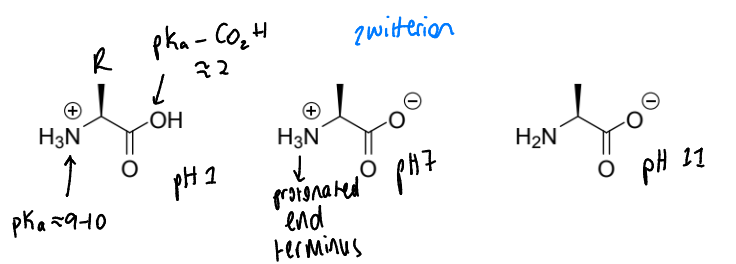
show how phosphate-containing compounds are charged at neutral pH
show how the energy of an interaction varies with distance (equation)
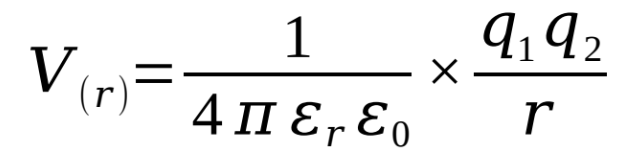
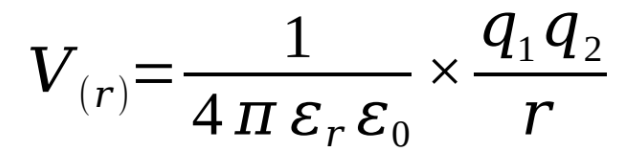
what are the terms

how does ε vary with solvent

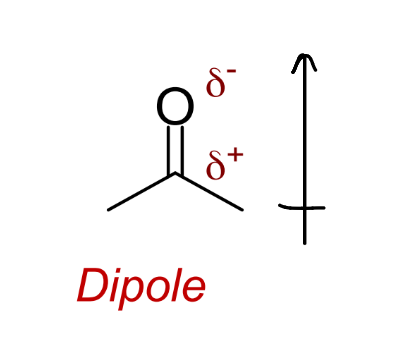
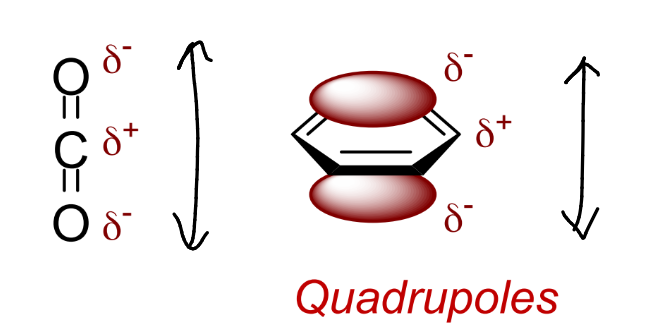
represent the dipole on this molecule both in terms of δ and arrow
show 2 examples of quadrupoles
what is a hydrogen bond
a hydrogen atom bridges the gap between two N or O atoms
bonding of the hydrogen atom in a hydrogen bond
formally bonded to the donor atom but much closer to the 2nd atom (acceptor atom) than normal
how close is the hydrogen atom to the acceptor atom
within the van der Waal radius
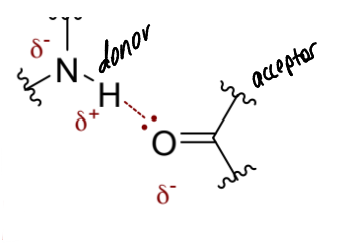
show an H bond between a secondary amine group and a carbonyl group
label donor/acceptor and show partial charges
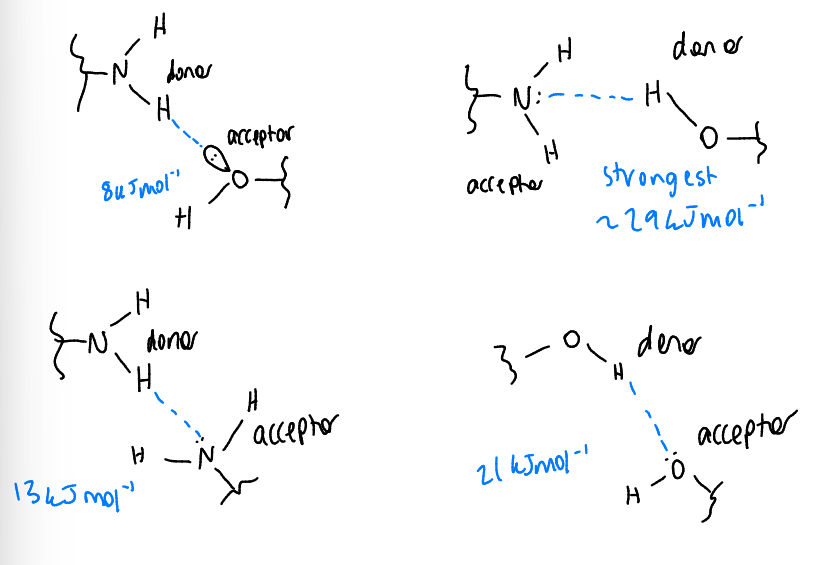
show 4 possible H bonding interactions between -NH2 and -OH
which is strongest/weakest?
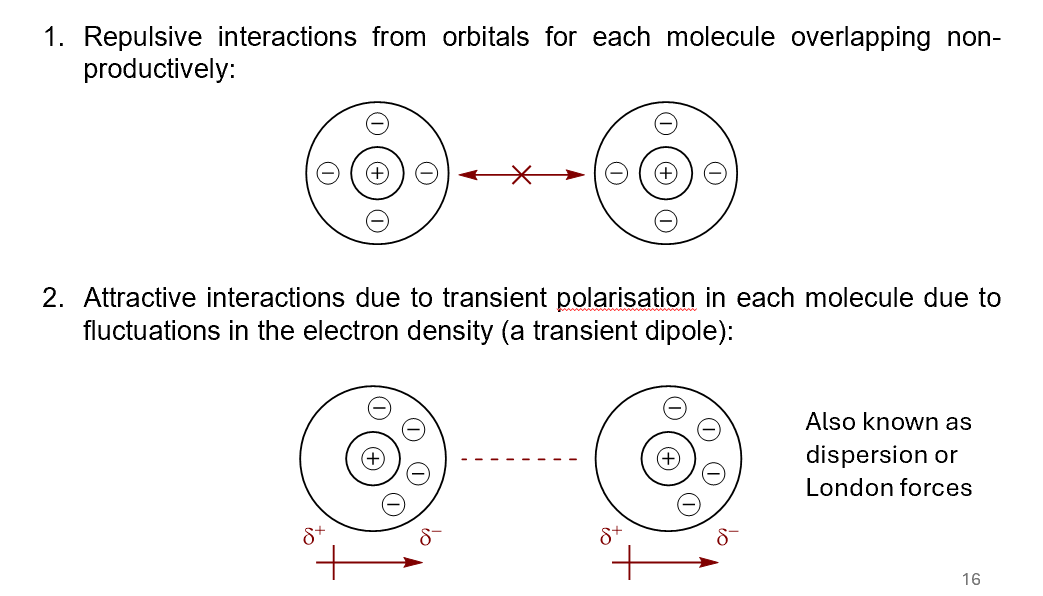
what are the 2 factors contributing to van der Waals interactions? show a diagram for each
show on an energy vs distance graph
the force response curve for van der Waals interactions
the graphs for repulsive forces and attractive forces separately
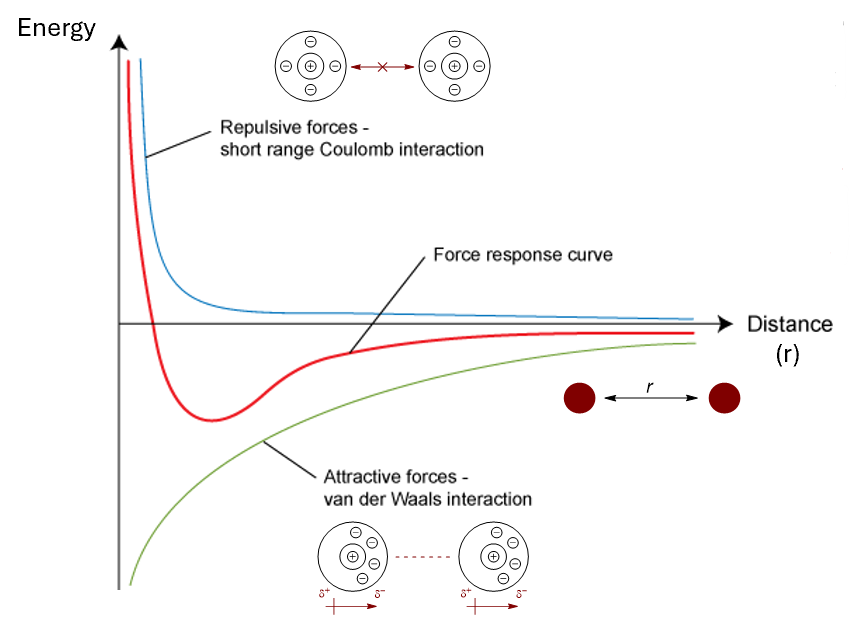
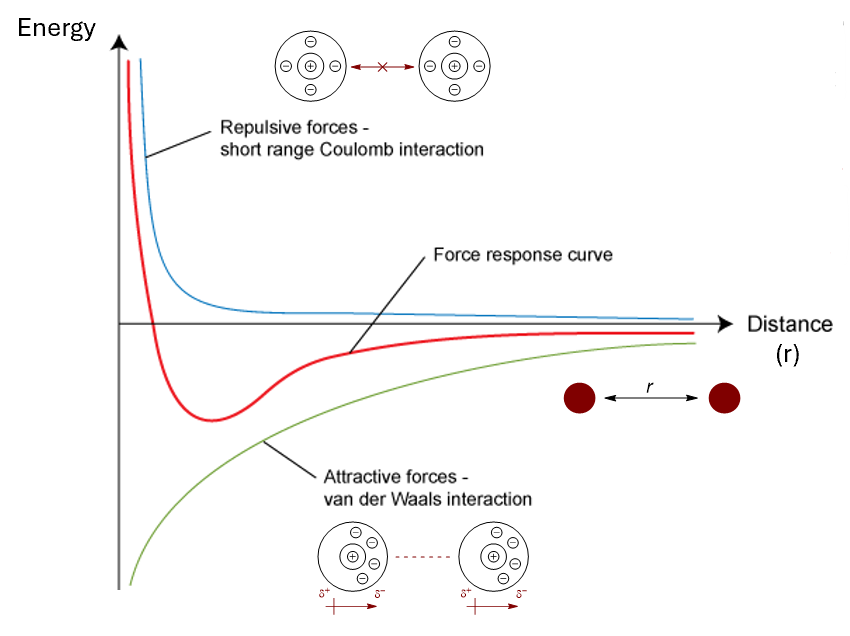
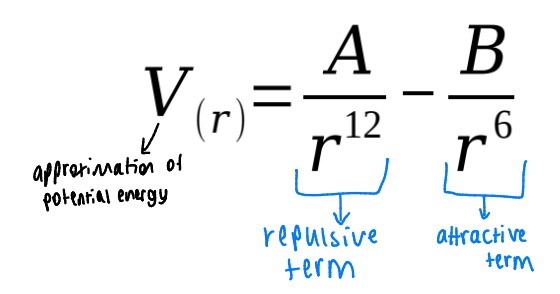
what equation represents this graph (red) and each of the repulsive and attractive sections
how significant are vdW forces
effect of changes in packing?
individual interaction energies are very small but collectively they can make a substantial contribution to interaction
a small change in packing can have a substantial effect
ideal binding interaction
maximal shape complementarity between the two binding partners so that the attractive interactions are maximised
dominant intermolecular force?
h bonding
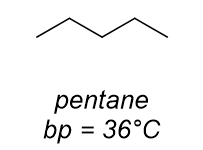
dominant intermolecular force?
vdW only
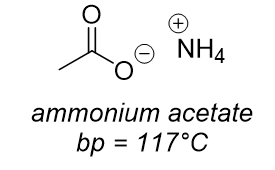
dominant intermolecular force?
charge-charge interaction
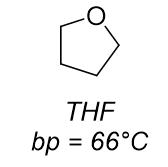
dominant intermolecular force?
dipole-dipole
rank the 4 intermolecular forces in order of least to most strong
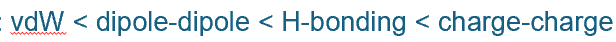
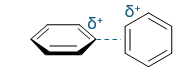
favourable or unfavourable?
+ and +, not favoured
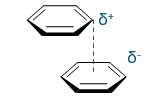
favourable or unfavourable?
name + type of interaction?
favourable (+ and -) - offset, face to face
π-stacking interaction - type of quadrupole interaction
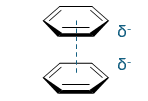
favourable or unfavourable?
describe
- and -, not favoured
face to face interaction
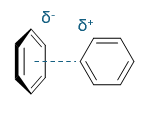
favourable or unfavourable?
describe
favoured, + and -
T shaped, edge to face
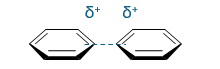
favourable or unfavourable?
not favoured - + and +
effect of substituting with EDG on quadrupole interactions
increases the quadrupole moment/strength of the interaction
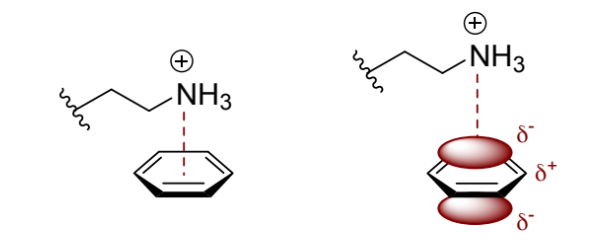
what is a cation-π interaction?
charge-quadrupole interaction
dependence of cation-π interactions on distance
1/r3 dependence
show the cation-π interaction between -N+H3 and benzene
positive charge interacts with the concentration of negative charge above or below the plane of the ring
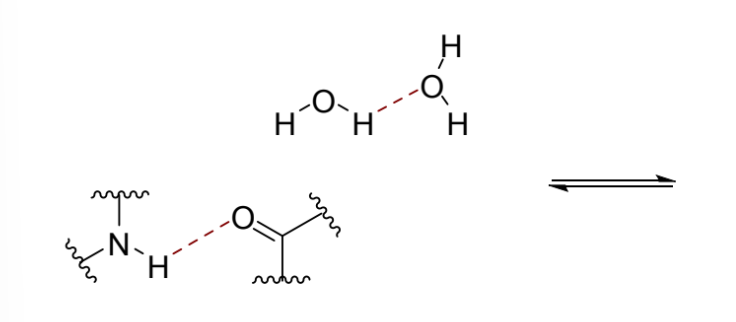
why might water as a solvent change the favourability of intermolecular interactions
hydrogen bonding with water can be as strong as hydrogen bonding between an acceptor and donor
?
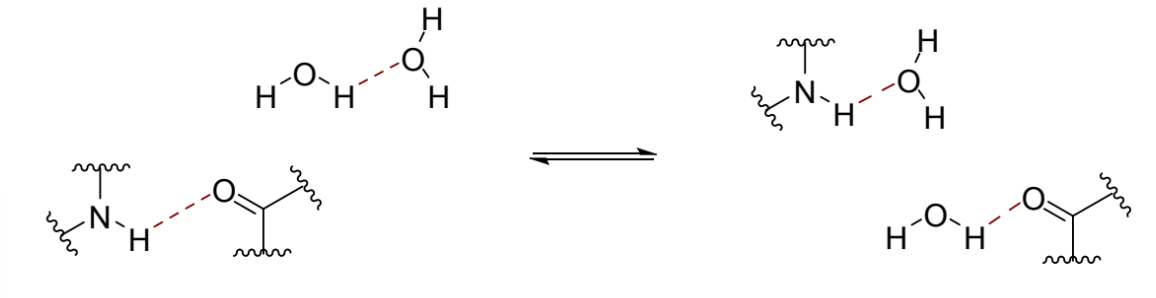
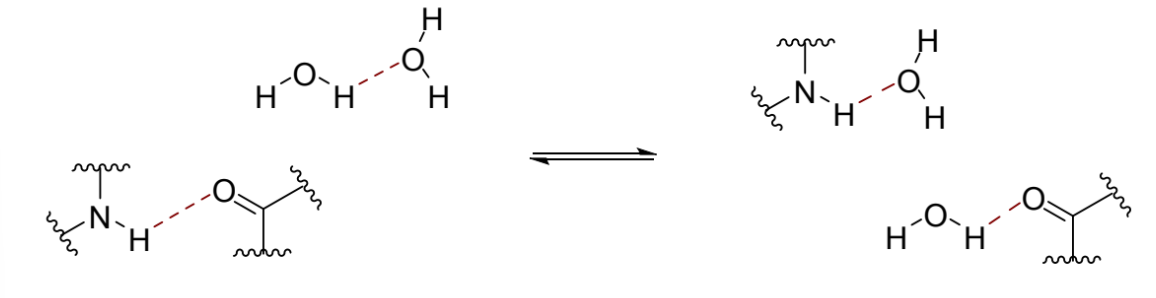
enthalpy in this situation
no enthalpic penalty for lost interactions in these cases - same number of total H bonds as water is H bonding to itself anyway
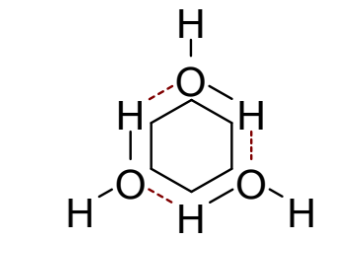
explain the hydrophobic effect using cyclohexane (include diagram)
cyclohexane cannot form H bonds with water - only vdW
for all the water molecules to form H bonds high order is needed so they form constrained networks around each molecule of hydrophobic cyclohexane
this is very entropically unfavourable
what does the hydrophobic effect refer to
how can this provide a driving force
refers to ‘hydrophobic’ portions of molecules packing together favourably - as water is immiscible with hydrophobic portions
release of water molecules into bulk solvent is a major entropic driving force for many interactions
what is ΔG for a biomolecular interaction
the sum of all the charges in binding interactions
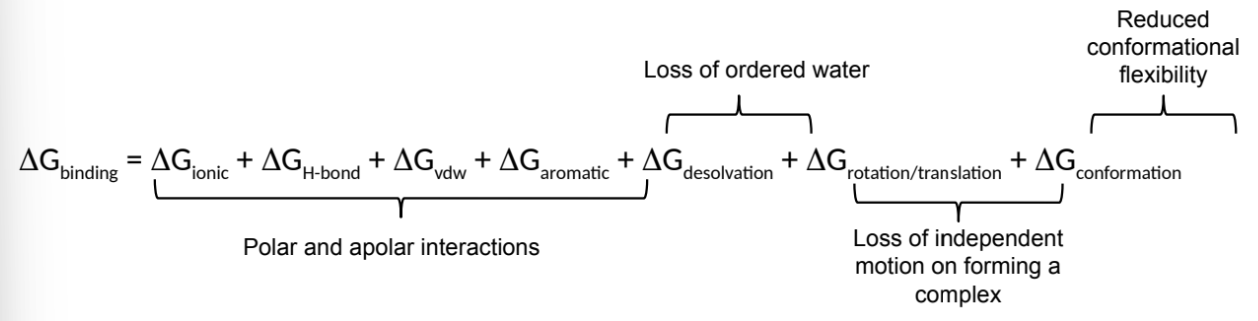
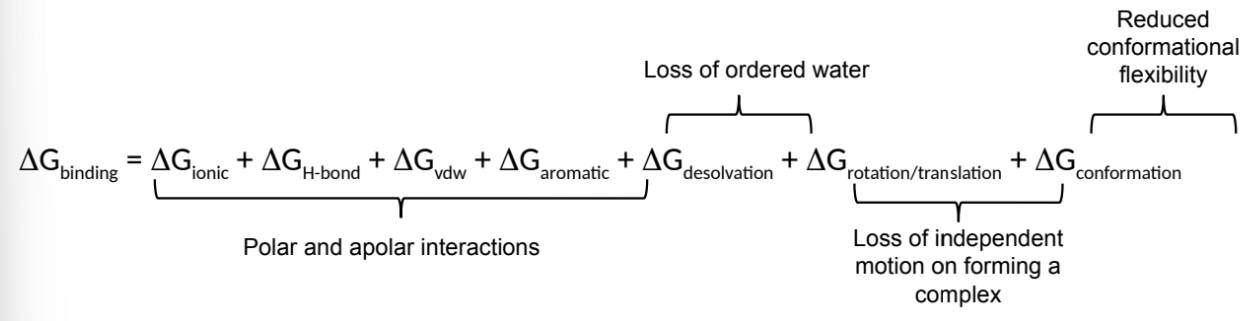
what is ΔGbinding a sum of? (7)
group the components into 4 sections
what is ΔGrotation/translation?
mostly an entropy penalty for bringing two molecules together to form a complex
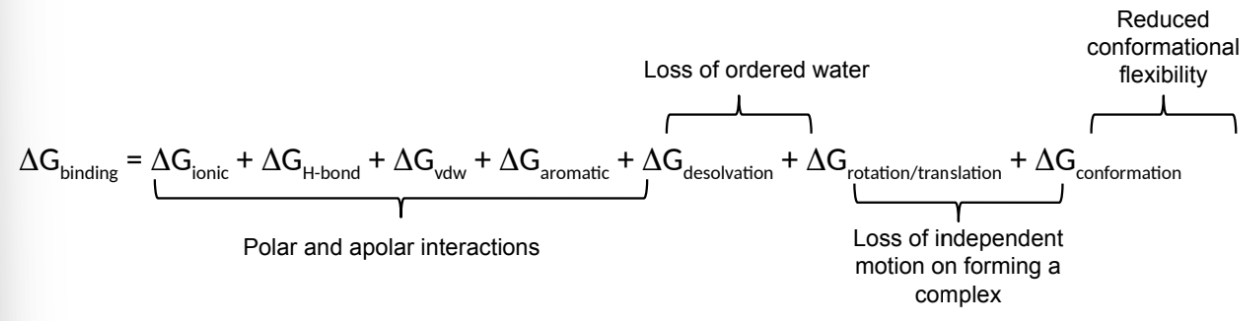
what is ΔGconformation?
mostly an entropy penalty for restricting conformational flexibility