Chemistry chapter 8
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Electron configuration
The distribution of electrons among the available subshells of an atom.
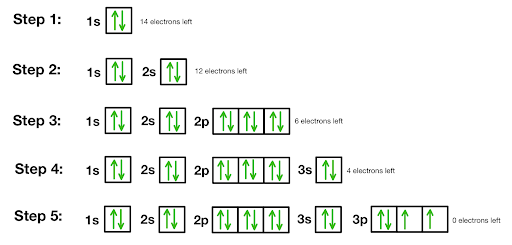
Orbital diagrams
Diagrams used to show how the orbitals of a subshell are occupied by electrons.
Pauli’s exclusion principle
No two electrons in an atom can have the same four quantum numbers.
Noble gas core
Inner-shell configuration corresponding to one of the noble gases.
Hund’s rule
The lowest-energy arrangement of electrons in a subshell is obtained by placing electrons into separate orbitals with the same spin before pairing.
Special electron configuration
Cu:[Ar} 4s1 3d10
Cr:[Ar] 4s1 3d5
Paramagnetic
A substance that is weakly attracted by a magnetic field, typically due to unpaired electrons.
Diamagnetic
A substance that is not attracted by a magnetic field or is very slightly repelled.
Atomic properties; Atomic radius
Period: radius tends to decrease with increasing atomic number
Group: radius increases with the period number
Atomic properties; ionization energies
within any period the value increases as the atomic number goes up; while in the group energy decreases
Atomic properties; electron affinities
The general trend is that the value increases from left to right, though 2A and 5A have smaller affinities, while group 8a has little to no affinity
Values becomes increasingly negative with group number
Atomic properties; effective nuclear charge
The higher the effective nuclear charge the smaller the atom
First ionization energy
The amount of energy required to remove an electron from an atom in the gaseous state, in inorder to create a positive ion.
Electron affinity
The energy required to add an electron to an atom in a gaseous state to form a negative ion.
Acidic oxide
An oxide that reacts with bases.
Basic oxide
An oxide that reacts with acids.
Amphoteric oxide
An oxide that has both basic and acidic properties.
Group 1 elements
Alkali metals (ns^1) become more reactive as you go down the column.
Make basic oxides
Formula of R2O
Group 2 elements
Alkaline earth metals (ns^2) are reactive but less so than alkali metals; reactivity increases down the group.
Make basic oxides
Formula RO
Group 3 elements (ns^2np^1)
boron is a metalloid, all others are metals
formula of R2O3
boron oxide (B2O3) is acidic
aluminum oxide (Al2O3) and gallium oxide (Ga2O3) are amphoteric
indium oxide (In2O3) and Thallium (Ti2O3) are basic
Group 4 element (ns2^np^2)
Includes Carbon nonmetal, silicon and germanium metalloid, Tin and lead metals
Formula of RO2
Carbon dioxide (CO2), silicon dioxide (SiO2), and germanium dioxide (GeO2) is acidic
Tin dioxide (SnO2) lead(IV) oxide (PbO2) is amphoteric
Group 5 elements
Includes nonmetals (nitrogen, phosphorus), metalloids (arsenic, antimony), and a metal (bismuth)
The empirical formula of R2O3 and R2O5
Molecular formula of R4O6 and R4O10
Nitrogen (N2O3, N2O5), phosphorus (P4O6, P4O10), arsenic (AS2O3, As2O5) are acidic
Antimony(Sb2O3, Sb2O5) is amphoteric
Bismuth(Bi2O3) is basic
Group 6 elements
Chalcogens (ns^2np^4) include oxygen, sulfur, selenium (nonmetals), tellurium (metalloid), and polonium (metal).
Formula of RO2 and RO3
All except for TeO2 are acidic
PoO2 is amphoteric though more basic than TeO2
Group 7 elements
Reactive non-metals
The molecular formula of X2 (X is a Halogen)
Generally unstable acidic oxides
Astatine might be expected to be a metalloid(have a concise half life)
Group 8 elements
Unreactive