Exam Q - ions in aq
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
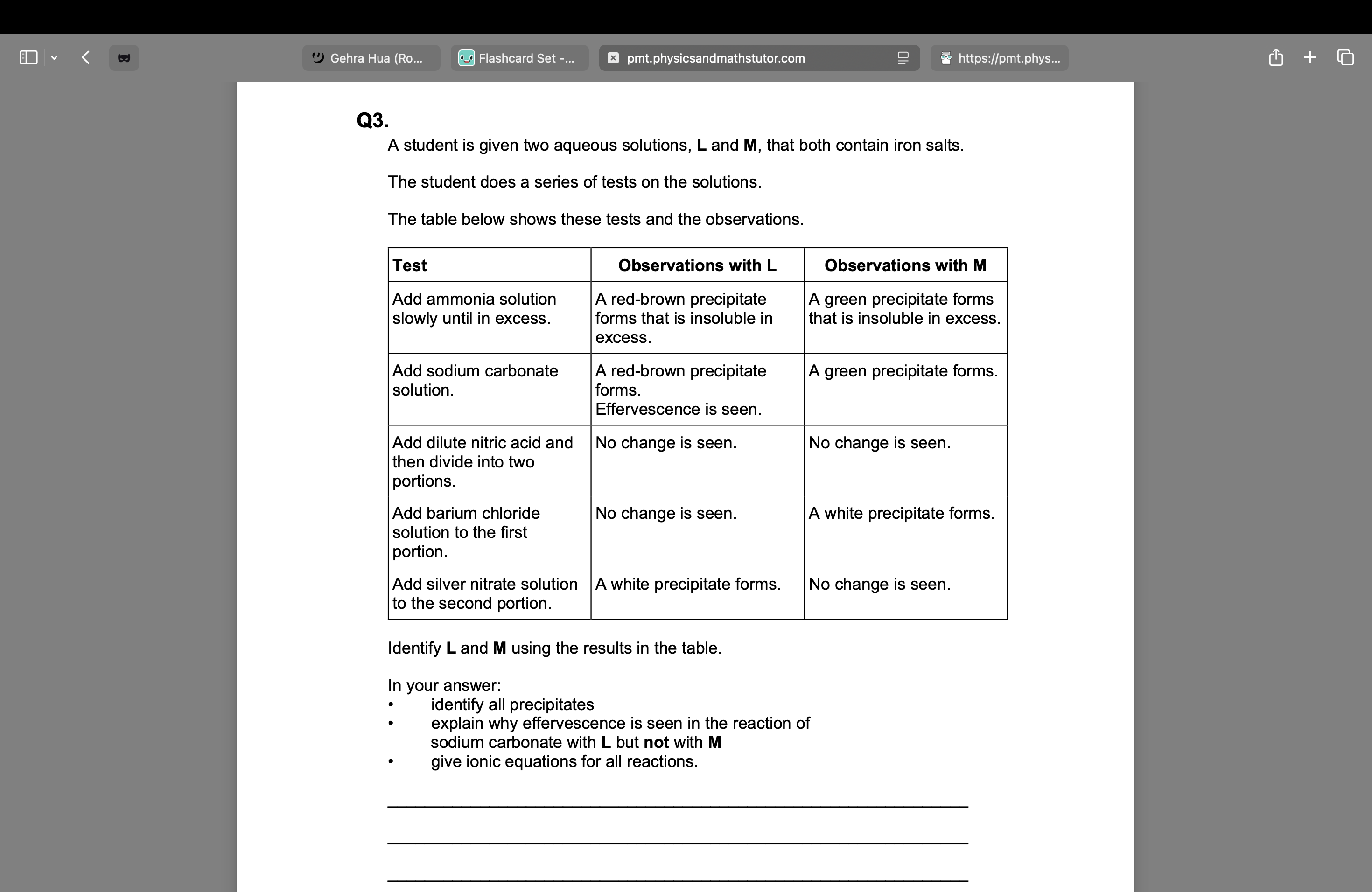
state a reagent for reaction from Fe3+ to Fe2+
XS Sn
Solution Y contains two different negative ions.
To a sample of solution Y in a test tube a student adds
• silver nitrate solution
• then an excess of dilute nitric acid
• finally an excess of concentrated ammonia solution.
The observations after each addition are recorded in the table.
Reagent added to solution Y | Observation |
silver nitrate solution | cream precipitate containing compound D and compound E |
excess dilute nitric acid | cream precipitate D and bubbles of gas F |
excess concentrated ammonia solution | colourless solution containing complex ion G |
Give the formulas of D, E and F.
Give an ionic equation to show the formation of E.
Give an equation to show the conversion of D into G.
D AgBr Ignore state symbols
1
E Ag2CO3
1
F CO2
1
2 Ag+ + CO32– → Ag2CO3
1
AgBr + 2 NH3 → Ag(NH3)2+ + Br –
Or → Ag(NH3)2Br
One mark for Ag(NH3)2+ and 1 mark for equation
If D = AgCl, then allow 2 marks for
AgCl + 2 NH3 → Ag(NH3)2+ + Cl–
2
When anhydrous aluminium chloride reacts with water, solution Y is formed that contains a complex aluminium ion, Z, and chloride ions.
Give an equation for this reaction.
___________________________________________________________________
(1)
(b) Give an equation to show how the complex ion Z can act as a Brønsted–Lowry acid with water.
(a) AlCl3 + 6H2O → [Al(H2O)6]3+ + 3Cl–
Allow
AlCl3 + 6H2O → Al(H2O)5(OH)2+ + H+ + 3Cl–
1
(b) [Al(H2O)6]3+ + H2O → [Al(H2O)5(OH)]2+ + H3O+
Solution A contains the compound [Cu(H2O)6]
(a) An excess of ammonia was added to a sample of solution A to form solution B.
Write an ionic equation for the reaction that occurs when solution A is converted into solution B and state the colour of solution B
(a) [Cu(H2O)6 ]2+ + 4NH3 → [Cu(NH3)4(H2O)2 ]2+ + 4H2O
1
Deep blue / Royal blue / Dark blue (solution)
Reagent D was added to another sample of solution A to form a yellow-green solution.
Identify reagent D and write an ionic equation for the reaction that occurs when the yellow-green solution is formed from solution A.
Solution A contains the compound [Cu(H2O)6]
HCl / hydrochloric acid
Allow soluble chloride salt
Also allow any reagent which leads to a change in colour of solution due to a change in ligands (e.g. NH2CH2CH2NH2) or change in oxidation state (e.g. SO2 ) and associated correct equations.
1
[Cu(H2O)6 ]2+ + 4Cl− → [CuCl4]2− + 6H2O
[Cu(H2O)6 ]2+ + 4HCl → [CuCl4]2− + 6H2O + 4H+
The following tests were carried out to identify an unknown green salt Y.
An aqueous solution of Y gave a cream precipitate of compound A when reacted with silver nitrate solution.
Compound A gave a colourless solution when reacted with concentrated ammonia solution.
Another aqueous solution of Y gave a green precipitate B when reacted with sodium carbonate solution.
The green precipitate B was filtered and dried and then reacted with sulfuric acid to give a pale green solution containing compound C and a colourless gas D.
(a) Identify by name or formula the compounds A, B, C, D and Y.
(c) Write the simplest ionic equation for the reaction that occurs between the green precipitate B and sulfuric acid.
(a) A Silver bromide / AgBr
1
B Iron(II) carbonate / FeCO3
1
C Iron(II) sulphate / FeSO4
1
D Carbon dioxide / CO2
1
Y Iron(II) bromide / FeBr2
(c) 2H+ + CO32− ⟶ H2O + CO2
Allow FeCO3 + 2H+ ⟶ Fe2+ + CO2 + H2O
Q6.
What forms when a solution of sodium carbonate is added to a solution of gallium(III) nitrate?
A A white precipitate of gallium(III) carbonate.
B A white precipitate of gallium(III) hydroxide.
C A white precipitate of gallium(III) carbonate and
bubbles of carbon dioxide.
D A white precipitate of gallium(III) hydroxide and
bubbles of carbon dioxide.
d
Which compound gives a colourless solution when an excess of dilute aqueous ammonia is added?
A MgCl2
B AgCl
C CuCl2
D AlCl3
b
What is the final species produced when an excess of aqueous ammonia is added to aqueous aluminium chloride?
A [Al(NH3)6]3+
B [Al(OH)3(H2O)3]
C [Al(OH)4(H2O)2]–
D [Al(OH)(H2O)5]2+
b
Describe what you would observe when dilute aqueous ammonia is added dropwise, to excess, to an aqueous solution containing copper(II) ions.
Write equations for the reactions that occur.
(a) Blue precipitate
1
Dissolves to give a dark blue solution
1
[Cu(H2O)6]2+ + 2NH3 Cu(H2O)4(OH)2 + 2NH4+
1
Cu(H2O)4(OH)2 + 4NH3 [Cu(NH3)4(H2O)2]2+ + 2OH– + 2H2O
This question is about aqueous ions of the metal iron.
When an aqueous [Fe(H2O)6]3+ ion reacts with ethanedioate ions, an iron(III)
complex ion X is formed.
The only ligands in X are ethanedioate ions.
state the charge.
3-
(b) The formation of X is an example of the chelate effect.
Explain the meaning of the chelate effect.
M1 When bidentate/multidentate ligands replace monodentate ligands (to
form a more stable complex)
M2 Because there is an increase in entropy/positive entropy
change/disorder or more particles formed (so ∆G is negative and ∆H is
approximately 0)
Outline how Fe2+ ions catalyse the reaction between S2O82– ions and I– ions
in aqueous solution.
In your answer you should include
• a sketch graph to show how the concentration of S2O82– ions changes
over time
• an explanation of how Fe2+ ions catalyse the reaction, including
equations
• an overall equation for the reaction.
_____________________________________
1a labelled axes
and concentration (of S2O82- ions) decreasing with
time (ignore units)
1b downwards curve of reducing steepness
stage 2 explanation
2a (reaction slow) because S2O82- and I- repel/high
Ea
OR
(reaction slow) because two negative ions
repel/high Ea
2b Fe2+ attracts the S2O82- so lower Ea
OR
Fe2+ and S2O82- oppositely charged so lower Ea
2c Iron/Fe has a variable oxidation state
OR
Fe2+ oxidised to Fe3+
OR
Fe2+ → Fe3+ + e-
stage 3 equations
3a 2 Fe2+ + S2O82- → 2 SO42- + 2 Fe3+
3b 2 Fe3+ + 2 I-
→ 2 Fe2+ + I2
3c S2O82- + 2 I-
→ 2 SO42- + I2
Allow equations with hexaaqua ions
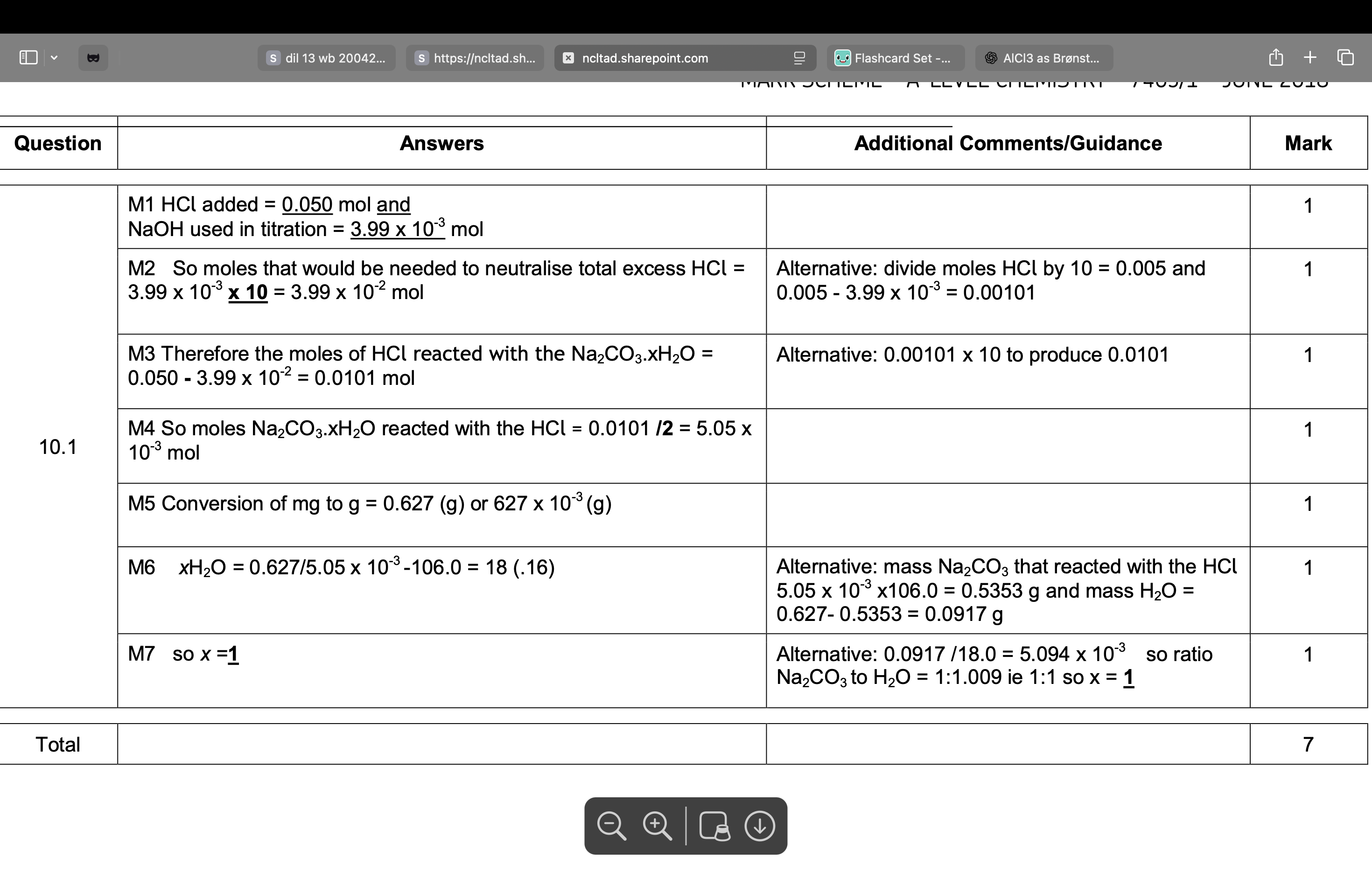
The student adds sodium carbonate solution to a solution containing
[Fe(H2O)6]2+ ions.
State one observation the student would make.
green ppt

Solution A contains the compound [Cu(H2O)6]Cl2
An excess of ammonia was added to a sample of solution A to form solution B.
Write an ionic equation for the reaction that occurs when solution A is
converted into solution B and state the colour of solution B.
[2 marks]
Reagent D was added to another sample of solution A to form a yellow-green
solution.
Identify reagent D and write an ionic equation for the reaction that occurs when
the yellow-green solution is formed from solution A.
Explain why colorimetry cannot be used to determine the concentration of
solutions containing [CuCl2]−
In your answer refer to the electron configuration of the metal ion.
[Cu(H2O)6 ]2+ + 4NH3 [Cu(NH3)4(H2O)2 ]2+ + 4H2O
1
Deep blue / Royal blue / Dark blue (solution)\
HCl/ hydrochloric acid
1
[Cu(H2O)6 ]2+ + 4Cl– [CuCl4]2– + 6H2O
[Cu(H2O)6 ]2+ + 4HCl [CuCl4]2– + 6H2O + 4H+
1
07.6 (3)d10 or has full (3)d (sub) shell/orbital It is colourless/cannot absorb (frequencies of) visible light
equation when ALCL3(aq) acts as a brostead lowery acid
[Al(H2O)6]3+ + H2O → [Al(H2O)5(OH)]2+ + H3O+


(e) [CuCl4]2− + 6H2O → [Cu(H2O)6]2+ + 4Cl−
1
(f) deep / dark blue
1
[CuCl4]2− + 4NH3 + 2H2O → [Cu(NH3)4(H2O)2]2+ + 4Cl−
Give the formula of the complex ion C.
State one condition needed for the formation of C from [Al(H2O)6]3+(aq) and
NaOH(aq).
Give an equation for this reaction.
[3 marks]
M1 C = [Al(OH)4]– OR [Al(H2O)2(OH)4]– OR [Al(OH)6]3–
M2 Excess NaOH
M3 [Al(H2O)6]3+ + 4 OH– [Al(OH)4]– + 6 H2O
OR
[Al(H2O)6]3+ + 4 OH– [Al(H2O)2(OH)4]– + 4 H2O
OR
[Al(H2O)6]3+ + 6 OH– [Al(OH)6]3– + 6 H2O
Explain, with the use of an equation, why a solution containing [Al(H2O)6]3+ has
a pH ˂7
[3 marks]
M1 [Al(H2O)6]3+ + H2O ⇌ [Al(H2O)5(OH)]2+ + H3O+ OR
[Al(H2O)6]3+ ⇌ [Al(H2O)5(OH)]2+ + H+
Al3+ has a small size and high charge OR has a high charge
Accept other equations
M2 density
M3 M2 Allow the aluminium ion has a small size and
high charge OR has a high charge density
Weakens the OH bond (in water) releasing H+ ions