pcs unit 3 - immune system
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
immune system
a distributed set of cells that fight disease in the blood, lump fluid, tissues, and organs throughout the body; a system of physiological defense that…
distinguishes self vs non-self
removes/makes harmless foreign substances
destroys cancerous cells
innate immune responses
defend against foreign substances without having to recognize their specific identities
adaptive immune responses
depend on the immune system recognizing specific pathogens; usually needs to be introduced to the pathogen before (i.e., getting sick or vaccination)
leukocytes
white blood cells
macrophages
natural killer cells (NKs)
neutrophils
lymphocytes
B and T cells
categories of innate defenses
physical barriers
phagocytes
natural killer cells
complement system
immune system
physical barriers
keep hazardous organisms and materials outside the body
ex: multilayered skin, hair, mucus linings, stomach acid, and antimicrobial chemicals (lysozomes)
phagocytes
cells that engulf pathogens and cellular debris
ex: macrophages and neutrophils
natural killer cells (NKs)
perform immune surveillance by recognizing and destroying cancer cells and virally infected cells by lysing the cells
complement system
a set of at least 30 proteins circulating in the plasma that assist in the destruction of pathogens
can recruit phagocytes (e.g., neutrophils and macrophages)
activates the membrane attack complex (MAC)
steps of inflammation
a number of types of cells, including epithelial cells and leukocytes, release signaling molecules that
increase capillary permeability → endothelial cells contract, widening spaces (intercellular clefts) between them to increase protein and leukocyte delivery to the injured area
dilate local arterioles → increase blood flow to the areas, increasing the delivery of proteins and WBCs (edema)
phagocytes such as neutrophils and macrophages, move out of the blood (across the endothelium of capillaries) to enter the inflamed area
chemotaxis
killing the pathogen
engulfment (then destruction) by a phagocyte cell, like neutrophils
complement system
tissue repair
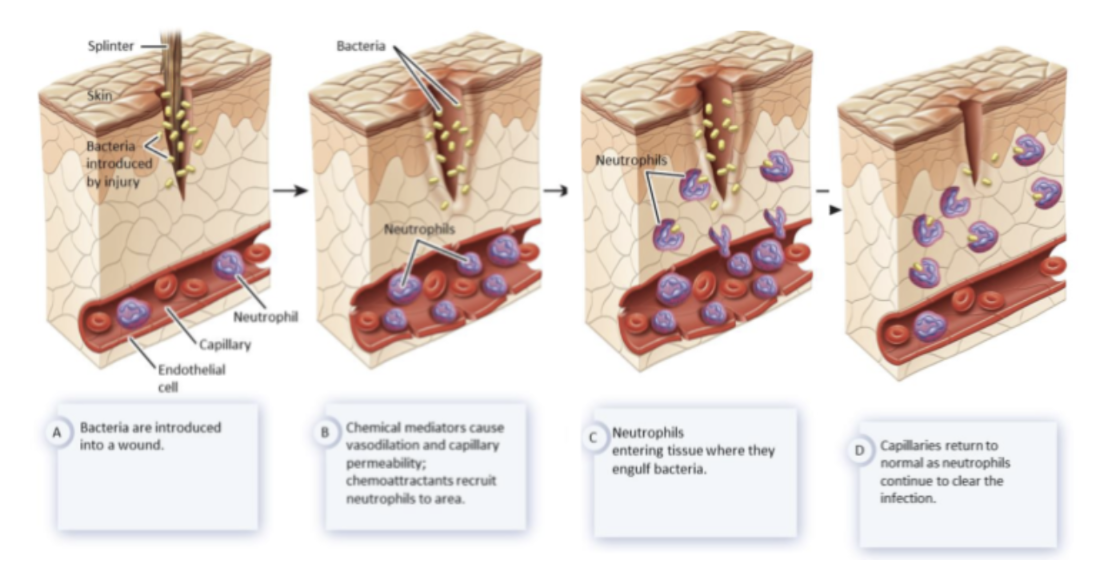
chemotaxis
directed, multistage migration of WBCs from the blood to an injury site in response to a chemical stimulus
C3B
one of the complement proteins; interacts with the pathogen membrane and marks it for destruction via phagocytosis or MAC
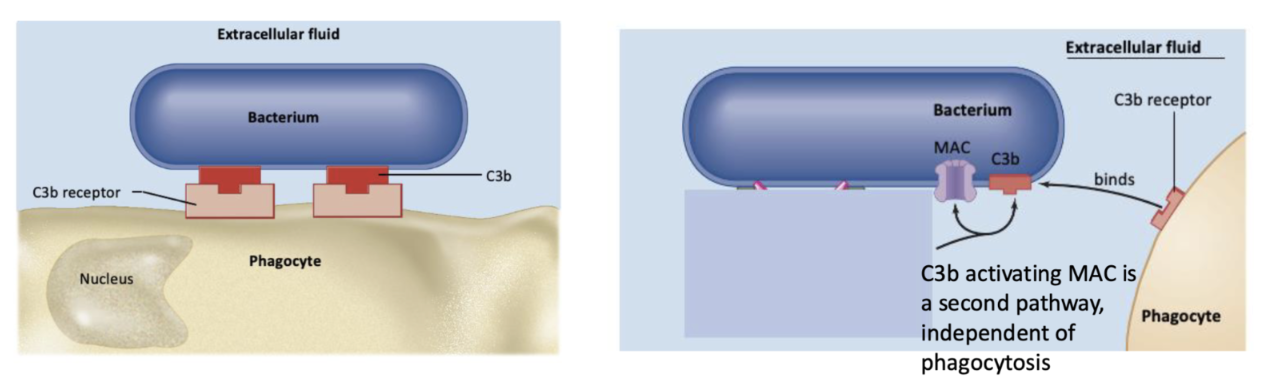
membrane attack complex (MAC)
a complex of complement proteins that embeds in the membrane of the pathogen and pokes holes in it
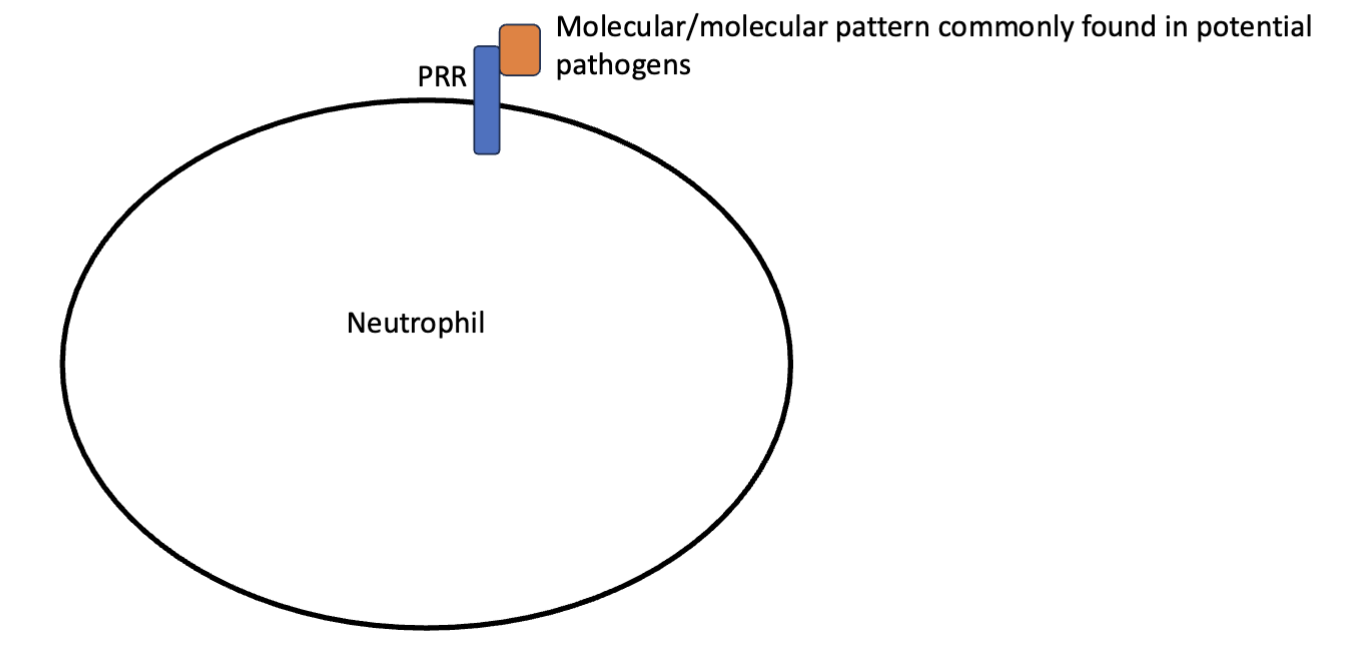
patten recognition receptors (PRRs)
transmembrane proteins on neutrophils that recognize molecular patterns commonly found in potential pathogens
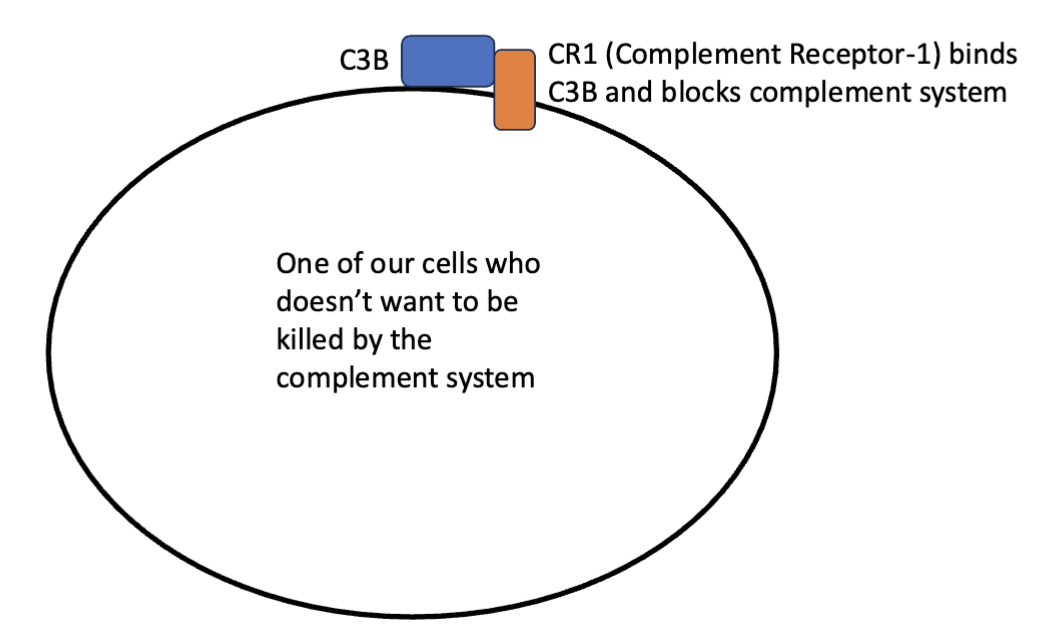
complement receptor-1 (CR1)
binds to C3B and blocks the complement system
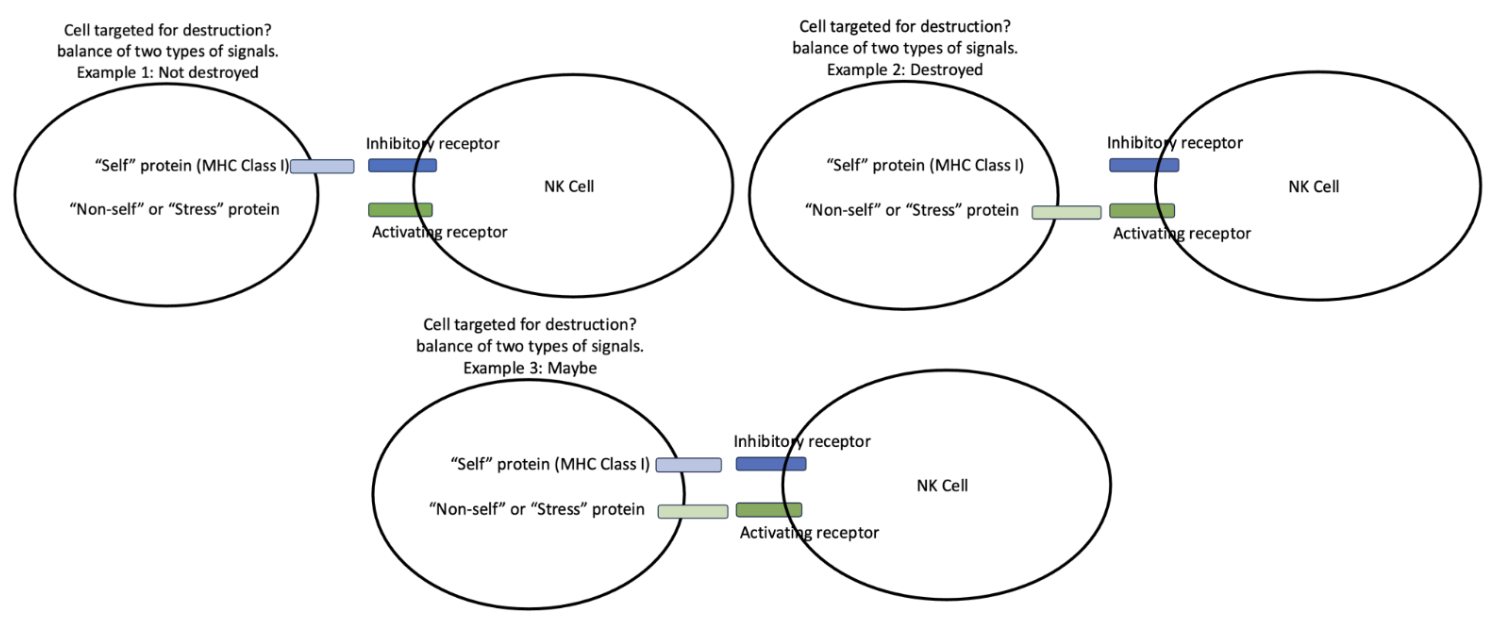
natural killer cells (NKs)
use a combination of activation and inhibitory receptors
activating receptors → send an activating signal, telling NKs to kill cells
inhibitory receptors → send an inhibitory signal, telling NKs not to kill cells
when both are present, cell destruction depends on how much of the activation receptor vs inhibitory receptor is present
lymphocytes
key cells in adaptive immune response; originate in the bone marrow, and can be found in the lymphatic system and the blood
B cells → plasma cells, memory B cells
helper T cells
cytotoxic T cells
lymphatic system
vessels that drain interstitial fluid into the veins
term and blood vessels are where most B and T cells reside
antigens
any kind of marker (typically proteins/sugars) that the immune system can recognize to generate an adaptive immune system; the key to specify in adaptive immune response
exist on viruses, bacteria, allergens, parasites, proteins, tumor cells (non-self), and normal cells (self) in our own bodies
B cells
express one antigen receptor (which can be secreted as an antibody) which recognizes one antigen
antigen binding to receptors cause division → creates plasma cells and memory term
antigen presentation is a second function
a type of immunoglobulin
immunoglobulin
multi-subunit proteins with a vairable region
plasma cells
secrete antibodies
memory B cells
store antigen receptors for future immune responses
antibodies
secreted by memory B cells
the Fc (stem) portion interacts with antigen receptor molecules on phagocytes, stimulating phagocytosis of pathogens
also activate the complement system
helper T cells
bind to antigens complexed with MHC Class II
specific to one antigen
require co-stimuli to fully activate and secrete signals that
activate themselves (autocrine signaling)
activate nearby B cells, cytotoxic T cells, and NK cells (paracrine signaling)
cytotoxic T cells
activated by antigen complexed with MHC Class I molecules
specific to one antigen
require signals from helper T cells for activation
MHC Class II
not antigen-specific
made by a macrophages, B cells, and other antigen-presenting cells
bind to Helper T cells
MHC Class I
not antigen-specific
expressed by all cells so cytotoxic T cells can target the destruction of any cell
DNA recombination
occurs in B cell receptors and T cell receptors
occurs during development for DNA coding antibodies/receptors
results in the coding of different versions of proteins to produce enormous diversity → allows cells to respond to different types of antigens
second exposure
bigger and faster than the first (peak response 7-10 days vs 2-5 days) due to more antibody production
immunological memory is mediated by memory T and B cells
after an infection
most activated lymphocytes undergo apoptosis
antibodies can last for days to months
memory B cells remain, some helper T cells and cytotoxic T cells also remain as memory cells
active immunity
resistance to a pathogen that occurs because of exposure to a pathogen or vaccination, causing an adaptive immune response
passive immunity
resistance to a pathogen that occurs because of direct transfer of antibodies from one individual to another
occurs during gestation, breastfeeding, and as therapy
encounter and antigen recognition (step 1 of adaptive response)
each lymphocyte expresses one type of surface receptor that could recognize an antigen
if the surface receptor binds an antigen, that is “recognition”
lymphocyte activation (step 2 of adaptive response)
binding of antigen activates lymphocytes, causing multiple rounds of cell division, clonal expansion, and stimulating their function in coordinating attack/immune memory
coordinate attack (step 3 of adaptive response)
antibodies target pathogens for destruction by macrophages and the complement system
cytotoxic T cells kill our cells presenting antigen with the MHC Class I molecule (most often virally infected cells)
helper T cells supply signals that participate in the activation of other immune cells
passing the infection (step 4 of adaptive response)
once the pathogen is destroyed, most activated lymphocytes undergo apoptosis
antibodies and memory B and T cells mean that we respond more quickly and robustly to the next infection
immune tolerance
the development of the prevention of lymphocytes from attacking our own cells
includes central and peripheral term
central tolerance
development of tolerance in the organs where developing B and T cells mature by clonal deletion
B cells mature in the bone marrow, T cells mature in the thymus (lymph organ above the heart)
clonal deletion in B cells
process of apoptosis when immature B cells with strongly reactive receptors to antigens encounter a wide range of self-antigens during maturation in the bone marrow
clonal deletion in T cells
process of apoptosis when immature Helper T cells and Cytotoxic T cells with strongly reactive receptors to antigens encounter a wide range of self-antigens during maturation in the bone marrow
mediated by TECs
thymic epithelial cells (TECs)
express both MHC Class I and II molecules and present antigens to both immature Helper T and Cytotoxic T cells
peripheral tolerance
recognition of specific antigens by Regulatory T cells to inactivate T cells
mediated by Regulatory T cells
regulatory T cells
central role occurs outside of the thymus and bone marrow
can recognize self antigen complexed with MHC Class I and II proteins
target both autoreactive Cytotoxic T cells and Helper T cells for inactivation
autoimmune disease
occurs when the immune system attacks antigens on our own cells
ex: multiple sclerosis (attacks myelin in the CNS), type 1 diabetes mellitus (attacks beta islet cells in the pancreas)
common pathways of autoimmune diseases
failure in clonal deletion and/or inactivation of autoreactive lymphocytes
low number or low activity of TECs or Regulatory T cells
exposure to pathogens containing antigens very similar to our own proteins can lead to cross-reactivity
innate vs adaptive immune responses
innate → rapid (hours, requires chemotaxis), non-specific, no immunological memory
adaptive → longer (days, requires clonal expansion), specific, has immunological memory
interaction of innate and immune responses
antibodies direct phagocytes and the complement system to pathogens
macrophages present antigens to helper T cells
Helper T cells supply signals that help activate NK cells
B and T cells arrive via chemotaxis to sites of inflammation