Week 11 - Infectious Diseases
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
Disease
A disturbance in normal functioning of an organism
Zoonotic diseases + examples
Infectious diseases from animals. Humans are usually dead-end hosts (ex. rabies, West Nile fever, Ebola).
Pathogens
Microbes frequently associated with disease production
Pathogenesis
Mechanism a microbe uses to cause the disease state, including infection and specific signs/symptoms
Primary pathogens vs opportunistic pathogens
Produce disease readily in healthy hosts vs only cause disease when displaced to an unusual site or when the host has a weak immune system
Virulence
Intensity of disease that a pathogen can induce
Case-to-infection (CI) ratio
Proportion of infected individuals who develop the disease
LD50 vs ID50 vs TCID50
LD50: amount of pathogen/toxin that kills 50% of subjects
ID50: amount of pathogen that causes infection in 50% of subjects
TCID50: amount of pathogen that causes infection in tissue culture 50% of the time
Attenuated strains
Weaker versions of viruses that are useful for vaccines (train your immune system)
Avirulent strains
No longer cause disease
Carrier (asymptomatic)
Individual infected with a pathogenic microbe who never exhibits signs or symptoms of the disease (asymptomatic), can still transmit the microbe to others
How do pathogens cause infections? (3)
Gain access to tissues
Evade/overcome host defences
Obtain nutrients from the host
Virulence factors
Produced by pathogens to help cause infections
E. coli (disease + virulence factors + action)
Causes hemorrhagic colitis and kidney failure
Intimin, tir, T3SS, shiga toxins
Attachment, receptor for attachment, injects tir for attachment, stops translation in host cells
Neisseria gonorrhoeae (disease + virulence factors + action)
Causes gonorrhea
Fimbriae, IgA protease, LOS (an endotoxin)
Attachment, immune evasion, destruction of IgA antibody, evokes inflammatory damage
Attachment mechanisms
Can occur through specific protein-protein interactions (ex. viruses) or through more general interactions (ex. rice blast spores stick to most cells)
Host range + how can it be expanded?
The group of organisms a pathogen can infect. Can be expanded via mutation (ex. FPLV in cats became CPV in dogs)
Antigenic variation/drift
Shift their surface protein structures to avoid host immunity
Capsules
Provides attachment
Interferes with opsonization (phagocytosis), antibody binding, and antibody stimulation
Latency
Ultimate evasion method, virus inserts its genome into host cells and goes dormant. Periodic reactivation → lytic replication may occur
Restriction endonucleases
Used by bacteria to digest phage DNA, a defence mechanism
Exotoxins vs endotoxins
Toxins produced/secreted vs toxins part of the microbial structure itself, both cause inflammation
Examples of endotoxins (2)
Lipopolysaccharide (LPS): most common endotoxin, gram-negative bacteria
LOS: lacks the O-antigen
Lipoteichoic acid: found in Gram-positive cells
Examples of routes of transmission (4)
Fecal-oral
Respiratory
Vector-borne
Sexual transmission
Direct vs indirect contact
Physical contact between hosts vs an inanimate object carries the agent between infected and susceptible hosts
Horizontal route of transmission + examples
Transmission between members of the same species, often via respiratory, fecal-oral, and sexual exit + entry points (ex. rhinovirus, influenza, polio)
Vertical route of transmission + examples
Transmission from parent to child, often in utero via placenta, birth, breast milk, and germ cells (ex. HIV, hep B + C)
Epidemiology
Study of patterns of disease in populations
Morbidity vs mortality rate
Rate of disease vs rate of death due to disease in a pop.
Incidence vs prevalence
Number of new cases appearing vs total number of cases overall
Endemic disease
Always present in the population, often results in cyclical patterns due to season, immunity status, etc
Epidemic
Incidence of a disease rises much more than the normally expected value
Outbreak
Unexpected cluster of cases in a short time in a localized population
Common-source epidemics
Single source of infection which the population is exposed to, rapid increase in incidence followed by decrease, cases tend to stay near the source of the problem (ex. food poisoning)
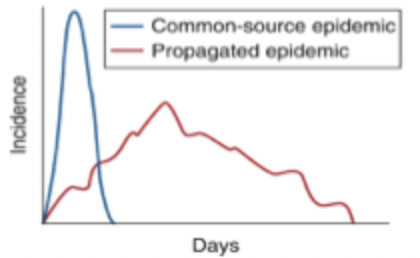
Propagated epidemic
Infection passing from one host to another, exhibits a gradual increase in incidence over time (ex. flu, measles, tuberculosis, COVID-19)
Koch’s postulates (4)
Used to show whether a specific microbe causes a specific disease:
Suspected microbe must be in every person with the disease, but not those without the disease
Pure culture of the suspected microbe is obtained
Experimental inoculation of the suspected microbe into a healthy test subject causes the same illness
Suspected microbe is recovered from the experimentally inoculated host organism
Gastric ulcers and Koch’s postulates
H. pylori was thought to cause stomach ulcers
But it was also found in many individuals without ulcers, and regular use of medication can also cause ulcers
Issue with Koch’s postulates
Not all individuals show the same degree of infection, and some things are genetically more susceptible to certain infections
Molecular Koch’s postulates
Sometimes, the classic postulates are not ethical or possible, so a modern adaptation was made:
Virulence factors should be present in the pathogen
Experimental inactivation of the vir factor gene should decrease virulence
Vir factor gene should be expressed in an infection
Immunity to the pathogen/resistance to the vir factor must provide protection
Virulence factors + genetic spread between pathogens
Include adhesion, invasion, or secretion factors
Can be passed via horizontal gene transfer, implanting pathogenicity islands
Emerging/reemerging diseases
Occurs when a pathogen encounters a new population (zoonotic transfer), ex. transfer of SIV in monkeys to HIV in humans
Lyme disease (microbe, vectors)
Caused by B. burgdorferi
Usually in deer and mice, but is transmitted to humans through ticks
Spread quickly through the US when humans spread into forests
Cause of pathogenic E. coli
Acquiring vir factor genes via horizontal gene transfer, producing shiga toxins and shutting down translation
How did methicillin-resistant S. aureus (MRSA) come to be?
Selective pressure from antibiotic overuse has led to acquisition of resistance traits against the drugs
Enterococcus faecium case study
Gastrointestinal bacteria that cause severe infections
Resistant E. faecium (VREfm) emerged due to high antibiotic use in hospitals
Sequenced REfm using Illumina to analyze factors contributing to resistance, found emerging strains inhibited the growth of old strains
Bacteriocin T8 production was found to be a driving feature of global VREfm strain emergence and persistence in healthcare settings
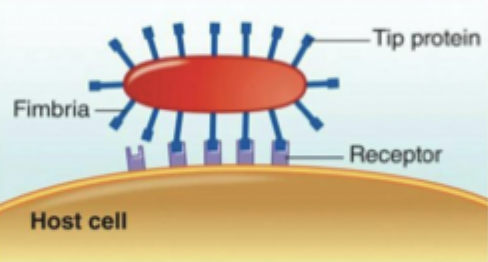
Fibronectin-binding proteins
Bind to fibronectin via fimbriae w/ adhesive tips, repulses negative charge on the host cell
Tir and intimin
Tir is injected into intestinal cells via T3SS, producing a pedestal that bacteria latch onto and feed from via intimin. This allows them to steal nutrients from intestinal cells
Methods that microbes use to deliver virulence factors (3)
Cell lysis (ex. C. botulinum releasing botulinum toxin)
Secretion to environment (ex. S. aureus and a-toxin)
Injection into host cell via T3SS and T4SS by gram-negative species (ex. E. coli and tir)
Iron-binding strategy by pathogens
Iron is a limiting nutrient sequestered by lacto+transferrin and locked inside host cells
Use siderophores to compete with host lactoferrin and transferrin
Lower pH at site of infection, impairing lacto and transferrin
Produce cytolysins that lyse host cells to steal iron
Tuberculosis + iron
Caused by M. tuberculosis, which requires iron to survive + steals it from RBCs
Exotoxins (3)
A-B toxins: B subunit binds to host cell receptors, causing a negative action in the cell
Cytolysins: act on cell membranes to burst cells
Superantigens: stimulate T cells to secrete lots of cytokines
A-B toxins
B binds to receptors on host cells → taken up by endocytosis → B dissociates and forms a channel into the cell → A enters through the channel and has toxic enzymatic activity inside host cells
Diphtheria toxin (bacteria + activity + disease)
A-B toxin released by Corynebacterium diphtheriae
Inhibits EF2 + protein translation
Causes diphtheria
Cholera toxin and pertussis
A-B toxin by Vibrio cholerae and Bordetella pertussis
Increase cAMP levels by making adenylate cyclase constitutively active
Cholera and whooping cough
Shiga toxins
A-B toxin by Shigella dysenteriae and E. coli
Cleaves rRNA + prevents ribosome synthesis
Hemolytic uremic syndrome
SNARE proteins and A-B neurotoxins
Cleave SNARE proteins, preventing neurotransmitter release (ex. botulism toxin prevents muscle contractions by blocking acetylcholine, tetanus toxin produces continuous muscle contraction by blocking glycine and GABA )
Cytolysins
Form pores or degrade phospholipids (ex. hemolysins lyse RBCs, S. aureus forms pores)
Phospholipase cytolysins
Degrade phospholipids, ex. lecithinases causing gangrene
Superantigens + related syndromes
Act on T cells, releasing a massive amount of cytokines that induce systemic inflammation. Cause toxic shock syndrome + food poisoning
Streptococcus pyogenes (attachment + overcome host defences)
Opportunistic
Attaches with fibronectin-binding proteins
Overcomes host defences with capsules and M proteins (bind antibodies)
Mycobacterium tuberculosis
Surround themselves with lipoarabinomannan (LAM), superoxide dismutase, and catalase to protect themselves from oxidative damage
Form granulomas that crack open and cause caseous necrosis, freeing the microbe
How do bacteria become pathogens?
Random mutation
Horizontal gene transfer
Conjugation, transposable elements, plasmids, and temperate (lysogenic) phages
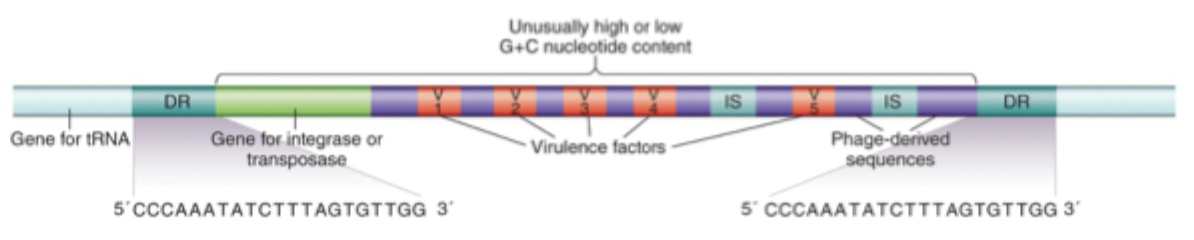
Pathogenicity islands (PAIs) (what is it + what they code for + how are they identified)
Large DNA sequences from foreign sources via HGT, not found in non-pathogenic strains
Adhesion/invasion molecules, toxins, T3/4SS, iron-binding proteins
Unusually high/low GC content
Contents of pathogenicity islands related to transposons, integrated phage genomes, and phage DNA integration mechanisms
Transposons: flanking direct repeat regions
Integrated phage genomes: multiple mobile genetic elements
Phage DNA integration: adjacent tRNA genes
Transduction
Transferring toxic genes via lysogenic phages to other microbial strains
Why would phages carry toxin genes against eukaryotic cells? + diphtheria toxin example
Kills competing eukaryotic cells, providing more nutrients to microbial hosts of phages → phages can thrive. Diphtheria toxin is expressed by C. diphtheriae in the human body, where iron levels are low, so that the toxin can lyse body cells for iron
Role of protozoa in pathogen spread to human
Pathogens that can survive and thrive and protozoa often thrive inside human cells as well
Productive vs abortive infections
New infectious viral particles are produced in host cells (hijacks host cell) vs few (if any) are produced
3 types of infections
Acute, latent, and persistent
Acute infections + example
Short duration → symptoms observed → infection is cleared and immunity is gained (ex. common cold)
Persistent/chronic infections + example
New viral particles are continuously produced, so the host doesn’t clear the virus in a reasonable timeframe (but symptoms may cease). May be due to weakened immune system or mutated virus (ex. hep B or C)
Latent infections
Acute infections → latency, replication of virus shuts down and may reactivate under stress (ex. herpesviruses, lambda phage)
Mechanism of herpesvirus in latent infections
Maintains an episome (its own circular genome) during latency + latency associated transcripts (LATs) which help maintain latency
Mechanical route of transmission + example
Transfer of virus from host to host via another vector, where the vector is not a reservoir but just a transport vehicle. Some insect vectors are required for virus replication cycles, while some are just passive vectors (ex. yellow fever, myxoma virus)
Apoptosis
Cell suicide to safely kill infected cells, usually doesn’t cause inflammation. DNA is degraded into small fragments and bits of cell are released as “blebs”
Necrosis + poliovirus and bunyavirus mechanisms
Cell bursts due to overfilling with new viruses or viral impairment of normal functions. Poliovirus hijacks translation and makes a ton of viral proteins, while bunyavirus cap-snatches host cell mRNA, degrading it, and puts the cap on viral mRNA instead
Syncytia and inclusion bodies
Viruses fuse host cells together or form clumps of viral proteins inside host cells, both of which kill host cells
How are immune responses responsible for the signs and symptoms of viral infection? (cytokines + Hep B + HBV)
Inflammatory cytokines of the common cold causes symptoms
Killing Hep B-infected hepatocytes, impairing liver damage
HBV antibody-antigen complexes deposited in the kidney, causing damage
Molecular mimicry + example
Viral proteins looking similar to host proteins, causing antiviral responses to target host proteins (ex. multiple sclerosis is caused by T cells destroying myelin)
3 events that occur as viruses reproduce, altering their virulence
Mutations, recombinations, and reassortments
Viral mutations
Many viruses lack DNA proofreading, leading to changes over time
Antigenic drift
Structural mutations in hemagglutinin (HA) and neuraminidase (NA) viral surface proteins that make our immune responses ineffective
Viral recombination
Fusion of two separate viral genomes to make a new hybrid genome, resulting in entirely new properties
Virus reassortment
Segmented genome from multiple viruses swapping segments to form new viruses. Causes antigenic shift, a dramatic change in antigens due to reassortment
Cultural influences on the evolution and virulence of pathogens (2)
Nurseries: hospital attendants, who touch many babies, can serve as vectors
WW1 trenches: extreme crowding, many sick and injured, and the monoculture of male soldiers made disease spread rampant