Chemistry - Ionic Bonding
1/11
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
When do ions form?
When an atom loses or gains electrons to create a full outershell
What does it happen with?
A metal and a non-metal
Hydrogen charge
H+
Hydroxide charge
OH-
Ammonium charge
NH4+
Carbonate charge
CO32-
Nitrate charge
NO3-
Sulfate charge
SO42-
Ionic bond diagram
Dot and cross diagram
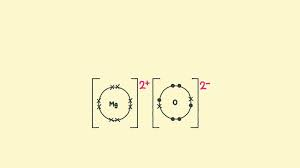
What is an ionic bond?
An electrostatic attraction between oppositely charge ions
Properties of Ionic Compounds
High melting and boiling points because the ionic bonds are strong and require lots of energy to break them
Do ionic compounds conduct electricity when solid and why?
They don’t because the ions are not free to move but do when molten (liquid) or dissolved in water because the ions are free to move