Atoms, Nuclei and radiation
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Experiment for existence and size of nucleus
Rutherford Scattering
Fired alpha particles to gold foil
Most pass through no deflection: atom mostly empty space
Some deflect by small angle: Nucleus positively charged
Very few reflected back: Very small and dense nucleus
Model of an atom
Nucleus: protons and neutrons
Orbital electrons
Nucleon number
Number of protons and neutrons
Proton number
Number of protons in an atom
Isotopes
Atoms of the same element, same number of protons, different number of neutrons (different nucleon number)
What happens to nucleon numbers and charges in nuclear processes
Conserved
No. nucelons (protons + neutrons) and leptons (electrons+neutrinos) of system conserved
Specific charge
Charge-mass ratio
Charge/Mass
What group of fundamental particles do baryons and mesons belong to
Hadrons
subatomic particles made up of quarks
Particles in baryons
3 quarks our 3 antiquarks
Particles that make up mesons
1 quark and 1 antiquark
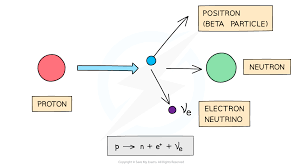
Beta-plus decay
Proton turns into a neutron, emit positron and electron neutrino
Up quark turns into down quark
Beta-minus decay
Neutron turns into a proton, emits electron and anti-electron neutrino
down quark turns into up quark
Composition of proton
uud
Neutron composition
udd
Difference between leptons and quarks
Quarks interact with strong nuclear force, quarks do
Compare the properties of a quark and an antiquark
Same mass
Same magnitude of charge
Both fundamental
Opposite charge
What is a fundamental particle
Particle which cant be broken down into anything smaller