IR - The effects of drug structure on analytical data
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
What are wavenumbers?
Number of waves per cm
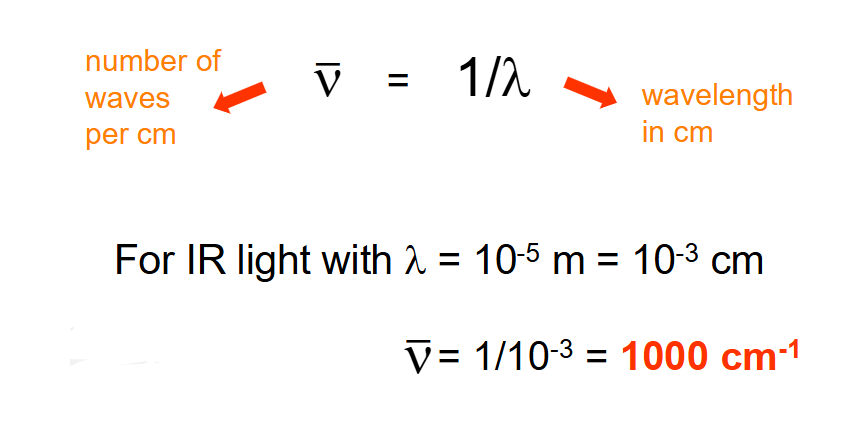
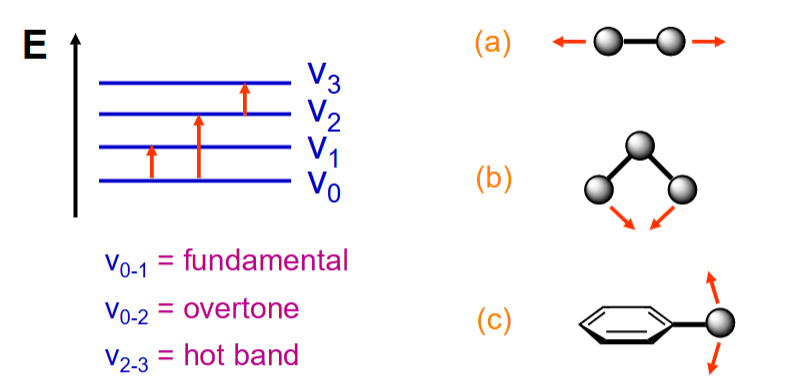
What are the 3 main types of vibration?
Bond stretching
Angle deformation
Out of plane deformation
Caused by IR light absorption
What is Hooke’s law?
Stronger bonds connecting lighter atoms absorb in the IR at higher frequency than weaker bonds connecting heavier atoms
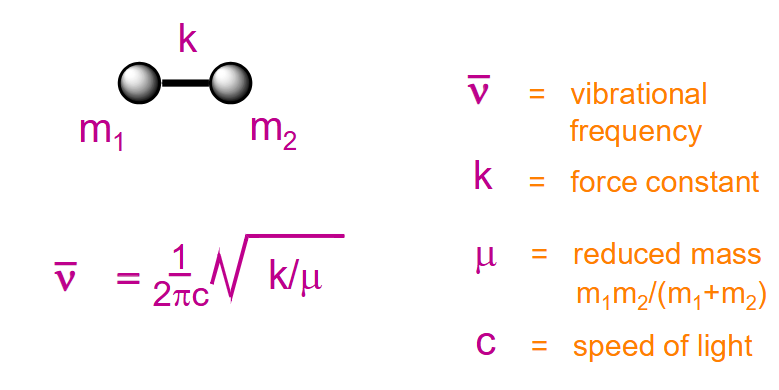
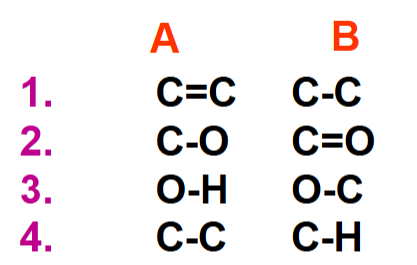
In 1 to 4 which has the higher vibrational frequency A or B?
C=C - double is stronger than single
C=O
O-H - H is lighter than C
C-H

Does vibrational frequency increase or decrease from left-to-right along this series?
Increases
Heavier from left to right
EN increases, pulling electron density toward X.
Bond polarity increases, strengthening the X–H bond.
Force constant increases, raising the vibrational frequency.
The reduced mass changes only slightly because all X atoms are light, so bond strength dominates the trend
What is used for calibration of IR?
Polystyrene C=C 1601 cm-1 band
Where are aromatic and aliphatic bands on IR?
Below 3000 - aliphatic
Above 3000 - aromatic
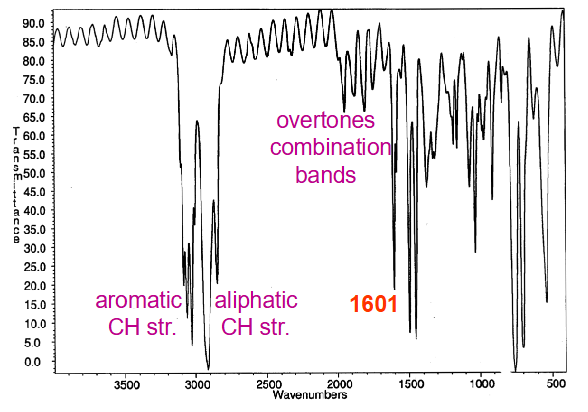
Why is hard to tell if there’s 1 or 2 forms of a bond, use amide as an example?
Two forms present in IR; free and H-bonded
2 x N-H str. at 3350 and 3200 cm-1
2 x C=O str. at 1690 and 1650 cm-1 sometimes merged
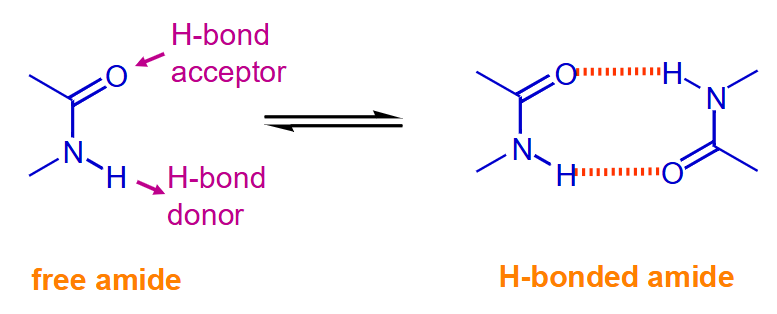
Resonance vs. Induction
What are the IR values of alkanes, alkenes & alkynes?
C-H saturated <3000 cm-1
C-H unsaturated >3000 cm-1
Bond length decreases with increasing S-character
Shorter/stronger bonds absorb in the IR at higher cm-1
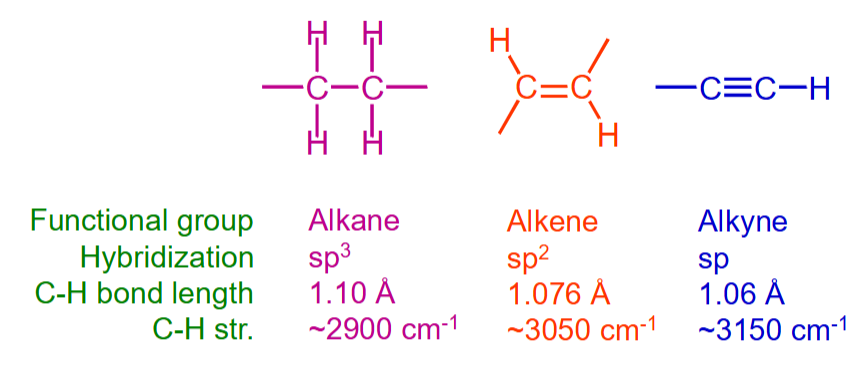
What bonds have wavelength 4000-3000 and ~3000cm-1?

What bonds have wavelength of C=O at ~1700 cm-1?

What bonds have wavelength 3000-1500 cm-1?
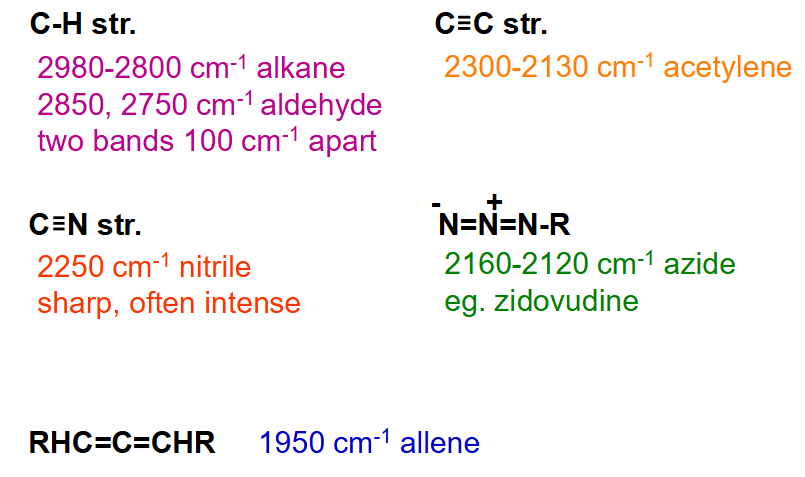
What bonds have wavelength 1600, 1500-900 cm-1?

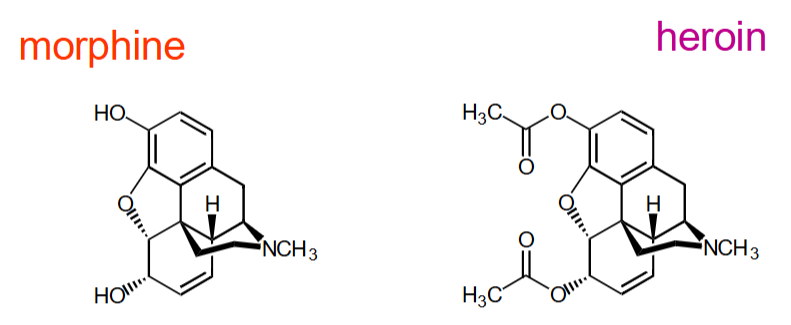
How are these two drugs different in the IR?
Morphine - 2 OH strr, ~3400 cm-1 but no C=O str
Heroin - 2 C=O str. ~1700 cm-1 but no OH str
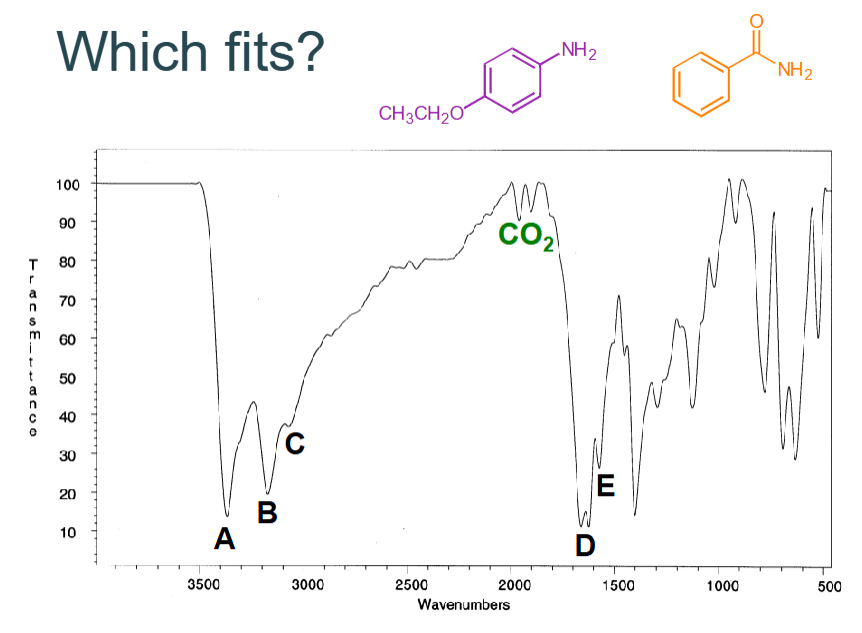
Which fits?
CO2 can be ignored
A - NH str
B - NH str
C - CH unsaturated
D - C=O str
Answer = orange
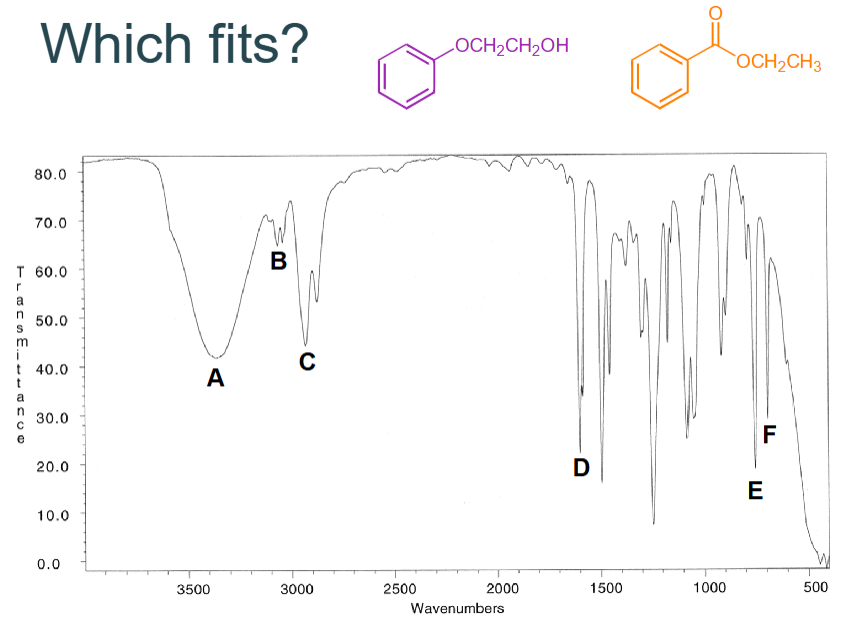
Which fits?
A - OH str
B - CH unsaturated
C - CH unsaturated
D - C=C str
E - benzene
No 1700 so not a carbonyl
Answer = purple
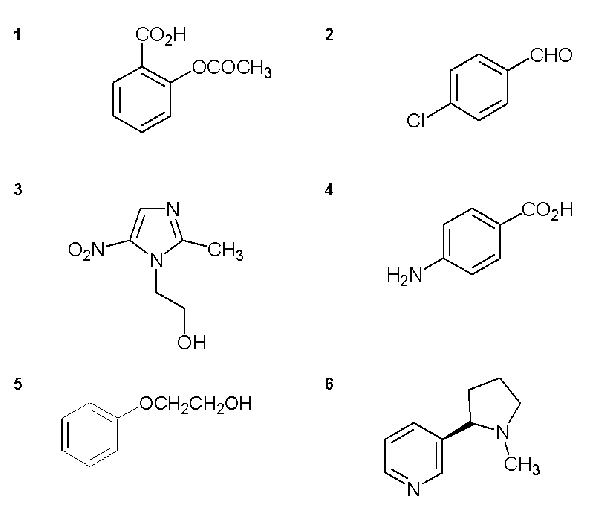
Infrared analysis of a particular compound (A) showed the following absorption bands at 3450, 3375, 3200-2550 (broad), 3030, 1675 and 1600 cm-1. The structure of compound (A) and five other compounds are given below.
Draw the structure of compound (A) and explain why your choice is consistent with the IR information given by assigning the IR data to the functional groups.
No band just below 3000 so not aliphatic (CH)
Answer = structure 4