Repro: L6,7,8 flashcards
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
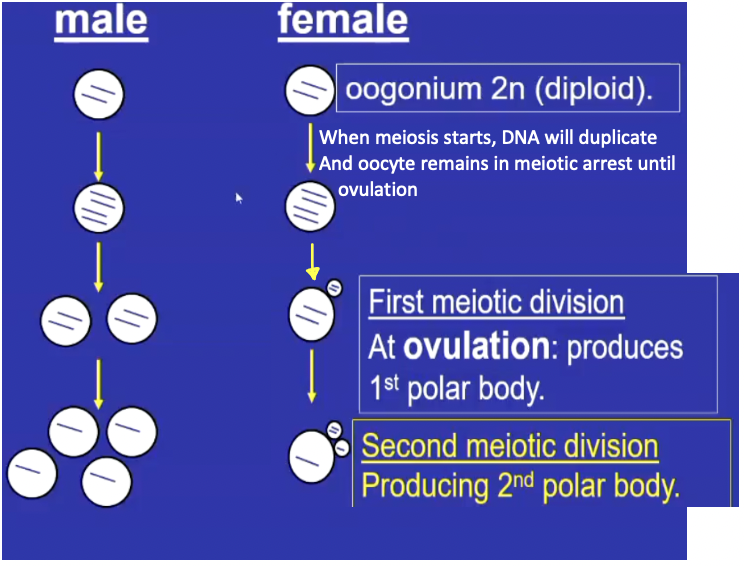
Main stages of germ cell development in females
Primordial germ cells (PGCs) → Oogonia → Oocytes → Follicles → Egg (ovum)
they are all the same cells at different stages:
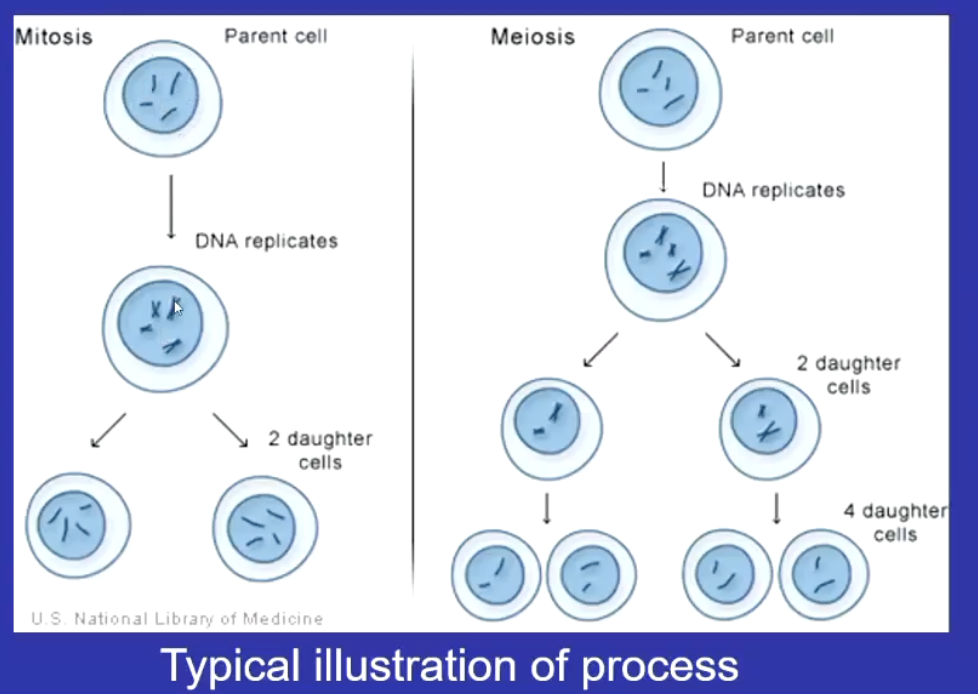
PGCs undergo mitosis: numbers increase as they multiply and is called an oogonia
Oocyte: when the oogonia stops undergoing mitosis and undergoes meiosis
Egg: when grows to maturity and ovulated
What happens to PGCs in migration
undergo mitosis and migrate to gonads via Growth factors such as TGFβ, SCF (stem cell factor), and bFGF (basic fibroblast growth factor).
end up as oogonia
What do oogonia do in the ovary
undergo mitosis and become surrounded by somatic cells, forming germ cell nests.
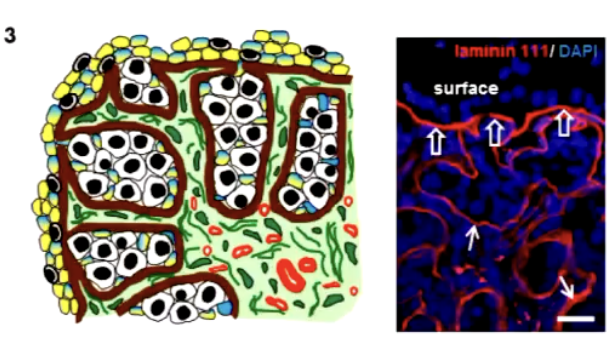
what are germ cell nests
Clusters of interconnected oogonia that share cytoplasmic bridges due to incomplete cytokinesis.
Cytoplasmic bridges: communication and sharing of resources, coordinating germ cell development.
What happens in oogonia > oocytes
oogonia stop dividing and enter meiosis and are now called oocytes
oocytes are finite
most oocytes die by apoptosis before follicle formation bc they have insufficient granulosa support or failure to reach meioti
What is meiotic arrest
A prolonged pause in meiosis that occurs in oocytes during fetal development and lasts until ovulation - oocyte remains activeb y:
Acquiring more components as it grows to resume meiosis around ovulation
Acquiring developmental competence to support embryo development and a healthy individual
essential for oocytes to remain viable for years and synchronizes maturation with follicle development and hormonal cycles.
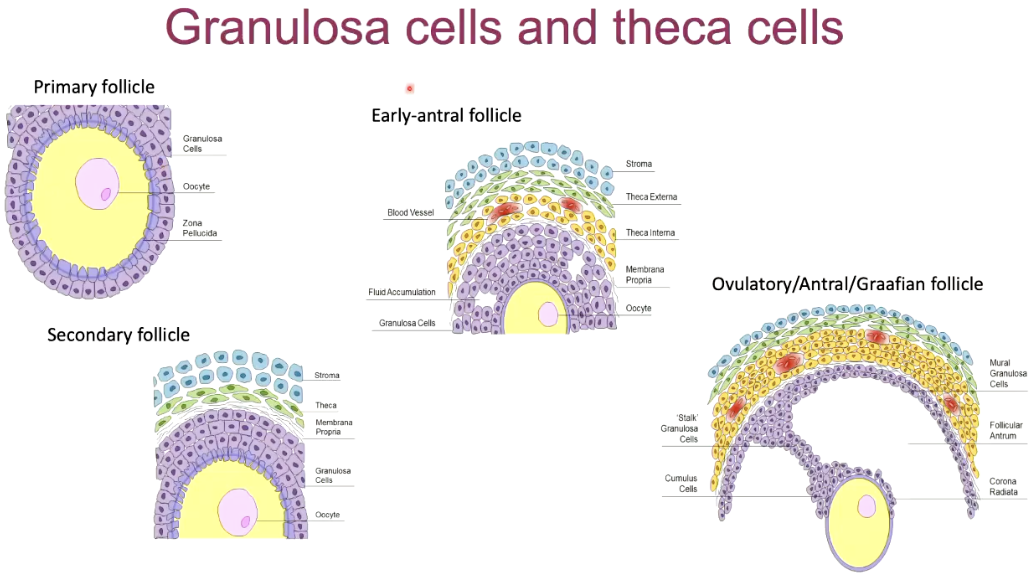
primordial follicles
form mid-pregnancy in foetal ovary (humans) or at birth in mice
structure is A primary oocyte surrounded by a single layer of flattened pre-granulosa cells.
The zona pellucida — a glycoprotein layer that persists around the egg even after ovulation.
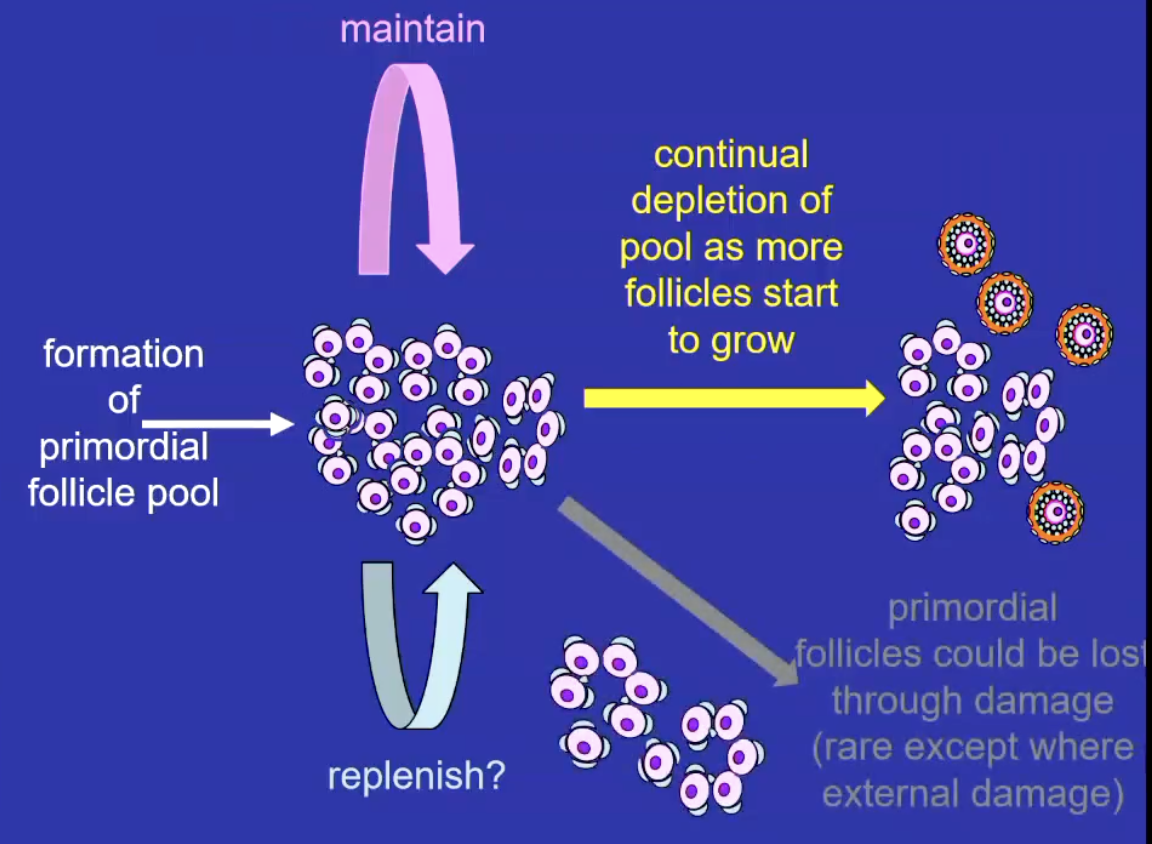
What is the primordial follicle pool
A finite reserve of resting follicles formed before birth; no new follicles are generated afterward.
Follicles either grow or die. (atresia) through reproductive life
The resting pool are quiescent follicles and if a follicle leaves they will grow and differentiate into more advanced follicles
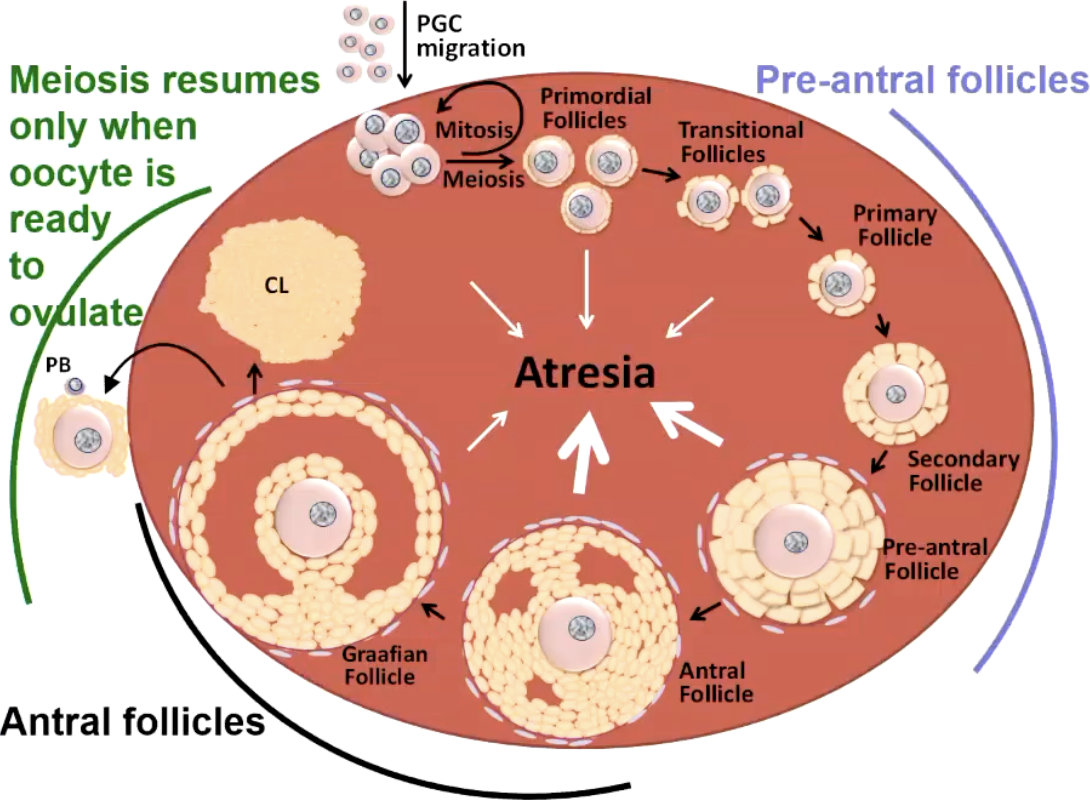
What is folliculogenesis?
Process where primordial follicles grow and develop into mature, ovulatory (Graafian) follicles
main stages are: Primordial → Primary → Secondary (Pre-antral) → Antral → Pre-ovulatory (Graafian).
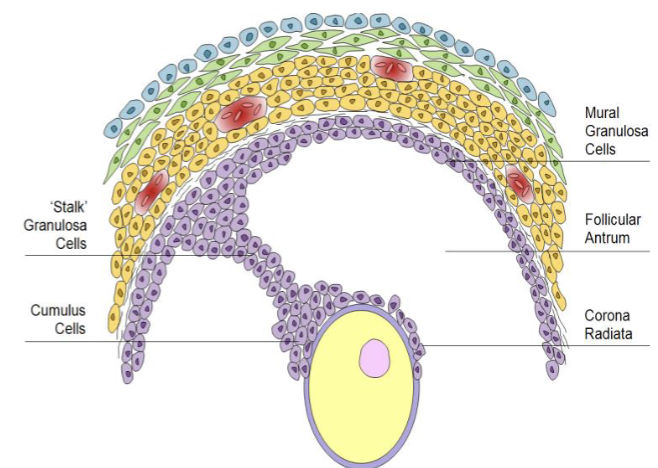
What is the strucure of granulosa cells associated with the oocyte as it ages
cumulus: loosely associated, some come out with oocyte in ovulation
corona radiate: attach to outside the zona pellucida
Stalk: suspend oocyte within follicular fluid
What hormones are involved with granulosa cells
They respond to FSH and produce Oestrogen and inhibin A/B
They influence the HPG axis (determines fertility and menstrual cycle
produce AMH as a marker of ovarian reserve
What are the main cell types in the follicle
Oocyte (germ cell)
Granulosa cells (somatic support cells)
Theca cells (hormone-producing outer layer)
What is the first visible sign of follicle activation
granulosa cells: squamous > cuboidal > then multilayerd
oocyte begins to grow
this is irreversible
Regulated by PTEN signalling pathway which keeps primordial follicles resting state
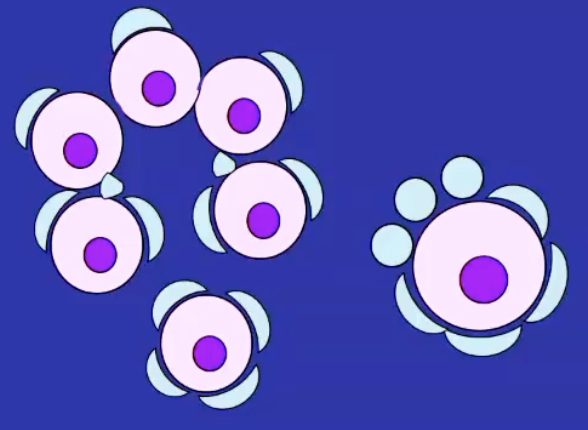
What happens if PTEN signalling is knocked out?
all follicles grow simultaneously = depletion of the ovarian reserve
What is a primary follicle
Follicle with growing oocyte and layer of cuboidal granulosa cells
What is a pre-antral follicle
follicle with multiple layers of granulosa cells and a basement membrane
forms the zona pellucida, a glycoprotein-rich ECM between oocute and granulosa cells
theca cell layer surrounds the granulosa layer externally
avascular inside the basement membrane
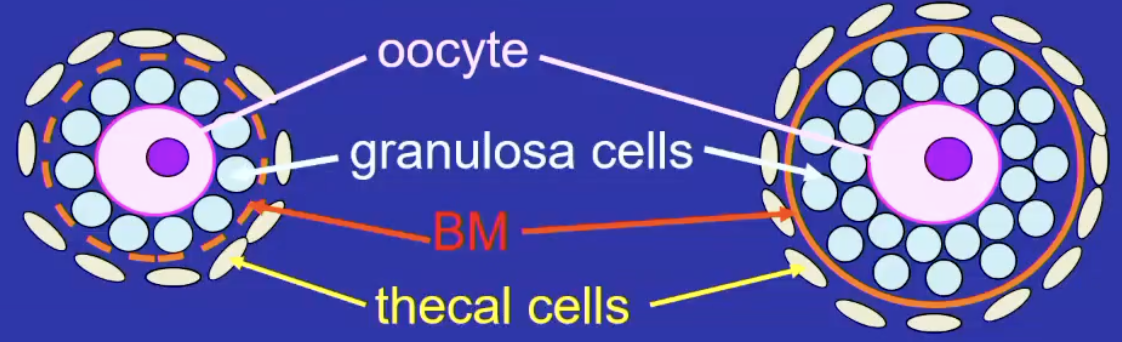
What are theca cells
Recruited from the ovarian stroma
essential for follicle growth
differentiate into theca interna and externa
the last to die in atresia
associated with PCOS (LH/FSH imbalance)
Theca interna
Respond to LH > produce androgens
> communicate hormonal messaes to the oocyte
only part of oocyte that has blood vessels (vascular)
Theca externa
Fibrous, for structural support
What is an antral follicle
formation of a fluid-fileld cavity (antrum) with the granulosa layers
antrum accumulates and supplies oocyte with nutrients and metabolic waste
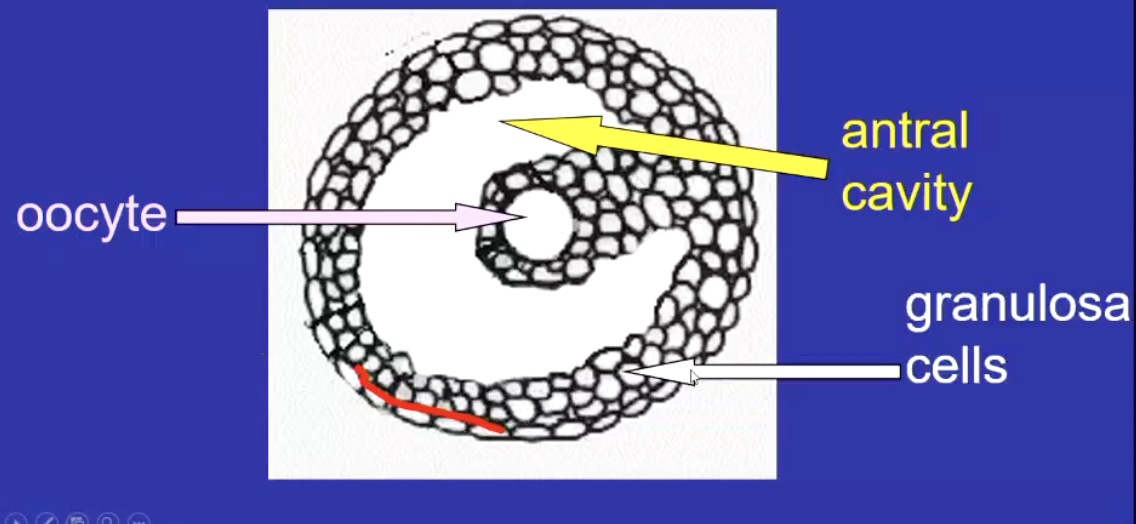
What happens to granulosa and oocyte as the antral follicle forms
oocyte grows and produces mRNAs needed for early embryonic development
granulosa cells proliferate and form specialised subtypes
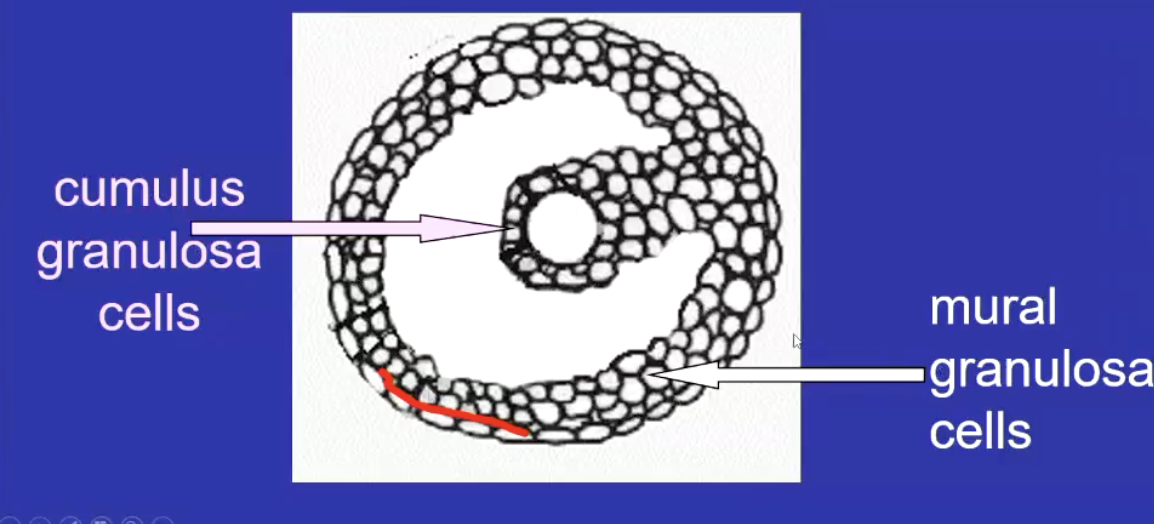
What are the 2 main types of granulosa in mature follicles
Cumulus granulosa: surround and communicate with oocyte
Mural granulosa: line the follicle wall, steroidogenically active, interface with the theca layer - produce steroids and nutrient and hormone exchange betweeen follicle and bloodstream
connected by gap junctions for transfer of ions and signalling molecules
What is the pre-ovulatory follicle
Large, fully mature follicle ready to release an oocyte during ovulation
stopped here until puberty
What occurs before ovulation
before ovulating, the follicle expands > oocyte resumes meiosis > cumulus and mural granulosa cell functions diverge
What occurs during ovulation
The oocyte + cumulus complex is released; meiosis continues to produce a haploid oocyte and a polar body.
The remaining follicle forms the corpus luteum which secretes hormones (prog) to support pregnancy
What is follicular atresia
degeneration and death of follicles that don’t complete development
90% of follicles undergo atresia
Hormone availability and competition for FSH determines if cell will undergo atresia
AMH and follicles
Produced by growing pre antral follicles, prevents premature follicle activation
High AMH levels inhibit recruitment of new follicles from resting pool
High AMH = fewer follicles activated, resting pool preserved
Low AMH: more follicles activate, reserve depleted faster
PTEN is better for suppressing follicle activation
Which hormones control ovarian follicle development?
GnRH from the hypothalamus stimulates secretion of FSH and LH from anterior pituitary
FSH: Stimulates granulosa cell proliferation and antral growth, Promotes estrogen synthesis in granulosa cells, Required for follicle survival beyond early stages.
LH: Stimulates theca cells to produce androgens (precursors to estrogen), Required for ovulation and corpus luteum formation.
Is FSH required for early follicle growth?
no
Does endocrine suppression (oral contraceptive) affect the primordial follicle pool
No — the primordial pool declines independently of circulating hormones.
When do follicles become hormonally dependant
At the antral stage, when they begin expressing FSH receptors on granulosa cells.
FSH stimulates aromatase expression in granulosa cells - converts theca-derived androgens into oestrogen
If follicles are FSH deprived, They undergo atresia and degenerate.
What does oestrogen do in follicles?
It promotes granulosa cell proliferation and positive feedback on follicle growth.
What is LH essential for?
Ovulation - triggers oocyte maturation and follicle rupture.
What is the LH surge
High levels of oestrogen from the dominant follicle trigger positive feedback on the hypothalamus and pituitary.
Oocyte completes meiosis I.
Follicular wall ruptures → ovulation.
Remaining granulosa and theca cells are luteinised to form the corpus luteum.
What occurs during luteinisation
Focus on production of progesterone over oestrogen
granulosa cells increase 10x in size
Corpus luteum become highly vascularised
Why is corpus luteum yellow
accumulation of glycogen granules and lutein pigments
What is follicle dominance?
The process where one (or a few) follicles are selected to continue development and ovulate, while others regress.
Occurs at mid-antral stage
Characteristics of dominant follicles?
More FSH receptors.
Better vascularisation of the theca layer (↑ FSH delivery).
Mural granulosa cells begin expressing LH receptors.
It has developed LH receptors and autocrine/paracrine signals that sustain growth even with lower FSH.
What hormone negatively feedbacks on FSH production
Ostrogen and inhibin from granulosa cells.
Why do different animals ovulate different amounts of oocytes?
Humans: only one follicle becomes dominant due to tight FSH regulation.
Sheep (2 oocyte): threshold for FSH decline allows two follicles to survive.
mice (6-10): Mice have weaker FSH suppression and multiple follicles can remain dominant.
What determines litter size in mammals
The number of dominant follicles that successfully ovulate.
Do follicles communicate with each other?
Yes — local paracrine signaling allows follicles to coordinate growth and regulate the number of dominant follicles.
Ensures the appropriate number of follicles mature each cycle to match species reproductive strategy.
What is the ovarian surface epithelium
thin layer outside the ovary
ruptures then repaires each month after ovulation - highly linked to cancer bc of the constant cell turnover (mutations)
Lots of stem-cell-like cells in this to help with repeated repair and rupture
What other structure is associated with ovarian cancers?
Fallopian tube
What is used to study ovarian mesenchymal stem cells
Pdgfr-alpha
isolated and studied with Flow cytometry (FACS) and single-cell sequencing
oMSC paracrine and protective effects:
Anti-apoptotic (protect granulosa cells)
Anti-inflammatory (reduce harmful inflammation)
Angiogenic (promote new blood vessels)
Stimulate follicles (activate dormant primordial follicles)
Anti-oxidant (reduce oxidative stress and ovarian aging)
What is the Extracellular matrix in the ovary
A 3D scaffold for follicles and stromal cells
made of water, proteoglycans, fibrous proteins
remodelled for follicle growth every month
becomes fibrous with age
Which pathway mediates tension signalling for follicle activation?
Hippo pathway
Where are perivascular cells found
Around blood vessels
What parts of the ovary is avascular or vasclar
cortex is avascular
medulla is vascularised which is essential for follicle growth and hormone transport
Identifying granulosa and theca diagramatically
granulosa: purple
theca: green
What is the Zona pellucida?
A Halo around the oocyte that is produced by the oocyte
Made up of glycoproteins secreted by oocyte and granulosa
ZP1,ZP2,ZP3 are structural components - repeated dimers
What is ZP3
Essential for preventing polyspermy after fertilisation
Changes conformation once sperm enters to block other sperm
Tried as birth control but it was not reversible and reduced the primordial follicle pool - scrapped
Oocyte activity and mRNA storage
Oocyte relies on stored mRNA - doesnt perform new transcription
Stored mRNA:
Regulates protein synthesis
Acts as a switch to turn gene expression on/off
Maintains meiotic arrest
Mitochondria in oocytes
inherited from mother
quality decreases with age due to being reactive to ROS + high mutation rate
bottleneck: Random segregation of healthy vs mutant mitochondria — no control over which primary oocytes get which
What is the lipid content in oocytes
Very high lipid levels (makes them dark in species like cows)
What are vesicles in oocytes?
Occupy 15–35% of volume; often move to the center during maturation; unclear function
What are cortical granules?
Secretory organelles from Golgi; react with ZP3 to block polyspermy
How do oocytes and granulosa cells support each other
Oocytes help granulosa cells expand
Granulosa cells help oocytes maintain meiotic arrest and support maturation
What are gap junctions
Specialised intercellular channels for exchanging ions, metabolites, signalling molecules
Maintain coordination between granulosa cells and the oocyte
What are the 2 key connexins (gap juncs)
Connexin 43 (Cx43)
Found between granulosa cells
Enables communication across follicular cell layers
Connexin 37 (Cx37)
Connects granulosa cells to the oocyte
Essential for oocyte-granulosa communciation
Function of gap junctions
Supports metabolic cooperation - oocyte depends on the granulosa for nutrients and energy
Maintain high cAMP in oocyte - prevents premature meiotic resumption
Facilitate signalling via oocyte-derived factors like GDF9 and BMP15 - promotes granulosa differentiation and function
What happens to ga[ junctions in ovulation
Supports metabolic cooperation - oocyte depends on the granulosa for nutrients and energy
Maintain high cAMP in oocyte - prevents premature meiotic resumption
Facilitate signalling via oocyte-derived factors like GDF9 and BMP15 - promotes granulosa differentiation and function
What are transzonal projections
Long, thing cytoplasmic extensions from granulosas - penetrate the ZP and contact the oocyte membrane
Contain gap junctions at their tips - reinforces the granulosa-oocyte communication
Unique structure
Bridge between oocyte and granulosa
Transfer of small molecules, growth factors, signals
Support oocyte metabolism and maintain meiotic arrest
What is genomic imprinting?
Epigenetic silencing where only one parental allele (maternal or paternal) is expressed
Maternal vs Paternal imprinting
Maternal: Mother’s gene inactive, father’s active
Paternal: Father’s gene inactive, mother’s active
imprinting occurs during sperm or egg development
What happens to the embryos genome after fertilisation (into zygote)
Global demethylation of DNA (except imprinted genes remain protected)
PGCs must remove imprinting patterns to later re-establish new imprinting during gametogenesis - ensures parent-specific gene expression
Cycle of imprinting
Fertilisation:
Zygote inherits imprints from both parents.
Some genes show methylation from one parent only.
Early Embryo:
Most of the genome becomes demethylated (methylation removed).
Imprinted genes remain methylated and retain their specific pattern.
Primordial Germ Cells (PGCs):
Later in embryonic development, PGCs undergo demethylation, including removal of previous imprints.
This “resets” the genome so that new sex-specific imprints can be established.
Gametogenesis:
As germ cells mature:
Female germ cells → acquire maternal imprints.
Male germ cells → acquire paternal imprints.
Cycle repeats with the next generation.
What stage are oocytes in meiotic arrest?
Dictyate stage of prophase I
meiotic arrest is maintained by GPCRs on the oocyte producing high cAMP levels
How does ovulation trigger meiosis to resume?
At ovulation, granulosa-oocyte contact decreases, GPCR activity stops, cAMP falls, and meiosis resumes
What weakens the follicle wall before ovulation?
Hyaluronan secreted by cumulus cells
phases of ovulation
Follicle Expansion — Hyaluronic acid causes swelling; essential for ovulation
Follicle Contraction — Smooth muscle in outer follicle contracts; inhibition stops ovulation
Follicle Rupture — Follicle surface bulges and ruptures, releasing egg and follicular fluid
What contraction of a cell is essential during ovulation?
muscle cell contraction is essential for egg release
oocyte survives 24 hours after ovulation