Williamson ether synthesis reagent
1/5
Earn XP
Description and Tags
Alkoxide (RO- Na+) as nucleophile (alkoxide ex) methoxide (CH4 O- Na+) or the sodium propoxide (CH3CH2CH2O-Na+) Alkyl halide as Electrophile
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
Williamson ether synthesis reagent
Alkoxide (RO- Na+) as nucleophile
Alkyl halide as Electrophile, typically a primary alkyl halide, is reacted with an alkoxide to produce ethers.
alkoxide
strong base and good nucleophile. ex) methoxide (CH4 O- Na+) or the sodium propoxide (CH3CH2CH2O-Na+)
williamson etherson mechanism
This reaction is an example of an SN2 mechanism, where the alkoxide attacks the electrophilic carbon of the alkyl halide, resulting in the formation of an ether and the release of a halide ion.
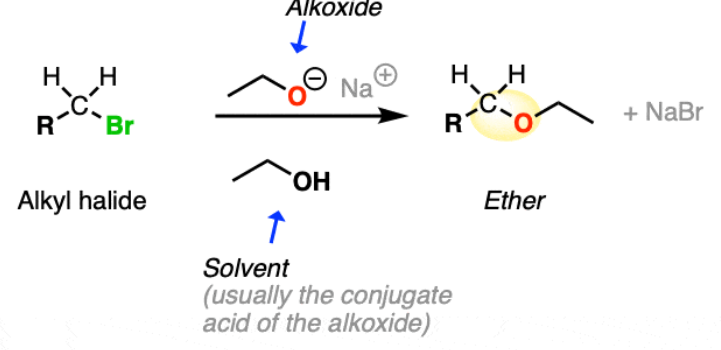
ether synthesis
Williamson ether synthesis
Acid-Catalyzed Dehydration
acid catalyzed dehydration
use alcohol instead of primary alkyl halide
Nucleophile: Alcohol (work as base) Electrophile: The H+ from the H2SO4 (the acid).