Nucleotides and nucleic acids
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
Draw + label the basic structure of a nucleotide
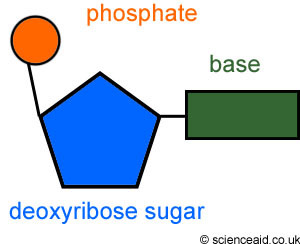
Draw a nucleotide showing the structure of the pentose sugar and where the phosphate group and nitrogenous base attach using the standard system for numbering the carbons in the sugar
Organic base = carbon 1
Phosphate group = carbon 5
What are the two main types of nucleic acid
RNA and DNA
Similarities and differences between DNA and RNA
DNA uses deoxyribose sugars
RNA uses ribose sugars
DNA has thymine
RNA has uracil
Basic nucleotide structure is the same
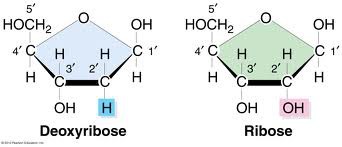
Draw the structures of ribose and deoxyribose and identify the difference between the two pentose sugars.
Deoxyribose has less oxygen
List all nitrogenous bases in nucleic acids
Adenine
Guanine
Thymine
Cytosine
Uracil
Name the two types of nitrogenous base and state which bases belong to which type
Purines: double ring structure - Adenine and Guanine
Pyrimidines: single ring structure - Cytosine, Thymine and Uracil
Define monomer
Individual molecules that make up a polymer
Define polymer
Long-chain molecules composed of bonded individual molecules (monomers) in a repeating pattern
Define nucleic acid
Large polymers formed from nucleotides. Contain the elements: carbon, hydrogen, nitrogen, phosphorous and oxygen
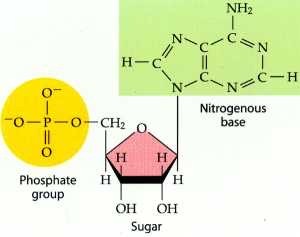
Define nucleotide
The monomers used to form nucleic acids.
Made up of a pentose monosaccharide, a phosphate group and a nitrogenous base
Define polynucleotide
A linear polymer composed of many nucleotide units, constituting a section of a nucleic acid molecule
Define phosphodiester bond
Covalent bonds formed between the phosphate group of one nucleotide and the hydroxyl group of another
Draw and label a diagram to show how nucleotides can link together to form polynucleotides (including the production of water).
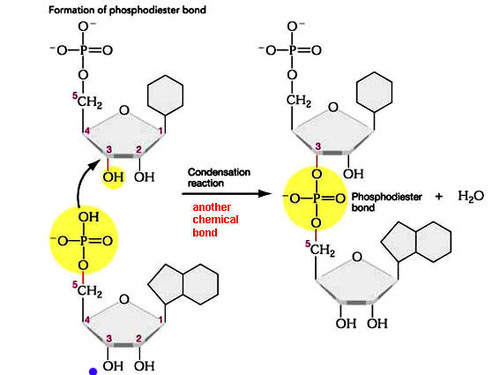
State the name of the reaction that joins nucleotides to other nucleotides and the name of the reaction that breaks phosphodiester bonds.
Condensation
State 3 main types of activity for which cells require energy with examples
Synthesis - e.g. making proteins
Transport - e.g. pumping molecules across membranes using active transport
Movement - e.g. protein fibres that cause muscle contraction
Draw + label diagram of ATP and ADP
ATP - ribose, adenine and 3 phosphates
ADP - ribose, adenine and 2 phosphates
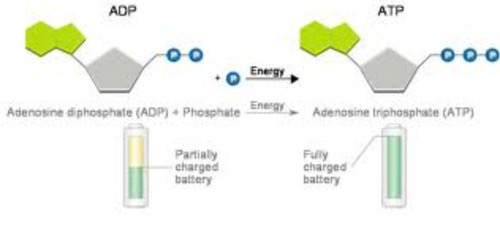
Give similarities and differences between the structure of ATP and DNA and RNA nucleotides
ATP uses ribose like RNA
Structure very similar but base is always adenine and has 3 phosphates instead of 1
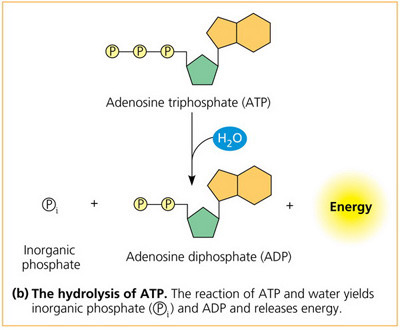
Draw a reaction to show how energy is released from ATP to provide energy for cellular activities.
A small amount of energy is needed to break bond holding last phosphate on but lots of energy given out when that ion then takes part in further reactions making bonds.
State 5 properties of ATP and explain why each makes it ideally suited to function as an energy transfer molecule
Small - moves easily into, out of and within cells
Water soluble - energy requiring processes happen in an aqueous environment
Contains bonds between phosphates of intermediate energy - enough to be useful but not so much that lots is wasted as heat
Releases energy in small quantities - suitable for cellular needs but not too much lost as heat
Easily regenerated - can be recharged with energy
Define the term "phosphorylation"
The addition of phosphate group to a molecule
Draw and label a diagram of the structure of DNA
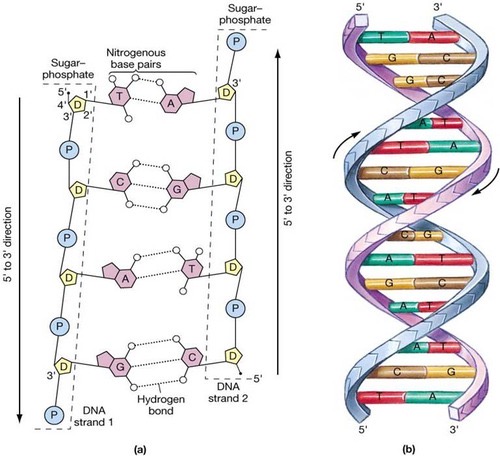
Define complimentary base pairing
Specific hydrogen bonding between nucleic acid bases. Adenine bonds to thymine or uracil and guanine bonds to cytosine
Define sugar-phosphate backbone
Forms the structural framework of nucleic acids composed of alternating sugar and phosphate groups
Define antiparallel
The two strands are parallel but run in opposite directions (one 3 to 5 the other 5 to 3)
Define double-helix
A pair of parallel helixes intertwined about a common axis
Define strand
One of the two polynucleotide chains that make up a DNA molecule
State the complementary base pairing rules, name the bond that holds them together, and state the number of bonds that hold each pair together
Adenine and Thymine are held by 2 hydrogen bonds
Guanine and Cytosine are held by 3 hydrogen bonds
Explain why a DNA molecule has equal amounts of adenine and thymine and equal amounts of cytosine and guanine
A and T are always paired together so will always appear in equal amounts. The same is true for C and G
Describe how purines and pyrimidines are arranged in the complementary base pairing rules
Always paired a pyrimidine with a purine as purines slightly bigger (two rings) and each rung must be the same length.
Describe the significance of the double stranded, complementary base paired nature of DNA for its function
The two strands allow it to be replicated accurately by semi-conservative replication and the complementary bases means once the strands have split, their is only one option to pair with each base to form the next strand
Describe the significance of the sequence of bases in a DNA strand for its function
The sequence of bases codes for amino acids and the full gene for a protein.
Describe, and explain the importance of the steps in the isolation and purification of DNA by precipitation
Grind up plant material with a pestle and mortar - breaks down the cell walls
Mix sample with detergent - breaks down cell membranes
Add salt to the solution - breaks hydrogen bonds between DNA and water
Add protease - breaks down proteins associated with DNA
Add layer of ethanol - causes DNA to precipitate between the two layers (ethanol on top, water on bottom)
The white strands of DNA can be picked up by spooling onto a glass rod