Chemistry Week 12
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
Valance Bond Theory
States that a covalent bond is just an overlap of atomic orbitals
Atoms want to be as close together as possible
- Limited by nuclei pushing each other away
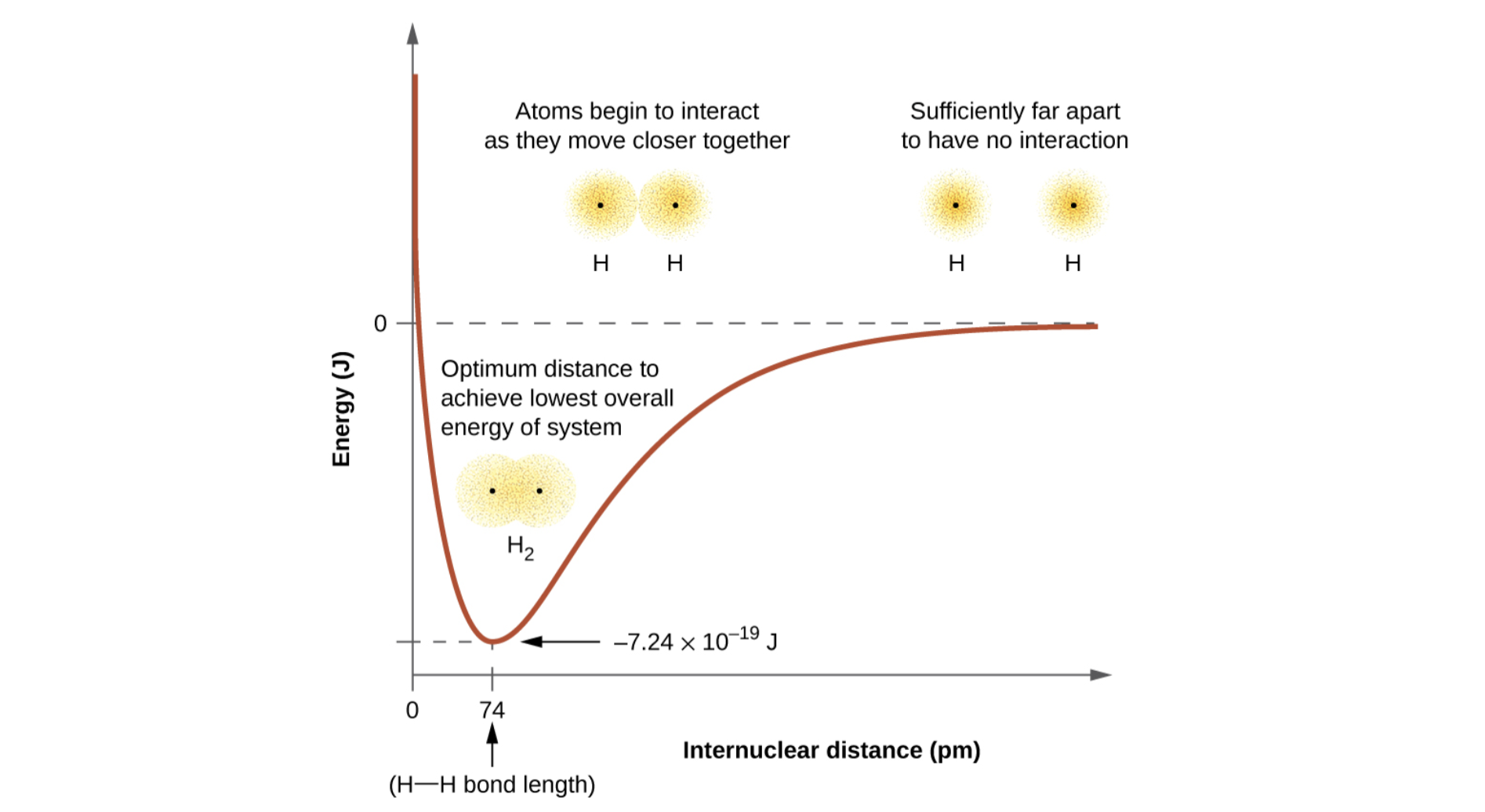
Energy slowly decreases as atoms move together, but then skyrockets as they get close enough for nuclei to be repulsive
Sigma Bond
Type of overlap of atomic orbitals
Happens when two s orbitals, an s and a p orbital, or the end to end overlap of 2 p orbitals occurs
Can be the only bond in a pair or the first bond of a multi bond
Bonds exist on the bonding access
- Bonds in one straight line of space, only capable of holding one electron pair
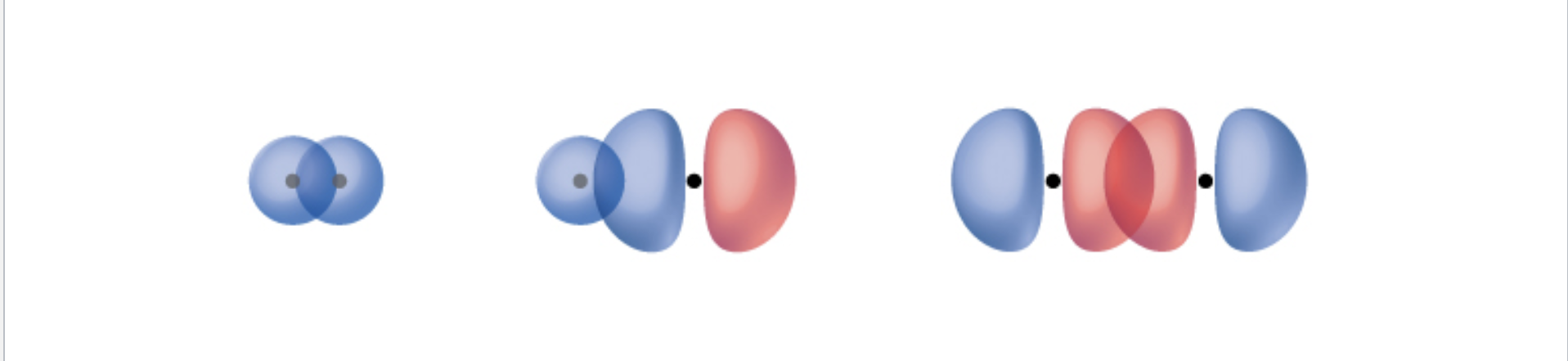
Pi Bond
Type of overlap of atomic orbitals
Happens when side by side p orbitals overlap
Bonds happen above and below the bonding axis
Seen as multiple bonds in lewis structures
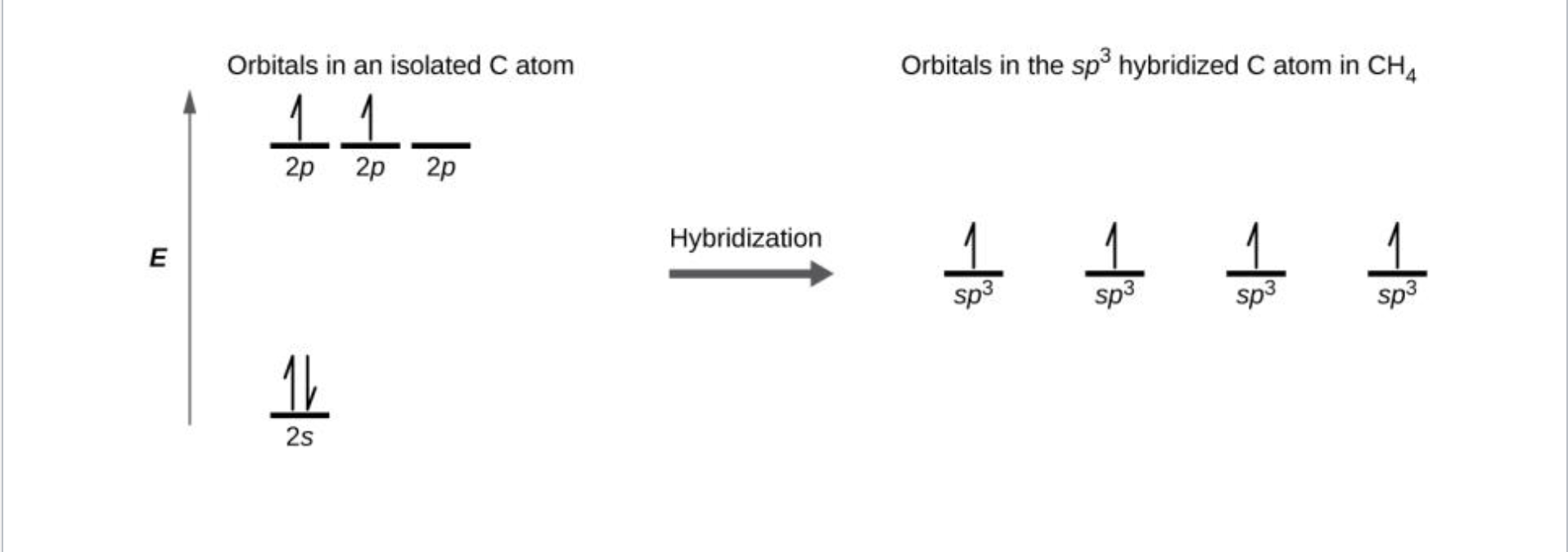
Hybrid Orbitals
S and P orbitals combine
- Notated as SP^x with x being the number of P orbitals that needed to be used
Done when multiple bonds are needed
- 1 Bond for every pair used
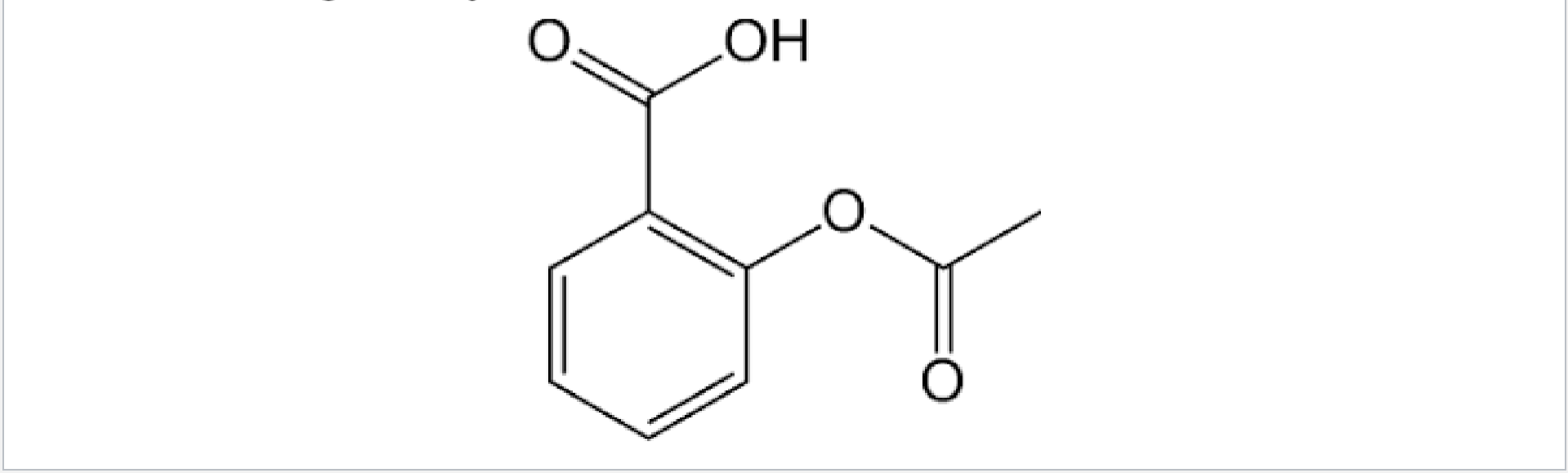
Lewis Structure Short Hand
Used in large organic models
C and H aren’t draw
- C is implied to be between bonds and at the end of bonds
- Every C is given an octet by adding as many H to it as necessary
Paramagnetism
Weak attractions form to externally applied magnetic force
Caused by unpaired electrons that exist in molecule
Diamagnetic
Weak repulsion to externally applied magnetic force
Caused by all electrons being paired
Molecular Orbital Theory
Describes electron distribution in a molecule using molecular orbitals
- Molecular orbitals formed through combining linear combinations of atomic orbitals (LCAO)