QA IN HEMATOLOGY
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
Laboratory Error
An occurrence or an event that has a negative impact on the laboratory which includes the personnel, patient results, machine, or the environment.
46 to 68%
Error Percentage of Pre-Analytical Phase
7 to 13%
Error Percentage of Analytical Phase
19 to 47%
Error Percentage of Post-Analytical Phase
Quality Management
This ensures the consistent delivery of accurate, reliable, and high-quality services.
Quality Assurance
Refers to the overall monitoring of the program that ensures that the final result reported by the laboratory is as correct and as timely as possible.
Quality Control
One component of the program includes standards for specimen collection, instrument maintenance, personnel training and competency, safety, data.
Designed to detect and correct deficiencies in the analytical process of the laboratory prior the release of the patient result, to ensure the quality of the work before it is reported.
Establish SOP
Define SOS
Outline QMS
Quality Planning
Regular Internal Audits
Proficiency Testing
Participation in External Quality Assessment Schemes
Quality Assurance
Use of Control Materials
to monitor equipment performance, verify test accuracy, and detect potential issues during testing.
Quality Control
Review Test Results
Identify Outliers
Implement Corrective or Preventative Actions
Quality Improvement
Clinical Laboratory Improvement Amendments [CLIA]
A set of regulatory standards in the United States that govern clinical laboratories like hematology.
Food and Drug Administration [FDA]
A federal agency responsible for regulating medical products, including in-vitro diagnostics and medical devices used in hematology.
International Organization Standard [ISO]
An independent, non-governmental international organization that develops and publishes standards to ensure quality, safety, and efficiency of products and systems.
ISO 15189
ISO that provides a framework for quality management systems in hematology laboratories.
National Accreditation Board for Testing and Calibration Laboratory [NABL]
An Indian organization responsible for assessing and accrediting laboratories based on their ability to produce accurate and reliable test results.
College of American Pathologists [CAP]
A professional organization for pathologists and laboratory professionals.
Department of Health [DOH]
The primary government agency responsible for promoting and protecting the health and well-being of Filipinos.
Philippine Society of Pathologists [PSP]
The counterpart of CAP in the Philippines. A professional organization for pathologists.
Philippine Society of Hematology and Blood Transfusion [PSHBT]
A professional organization that focuses on hematology and blood transfusion in the Philippines.
Philippine Association of Blood Banks [PABB]
A non-profit organization composed of blood banks and blood centers.
Professional Regulation Commission [PRC]
A government agency responsible for regulating and licensing professionals in the Philippines.
Sensitivity
Specificity
Predictive Value
Accuracy
Precision/Reproducibility
Practicality
Reliability
7 Parameters of Quality Control
Sensitivity
Ability of the test to detect even the smallest amount or concentration of analyte in a sample.
Analytical Sensitivity
This measures the minute concentrations of an analyte.
Diagnostic Sensitivity
This test must always give a positive result in the presence of a disease; True Positive.
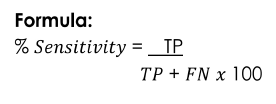
Formula for Se[N]sitivity
Specificity
Ability of a test or a method to measure only the specific analyte of interest without the interferences of other substances present in the sample.
Analytical Specificity
This measures only one unknown substance of interest.
Diagnostic Specificity
This gives a negative result in the absence of the disease; True Negative.
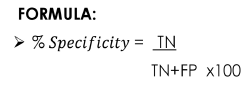
Formula for S[P]ecificity
Positive Predictive Value [PPV]
The probability that subjects or patients with positive screening tests truly harbor the disease.
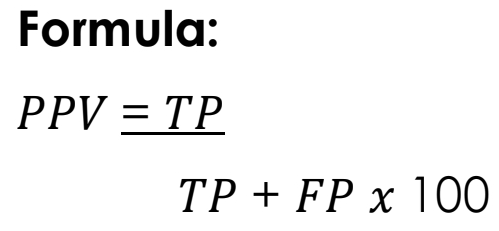
PPV Formula
Negative Predictive Value [NPV]
The probability that subjects or patients with a negative screening test truly do not have the disease.
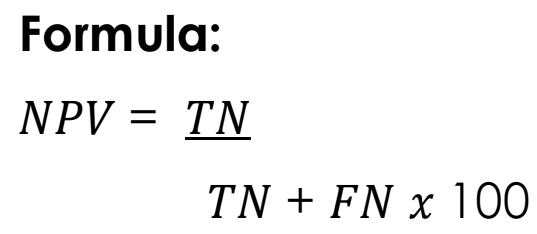
NPV Formula
Accuracy
How close or how near you are to the test value.
Precision / Reproducibility
The ability to producce the same results.
Reliability
The ability of an analytical method to maintain accuracy (valid) and precision (consistent) over an extended period of time.
Practicality
A degree by which a method could be easily repeated.
Internal Quality Control
● Involves running known samples with anticipated results to monitor the accuracy and precision of testing procedures
● Internal Quality Control Materials can either be included in the test kits or third party controls
● Control samples can be lyophilized or powdered form and can be diluted by distilled water.
External Quality Control
● Also called PROFICIENCY TESTING
● Participating in proficiency testing programs allows laboratories to assess their performance against established standards
National Kidney and Transplant Institute [NKTI]
They’re the ones who conduct the proficiency testing here in the Philippines; Hematology NRL.
Dr. James Westgard
Developed Westgard Multi-Rule System
Control Rules
Category of Rules that will require an immediate corrective action if the analytical process is out of control.
Warning Rules
Category of Rules that will give us a clue of a potential issue that should be investigated further.
1 cv outside 2SD
Random Error
Accept [Warning Rule]
1 2s Description, Type of Error, and Action
1 cv outside 3SD
Random Error
Reject
1 3s Description, Type of Error, and Action
2 consecutive control value outside 2SD on the same side
Systematic Error
Reject
2 2s Description, Type of Error, and Action
2 Consecutive control values outside 2SD on opposite sides.
Random Error
Reject
R 4s Description, Type of Error, and Action
4 consecutive control values outside 1SD on the same side.
Systematic Error
Reject
4 1s Description, Type of Error, and Action
10 consecutive control values on the same side of the mean.
Systematic Error
Reject
10x Description, Type of Error, and Action