Unit D - Energy & Rates
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
How does how effectively transformation energy affect human civilization.
Our ability to harness energy has greatly, contributed to the development of human civilization, but how efficiently we transform energy determines the sustainability and
effectiveness of those advancements.
Thermochemistry
The study of energy changes during physical or chemical changes in matter is called
Energy
the ability to do work (Joules or J)
Work
the amount of energy transferred by a force over a distance (J)
All forms of energy can be classified as either…
kinetic energy or potential energy
Potential energy:
the energy of a body or system due to its position or composition:
- Think of this as stored energy, like water at the top
of a dam,
- or energy stored in a battery
- Or energy stored between atoms in their bonds
Kinetic energy
the energy of an object due to its motion
thermal energy
The total quantity of potential energy of a substance
Heat
The transfer of thermal energy from a warm object to a cooler object.
- When you heat water in a kettle, you transfer thermal energy from the burner to the kettle on the water.
- When you cool an object such as letting the kettle rest, the energy is transferred to its surroundings.
Temperature
a measure of the average kinetic energy of entities in a substance
Temperatures relation to kinetic energy
The higher the temperature the higher the kinetic energy
Law of Conservation of Energy, What does is tell us about Chemical Systems?
Energy cannot be created or destroyed, It just changes from.
From the law of conservation fo energy, you know that the total energy in a chemical system and its surrounds must be the same before and after the reaction
Chemical System
a group of reactants and products being studied
Surrounding
All the matter that is not part of the system
Types of Reactions
Exothermic
Endothermic
Exothermic
releasing energy to the surroundings
Eng on product side
In a chemical reaction, if more energy is released from the formation fo new bonds in the products than is required to break bonds in the reactants, then some energy is released to the surroundings
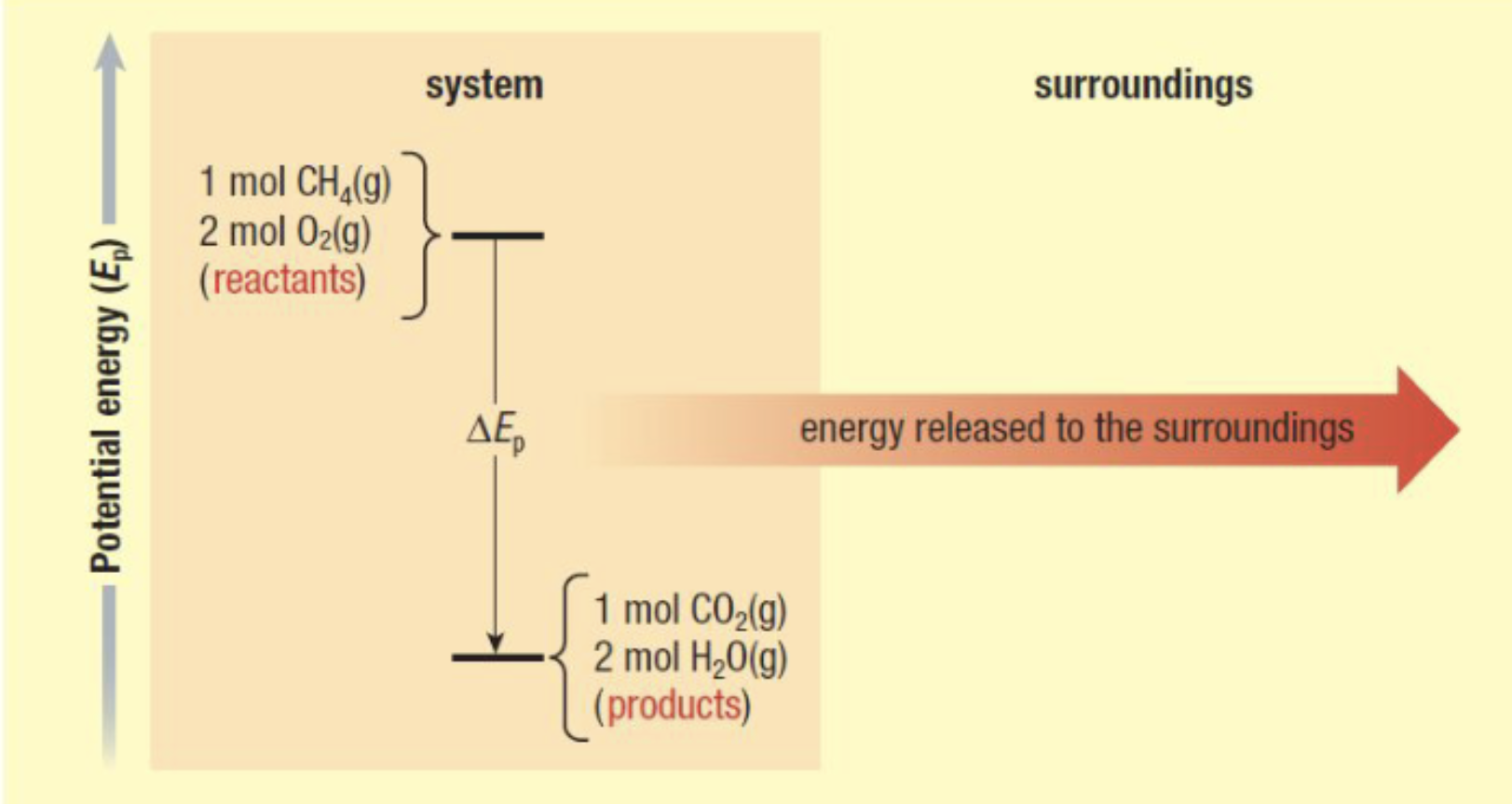
Endothermic or Exothermic?
Exothermic
Endothermic
Absorbing energy from the surroundings
Energy on reactant side
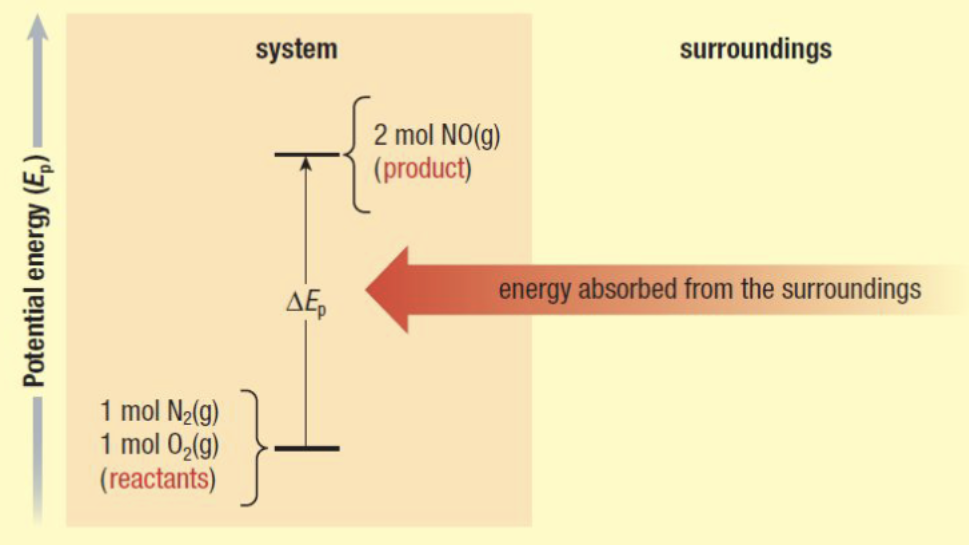
Endothermic or Exothermic?
Endothermic
Nuclear Energy (3)
all nuclear reactions are exothermic.
Per unit of mass, nuclear reactions release much more energy than standard exothermic chemical reactions.
There a two types of nuclear reactions: Fusion & Fission
Fusion (3)
the process of combining two or more nuclei of low atomic mass to form a heavier, more stable nucleus
Currently the sun is undergoing this kind of fusion reaction making helium. As planets get older they will start fusing heavier elements. Hydrogen > Helium > Lithium etc
Fusion reactions are extremely energy efficient
Fission
the process of using a neutron to split a nucleus of high atomic mass into two nuclei with smaller masses
Fusion reactions are 3-4x more efficient than fission reactions
Fusion is 4 million times more efficient than gas
Specific heat capacity
(c) The quantity of thermalenergy required to raise the temperature of 1g of substance by 1C; J/(g.C)
- In simple terms it tells you how resistant a material is to changing temperature. How “stubborn” the substance is.
Lower specific heat → ______ resistant to change
Less
Higher specific heat→ ______ resistant to change
more
Specific heat capacity (Water)
4.18 J/g.c
What do scientists use calorimetry for?
study the thermal energy transfer
Calorimetry
the process of measuring energy changes during physical/chemical changes
Calorimeter
a device that is used to measure thermal energy change in physical/chemical changes
specific heat capacity formula or the heat energy formula.
Q=MCT
q: the total amount of thermal energy absorbed or released by a chemical system (J)
m: mass in grams
c: specific heat capacity (J/g x C)
ΔT: change in temperature (C)
ΔT = Tfinal - Tinitial
Enthalpy (H)
The total amount of thermal energy in a substance/system
Enthalpy Change (ΔH):
The energy released to or absorbed from the surroundings during a chemical or physical change
Enthalpy change represents the heat energy transferred into or out of a chemical system during a reaction.
H is neg (exothermic)
H is pos (endothermic)
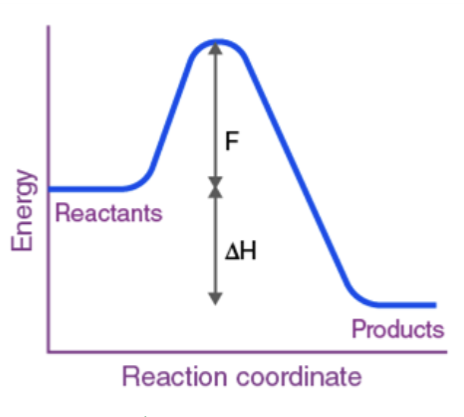
Endothermic or Exothermic?
Exothermic
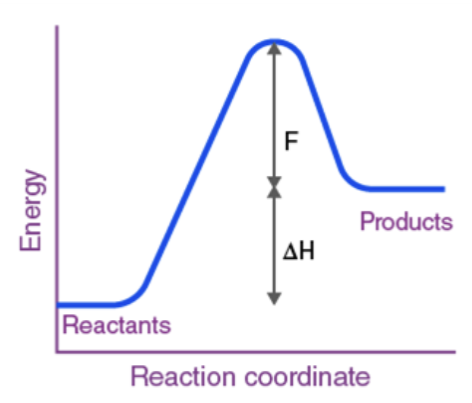
Endothermic or Exothermic?
Endothermic
Change in Hr
ΔHr: The enthalpy change associated with a physical, chemical, or nuclear change involving 1 mol of a substance, J/mol
We use this to determine how much energy might be produced or absorbed in a given chemical reaction
Change in Hr for solution, combustion, vaporization, formation, neutralization
Hsol
Hc
Hvap
Hf
Hneut
Always set to one mol
total enthalpy change
ΔH=nΔHr
ΔH = total enthalpy change of the reaction (kJ)
n = number of moles (mol)
ΔHr = molar enthalpy change (enthalpy change per mole of reaction) (kJ/mol)
ΔHsystem = −qsurroundings
It shows that heat energy is conserved. The heat lost by the system is gained by the surroundings (and vice versa). If the system releases heat, the surroundings absorb the same amount of heat, and if the system absorbs heat, the surroundings lose it.
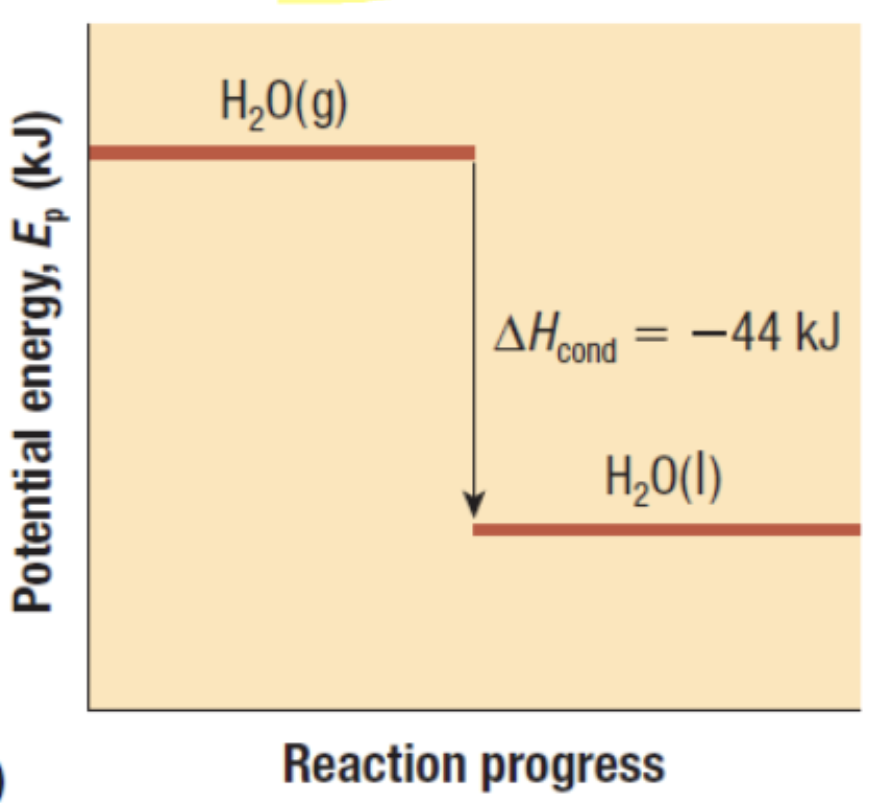
Exothermic or Endothermic?
Exothermic
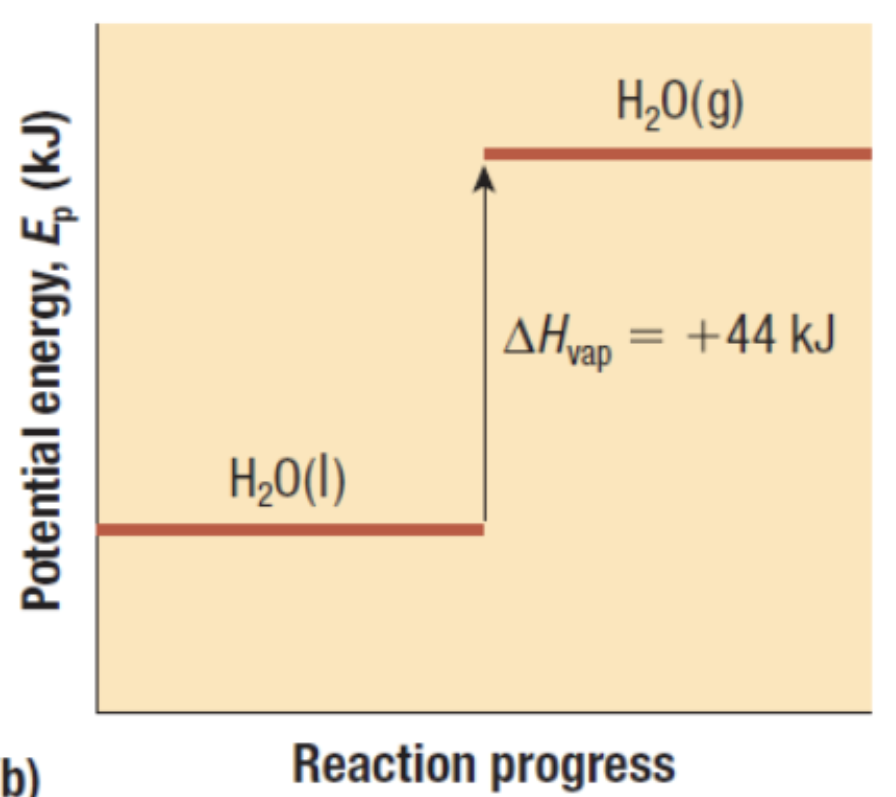
Exothermic or Endothermic?
Endothermic
How do we know how much energy will be released in a molecule?
A covalent bond between 2 atoms will break if enough energy is supplied
bond dissociation energy
The quantity of energy required to break a chemical bond
They all have positive values
- All for 1 mol
(kJ/mol)
Bond dissociation energy for C = O
799 kJ/mol
Bond Energies
Bond energies can be used to calculate approximate enthalpy changes
ΔH=∑Ebonds broken−∑Ebonds formed
Bonds broken → energy is absorbed (positive)
Bonds formed → energy is released (negative)
D is the bond energy taken from the tables
Hess’s Law
The enthalpy change for the conversion of reactants to products is the same whether the conversion occurs in one step or several steps
Standard Enthalpy of Formation (ΔHf)
The Standard Enthalpy of Formation (ΔHfO) of a compound is the change in enthalpy that occurs when 1 mol of compound is formed from its elements, with all substances in their standard states. (kJ/mol)
Standard State
The most stable form of a substance under standard conditions 25oC and 100kPa
At Standard State… (3)
HOFNCl and Nobles gases are gases
Mercury and Bromine are liquids
All others are solids
Chemical Kinetics
the branch of chemistry that deals with rates of reactions
what is the reaction rate of a chemical reaction
The reaction rate of a chemical reaction is the change in concentration of a reactant or product per unit of time
The stems from the concept that different reactions have different speeds.
How do chemists measure reaction rates if there is no “speedometer” for reactions?
Chemists measure reaction rates by tracking how fast reactants are used up or products are formed over time. This is similar to calculating average speed (change in amount ÷ time). Because reactions don’t have a clear “end point,” scientists measure changes over time to determine the average rate of reaction.
Average Reaction Rate
the change of reactant or product concentration over a given time interval
rate A = Conentration of A at time t2 - concentration of A at time 1 / t2- - t1
you would always want a positive reaction rate
rate a = change in (A)/change in T
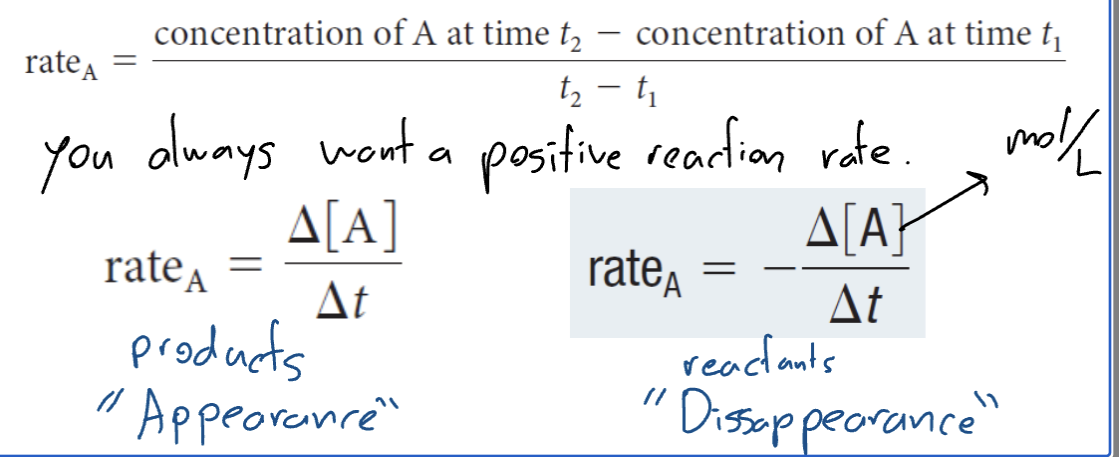
Determining Average Reaction Rate using Graphical Data
change in y (concentration) /change in x (time)
(rise over run)
Factors Affecting Reaction Rates
Chemical Properties
Concentration of reactants
Surface Area
Temperature
Presence of a catalyst
Catalyst
a substance that alters the rate of a chemical reaction without itself being permanently changed
Why are reaction rates important in industry, and what role do catalysts play?
Reaction rates are important in industry because they make manufacturing processes faster, cheaper, and more efficient. Catalysts are substances that speed up reactions without being used up. They are used in real-life applications like catalytic converters and in food production (e.g., blue cheese).
Collision Theory
Collision Theory states that chemical reactions can occur only if reactants collide with proper
orientation and with enough kinetic energy. (activation energy)
Enough to break the bonds and form products By adjusting our 5 factors of reaction rate, we are
increasing the amount of collisions that can occur making the reaction happen
“Enough kinetic energy” → Activation Energy
The minimum energy that reactant molecules must possess for a reaction to be successful.(Ea)
Think of the activation energy as a hill or barrier that the reactants must undergo to perform the reaction (hump on graph)
Temperature of the Reaction System
When temperature is increased, there is higher kinetic energy in the system, thus a higher likelihood of collisions
Faster reaction rate
Concentration affect on the rate of reaction:
As concentration increases → higher likelihood of collisions → faster reaction
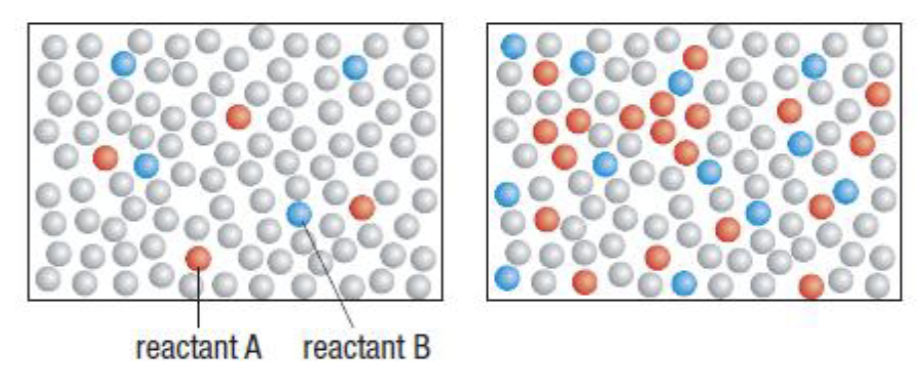
Where would the reaction be slower, wb faster? Why?
slow, fast - concentration because there are more particles in the same volume.
Surface Area affect on the rate of reaction:
As Surface Area increases → more opportunities for collisions → faster reaction
Two types of Catalyst:
Heterogeneous Catalyst
Homogeneous Catalyst
Heterogeneous Catalyst
a catalyst in a reaction in which the reactants and the catalyst are in different physical states
Homogeneous Catalyst
a catalyst in a reaction in which the reactants and the catalysts are in the same physical state
How do catalysts work
For any reaction to occur, the kinetic energy of colliding reactant entities must be equal to or greater than the activation energy. However, Catalysts don’t affect either of these things
Catalysts provides an alternative pathway for the reaction, which has a lower activation energy
Biological catalysts
A catalyst made by a living system, usually an enzyme
- eg) lactase to break down the sugars in milk products for someone who is lactose intolerant
- To speed up fermentation of alcohols, cheeses, yogurts etc