T&F Systems Test #1 (practice and official T1 solution)
1/29
Earn XP
Description and Tags
Lectures 1-12
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
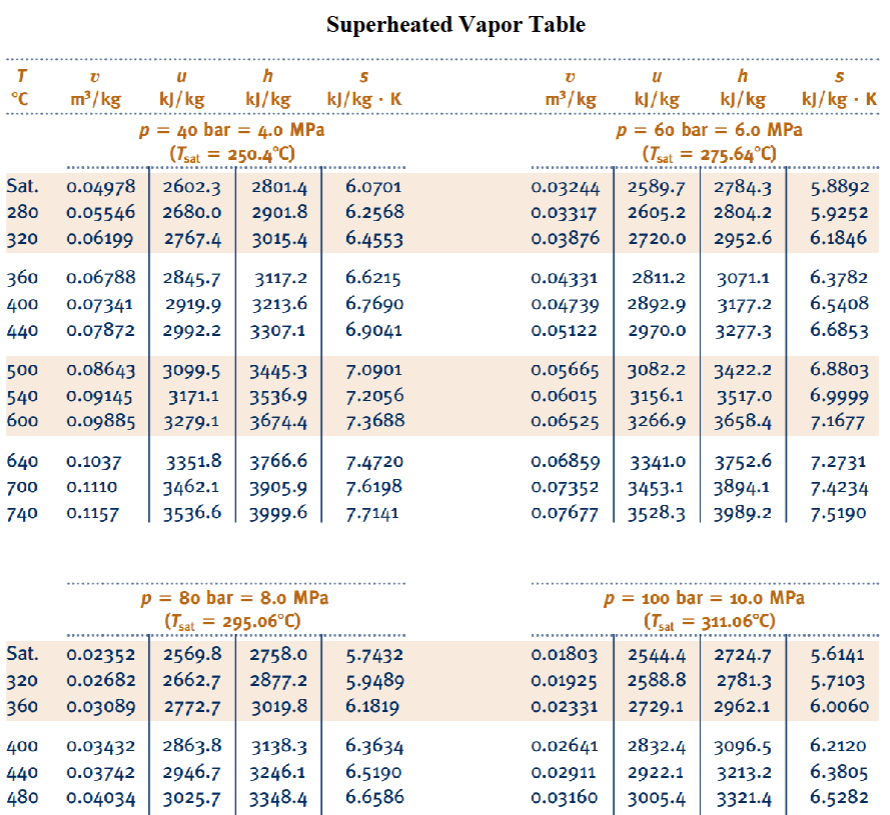
Select all intensive properties.
volume
quality
internal energy
pressure
energy
boiling point
temperature
density
enthalpy
specific enthalpy
concentration
mass
specific volume
material hardness
area
quality
pressure
boiling point
temperature
density
specific enthalpy
specific volume
concentration
material hardness
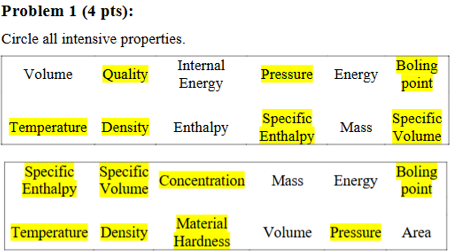
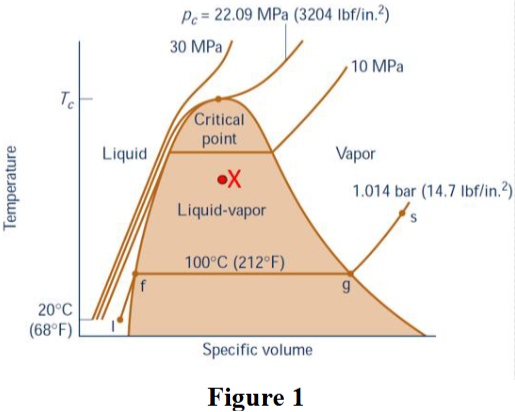
Figure 1 shows a typical water T-v phase diagram. If a system is moving from point f to g, select all equilibrium states during this process.
Thermal equilibrium
Mechanical equilibrium
Phase equilibrium
Chemical equilibrium
Thermal equilibrium, Mechanical equilibrium, and Chemical equilibrium
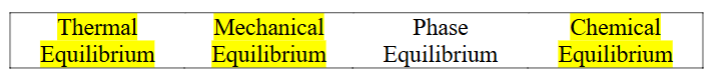
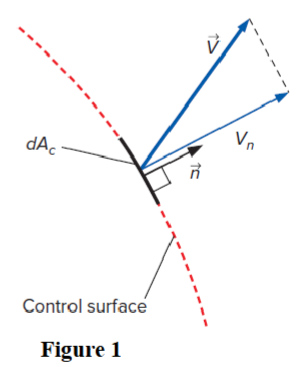
Figure 1 shows a control surface of a control volume. What is the equation calculating the mass flow rate (δm) in or out of this differential surface dAc:
δm = (p)(Vn)(dAc)
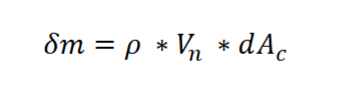
Figure 1 shows a typical water T-v phase diagram. If an isolated system locates at point X, select all the equilibrium states this system has.
Thermal equilibrium
Mechanical equilibrium
Phase equilibrium
Chemical equilibrium
Thermal equilibrium, Mechanical equilibrium, Phase equilibrium, and Chemical equilibrium

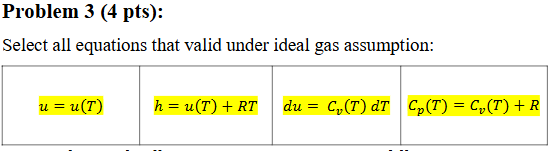
Select all equations that are valid under the ideal gas assumption:
u = u(T)
h = u(T) + RT
du = Cv(T) dT
Cp(T) = Cv(T) + R
All of the above
In the first law of thermodynamics, heat transfer, and work depend on the end state and the path it follows. Therefore, they are path functions.
What is the mathematical difference between a path function and a point function?
Path Function has no exact differential and a point function has an exact differential
What is the mathematical definition of steady state and uniform conditions?
δ(any properties)/δ(t) = 0, δ(any properties)/δ(X,Y,Z) = 0

Write down the definition for the state postulate.
A simple compressible system completely specified by two independent intensive properties.
T/F: A system is considered simple compressible when in absence of electrical, magnetic, gravitational, motion and surface tension effects.
True
Select all substance(s) that are pure substance(s).
Air
Water
Mixture of ice and water
Mixture of oil and water
Air, water, and mixture of ice and water
![<p>A u-tube pressure measuring device that has six different mediums is shown in Figure.2. Determine the pressure difference in [Pa] between points A and B.</p><p>𝜌<sub>𝐵𝑒𝑛𝑧𝑒𝑛𝑒</sub> = 870 𝑘𝑔/𝑚<sup>3</sup></p><p>𝜌<sub>𝑀𝑒𝑟𝑐𝑢𝑟𝑦</sub> = 13500 𝑘𝑔/𝑚<sup>3</sup></p><p>𝜌<sub>𝑊𝑎𝑡𝑒𝑟</sub> = 1000 𝑘𝑔/𝑚<sup>3</sup></p><p>𝜌<sub>𝐾𝑒𝑟𝑜𝑠𝑒𝑛𝑒</sub> = 820 𝑘𝑔/𝑚<sup>3</sup></p><p>𝜌<sub>𝑂𝑖𝑙</sub> = 600 𝑘𝑔/𝑚<sup>3</sup></p><p>𝑔 = 9.8 𝑚/𝑠<sup>2</sup></p><p>Neglect gravitational force for air.</p>](https://knowt-user-attachments.s3.amazonaws.com/e5112909-4568-43e5-88ef-d19cfe1f10d4.png)
A u-tube pressure measuring device that has six different mediums is shown in Figure.2. Determine the pressure difference in [Pa] between points A and B.
𝜌𝐵𝑒𝑛𝑧𝑒𝑛𝑒 = 870 𝑘𝑔/𝑚3
𝜌𝑀𝑒𝑟𝑐𝑢𝑟𝑦 = 13500 𝑘𝑔/𝑚3
𝜌𝑊𝑎𝑡𝑒𝑟 = 1000 𝑘𝑔/𝑚3
𝜌𝐾𝑒𝑟𝑜𝑠𝑒𝑛𝑒 = 820 𝑘𝑔/𝑚3
𝜌𝑂𝑖𝑙 = 600 𝑘𝑔/𝑚3
𝑔 = 9.8 𝑚/𝑠2
Neglect gravitational force for air.
PA-PB = 10470.32 Pa

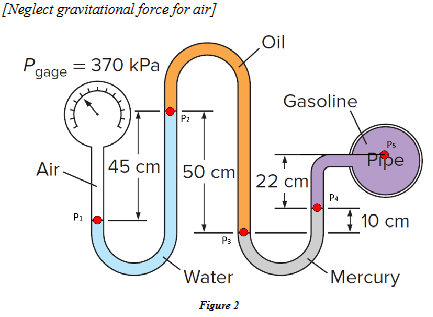
A gasoline line is connected to a pressure gage through a double-U manometer, as shown in Fig.2. If the reading of the pressure gage is 370 kPa, determine the gage pressure at five positions (P1, P2, P3, P4, P5). Neglect gravitational force for air.
𝜌𝑀𝑒𝑟𝑐𝑢𝑟𝑦 = 13500 𝑘𝑔/𝑚3
𝜌𝑊𝑎𝑡𝑒𝑟 = 1000 𝑘𝑔/𝑚3
𝜌𝑂𝑖𝑙 = 600 𝑘𝑔/𝑚3
𝜌Gasoline = 750 𝑘𝑔/𝑚3
𝑔 = 9.8 𝑚/𝑠2
Hint: ΔP = -pgΔZ
𝑃1= 370 𝑘𝑃𝑎, 𝑃2= 365.6 𝑘𝑃𝑎, 𝑃3= 368.5 𝑘𝑃𝑎, 𝑃4= 355.25 𝑘𝑃𝑎, and 𝑃5= 353.63 𝑘𝑃𝑎

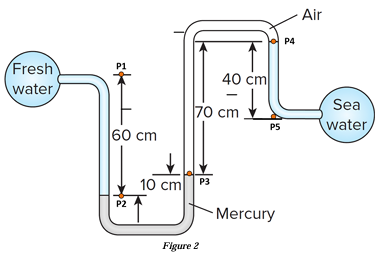
Freshwater and seawater flowing in parallel horizontal pipelines are connected to each other by a double U-tube manometer, as shown in Fig.2. Determine the pressure difference (𝑃1 − 𝑃5) between the two pipe centerlines.
𝜌Saltwater = 13500 𝑘𝑔/𝑚3
𝜌𝑀𝑒𝑟𝑐𝑢𝑟𝑦 = 13500 𝑘𝑔/𝑚3
𝜌Water = 13500 𝑘𝑔/𝑚3
𝜌Air = 13500 𝑘𝑔/𝑚3
g = 9.8 m/s2
Hint: ΔP = -pgΔZ
P1-P5 = 3300.35 Pa (Similar to the attached SS but diff. #’s)

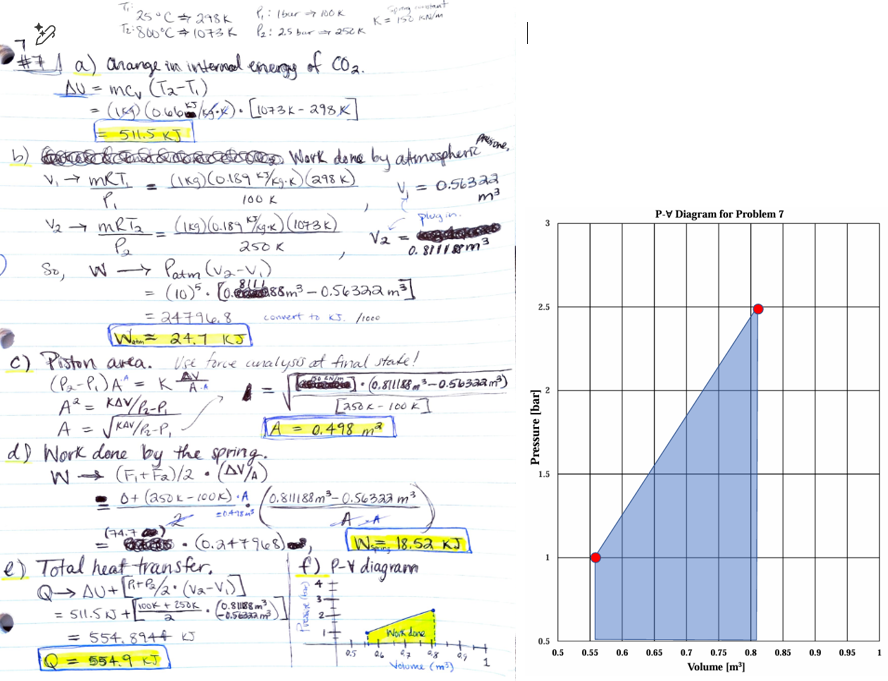
![<p>A linear spring-loaded piston-cylinder device contains 1 kg of ideal carbon dioxide. This system is slowly heated from 1 Bar and 25°C to 2.5 Bar and 800°C. Assume the spring is initially hanged with no force and neglects the piston kinetic, internal, and potential energy, and the spring constant is 150 kN/m.</p><p>(a). Determine the internal energy change of the CO<sub>2</sub> (10 pts)</p><p>(b). Determine the work done by atmosphere pressure (5 pts)</p><p>(c). Determine the piston area (5 pts)</p><p><strong><em>Hint: Do a force analysis at the final state to calculate the piston area.</em></strong></p><p>(d). Determine the work done by spring (5 pts)</p><p>(e). Determine the total heat transfer to this system. (10 pts)</p><p>(f). Extra Credit (15 pts), Sketch this entire process on a P-∀ diagram in Fig.4 and highlight the total work during this process.</p><p></p><p><em>[Carbon dioxide has a constant specific heat 𝐶<sub>𝑣</sub> = 0.66 𝑘𝐽/𝑘𝑔∗𝐾, 𝑅<sub>CO2</sub> =0.189 𝑘𝐽/𝑘𝑔∗𝐾, and the atmospheric pressure is 1 bar.]</em><strong><em> Use Appendix A&B for reference.</em></strong></p>](https://knowt-user-attachments.s3.amazonaws.com/919574f8-9f76-489c-9b44-1b566a0c798b.png)
A linear spring-loaded piston-cylinder device contains 1 kg of ideal carbon dioxide. This system is slowly heated from 1 Bar and 25°C to 2.5 Bar and 800°C. Assume the spring is initially hanged with no force and neglects the piston kinetic, internal, and potential energy, and the spring constant is 150 kN/m.
(a). Determine the internal energy change of the CO2 (10 pts)
(b). Determine the work done by atmosphere pressure (5 pts)
(c). Determine the piston area (5 pts)
Hint: Do a force analysis at the final state to calculate the piston area.
(d). Determine the work done by spring (5 pts)
(e). Determine the total heat transfer to this system. (10 pts)
(f). Extra Credit (15 pts), Sketch this entire process on a P-∀ diagram in Fig.4 and highlight the total work during this process.
[Carbon dioxide has a constant specific heat 𝐶𝑣 = 0.66 𝑘𝐽/𝑘𝑔∗𝐾, 𝑅CO2 =0.189 𝑘𝐽/𝑘𝑔∗𝐾, and the atmospheric pressure is 1 bar.] Use Appendix A&B for reference.
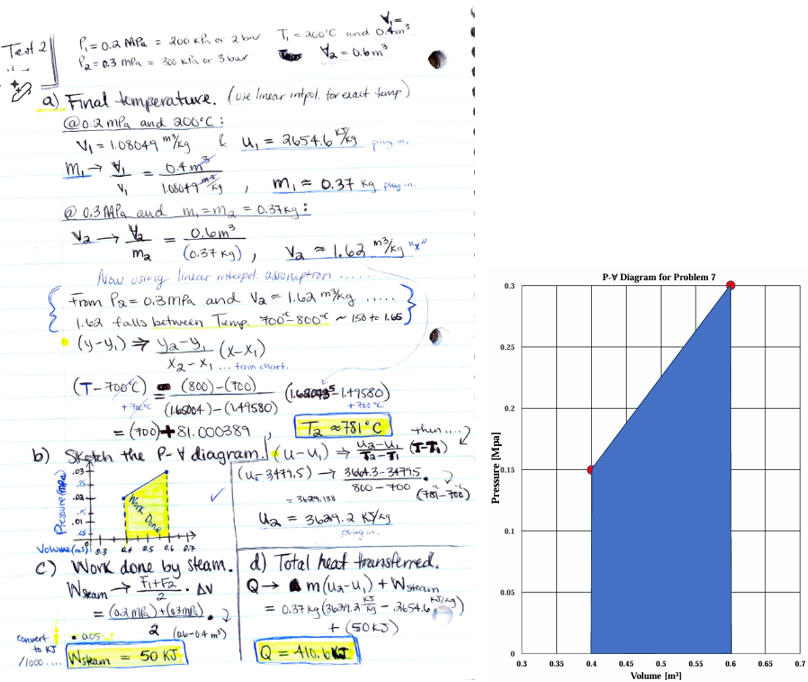
![<p>A piston–cylinder device initially contains superheated vapor at 0.2 MPa, 200°C, and 0.4 m3. At this state, a linear spring is touching the piston but exerts no force on it. Heat is now slowly transferred to the steam, causing the pressure and the volume to rise to 0.3MPa and 0.6 m3, respectively.</p><p>(a) The final temperature.</p><p><strong><em>Hint: using linear interpolation to find out the exact temperature</em></strong></p><p>(b) Sketch this process on a P-V Diagram, and highlight the total work.</p><p>(c) The work done by the steam.</p><p><strong><em>Hint: using the P-V diagram to find out the work]</em></strong></p><p>(d) The total heat transferred. </p>](https://knowt-user-attachments.s3.amazonaws.com/c768380b-4941-439a-a544-add78c163d9f.png)
A piston–cylinder device initially contains superheated vapor at 0.2 MPa, 200°C, and 0.4 m3. At this state, a linear spring is touching the piston but exerts no force on it. Heat is now slowly transferred to the steam, causing the pressure and the volume to rise to 0.3MPa and 0.6 m3, respectively.
(a) The final temperature.
Hint: using linear interpolation to find out the exact temperature
(b) Sketch this process on a P-V Diagram, and highlight the total work.
(c) The work done by the steam.
Hint: using the P-V diagram to find out the work]
(d) The total heat transferred.
Which of the following properties is classified as an extensive property?
a. Temp.
b. Pressure
c. Mass
d. Density
C

B

a. -70 KJ
b. 70 KJ
c. 30 KJ
d. -30KJ
C
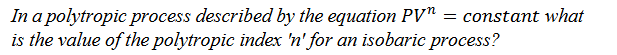
a. + inf.
b. 0
c. 1
d. - inf.
B

B

What is the Zeroth Law of Thermodynamics?
D

D

C

C
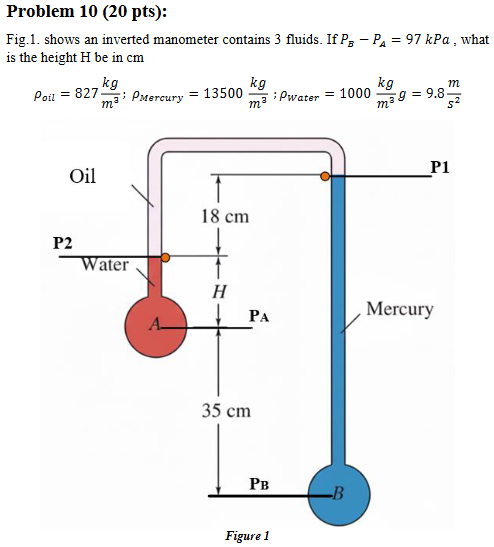
What is the Height (h)?
Hint: 4 steps
Formula: ΔP = -pgΔZ
Water: P2-PA = -g*pwater*(Z2-ZA = ”h”)
Oil: P2-P1 = -g*poil*(Z2-Z1)
Mercury: P1-PB = -g*pmercury*(Z1-ZB)
→ PB-PA= 97 kPa = 97×10² Pa
→ Rearrange setup to find h
h = 23 cm

![<p>a. Initial temp, in K.</p><p>b. Final temp, in K.</p><p>c. Internal energy change, in kJ.</p><p>d. Heat transfer for the process, in kJ.</p><p>e. Plot this process on a <em>P-V</em> diagram (extra credit)</p><p></p><p><strong><em>Hint: 4 steps</em></strong></p><ol><li><p><em>define boundary</em></p></li><li><p><em>assumptions</em></p></li><li><p><em>define properties at the initial state and final state</em></p></li><li><p><em>write the energy balance eq. and evaluate each term [assume a constant pressure process]</em></p></li></ol><p></p>](https://knowt-user-attachments.s3.amazonaws.com/b8288623-46f7-4817-85eb-6bedf7f742f1.png)
a. Initial temp, in K.
b. Final temp, in K.
c. Internal energy change, in kJ.
d. Heat transfer for the process, in kJ.
e. Plot this process on a P-V diagram (extra credit)
Hint: 4 steps
define boundary
assumptions
define properties at the initial state and final state
write the energy balance eq. and evaluate each term [assume a constant pressure process]
a. 337 K
b. 674 K
c. 250 kJ
d. 350 kJ
e. Shaded area [P1=P2= 100 | V1= 1m³ to V2= 2m³]
![<p>a. 337 K</p><p>b. 674 K</p><p>c. 250 kJ</p><p>d. 350 kJ</p><p>e. Shaded area [P1=P2= 100 | V1= 1m³ to V2= 2m³]</p>](https://knowt-user-attachments.s3.amazonaws.com/9c5bd6e4-6d87-4bed-8ade-8dec904c8aae.png)
![<p>a. Avg. velocity at each exit and inlet.</p><p>b. Neglecting the potential energy change and heat transfer from the turbine, what is the work (W<sub>cv</sub>) developed by the turbine?</p><p><strong><em>Hint: 5 steps</em></strong></p><ol><li><p><em>define boundary</em></p></li><li><p><em>write general CV eqs.</em></p></li><li><p><em>assumptions and given conditions</em></p></li><li><p><em>simplify CV eqs. for the boundary containing all systems</em></p></li><li><p><em>evaluate properties at inlets/outlets [use superheated vapor table for T<sub>1</sub>>T<sub>sat|@80bar</sub>…T<sub>2</sub>>T<sub>sat|@60bar</sub>] [saturated mix @position-3]</em></p></li></ol><p></p>](https://knowt-user-attachments.s3.amazonaws.com/513e73bb-d077-4b60-8351-1577287e0fe0.png)
a. Avg. velocity at each exit and inlet.
b. Neglecting the potential energy change and heat transfer from the turbine, what is the work (Wcv) developed by the turbine?
Hint: 5 steps
define boundary
write general CV eqs.
assumptions and given conditions
simplify CV eqs. for the boundary containing all systems
evaluate properties at inlets/outlets [use superheated vapor table for T1>Tsat|@80bar…T2>Tsat|@60bar] [saturated mix @position-3]
a. Vavg1= 40.9 m/s , Vavg2= 20.27 m/s, Vavg3= 101.189 m/s
b. Wcv= 69512 kJ

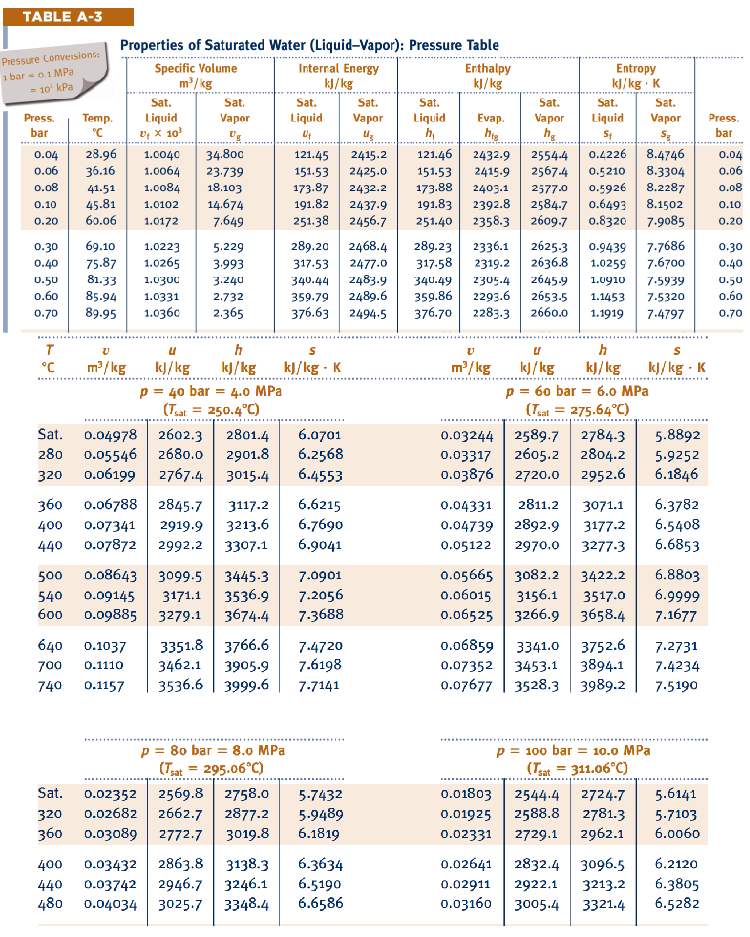
Appendix A. —> Table A-3
Properties of Saturated Water (Liquid-Vapor): Pressure Table
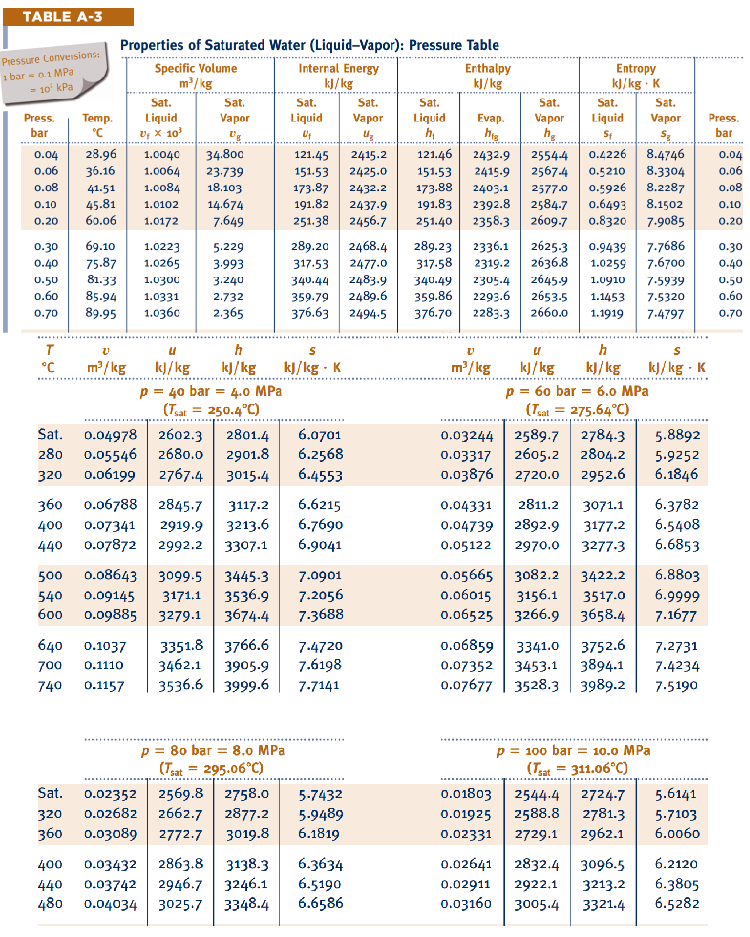
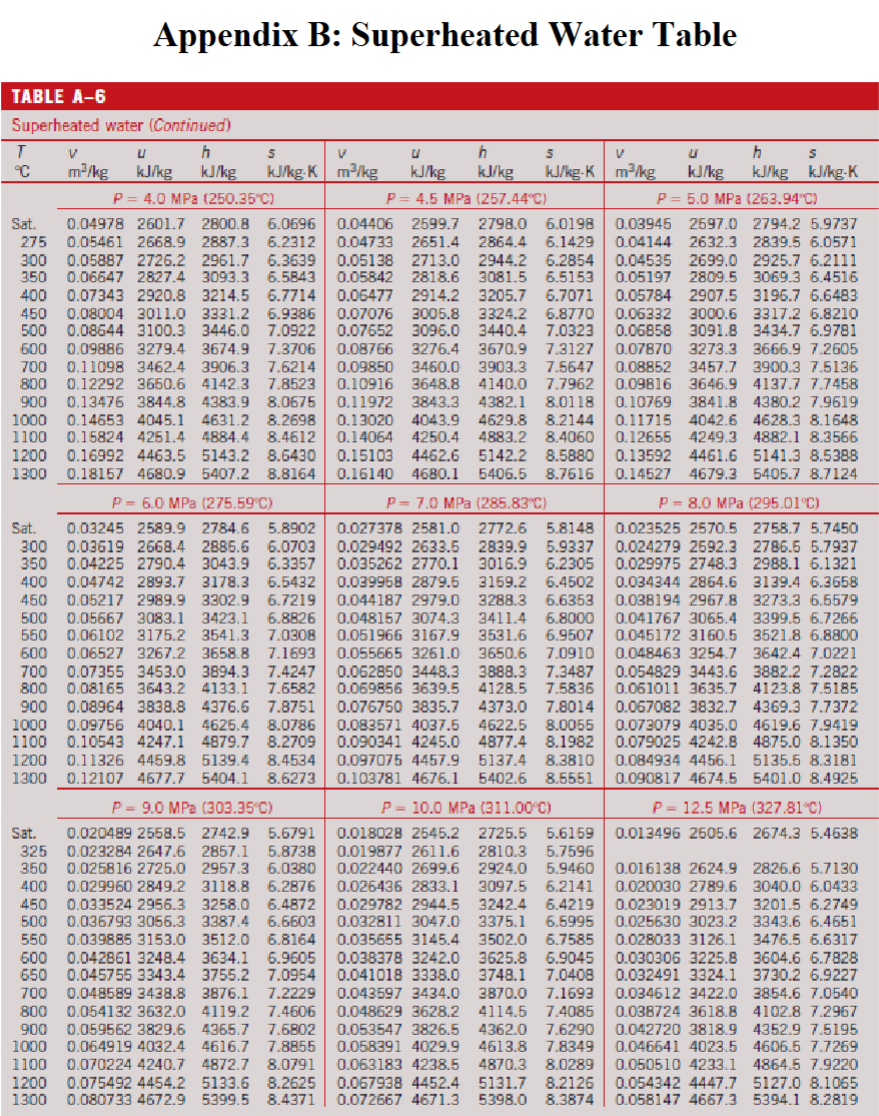
Appendix B. —> Table A-6
Properties of Superheated Water Table
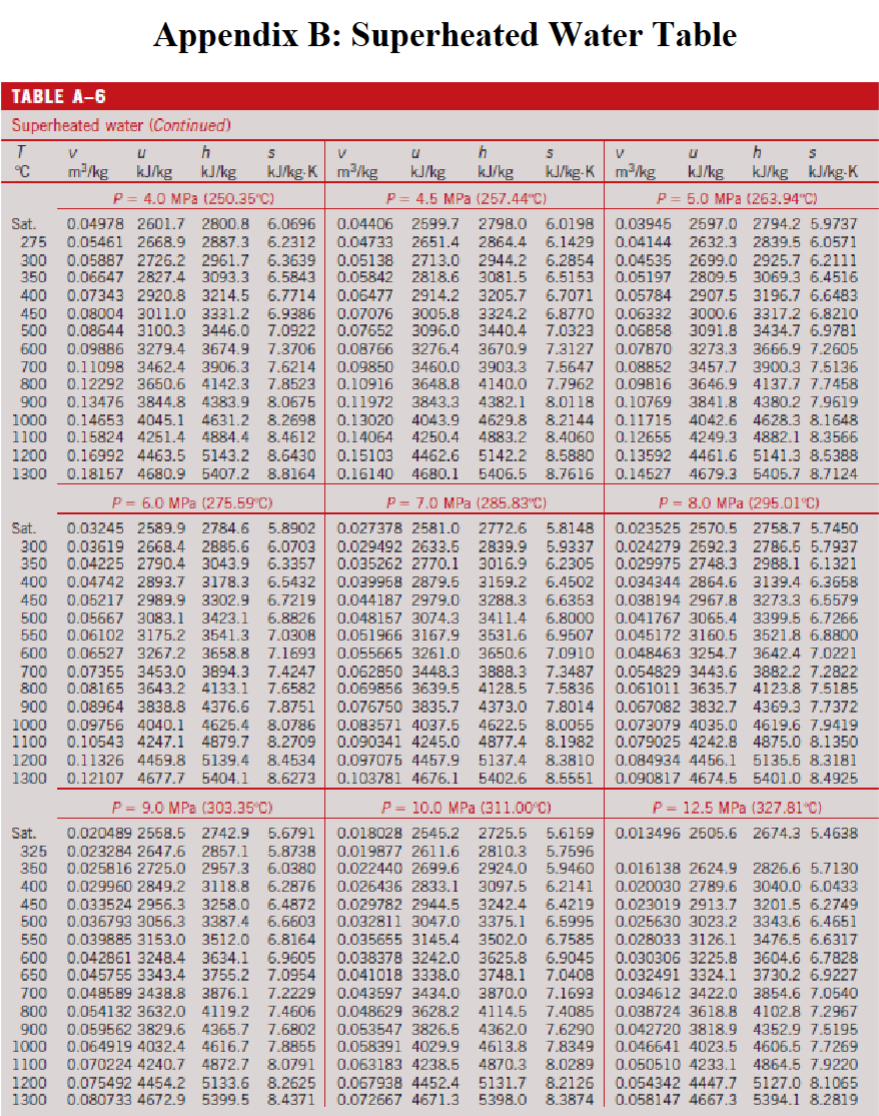
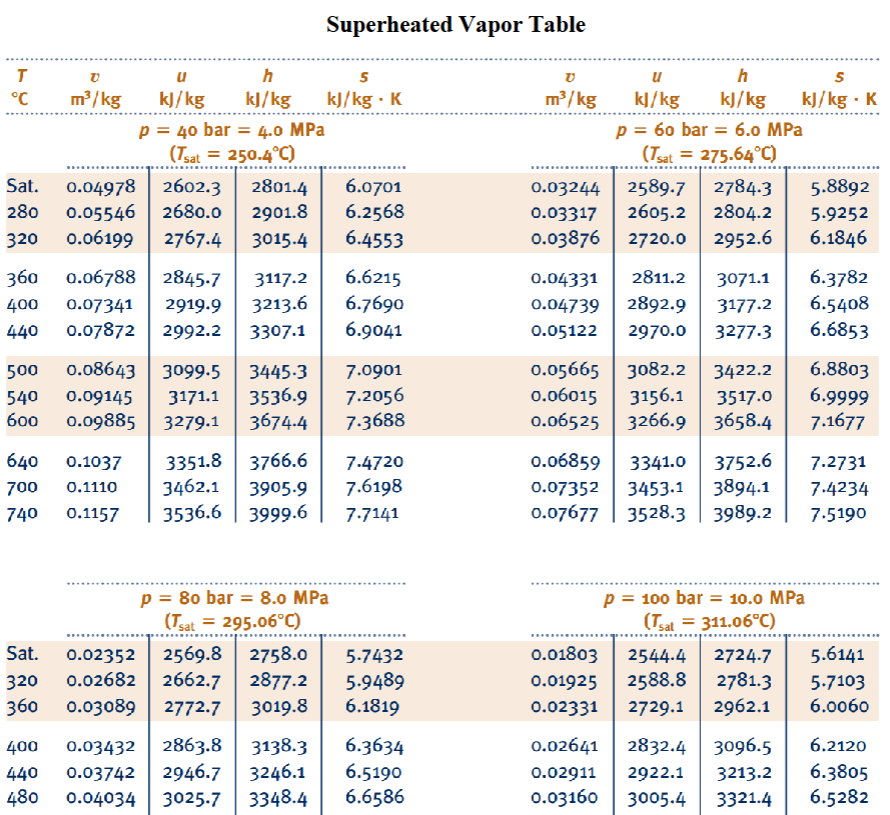
Appendix C. —> Table A-6
Properties of Superheated Vapor Table