Chem Lab 102 Final Review
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Error Analysis:
More precision and replication = reduces uncertainty
Measurement: our attempt to determine AND communicate the “true” value ~ (best estimate) ± (uncertainty)(units) ~ average and standard deviation
Uncertainty: cumulative effect of experimental fluctuations on the data’s precisions
Error: experimental fluctuations that distort the closeness of the measurements to the “true value”
Uncertainty is about how spread out or imprecise a measurement could be, while error is about how far a measurement is from the true value.
Error Analysis: Precision versus accuracy in measurements
Precision: the reproducibility of the measured value using the same experimental conditions
Accuracy: Closeness of the measured value to the “true” value
Error Analysis: Difference between random error (or uncertainty) and systematic error; in general, how they arise, how they affect measurements and successive calculations, and how they may be mitigated and/or identified in experimental procedure
Random Error: Fluctuations that arise from uncontrolled, and often uncontrolled variables
Equal probability of being ± “true”
Detectable by statistics ~ reduced by replication
Examples: Slight fluctuations when reading a meniscus in a buret, Human reaction time when starting/stopping a stopwatch, Small variations in balance readings due to air currents
Systematic Error: Fluctuations that arise from an artifacts or flaw in the experiment (reproducible)
Usually affects the result in one direction
Detectable through experimentation ~ Often difficult to detect
Examples: A balance that is not calibrated and always reads +0.05 g too high, A thermometer that is miscalibrated and consistently reads 2°C too low, Consistently misreading the meniscus from the wrong angle (parallax error in one direction)
Error Analysis: Equal probability of measurement being too high or too low—affects precision
Precision is limited by uncertainty/random error usually resulting from random fluctuations and is described by relative uncertainty
Error Analysis: Measurements repeatedly skewed in one direction across trials—affects accuracy
Accuracy is limited by error usually resulting from systematic fluctuations and is described by relative error (or percent error)
Error Analysis: Calculation of relative error and relative uncertainty
Relative error: measures how far a measured value is from the accepted (true) value, showing accuracy as a fraction or precent
Relative uncertainty: measures the size of the uncertainty compared to the measured value, showing precision as a fraction or precent
(value)/(reference) x 100 = %
Difference: relative error compares to true value, relative compares to measured value
Error Analysis: Labflow
Systematic: faulty equipment or miscalibration and ONE direction VS. different directions
Systematic: accuracy VS Random: precision
———————————————————————————————————————————
Percent Error: (|measured - true| / true) x100
Standard deviation:
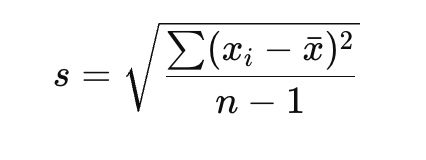
Error Analysis: Possible sources of random or systematic error in the 102L experiments you performed (THR, COL, RK1-2, EQB, ST1-3)
Thermochemistry: Manual
Thermochemistry: the study of the heat energy involved in chemical reactions and changes of physical state
Bond FORMATION: exothermic VS. Bond FORMING: endothermic
First law of thermodynamics: total energy of universe must remain constant
Units of thermochemistry: Joules / Kilojoules
Calorimetry: (units = Kelvin): using temperature change of a liquid (gaining/losing heat from reaction) to determine the heat change of the reaction
qcal = constant x change in temp
Delta H = qrxn = - (qsoln + qcal)
Thermochemistry: Exothermic vs Endothermic reactions
System releases heat energy, surrounding temp increase = exothermic = - enthalpy
System absorbs heat energy from surrounding, surrounding temp decrease = endothermic
Exothermic: more reactants AND Endothermic: more products
Thermochemistry: Calculation of heat change q using temperature change and specific heat, heat formation, delta Hf, standard heat formation delta*Hf, heat of reaction delta H*rxn
Heat change (q): qsoln=mc(delta)T ~ (mass)(specific heat)(Tfinal-Tinitial)
Specific Heat: amount of energy needed to raise 1 gram of a substance by 1C
Heat of reaction (Hrxn): -qsolution - heat lost by solution = gained by reaction
qrxn = -(qsoln + qcal)
Standard heat of formation: Heat change when one mole of a compound forms from its elements in standard states
Reaction enthalpy: Hf (products) - Hf (reactants)
Thermochemistry: Correct format of molecular equations and net ionic equations
An ionic compound is solid form may be included in a reaction’s net ionic equation if the reaction involves the solid’s dissolution into or precipitation out of its constituent ions
Net ionic equation: split compounds into ions unless solid or liquid and cancel out spectator ions
Thermochemistry: Hess’ law as a state function, implications for thermodynamics of separate reactions which combine multiple smaller processes (I.e. dissolution of solid NaOH into an aqueous solution of HCl)
State function: enthalpy change depends on the initial and final states, not path taken
Have to separate into smaller steps to find total heat of a complex process\
Example: NaOH in HCl system:
NaOH (s) → Na+ (aq) + OH- (aq)
HCl → H+ (aq) + Cl- (aq)
H+(aq) + OH- (aq) → H2O
total heat = sum of all reactions
Thermochemistry: Interpretation of Temp vs. Time calorimetry data graph & trend lines
After mixing point, reaction starts (temp rises = exothermic, temp falls = endothermic)
trend line drawn before and after mixing to
Thermochemistry: Prelab
Exothermic = negative enthalpy change (more products)
Hess's Law states that if a reaction can be written as the sum of two or more other reactions, the enthalpy change (ΔH) for the overall process must be the sum values of the constituent reactions.
enthalpy is a STATE function
NaOH: weigh by difference bc NaOH absorbs water from air, affecting mass
*given values for each ion* then product values - reactant values
Colligative Properties: Proportionality of colligative properties (freezing point depression, boiling point elevation, vapor-pressure lowering, and osmotic pressure) to van’t Hoff factor of solute and molality of solute
Colligative Properties: Calculation of an unknown solute’s molar mass based on its van’t Hoff factor, the known solvent’s given molal-freezing-point-depression constant, and its calculated molality (mols solute/kgs solvent) in solution.
Colligative Properties: Interpretation of a plotted warming or cooling curve to identify freezing point or boiling point
Colligative Properties: Manual
Behavior of a solution (solute and solvent) compared to an individual compound
Solvent is major component, solute is the minor component
Physical properties depend on QUANTITY of solute particles present (not identity)
Properties:
Freezing-point depression
Boiling-point elevation
Vapor-pressure lowering
Osmotic pressure
Molality (m): number of moles of solute per kg of solvent
as m increases, the colligative properties effect increases
Van’t Hoff: number of ions/particles a solute dissociates into solution ~ more ions = stronger
i=1 is a non-electrolyte
Delta Tf = -iKfm
Tf = Tf (solution) - Tf (pure)