A + P Exam 4 Final Learning Objective
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
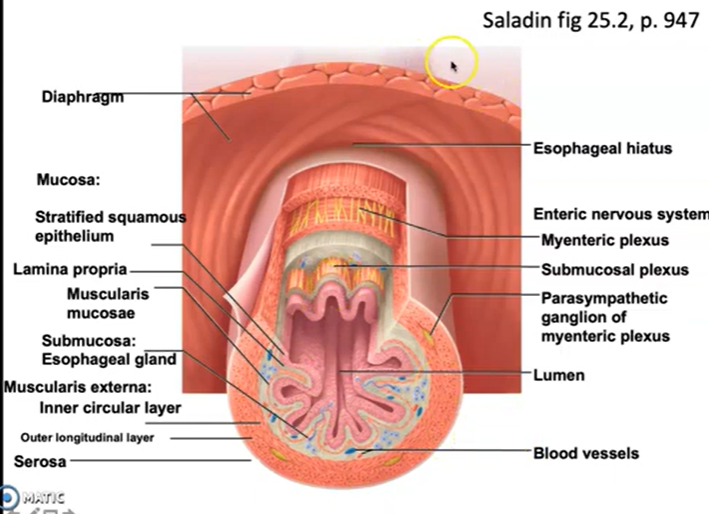
Describe the basic histological plan for the alimentary canal, and modifications that are seen in various regions (stomach, small intestine, large intestine) that facilitate function
Histological Plan of the Alimentary Canal Universal Wall Structure (4 Layers, Innermost → Outermost)
1. Mucosa
Innermost layer; has 3 sub-components:
Epithelium — faces the lumen directly; most functionally variable layer; its type reflects the organ's function and is the most important unifying concept for understanding the digestive system
Lamina propria — thin loose connective tissue beneath the epithelium; contains blood vessels, lymphatics, and immune cells
Muscularis mucosae — thin smooth muscle layer; produces small movements of the mucosal surface to expose epithelium to luminal contents
2. Submucosa
Thick connective tissue layer outside the mucosa
Contains larger blood vessels and lymphatics
Contains Meissner's plexus (submucosal nerve plexus) — part of the enteric nervous system; allows the digestive tract to function semi-independently of the CNS
3. Muscularis Externa
Smooth muscle responsible for propulsion and mixing of food
Standard two layers throughout most of the canal:
Inner circular layer — fibers run circumferentially; contraction narrows the lumen
Outer longitudinal layer — fibers run along the length; contraction shortens the tube
Myenteric plexus (Auerbach's plexus) sits between these two layers; coordinates peristalsis and mixing movements
Together Meissner's and Auerbach's plexuses form the enteric nervous system — the fact that the digestive system has its own dedicated nervous system reflects how critical digestion is to survival
4. Serosa or Adventitia
Serosa — smooth serous membrane (part of the peritoneum); covers organs within the peritoneal cavity; reduces friction as organs move
Adventitia — connective tissue (not a serous membrane); covers organs that are retroperitoneal or outside the abdominal cavity (e.g., esophagus)
The Mouth (Oral Cavity) Epithelium
Lined with non-keratinized stratified squamous epithelium
Same tissue type as the esophagus — reflects the shared function of mechanical protection from abrasion
Multi-layered tissue that withstands the physical forces of chewing, grinding, and food manipulation
Unlike the esophagus, the oral cavity also has specialized regions (tongue, taste buds) but the general lining epithelium throughout is stratified squamous
Function
Mechanical digestion — teeth physically break food into smaller pieces (mastication); tongue manipulates food and forms the bolus
Chemical digestion begins here:
Salivary amylase secreted by salivary glands begins breaking down starch → sugars
Basis of the cracker experiment — holding a plain cracker on the tongue causes it to taste sweet as amylase digests the starch
Lingual lipase also secreted here — begins minor fat digestion
Bolus formation — food is mixed with saliva, lubricated, and shaped into a bolus for swallowing
No significant absorption occurs in the oral cavity
Salivary Glands
Accessory structures of the oral cavity — not part of the alimentary canal wall itself but secrete into it via ducts
Three major pairs:
Gland | Location | Secretion Type | Notes |
|---|---|---|---|
Parotid | Near the ear, sides of face | Primarily serous (watery, enzyme-rich) | Largest salivary gland; become inflamed and swollen in mumps → characteristic chipmunk appearance |
Submandibular | Beneath the mandible (jaw) | Mixed — serous and mucous | Produces the largest volume of saliva overall |
Sublingual | Beneath the tongue | Primarily mucous (thick, lubricating) | Smallest of the three major pairs |
All are exocrine glands — secrete via ducts into the oral cavity
Histologically contain two visible secretory cell types:
Serous acini — produce thin, watery enzyme-containing secretion
Mucous acini — produce thick, lubricating mucus
Composition and Functions of Saliva
Saliva is mostly water but also contains:
Mucus — lubricates food bolus for smooth passage through esophagus and GI tract
Salivary amylase — initiates starch digestion
Lingual lipase — minor fat digestion begins
Antimicrobial substances — inhibit bacterial growth; protect teeth and oral tissues
Functions:
Dissolves food particles → enables taste (dissolved molecules reach taste receptors)
Lubricates oral cavity for speech, singing, playing instruments
Lubricates food bolus for swallowing and transit through the esophagus
Initiates chemical digestion (amylase, lipase)
Antimicrobial protection
Consequences of Insufficient Saliva — Xerostomia (Dry Mouth)
Food does not taste as good — dissolved molecules cannot reach taste receptors efficiently
Speech becomes difficult — tongue sticks to oral surfaces
Swallowing becomes harder — bolus not adequately lubricated
Dental cavities and oral infections increase — antimicrobial protection is lost
Particularly relevant in older adults taking multiple medications with anticholinergic side effects that reduce salivary secretion
Older antidepressants especially notorious for causing dry mouth → patients may stop taking medications as a result
Autonomic Control of Salivation
Salivation is controlled by the autonomic nervous system:
Parasympathetic stimulation → increases salivation
The S in SLUD mnemonic (Salivation, Lacrimation, Urination, Defecation)
Triggered by sight, smell, taste, or thought of food (cephalic phase)
Sympathetic stimulation → decreases salivation
Constricts arterioles supplying salivary glands → reduces blood flow → reduces secretion
Dry mouth during public speaking, job interviews, or high-stress situations = direct sympathetic effect
Histological Note — How the Mouth Fits the Universal Plan
The oral cavity does not follow the full four-layer wall plan seen in the rest of the alimentary canal
It lacks a muscularis mucosae, submucosa, muscularis externa, and serosa in the conventional sense
It is better understood as the entry point to the canal — where the stratified squamous epithelium of the outer body surface transitions into the digestive tube
The full four-layer histological plan begins properly at the esophagus and continues to the anus
The key histological point for the mouth: stratified squamous epithelium throughout — matched to its function of mechanical protection and food processing
Regional Modifications Esophagus
Epithelium:
Non-keratinized stratified squamous epithelium
Multi-layered protective tissue that resists mechanical abrasion from passing food boluses
Same tissue type found in the oral cavity and vaginal canal
Transitions abruptly at the gastroesophageal junction to simple columnar epithelium of the stomach — this sharp boundary is clearly visible under the microscope and is a key histological landmark
Muscularis Externa:
Standard two layers (inner circular + outer longitudinal)
Outermost Layer:
Adventitia (not serosa) — because the esophagus sits outside the peritoneal cavity
Function:
Purely a conduit — transports food boluses to the stomach one swallow at a time
No significant digestion or absorption occurs here
Stomach
Epithelium:
Simple columnar epithelium throughout
Specialized for secretion, not protection — which is why additional protective mechanisms (mucus) are critical
Single-layered and cannot withstand acid on its own
Mucosal Modifications — Rugae:
Longitudinal folds of the mucosa called rugae
Allow the stomach to distend as it fills — when empty, rugae are prominent; as stomach fills, they flatten
Primary role is distensibility, not increased surface area (unlike plicae of small intestine)
Mucosal Modifications — Gastric Pits and Glands:
Mucosal surface is punctuated by millions of gastric pits, each leading into one or more gastric glands
Gastric glands contain multiple specialized cell types:
Cell Type | Secretion | Function |
|---|---|---|
Surface mucous cells | Thick alkaline mucus | Protects stomach lining from HCl and enzymes |
Mucous neck cells | Less alkaline mucus | Additional mucosal lubrication and protection |
Chief cells (zymogenic) | Pepsinogen + gastric lipase | Pepsinogen activated by HCl → pepsin (protein digestion); lipase begins fat digestion |
Parietal cells | HCl + intrinsic factor | HCl activates pepsinogen, kills pathogens, promotes Ca²⁺/Fe²⁺ absorption; intrinsic factor required for B12 absorption |
G cells (enteroendocrine) | Gastrin (into bloodstream) | Stimulates parietal and chief cells; enhances gastric motility |
Muscularis Externa:
Three layers instead of the standard two — unique to the stomach:
Inner oblique layer (innermost, added layer)
Middle circular layer
Outer longitudinal layer
Three fiber directions produce complex churning and grinding movements essential for mixing food with gastric juice to form chyme
Once chyme is processed, muscularis propels it through the pyloric sphincter into the duodenum
Outermost Layer:
Serosa (stomach is within the peritoneal cavity)
Small Intestine
Overview:
Primary site of both chemical digestion and nutrient absorption
Has the most elaborate surface area modifications in the entire canal
Three regions: duodenum (receives bile + pancreatic juice), jejunum (primary absorption zone), ileum (connects to large intestine at ileocecal valve)
Epithelium:
Simple columnar epithelium
Populated heavily with absorptive cells and goblet cells
Three Levels of Surface Area Modification:
Level 1 — Plicae Circulares (Circular Folds):
Permanent semicircular folds of both mucosa and submucosa projecting into the lumen
Unlike rugae, they do not flatten when the intestine fills — always present
Force chyme to swirl in a circular, rifling pattern — slows transit and maximizes contact with absorptive surface
Carry embedded enzymes and absorptive cells; contact with them promotes both digestion and absorption
Analogous to large folds of a crumpled bath towel
Level 2 — Villi:
Finger-like projections of the mucosa arising from the plicae circulares
Covered by simple columnar epithelium
Each villus contains internally:
A capillary bed (arteriole → capillaries → venule) — absorbs sugars and amino acids → mesenteric veins → hepatic portal vein → liver
A lacteal (lymphatic vessel) — receives chylomicrons carrying reassembled dietary fat; drains into lymphatic system → subclavian vein
Analogous to the loops on a terrycloth towel
Level 3 — Microvilli (Brush Border):
Tiny projections covering the apical surface of each individual epithelial cell on the villi
Collectively called the brush border
Massively increase surface area at the cellular level
Host brush border enzymes embedded directly in the cell membrane (not secreted into lumen):
Sucrase, lactase, maltase, dextrinase, glucoamylase — complete carbohydrate digestion
Dipeptidase, aminopeptidase — complete peptide digestion to free amino acids
Final-stage digestion occurs right at the absorptive surface
Muscularis Externa:
Returns to standard two-layer arrangement (inner circular + outer longitudinal)
Outermost Layer:
Serosa
Large Intestine
Overview:
Primary roles: water reabsorption, housing the microbiome, compacting and expelling feces, Site of vitamin synthesis and absorption (by bacteria).
Nothing left to absorb in terms of nutrients — histology reflects this
Epithelium:
Simple columnar epithelium
No villi — the mucosa is flat; this is the single most important histological distinction from the small intestine
Contains intestinal crypts (glands) with openings visible in the mucosa — these can be mistaken for villi on a slide but are not
Goblet cells are extremely abundant — far more than anywhere else in the canal
As the colon progressively dehydrates fecal material, enormous amounts of lubricating mucus are needed to allow compacted feces to move through and be expelled without damaging the epithelium
Muscularis Externa — Unique Modifications:
The outer longitudinal layer is condensed into three narrow bands called taeniae coli rather than forming a complete layer
Because the taeniae coli are shorter than the colon itself, they gather the wall into characteristic pouches called haustra — giving the colon its segmented, bumpy appearance
Chyme moves through the large intestine haustrum by haustrum in a stepwise pattern
Outermost Layer:
Serosa (for intraperitoneal portions) or adventitia (for retroperitoneal portions)
Key Comparative Summary
Feature | Esophagus | Stomach | Small Intestine | Large Intestine |
|---|---|---|---|---|
Epithelium | Non-keratinized stratified squamous | Simple columnar | Simple columnar | Simple columnar |
Surface modifications | None | Rugae (distensibility) | Plicae, villi, microvilli (absorption) | No villi; crypts only |
Muscularis externa | 2 layers | 3 layers (+ oblique) | 2 layers | 2 layers (taeniae coli) |
Goblet cells | Few | Few | Present | Extremely abundant |
Outermost layer | Adventitia | Serosa | Serosa | Serosa/adventitia |
Primary function | Transport | Churning, digestion, secretion | Digestion + absorption | Water reabsorption, fecal compaction |
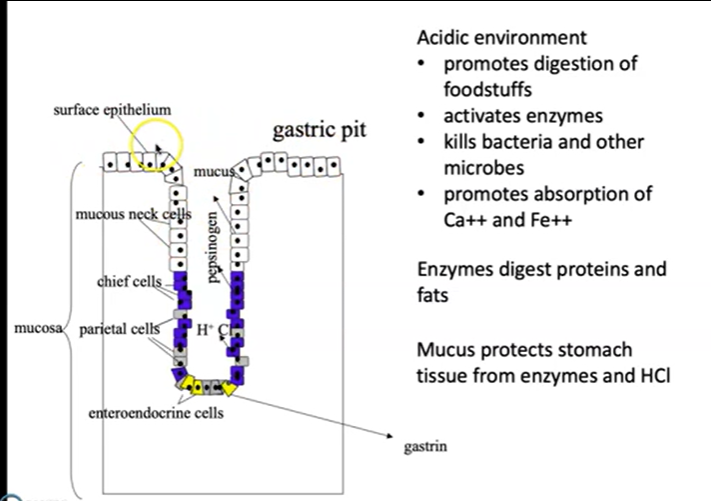
Describe the mechanism by which acid is secreted by the stomach. Write the chemical equation by which H+ are generated
Cell Types of the Gastric Glands
Surface Mucous Cells
Line the surface of the stomach and the upper portion of the gastric pits
Secrete a thick, alkaline mucus that coats the stomach lining
Protects the stomach from both the highly acidic gastric juice and the digestive enzymes being secreted into the lumen
Without this protective mucus layer the stomach would digest itself
Mucous Neck Cells
Found in the neck (upper) region of the gastric glands
Secrete a less alkaline form of mucus compared to surface mucous cells
Together with surface mucous cells they provide substantial mucus coverage throughout the stomach lining
Chief Cells (Zymogenic Cells)
Secrete two substances:
Pepsinogen — an inactive proenzyme (zymogen) that is converted to the active enzyme pepsin in the presence of HCl; pepsin is the primary protease of the stomach and initiates protein digestion; secreted in inactive form to prevent the chief cells from digesting themselves
Gastric lipase — an enzyme that begins the digestion of lipids, particularly triglycerides, in the stomach; contribution is modest compared to pancreatic lipase
Parietal Cells
Responsible for two critically important secretions:
Hydrochloric acid (HCl)
Creates the highly acidic environment of the stomach — gastric pH can reach as low as 1.5 to 2.0
Activates pepsinogen to pepsin
Promotes chemical digestion of foodstuffs
Kills ingested bacteria and pathogens — first-line defense against microorganisms entering through food and drink
Promotes absorption of minerals such as calcium and iron; this is why taking iron supplements with orange juice (vitamin C/ascorbic acid) is recommended — the acidity helps solubilize iron for better absorption
Intrinsic factor
A glycoprotein required for the absorption of vitamin B12 in the small intestine
Deficiency in intrinsic factor leads to pernicious anemia
Mechanism of HCl secretion:
CO2 diffuses into the parietal cell and combines with water via carbonic anhydrase to form carbonic acid (H2CO3)
Carbonic acid dissociates into bicarbonate (HCO3-) and a hydrogen ion (H+)
H+ is actively pumped into the gastric lumen via the H+/K+ ATPase (the proton pump) — for each H+ pumped out, a K+ is pumped in
HCO3- is exchanged for Cl- across the basolateral membrane; Cl- then passively moves into the gastric lumen
H+ and Cl- combine in the lumen to form HCl
The proton pump is the pharmacological target of proton pump inhibitors (PPIs) such as omeprazole (Prilosec), used for GERD and peptic ulcer disease
Three signals stimulate parietal cells to secrete more HCl:
Acetylcholine (ACh) — released by parasympathetic (vagal) nerve fibers
Gastrin — secreted by G cells, enters the bloodstream and returns to stimulate parietal cells
Histamine — released from immune cells and mast cells near the stomach; the same histamine involved in allergic responses — antihistamines taken for allergies can also affect gastric acid production
The alkaline tide — for every H+ secreted into the gastric lumen, one HCO3- is released into the bloodstream; blood draining from the stomach becomes slightly more basic during a meal; this temporary rise in blood pH following a large meal is called the alkaline tide
G Cells (Enteroendocrine Cells)
Located in the pyloric region of the stomach
A type of enteroendocrine cell
Secrete the hormone gastrin directly into the bloodstream — not into the gastric lumen, making this an endocrine secretion
Gastrin travels through the blood and:
Stimulates parietal cells to increase HCl secretion
Stimulates chief cells to increase enzyme secretion
Enhances gastric motility
Stimulated by:
Stomach distension
Presence of partially digested proteins in the stomach
Caffeine
Rising pH within the stomach (less acid present temporarily buffers the lumen)
The stomach is not the only digestive organ with enteroendocrine cells — the small and large intestines have them too, allowing all digestive organs to communicate with each other
Mechanism of Gastric Acid (HCl) Secretion by Parietal Cells Overview
HCl is secreted by parietal cells located in the gastric glands of the stomach mucosa
Gastric pH can reach as low as 1.5–2.0 — achieving this requires actively pumping H⁺ ions against an extremely steep concentration gradient
The entire mechanism relies on a multi-step process involving carbonic anhydrase, ion exchangers, and a primary active transporter
The Chemical Equation: Generation of H⁺ CO2+H2O→carbonic anhydrase H2CO3→H++HCO3−CO_2 + H_2O
CO₂ diffuses into the parietal cell from the blood
Combines with H₂O via the enzyme carbonic anhydrase
Forms carbonic acid (H₂CO₃), which rapidly and spontaneously dissociates into:
H⁺ (hydrogen ion) → pumped into the gastric lumen to form HCl
HCO₃⁻ (bicarbonate ion) → exits the cell at the basolateral membrane in exchange for Cl⁻
Note: This is the same carbonic anhydrase reaction encountered in respiratory gas transport and blood buffering — it is a central multipurpose reaction throughout physiology
Step-by-Step Mechanism Step 1 — CO₂ Enters the Parietal Cell
CO₂ diffuses freely from the arterial blood into the parietal cell down its concentration gradient
No active transport needed — CO₂ is lipid soluble and crosses membranes easily
Step 2 — Carbonic Anhydrase Reaction
Inside the parietal cell, CO₂ combines with H₂O via carbonic anhydrase
Produces carbonic acid (H₂CO₃)
H₂CO₃ immediately dissociates into H⁺ and HCO₃⁻
This is the source of the hydrogen ions that will be secreted into the stomach lumen
Step 3 — H⁺ is Pumped into the Gastric Lumen (Apical Membrane)
H⁺ is actively transported out of the parietal cell across the apical membrane into the gastric lumen via the H⁺/K⁺ ATPase (the proton pump)
This is primary active transport — directly uses ATP
For every H⁺ pumped out into the lumen, one K⁺ is pumped in to the cell
This is the step that creates the extreme acidity of the stomach
The H⁺/K⁺ ATPase is the pharmacological target of proton pump inhibitors (PPIs) such as omeprazole (Prilosec) — drugs used to treat GERD and peptic ulcer disease
Step 4 — HCO₃⁻ Exits at the Basolateral Membrane (The "Alkaline Tide")
The HCO₃⁻ generated in Step 2 is antiported (exchanged) for a Cl⁻ ion across the basolateral membrane
HCO₃⁻ exits the cell → enters the bloodstream
Cl⁻ enters the cell from the blood
This release of HCO₃⁻ into the blood during a meal causes a temporary rise in blood pH called the alkaline tide — a counterintuitive consequence of acid production where the blood draining from the stomach becomes slightly more basic as the stomach lumen becomes more acidic
Step 5 — Cl⁻ Moves into the Gastric Lumen (Apical Membrane)
The Cl⁻ that entered the cell at the basolateral membrane in Step 4 passively diffuses across the apical membrane into the gastric lumen
Step 6 — HCl is Formed in the Lumen
H⁺ (from Step 3) and Cl⁻ (from Step 5) combine in the gastric lumen to form HCl
Result: highly acidic gastric juice with pH as low as 1.5–2.0
Summary Diagram of Ion Movements
Location | Movement | Mechanism |
|---|---|---|
Blood → parietal cell | CO₂ diffuses in | Passive (lipid soluble) |
Inside cell | CO₂ + H₂O → H⁺ + HCO₃⁻ | Carbonic anhydrase enzyme |
Parietal cell → lumen (apical) | H⁺ pumped out, K⁺ pumped in | H⁺/K⁺ ATPase (primary active transport) |
Parietal cell → blood (basolateral) | HCO₃⁻ exits, Cl⁻ enters | Antiporter (secondary active transport) |
Parietal cell → lumen (apical) | Cl⁻ diffuses out | Passive diffusion |
Gastric lumen | H⁺ + Cl⁻ → HCl | Chemical combination |
Three Stimulants of Parietal Cell Acid Secretion
Parietal cells have receptors for three major signals, each acting independently to stimulate HCl secretion:
Acetylcholine (ACh) — released by parasympathetic (vagal) nerve fibers; the "rest and digest" neurotransmitter; vagus nerve stimulation increases acid secretion
Gastrin — secreted by G cells in the gastric glands; enters the bloodstream and returns to bind receptors on parietal cells
Histamine — released from immune and mast cells near the stomach; the same histamine involved in allergic responses — which is why antihistamines taken for allergies can also reduce gastric acid production by blocking this same receptor
All three signals converge on the parietal cell and stimulate the H⁺/K⁺ ATPase to secrete more H⁺
Pharmacological Targets Along This Pathway
Proton Pump Inhibitors (PPIs) — e.g., omeprazole (Prilosec): directly block the H⁺/K⁺ ATPase; most powerful acid-reducing drugs; used for GERD and peptic ulcer disease
H2 receptor antagonists — e.g., cimetidine (Tagamet): block the histamine receptor on parietal cells; reduce acid secretion; among the first rationally designed drugs in pharmacology; now less commonly used due to significant drug-drug interactions by altering metabolism of many other medications
Muscarinic receptor blockers — block the ACh receptor on parietal cells; reduce vagal stimulation of acid secretion; listed among effective GERD treatments
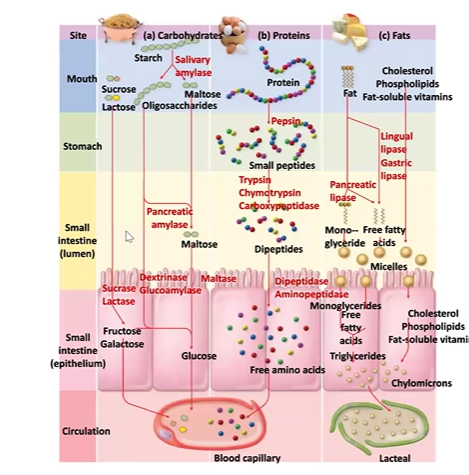
Describe the absorption of nutrients (simple sugars, amino acids, long and shortchain fatty acids) in the small intestine
Overview
The small intestine is the primary site of nutrient absorption in the body
Absorption occurs primarily in the jejunum — the middle segment — which has the most developed villi and brush border
The ileum continues absorption of remaining nutrients and specifically absorbs vitamin B12 and bile salts
Three levels of structural modification dramatically increase surface area for absorption:
Plicae circulares — permanent circular folds of mucosa and submucosa; slow transit time and force chyme into circular contact with absorptive surface
Villi — finger-like projections of the mucosa; each contains a capillary bed and a lacteal
Brush border (microvilli) — on the apical surface of each epithelial cell; hosts brush border enzymes; final-stage digestion occurs here
Different nutrients follow completely different absorption pathways — simple sugars and amino acids enter the blood capillaries and travel via the hepatic portal vein directly to the liver; fats follow a completely different indirect route through the lymphatic system
Structural Features Essential for Absorption
Villi
Finger-like projections of the mucosa projecting into the lumen
Each villus is covered by simple columnar epithelium
Each villus contains internally:
A capillary bed — arteriole, capillary network, venule; absorbs monosaccharides and amino acids
A lacteal — a lymphatic vessel; absorbs chylomicrons (packaged fat) which are too large to enter blood capillaries
The presence of villi is the key histological feature distinguishing the small intestine from the large intestine — the large intestine has no villi
Brush Border
Microvilli on the apical surface of each epithelial cell
Hosts brush border enzymes that perform final-stage digestion at the point of absorption
Digestion and absorption occur simultaneously at the same location — highly efficient arrangement
Two Pathways Out of the Epithelial Cell
Apical membrane — faces the intestinal lumen; where nutrients enter the epithelial cell from the lumen
Basolateral membrane — faces the interstitial fluid and capillaries; where nutrients exit the epithelial cell into the blood or lymph
All absorbed nutrients must cross both membranes to enter the body proper
Absorption of Simple Sugars
Prior Digestion
Carbohydrate digestion begins in the mouth with salivary amylase breaking starch into smaller polysaccharides
Continues in the small intestine with pancreatic amylase breaking polysaccharides into disaccharides and monosaccharides
Completed at the cell surface by brush border enzymes:
Sucrase — breaks sucrose into glucose and fructose
Lactase — breaks lactose into glucose and galactose
Maltase — breaks maltose into two glucose units
Dextrinase and glucoamylase — complete starch digestion to individual glucose units
Only monosaccharides can be absorbed — glucose, fructose, and galactose
Glucose and Galactose Absorption
Absorbed via secondary active transport on the apical membrane
Use sodium-glucose cotransporters (SGLT transporters) on the apical surface
Glucose and galactose hitch a ride as Na+ moves down its concentration gradient into the cell
The Na+ gradient driving this cotransport is maintained by the Na+/K+ ATPase on the basolateral membrane — which continuously pumps Na+ out of the cell keeping intracellular Na+ low
This is secondary active transport — glucose moves against its concentration gradient by coupling to the downhill movement of Na+; ATP is not used directly but powers the Na+/K+ ATPase that maintains the gradient
Once inside the epithelial cell glucose and galactose exit through the basolateral membrane via facilitated diffusion transporters (GLUT transporters) — moving down their concentration gradient into the interstitial fluid
From there they diffuse into the capillary network within the villus
Fructose Absorption
Uses a different and simpler mechanism than glucose and galactose
Absorbed via facilitated diffusion on the apical membrane using GLUT5 transporters — does not require sodium cotransport
Exits through the basolateral membrane via GLUT transporters into the capillary network
Absorbed more slowly than glucose and galactose because it relies on facilitated diffusion rather than active transport
Route to the Liver — All Simple Sugars
All monosaccharides (glucose, galactose, fructose) follow the same route after entering the capillary network:
Capillary bed of the villus → mesenteric veins → hepatic portal vein → hepatic sinusoids → hepatic veins → inferior vena cava → heart → systemic circulation
The liver receives all absorbed sugars first via the portal system before they reach systemic circulation
The liver regulates blood glucose levels — storing excess glucose as glycogen or releasing glucose when blood levels fall
Absorption of Amino Acids
Prior Digestion
Protein digestion begins in the stomach where pepsinogen is activated to pepsin by HCl; pepsin is the primary protease of the stomach and initiates protein digestion by cleaving peptide bonds
Continues in the small intestine with pancreatic proteases arriving from the pancreas:
Trypsin (activated from trypsinogen by enterokinase at the brush border) — cleaves interior peptide bonds
Chymotrypsin (activated by trypsin) — cleaves peptide bonds at different sites than trypsin
Carboxypeptidase (activated by trypsin) — removes amino acids from the carboxyl end of peptide chains
Completed at the cell surface by brush border enzymes:
Dipeptidase — breaks dipeptides into free amino acids
Aminopeptidase — removes amino acids from the amino end of peptide chains
Only free amino acids and very small peptides (dipeptides and tripeptides) can be absorbed
Amino Acid Absorption
Free amino acids are absorbed via sodium-coupled cotransporters on the apical membrane — similar mechanism to glucose and galactose
Multiple different carrier proteins exist for different classes of amino acids (acidic, basic, neutral) — each class has its own specific transporter
Amino acids move into the cell coupled to the downhill movement of Na+ — secondary active transport driven by the Na+ gradient maintained by basolateral Na+/K+ ATPase
Small dipeptides and tripeptides can also be absorbed directly via H+-coupled cotransporters on the apical membrane — they are then broken down to free amino acids inside the cell
Free amino acids exit through the basolateral membrane via facilitated diffusion carriers into the interstitial fluid
Diffuse into the capillary network within the villus
Route to the Liver — Amino Acids
Follow the same direct route as simple sugars:
Capillary bed of the villus → mesenteric veins → hepatic portal vein → hepatic sinusoids → hepatic veins → inferior vena cava → heart → systemic circulation
The liver receives all absorbed amino acids first via the portal system
The liver regulates amino acid levels — synthesizing plasma proteins, converting excess amino acids to glucose or fat, and converting ammonia (a byproduct of amino acid metabolism) to urea for renal excretion
Absorption of Long-Chain Fatty Acids
Overview
Long-chain fatty acids are the primary component of dietary fat
Fat absorption is considerably more complex than that of sugars and amino acids and follows a completely different and indirect pathway
The route is counterintuitive — fat bypasses the hepatic portal system entirely and instead travels through the lymphatic system before eventually reaching the liver via arterial circulation
Prior Digestion
Some gastric lipase begins fat digestion in the stomach but this contribution is modest
The bulk of fat digestion occurs in the small intestine:
Bile salts (from the liver and gallbladder) emulsify large fat globules into tiny micelles — bile salts are amphipathic and surround fat droplets dispersing them throughout the aqueous intestinal fluid dramatically increasing surface area
Pancreatic lipase acts within the micelles to break triglycerides into monoglycerides and free fatty acids
Bile salts are emulsifiers not enzymes — they make fat accessible to lipase but do not chemically break it down themselves
Step 1 — Micelle Migration to the Brush Border
Micelles (tiny fat droplets surrounded by bile salts) migrate through the intestinal fluid to the brush border of the epithelial cells
The hydrophilic exterior of the micelles allows them to move through the aqueous environment
Step 2 — Diffusion Across the Apical Membrane
Fatty acid components (monoglycerides and free fatty acids) diffuse out of the micelles and across the plasma membrane directly into the epithelial cell
Long-chain fatty acids are lipid-soluble — they can pass directly through the lipid bilayer of the plasma membrane by simple diffusion
Bile salts remain in the lumen — they are not absorbed here; they continue to form new micelles and facilitate further fat absorption
Bile salts are eventually reabsorbed specifically in the ileum and returned to the liver via the hepatic portal vein for recycling — this is called enterohepatic circulation
Step 3 — Reassembly of Triglycerides Inside the Epithelial Cell
Inside the epithelial cell the monoglycerides and long-chain fatty acids are transported to the smooth endoplasmic reticulum
They are reassembled back into triglycerides within the smooth ER
This reassembly step is unique to long-chain fatty acids — short-chain fatty acids do not undergo this step as described below
Step 4 — Chylomicron Formation
The reassembled triglycerides are packaged in the Golgi apparatus along with:
Cholesterol
Phospholipids
Proteins (apoproteins)
Into large lipoprotein particles called chylomicrons
Chylomicrons are the transport vehicles that carry fat through the lymphatic system
They are too large to enter blood capillaries directly — the tight junctions of blood capillary walls prevent their entry
Step 5 — Exocytosis into the Lacteal
Chylomicrons exit the epithelial cell by exocytosis through the basolateral membrane
They enter the lacteal — the lymphatic vessel running through the core of each villus
The endothelial cells of lacteals overlap loosely unlike the tight junctions of blood capillaries — this loose arrangement allows the large chylomicrons to squeeze through and enter the lymphatic vessel
Step 6 — Travel Through the Lymphatic System
Chylomicrons travel through the lymphatic vessels
Pass through lymph nodes
Eventually reach the thoracic duct — the largest lymphatic vessel in the body
Step 7 — Entry into the Bloodstream
The thoracic duct drains into the subclavian vein
Chylomicrons enter the venous bloodstream at the subclavian vein
Travel through the heart
Enter systemic arterial circulation
Eventually reach the liver via arterial circulation — after passing through multiple tissues first
This is a remarkably indirect route — fat travels through the lymphatic system and heart before eventually reaching the liver via arterial circulation
Contrast with glucose and amino acids which reach the liver directly and rapidly through the portal system
Why Long-Chain Fatty Acids Take This Indirect Route
Long-chain fatty acids must be reassembled into triglycerides and packaged into chylomicrons because they are insoluble in the aqueous blood plasma
Chylomicrons are too large for blood capillaries — they can only enter the larger and more permeable lacteals
The lymphatic route allows fat to bypass the liver initially — giving peripheral tissues (muscle, adipose) first access to dietary fat for energy and storage
Absorption of Short-Chain Fatty Acids
What They Are
Short-chain fatty acids have fewer carbon atoms than long-chain fatty acids (fewer than 6 carbons)
Produced primarily by bacterial fermentation of dietary fiber (cellulose and other plant-based polymers) in the large intestine
Examples include acetate, propionate, and butyrate
Not a major component of dietary fat — primarily a byproduct of microbial activity in the large intestine
How They Differ from Long-Chain Fatty Acids
Short-chain fatty acids are water-soluble — unlike long-chain fatty acids which are lipid-soluble and insoluble in water
Because they are water-soluble they do not need to be packaged into micelles for absorption
They do not need to be reassembled into triglycerides inside the epithelial cell
They do not need to be packaged into chylomicrons
They do not enter the lymphatic system
Absorption Mechanism
Absorbed directly across the epithelial cell membrane by simple diffusion — their small size and water solubility allow this
Can also be absorbed via specific transporters on the apical membrane
Once inside the epithelial cell they exit directly through the basolateral membrane into the capillary network
Route to the Liver
Enter the capillary network directly — unlike long-chain fatty acids
Follow the same direct route as glucose and amino acids:
Capillary network → mesenteric veins → hepatic portal vein → liver
Reach the liver directly via the portal system — not through the lymphatic system
This is the key distinction from long-chain fatty acids — short-chain fatty acids take the direct portal route while long-chain fatty acids take the indirect lymphatic route
Summary Comparison of Absorption Pathways
Simple Sugars (Glucose, Galactose)
Mechanism — sodium cotransport (SGLT) on apical membrane; facilitated diffusion (GLUT) on basolateral membrane
Route — capillary bed → mesenteric veins → hepatic portal vein → liver
Direct portal route
Simple Sugars (Fructose)
Mechanism — facilitated diffusion (GLUT5) on apical membrane; facilitated diffusion (GLUT) on basolateral membrane
Route — capillary bed → mesenteric veins → hepatic portal vein → liver
Direct portal route
Amino Acids
Mechanism — sodium-coupled cotransporters on apical membrane; facilitated diffusion on basolateral membrane; small peptides via H+-coupled cotransporters then broken down intracellularly
Route — capillary bed → mesenteric veins → hepatic portal vein → liver
Direct portal route
Long-Chain Fatty Acids
Mechanism — simple diffusion across apical membrane; reassembled into triglycerides; packaged into chylomicrons; exocytosis into lacteal
Route — lacteal → lymphatic vessels → thoracic duct → subclavian vein → heart → arterial circulation → liver
Indirect lymphatic route — bypasses portal system
Short-Chain Fatty Acids
Mechanism — simple diffusion or specific transporters across apical membrane; exit directly through basolateral membrane
Route — capillary bed → mesenteric veins → hepatic portal vein → liver
Direct portal route — same as sugars and amino acids; does not require chylomicron packaging or lymphatic transport
Distinguish between the exocrine and endocrine functions of the pancreas.
What hormones come from the pancreas and what are their functions?
What enzymes come from the pancreas and what are their functions?
Describe the composition and function(s) of pancreatic juice
Exocrine vs. Endocrine Functions of the Pancreas Overview
The pancreas is unique in that it serves both exocrine and endocrine roles — making it a mixed gland
It is classified as an accessory digestive organ — not part of the alimentary canal itself, but secretes substances into it via ducts
Histologically, the two functional portions look distinctly different from one another under the microscope
Exocrine vs. Endocrine: Key Distinctions
Feature | Exocrine | Endocrine |
|---|---|---|
Secretion destination | Into ducts → duodenum lumen | Directly into the bloodstream |
Structural unit | Acinar cells | Islets of Langerhans |
Products | Digestive enzymes + bicarbonate | Hormones (insulin, glucagon, somatostatin) |
Function | Chemical digestion in small intestine | Regulation of blood glucose and metabolism |
Appearance (histology) | Dominant tissue; surrounds islets | Islets appear as pale, distinct clusters amid acinar tissue |
Exocrine Function of the Pancreas Anatomy of the Exocrine Pancreas
Pancreatic acinar cells produce enzyme-rich secretions
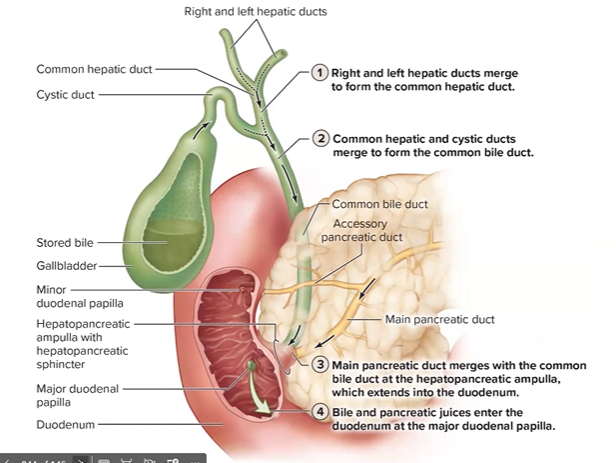
These secretions drain into the pancreatic duct
The pancreatic duct merges with the common bile duct at the hepatopancreatic ampulla (ampulla of Vater)
Both bile and pancreatic juice are delivered simultaneously into the duodenum at the major duodenal papilla
This junction means fat digestion (bile) and enzymatic digestion (pancreatic enzymes) are delivered to the same location at the same time — a highly coordinated arrangement
Composition of Pancreatic Juice
Pancreatic juice has two major components:
1. Bicarbonate (HCO₃⁻)
Most abundant component by volume
Produced by ductal cells of the pancreas
Function:
Neutralizes the highly acidic chyme arriving from the stomach into the duodenum
Protects the duodenal wall from erosion by stomach acid
Raises pH to approximately 7–8, establishing the appropriate environment for intestinal enzyme activity
Pancreatic enzymes work optimally at neutral-to-slightly-alkaline pH — they would be destroyed in acidic conditions
Regulation: Secretion of bicarbonate is triggered by secretin, which is released by the small intestine in response to acidic chyme entering the duodenum — a logical feedback loop: more acid arriving = more buffering needed
2. Digestive Enzymes
Produced by acinar cells
Represent the full complement of digestive enzymes needed for chemical digestion in the small intestine
Many are secreted as inactive zymogens (inactive precursor forms) to prevent autodigestion of the pancreas itself
Delivered via the pancreatic duct into the duodenum where they are activated
Pancreatic Enzymes and Their Functions Proteases (Protein-Digesting Enzymes)
All secreted as inactive zymogens — critical safety mechanism to prevent the pancreas from digesting itself
Enzyme | Secreted As | Activated By | Function |
|---|---|---|---|
Trypsin | Trypsinogen | Enterokinase (brush border enzyme in duodenum) | Cleaves peptide bonds; also activates other zymogens |
Chymotrypsin | Chymotrypsinogen | Trypsin | Cleaves peptide bonds at different sites than trypsin |
Carboxypeptidase | Procarboxypeptidase | Trypsin | Cleaves amino acids from the carboxyl end of peptide chains |
Together these proteases break proteins and large polypeptides down into small peptides and amino acids
Final digestion to free amino acids is completed by brush border enzymes (aminopeptidase, dipeptidase) at the surface of intestinal epithelial cells
Carbohydrate-Digesting Enzyme
Enzyme | Function |
|---|---|
Pancreatic amylase | Breaks down polysaccharides (starches) into disaccharides and short oligosaccharides; continues carbohydrate digestion begun by salivary amylase in the mouth; final digestion to monosaccharides completed by brush border enzymes |
Pancreatic amylase is secreted in active form — no zymogen form needed because carbohydrates do not threaten to digest pancreatic tissue
Fat-Digesting Enzyme
Enzyme | Function |
|---|---|
Pancreatic lipase | Primary fat-digesting enzyme in the entire body; breaks triglycerides down into monoglycerides and free fatty acids within micelles; works in conjunction with bile salts which emulsify fat to dramatically increase surface area available to lipase |
Also secreted in active form
Works inside micelles formed by bile salts — bile salts are not enzymes, they do not digest fat, but they make fat far more accessible to lipase by dispersing large fat globules into tiny droplets
Without bile salt emulsification, pancreatic lipase would have dramatically reduced efficiency
Endocrine Function of the Pancreas Anatomy of the Endocrine Pancreas
Endocrine cells are organized into clusters called Islets of Langerhans
These islets are scattered throughout the pancreatic tissue
Histologically they appear as pale, distinct oval clusters amid the darker surrounding acinar tissue — clearly distinguishable under the microscope
Islet cells secrete hormones directly into the bloodstream — no ducts involved
Pancreatic Hormones and Their Functions
Hormone | Secreted By | Stimulus for Release | Function |
|---|---|---|---|
Insulin | Beta (β) cells | Rising blood glucose (after a meal) | Lowers blood glucose — promotes uptake of glucose into cells, promotes glycogen synthesis in liver and muscle, promotes fat storage; anabolic hormone |
Glucagon | Alpha (α) cells | Falling blood glucose (fasting/between meals) | Raises blood glucose — stimulates glycogenolysis (breakdown of glycogen) and gluconeogenesis (synthesis of new glucose) in the liver; catabolic hormone |
Somatostatin | Delta (δ) cells | Rising blood glucose and amino acids | Inhibits release of both insulin and glucagon; also inhibits digestive secretions; acts as a general brake on digestion and metabolism; fine-tunes the balance between insulin and glucagon |
Regulation of Pancreatic Exocrine Secretion
Pancreatic juice secretion is coordinated by three mechanisms:
Vagal stimulation (parasympathetic — cranial nerve X):
Promotes pancreatic juice secretion in anticipation of and during a meal
Part of the cephalic and gastric phases of digestive regulation
Secretin:
Released by enteroendocrine cells of the small intestine in response to acidic chyme entering the duodenum
Stimulates pancreatic ductal cells to secrete bicarbonate-rich juice
Logical feedback: more acid arriving → more bicarbonate needed to neutralize it
Cholecystokinin (CCK):
Released by enteroendocrine cells of the small intestine in response to fatty chyme entering the duodenum
Stimulates acinar cells to secrete enzyme-rich pancreatic juice
Logical feedback: more fat arriving → more lipase needed
CCK also simultaneously triggers gallbladder contraction to release bile — coordinating enzyme delivery with emulsification
Key pattern: Secretin and CCK are "repeat offenders" — they appear again and again throughout digestive physiology, regulating the stomach, pancreas, liver, and gallbladder in a highly coordinated fashion
Why Zymogens Matter — Clinical Connection
Proteases are secreted as inactive zymogens because if they were active inside the pancreas, they would digest the organ itself
In acute pancreatitis, these zymogens become prematurely activated inside the pancreas
The pancreas begins autodigesting — an extremely painful and potentially life-threatening condition
Common causes include gallstones obstructing the pancreatic duct and chronic alcohol use
Summary
Component | Type | Source | Destination | Function |
|---|---|---|---|---|
Bicarbonate | Exocrine | Ductal cells | Duodenum lumen | Neutralizes acid, optimizes pH for enzyme activity |
Pancreatic amylase | Exocrine | Acinar cells | Duodenum lumen | Starch → disaccharides |
Pancreatic lipase | Exocrine | Acinar cells | Duodenum lumen | Triglycerides → monoglycerides + fatty acids |
Trypsin | Exocrine | Acinar cells (as trypsinogen) | Duodenum lumen | Protein/peptide digestion; activates other zymogens |
Chymotrypsin | Exocrine | Acinar cells (as chymotrypsinogen) | Duodenum lumen | Protein/peptide digestion |
Carboxypeptidase | Exocrine | Acinar cells (as procarboxypeptidase) | Duodenum lumen | Cleaves terminal amino acids from peptides |
Insulin | Endocrine | Beta cells of islets | Bloodstream | Lowers blood glucose; promotes storage |
Glucagon | Endocrine | Alpha cells of islets | Bloodstream | Raises blood glucose; promotes breakdown |
Somatostatin | Endocrine | Delta cells of islets | Bloodstream | Inhibits insulin/glucagon; brakes digestion |
Describe the hepatic portal system, and outline briefly the flow of blood from digestive organs – liver – heart. Why is this circulatory system important?
The Hepatic Portal System Overview
The hepatic portal system is a specialized circulatory arrangement in which blood travels from one capillary bed through a portal vein to a second capillary bed before returning to the heart
A portal system by definition means: capillaries → portal vein → capillaries (rather than the usual capillaries → vein → heart)
The liver exemplifies this perfectly: digestive organ capillaries → hepatic portal vein → hepatic sinusoids (liver capillaries) → hepatic veins → heart
This arrangement gives the liver first access to everything absorbed from the gut before it reaches systemic circulation
The liver receives blood from two sources simultaneously — a unique feature among organs in the body
Two Blood Supplies to the Liver
Source | Vessel | Type of Blood | Origin |
|---|---|---|---|
Hepatic portal vein | Portal vein from digestive organs | Nutrient-rich, deoxygenated | Capillary beds of stomach, small intestine, large intestine, spleen, pancreas |
Hepatic artery | Branch of celiac artery | Oxygenated | Systemic circulation |
Both sources mix together within the hepatic sinusoids — the leaky, highly permeable capillary-like channels within the liver
This is one of the only places in adult circulation where oxygenated and deoxygenated blood intentionally mix (the other being fetal circulation)
Complete Blood Flow Pathway: Digestive Organs → Liver → Heart Step-by-Step Flow
Step 1 — Absorption in digestive organ capillaries:
Nutrients (sugars, amino acids), drugs, toxins, and anything else absorbed from the gut lumen enter the capillary beds of the digestive organs
These include capillary beds of the stomach, small intestine, large intestine, spleen, and pancreas
Fat is the exception — dietary fat packaged as chylomicrons bypasses the portal system entirely, entering lacteals → lymphatic vessels → thoracic duct → subclavian vein (discussed below)
Step 2 — Drainage into the hepatic portal vein:
Blood from the capillary beds of the digestive organs drains into the mesenteric veins
These converge to form the hepatic portal vein
The hepatic portal vein carries nutrient-rich but deoxygenated blood directly to the liver
Step 3 — Entry into the liver:
The hepatic portal vein enters the liver at the hilum
At the same time, the hepatic artery delivers oxygenated blood to the liver
Both vessels branch repeatedly within the liver, ultimately delivering blood to the portal triads at the corners of each hepatic lobule
Step 4 — Flow through the hepatic sinusoids:
Blood from both the portal vein branches and hepatic artery branches flows into the sinusoids
Sinusoids are leaky, highly permeable capillaries running between plates of hepatocytes
Because sinusoids are so permeable, blood components interact closely with hepatocytes as they flow through
Kupffer cells (resident macrophages) are embedded in the sinusoid walls — they phagocytose old/damaged red blood cells, intercept microbes, and present antigens to the immune system
Blood flows from the portal triads at the periphery inward toward the central vein at the center of each lobule
Step 5 — Drainage into the central vein:
Blood percolates through the sinusoids and drains into the central vein at the center of each hepatic lobule
This is where blood from the hepatic portal vein and hepatic artery have fully mixed and been processed by hepatocytes
Step 6 — Exit via hepatic veins:
Central veins from multiple lobules converge into the hepatic veins
The hepatic veins drain into the inferior vena cava (IVC)
Step 7 — Return to the heart:
Blood travels via the IVC to the right atrium of the heart
From there it enters pulmonary circulation, returns to the left heart, and enters systemic circulation
Simplified Flow Summary
Digestive organ capillaries → mesenteric veins → hepatic portal vein → portal triads → hepatic sinusoids → central vein → hepatic veins → inferior vena cava → right atrium
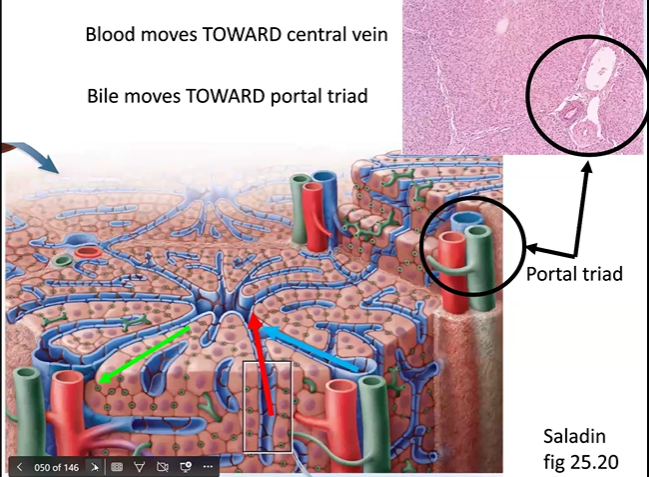
Microscopic Structure of the Liver — The Hepatic Lobule
The hepatic lobule is the functional unit of the liver
Appears roughly hexagonal in cross-section
Key landmarks:
Portal Triads
Located at each corner of the hexagon (6 per lobule)
Each portal triad contains three structures:
Branch of the hepatic portal vein
Branch of the hepatic artery
Bile ductule
Blood flows inward from the portal triads toward the central vein
Bile flows outward from hepatocytes toward the bile ductules at the portal triads
Blood and bile flow in opposite directions and remain in completely separate channels — they never mix
Central Vein
Runs through the center of each lobule
Blood flows from the periphery (portal triads) inward through sinusoids toward the central vein
Central veins ultimately drain into the hepatic veins → IVC
Hepatocytes
Main liver cells surrounding the sinusoids
Perform virtually all of the liver's metabolic, synthetic, and detoxification functions
As blood passes through the sinusoids, hepatocytes monitor and process everything arriving from the gut — giving the liver an early-detection and regulation role for nutrient and substance levels
Sinusoids
Specialized leaky capillaries between plates of hepatocytes
Highly permeable — allow close interaction between blood and hepatocytes
Contain Kupffer cells (resident macrophages) embedded in their walls
The Special Case of Dietary Fat — Why It Bypasses the Portal System
Dietary fat cannot enter blood capillaries directly because chylomicrons are too large
Instead: fat → reassembled into triglycerides → packaged into chylomicrons inside intestinal epithelial cells → exit by exocytosis into lacteals (lymphatic vessels in each villus)
Chylomicrons travel through lymphatic vessels → thoracic duct → drain into the subclavian vein → enter the heart → reach the liver via arterial circulation after passing through multiple tissues first
This is a remarkably indirect route compared to glucose and amino acids
Fat therefore arrives at the liver last, via the hepatic artery, rather than first via the portal vein
Nutrient | Route to Liver |
|---|---|
Glucose | Capillary → mesenteric vein → hepatic portal vein → sinusoids → liver (arrives first) |
Amino acids | Capillary → mesenteric vein → hepatic portal vein → sinusoids → liver (arrives first) |
Dietary fat (chylomicrons) | Lacteal → lymphatics → thoracic duct → subclavian vein → heart → arterial circulation → liver (arrives last, indirectly) |
Why the Hepatic Portal System Is Important
1. First-Pass Nutrient Regulation
The liver receives all absorbed nutrients first before they reach any other tissue or organ
This allows the liver to immediately regulate blood glucose, amino acid levels, and lipid metabolism
Example: after a carbohydrate-rich meal, the liver intercepts the surge of glucose from the portal blood and converts excess glucose to glycogen (glycogenesis) or fat — preventing dangerous hyperglycemia in systemic circulation
2. First-Pass Detoxification
Everything absorbed from the gut — including drugs, alcohol, toxins, and bacterial products — must pass through the liver before reaching systemic circulation
The liver's CYP450 (cytochrome P450) enzyme systems chemically modify these substances, typically converting lipid-soluble compounds into more water-soluble forms for renal excretion
This is called the first-pass effect — clinically significant because oral drugs can be substantially metabolized before ever reaching their target tissues
This is why some drugs must be given at higher oral doses than intravenous doses
This is also why some drugs cannot be given orally at all — they are completely inactivated by first-pass metabolism
Important caveat: detoxification does not always mean making a substance safer — sometimes liver enzymes convert a harmless compound into a toxic metabolite
Classic example: in patients with liver dysfunction (e.g., alcoholic liver disease), CYP450 enzymes produce a toxic metabolite of acetaminophen that can cause severe liver damage and cardiovascular collapse — which is why acetaminophen must be used cautiously in patients with liver disease
3. Immune Surveillance
Kupffer cells in the sinusoids act as sentinels
Because the sinusoids are so leaky, immune cells are positioned directly in the path of all portal blood
They phagocytose bacteria, bacterial products, and foreign antigens that may have entered the blood from the gut
Present antigens to the immune system — the liver plays an important role in immune defense against gut-derived pathogens
4. Waste Processing
Old and damaged red blood cells are broken down by Kupffer cells
Waste products arriving from digestive organ metabolism are filtered and prepared for excretion
Ammonia (a toxic byproduct of protein metabolism) is converted to urea in the liver and excreted by the kidneys
5. Plasma Protein Synthesis
The liver synthesizes most plasma proteins including albumin (the primary protein responsible for blood colloid osmotic pressure)
In liver failure, albumin synthesis drops → blood colloid osmotic pressure falls → fluid leaks out of capillaries into tissues → edema and ascites (fluid accumulation in the abdomen)
6. Vitamin and Mineral Storage
The liver stores fat-soluble vitamins (A, D, E, K), glycogen, and iron
All of these are sourced from nutrients arriving via the portal system
Clinical Correlations Portal Hypertension
If blood flow through the liver is obstructed (e.g., in cirrhosis, where scar tissue replaces normal liver tissue), pressure builds up in the hepatic portal vein
This is called portal hypertension
Consequences include:
Esophageal varices — portal blood backs up into esophageal veins, which dilate and can rupture, causing life-threatening bleeding
Ascites — increased portal pressure forces fluid out of capillaries into the abdominal cavity
Splenomegaly — the spleen enlarges as portal blood backs up
Liver Cirrhosis and Drug Metabolism
Cirrhosis disrupts normal hepatocyte function and CYP450 enzyme activity
Drug metabolism is impaired — drugs accumulate to toxic levels or are metabolized abnormally
Acetaminophen toxicity risk is dramatically elevated in cirrhotic patients
Urinalysis as a Window into Liver and Portal Function
Substances that should be processed by the liver (e.g., bilirubin from red blood cell breakdown) can appear in urine when liver function is impaired
Jaundice (yellowing of skin and eyes) reflects failure to process bilirubin arriving via the portal system
Summary Table
Feature | Detail |
|---|---|
Definition of portal system | Blood travels capillaries → portal vein → second capillary bed before returning to heart |
Portal vein carries | Nutrient-rich, deoxygenated blood from digestive organs |
Hepatic artery carries | Oxygenated blood from systemic circulation |
Where they mix | Hepatic sinusoids |
Direction of blood flow in lobule | Portal triads (periphery) → sinusoids → central vein |
Direction of bile flow in lobule | Hepatocytes → bile ductules → portal triads (opposite to blood) |
Blood and bile mix? | Never — completely separate channels |
Importance | First-pass nutrient regulation, detoxification, immune surveillance, waste processing, plasma protein synthesis |
Describe hormonal (via histamine, secretin, gastrin, and cholecystokinin) and neural control of secretion and motility in the various organs / glands of the digestive system
Hormonal and Neural Control of Digestive Secretion and Motility Overview
The digestive system is regulated by a highly coordinated combination of neural and hormonal signals
These signals ensure that the right secretions are produced, in the right amounts, at the right time, in the right organ
The same hormones appear repeatedly across multiple organs — earning some of them the label "repeat offenders" (particularly secretin and CCK)
Control is organized into three overlapping phases: cephalic, gastric, and intestinal
The Key Regulatory Signals Neural Control — The Two Systems 1. Autonomic Nervous System (Extrinsic)
Parasympathetic (Vagus Nerve — Cranial Nerve X):
Dominant neural driver of digestive activity — "rest and digest"
Vagal stimulation generally increases secretion and motility throughout the digestive system
Releases acetylcholine (ACh) at target organs
Innervates the stomach, small intestine, pancreas, liver, and gallbladder
Responsible for the cephalic phase — the sight, smell, taste, or thought of food activates the vagus nerve and primes the entire digestive system before food even enters the mouth
Mnemonic: SLUD — Salivation, Lacrimation, Urination, Defecation — all parasympathetic functions
Sympathetic Nervous System:
Generally inhibits digestive secretion and motility — "fight or flight"
Constricts arterioles supplying digestive organs, reducing blood flow and secretory activity
Example: dry mouth during public speaking or stress = sympathetic constriction of salivary gland arterioles
Suppresses gastric activity during the intestinal phase
Reduces urine output, saliva, and GI motility during acute stress
2. Enteric Nervous System (Intrinsic)
The digestive tract has its own dedicated nervous system embedded in the wall of the alimentary canal
Two plexuses:
Meissner's plexus (submucosal) — regulates secretion
Auerbach's plexus (myenteric) — coordinates motility and peristalsis
Allows digestive organs to regulate themselves semi-independently of the brain and spinal cord
Mediates short (myenteric) reflexes — purely local responses within the gut wall
Also mediates long (vagovagal) reflexes — signals travel to the medulla and back via the vagus nerve
The Four Key Hormones
Hormone | Produced By | Released In Response To | Primary Targets |
|---|---|---|---|
Histamine | Mast cells / immune cells in stomach wall | Presence of food, ACh, gastrin | Parietal cells of stomach |
Gastrin | G cells (enteroendocrine) of pyloric stomach | Stomach distension, proteins, caffeine, rising pH | Parietal cells, chief cells, stomach muscularis |
Secretin | Enteroendocrine cells of small intestine | Acidic chyme in duodenum | Pancreas (ductal cells), liver, stomach |
Cholecystokinin (CCK) | Enteroendocrine cells of small intestine | Fatty chyme in duodenum | Pancreas (acinar cells), gallbladder, stomach |
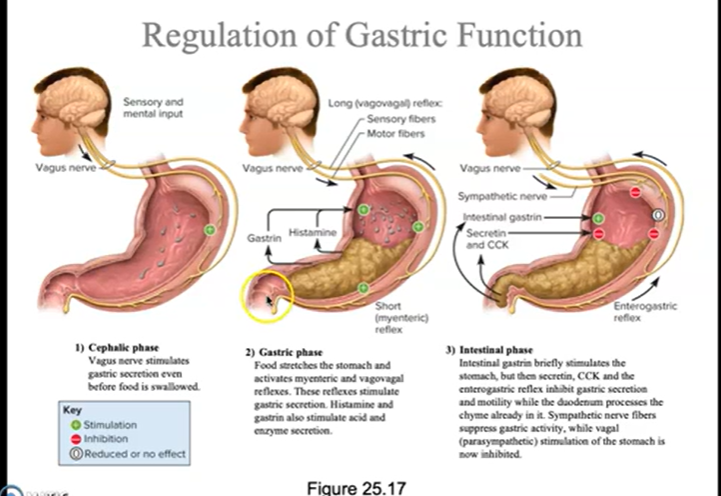
Control Organized by Phase Phase 1 — Cephalic Phase
Trigger: Sight, smell, taste, thought, or anticipation of food — before food even enters the mouth
Neural Control:
Higher brain centers (cerebral cortex, hypothalamus) activate the vagus nerve (CN X)
Vagal stimulation releases ACh at target organs throughout the digestive system
Primes the entire digestive system in anticipation of incoming food
Effects on Each Organ:
Salivary Glands:
Parasympathetic stimulation → increases salivation
Prepares oral cavity for food — lubricates, dissolves food particles, begins starch digestion via salivary amylase
Sympathetic activation does the opposite → decreases salivation → dry mouth during stress
Stomach:
Vagal stimulation directly stimulates parietal cells → increased HCl secretion
Vagal stimulation directly stimulates chief cells → increased pepsinogen secretion
Vagal stimulation stimulates G cells → release of gastrin into the bloodstream
Gastrin then travels back to stomach and amplifies acid and enzyme secretion
Stomach begins producing gastric juice before food arrives — physiological basis for stomach rumbling and salivation when smelling a home-cooked meal
Pancreas:
Vagal stimulation promotes pancreatic juice secretion in anticipation of incoming food
Liver/Gallbladder:
Vagal stimulation promotes bile secretion and prepares gallbladder for contraction
Phase 2 — Gastric Phase
Trigger: Food enters and distends the stomach; chemical content of food detected
Neural Control:
Stomach distension activates mechanoreceptors in the stomach wall
Triggers both:
Short (myenteric) reflexes — purely local enteric nervous system responses
Long (vagovagal) reflexes — signals travel via vagus nerve to medulla and back
Both reflex types further stimulate gastric secretion and motility
Hormonal Control — Gastrin:
What it is:
Peptide hormone secreted by G cells (enteroendocrine cells) in the pyloric region of the stomach
Released into the bloodstream (endocrine secretion — not into the gastric lumen)
Travels systemically and returns to act on target cells
What triggers its release:
Stomach distension (stretching of stomach wall)
Presence of partially digested proteins in the stomach
Caffeine in the stomach
Rising pH in the stomach (i.e., food buffers the acid temporarily, raising pH, which removes the inhibition on G cells)
Vagal stimulation (ACh directly stimulates G cells)
Effects of Gastrin:
Target | Effect |
|---|---|
Parietal cells | Stimulates HCl secretion — primary gastric effect |
Chief cells | Stimulates pepsinogen secretion |
Stomach muscularis | Enhances gastric motility and churning |
Lower esophageal sphincter | Increases tone — prevents acid reflux |
Small intestine and colon | Mild stimulation of motility |
Hormonal Control — Histamine:
What it is:
Paracrine signaling molecule released by mast cells and immune cells in the stomach wall
Acts locally on nearby parietal cells — does not need to enter the bloodstream to exert its effect
Same histamine molecule involved in allergic responses — which is why antihistamines for allergies can also affect gastric acid production
What triggers its release:
Presence of food in the stomach
ACh (vagal stimulation)
Gastrin
Effects of Histamine:
Target | Effect |
|---|---|
Parietal cells | Potently stimulates HCl secretion via H₂ receptors |
Acts synergistically with gastrin and ACh — all three signals converge on parietal cells simultaneously and amplify each other's effects
H2 receptor antagonists (e.g., cimetidine/Tagamet) block this receptor and reduce acid secretion — among the first rationally designed drugs in pharmacology
Summary of the Three Stimulants of Parietal Cells:
Signal | Type | Receptor on Parietal Cell |
|---|---|---|
Acetylcholine (ACh) | Neurotransmitter (vagal) | Muscarinic receptor |
Gastrin | Hormone (bloodstream) | Gastrin/CCK-B receptor |
Histamine | Paracrine (local) | H₂ receptor |
All three ultimately activate the H⁺/K⁺ ATPase (proton pump) to secrete more H⁺ into the gastric lumen
Self-Limiting Feedback Within the Gastric Phase:
As HCl accumulates and gastric pH drops very low, this directly inhibits G cells from releasing more gastrin
Classic negative feedback — excessive acid in the stomach inhibits further acid production
Prevents runaway acid production
Phase 3 — Intestinal Phase
Trigger: Acidic, fatty chyme enters the duodenum
This phase has two components:
Brief stimulatory component — intestinal gastrin momentarily stimulates the stomach
Sustained inhibitory component — the dominant effect; protects the duodenum from being overwhelmed
Hormonal Control — Secretin:
What it is:
Peptide hormone released by enteroendocrine cells of the small intestine (duodenum)
Released into the bloodstream; travels to multiple target organs
What triggers its release:
Acidic chyme entering the duodenum (low pH is the primary trigger)
Logical: more acid arriving = more neutralization needed
Effects of Secretin:
Target Organ | Effect | Why It Makes Sense |
|---|---|---|
Pancreas (ductal cells) | Stimulates bicarbonate-rich pancreatic juice secretion | Bicarbonate neutralizes acid in duodenum |
Liver | Stimulates bile secretion | Prepares for fat digestion |
Stomach — parietal cells | Inhibits HCl secretion | Prevents more acid from arriving in already-acidic duodenum |
Stomach — muscularis | Decreases gastric motility | Slows delivery of more chyme to duodenum |
Secretin essentially tells the stomach: "slow down, the duodenum is already acidic enough"
While simultaneously telling the pancreas: "send bicarbonate to neutralize the acid that's already here"
Hormonal Control — Cholecystokinin (CCK):
What it is:
Peptide hormone released by enteroendocrine cells of the small intestine (duodenum and jejunum)
Name encodes its function: "cholecysto" = gallbladder, "kinin" = movement/stimulation
Released into the bloodstream; travels to multiple target organs
What triggers its release:
Fatty chyme entering the duodenum (fat is the primary trigger)
Partially digested proteins in the duodenum also stimulate CCK release
Effects of CCK:
Target Organ | Effect | Why It Makes Sense |
|---|---|---|
Pancreas (acinar cells) | Stimulates enzyme-rich pancreatic juice secretion (lipase, proteases, amylase) | More fat/protein arriving = more enzymes needed |
Gallbladder | Triggers contraction → releases stored bile into duodenum | Bile salts emulsify fat, increasing surface area for lipase |
Sphincter of Oddi | Causes relaxation → allows bile and pancreatic juice to flow into duodenum | Opens the gate at the hepatopancreatic ampulla |
Stomach — secretion | Inhibits gastric acid and enzyme secretion | Duodenum already processing a load — no more needed |
Stomach — muscularis | Decreases gastric motility and slows emptying | Prevents overwhelming duodenum with more chyme |
Brain | Contributes to satiety (feeling of fullness) | Signals that digestion is underway; reduces appetite |
CCK coordinates the simultaneous delivery of both bile and pancreatic enzymes to the duodenum — perfectly timed for fat and protein digestion
Neural Control — Enterogastric Reflex:
When acidic or fatty chyme enters the duodenum, stretch receptors and chemoreceptors in the duodenal wall send inhibitory signals back to the stomach
This reflex inhibits gastric secretion and motility via both the enteric nervous system and sympathetic fibers
Works in parallel with secretin and CCK to slow gastric emptying
Ensures the duodenum is not overwhelmed with more chyme than it can process, neutralize, and absorb
Organ-by-Organ Summary of Control Salivary Glands
Signal | Effect |
|---|---|
Parasympathetic (ACh) | Increases salivation — primary control of saliva |
Sympathetic | Decreases salivation — constricts arterioles to glands |
Sight/smell/taste of food | Activates parasympathetic → increased saliva (cephalic phase) |
No significant hormonal control of salivation
Older antidepressants with anticholinergic side effects block muscarinic receptors → xerostomia (dry mouth) → reduced antimicrobial protection → increased dental cavities and oral infections
Stomach
Signal | Type | Effect on Secretion | Effect on Motility |
|---|---|---|---|
Vagus nerve (ACh) | Neural (parasympathetic) | ↑ HCl, ↑ pepsinogen | ↑ churning and mixing |
Gastrin | Hormone | ↑ HCl, ↑ pepsinogen | ↑ motility |
Histamine | Paracrine | ↑ HCl (via H₂ receptor) | No significant effect |
Secretin | Hormone | ↓ HCl | ↓ motility |
CCK | Hormone | ↓ HCl | ↓ motility, slows emptying |
Enterogastric reflex | Neural (sympathetic/enteric) | ↓ secretion | ↓ motility |
Excess acid (low pH) | Local feedback | ↓ gastrin release → ↓ HCl | No direct effect |
Sympathetic activation | Neural | ↓ secretion | ↓ motility |
Pancreas
Signal | Type | Effect |
|---|---|---|
Vagus nerve (ACh) | Neural (parasympathetic) | ↑ pancreatic juice secretion (enzymes + bicarbonate) |
Secretin | Hormone | ↑ bicarbonate-rich juice specifically |
CCK | Hormone | ↑ enzyme-rich juice specifically |
Liver and Gallbladder
Signal | Type | Effect |
|---|---|---|
Vagus nerve (ACh) | Neural (parasympathetic) | ↑ bile secretion from liver; prepares gallbladder |
Secretin | Hormone | ↑ bile secretion from liver |
CCK | Hormone | Gallbladder contraction → bile released into duodenum; relaxes sphincter of Oddi |
Summary of the "Repeat Offenders" — Secretin and CCK
Feature | Secretin | CCK |
|---|---|---|
Produced by | Small intestine enteroendocrine cells | Small intestine enteroendocrine cells |
Released in response to | Acidic chyme | Fatty/protein-rich chyme |
Effect on pancreas | ↑ Bicarbonate secretion | ↑ Enzyme secretion |
Effect on gallbladder | ↑ Bile secretion | Contracts gallbladder → releases bile |
Effect on stomach secretion | ↓ HCl | ↓ HCl |
Effect on gastric motility | ↓ Slows emptying | ↓ Slows emptying |
Overall theme | Neutralize acid | Digest fat and protein |
Master Summary Table — All Phases
Phase | Trigger | Key Signals | Organs Affected | Net Effect |
|---|---|---|---|---|
Cephalic | Sight/smell/thought of food | Vagus nerve (ACh), gastrin | Salivary glands, stomach, pancreas, gallbladder | ↑ Secretion and motility throughout; primes digestive system |
Gastric | Food in stomach; distension; proteins; caffeine | Gastrin, histamine, ACh, myenteric/vagovagal reflexes | Stomach primarily | ↑ HCl, ↑ pepsinogen, ↑ motility; self-limited by low pH |
Intestinal | Acidic + fatty chyme in duodenum | Secretin, CCK, enterogastric reflex | Stomach (inhibit), pancreas, liver, gallbladder (stimulate) | ↓ Gastric activity; ↑ bicarbonate, ↑ enzymes, ↑ bile released |
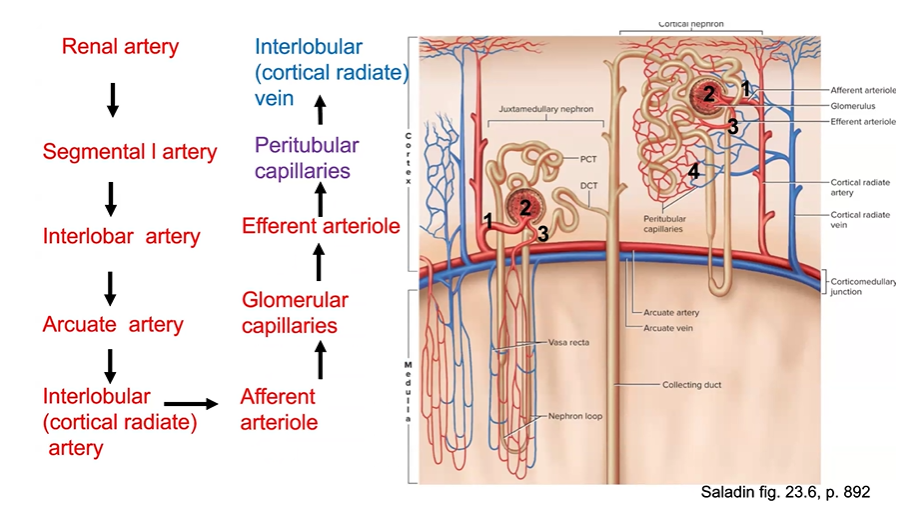
Trace the path taken by a drop of blood as it leaves the renal artery and travels through the kidney, exiting through the renal vein. Trace the path taken by a drop of urine as it leaves the renal papilla and exits the kidney through the ureter.
Functions of the Kidneys Overview
One of the most physiologically important organs — kidney function directly reflects and maintains virtually every aspect of internal homeostasis
A basic urinalysis (urine dipstick) is standard in routine checkups because abnormalities can reveal kidney disease, metabolic disorders, and infections non-invasively
1. Excretion and Elimination of Waste Products
Excretion — removes organic waste from body fluids
Elimination — expels waste from the body in urine
Waste products removed:
Urea — from amino acid breakdown; toxic if accumulated
Creatinine — from muscle metabolism
Uric acid — from nucleic acid breakdown
Bilirubin metabolites — from RBC breakdown
Drug metabolites and toxins
Accomplished via three processes:
Filtration — pressure-driven; pushes everything small out of blood at the glomerulus
Reabsorption — reclaims useful substances from filtrate back into blood
Secretion — moves substances from blood into tubular fluid for elimination
2. Regulation of Blood Pressure, Volume, and Solute Concentration
Kidneys regulate Na⁺, K⁺, and Cl⁻ — sodium balance governs blood volume and pressure (water follows sodium)
Slower than respiratory/cardiovascular regulation but essential for long-term stability
Key mechanisms:
Mechanism | Trigger | Effect |
|---|---|---|
RAAS | ↓ BP/volume → renin → angiotensin II | Vasoconstriction; ↑ aldosterone; ↑ ADH; triggers thirst |
Aldosterone | Angiotensin II or ↑ K⁺ | ↑ Na⁺ reabsorption + K⁺ secretion in late DCT/collecting duct → ↑ blood volume |
ADH | ↑ Blood osmolarity or ↓ volume | Inserts AQP2 → ↑ water reabsorption → concentrated urine |
ANP | ↑ Blood volume (atrial stretch) | ↑ GFR; inhibits RAAS/ADH; ↑ Na⁺ and water loss in urine |
3. Stabilizing Blood pH
Works alongside the respiratory system to maintain blood pH within 7.35–7.45
Lungs adjust pH quickly (CO₂); kidneys make slower, sustained corrections
Mechanisms:
H⁺ secretion — via Na⁺/H⁺ antiporters (PCT) and H⁺ pumps (collecting duct)
HCO₃⁻ reabsorption — via carbonic anhydrase mechanism in PCT; retains primary pH buffer
NH₄⁺ secretion — in PCT; additional route for acid elimination
Aldosterone also promotes H⁺ secretion in collecting duct — excess can cause metabolic alkalosis
The carbonic anhydrase reaction is central here — same reaction seen in respiratory gas transport and gastric HCl secretion
4. Detoxification
Kidneys convert lipid-soluble compounds into more water-soluble forms for urinary excretion
The PCT actively secretes drugs and toxins into tubular fluid — a major route for pharmaceutical clearance
When two drugs share the same secretory transporter → they compete → transport maximum (Tm) for each is reduced → clinically significant drug interactions
Creatinine clearance estimates GFR clinically — freely filtered, not reabsorbed or secreted
5. Conservation of Nutrients
Useful filtered substances are reabsorbed and returned to blood:
Glucose — entirely reabsorbed in PCT via SGLT; if blood glucose exceeds Tm → glucosuria (classic sign of uncontrolled diabetes)
Amino acids — Na⁺-coupled cotransport in PCT
Electrolytes — Na⁺, Cl⁻, K⁺, Ca²⁺, Mg²⁺, phosphate throughout the nephron
HCO₃⁻ — PCT and collecting duct
Water — obligatory in PCT (AQP1); regulated by ADH in DCT and collecting duct
6. Water Balance — Concentrated or Dilute Urine
Urine can range from 65–100 mOsm/L (dilute) to 1200 mOsm/L (concentrated)
Made possible by:
Countercurrent multiplier (loop of Henle) — builds medullary osmotic gradient (300 → 1200 mOsm/L)
Vasa recta — preserves gradient without washing it away
ADH present → AQP2 inserted → water reabsorbed → small concentrated urine
ADH absent → collecting duct impermeable → large dilute urine
Key principle: the kidney cannot replace lost water — it can only conserve what remains
7. Calcium and Phosphate Regulation
PTH released when blood Ca²⁺ falls:
Opens Ca²⁺ channels in DCT → ↑ Ca²⁺ reabsorption
Inhibits phosphate reabsorption in PCT → phosphate lost in urine
Also acts on bone (releases Ca²⁺) and intestine (↑ Ca²⁺ absorption)
Kidneys activate vitamin D → converts inactive form to calcitriol → promotes intestinal Ca²⁺ absorption
Kidney disease impairs this → Ca²⁺ deficiency and bone disease
8. Erythropoiesis Regulation
Kidneys produce erythropoietin (EPO) when low blood oxygen (hypoxia) is detected
EPO travels to bone marrow → stimulates RBC production and maturation → restores oxygen-carrying capacity
Classic negative feedback loop
Clinical relevance:
Chronic kidney disease → impaired EPO → anemia of chronic kidney disease
Treated with synthetic EPO (epoetin)
EPO famously abused by endurance athletes for blood doping
Blood and Urine Pathways Through the Kidney Overview
The kidney has a precisely organized vascular architecture with a consistent naming pattern shared between arteries and veins
One critical exception: there is no segmental vein — the venous pathway skips directly from interlobar vein to renal vein
The kidney uniquely features two capillary beds in series — the glomerular capillaries and the peritubular capillaries — separated by arterioles on both sides
Urine and blood travel through entirely separate pathways that never mix
Part 1 — Path of a Drop of Blood: Renal Artery → Renal Vein The Unique Feature: Two Capillary Beds in Series
Unlike typical systemic circulation (artery → capillaries → vein), the kidney has:
Afferent arteriole → glomerular capillaries → efferent arteriole → peritubular capillaries
This means blood passes through an arteriole on both sides of the first capillary bed
This arrangement allows independent regulation of pressure within the glomerulus
The efferent arteriole has a naturally smaller diameter than the afferent arteriole — one structural reason why glomerular pressure is inherently higher than in typical capillaries
Step-by-Step Arterial Path (Oxygenated Blood In)
Step 1 — Renal Artery:
Oxygenated blood enters the kidney at the hilum via the renal artery
The hilum is the medial indentation where the renal artery enters and the renal vein and ureter exit
Step 2 — Segmental Arteries:
The renal artery immediately branches into segmental arteries
Each segmental artery supplies a distinct segment of the kidney
NOTE: There is NO segmental vein on the venous side — this is a common exam trap
Step 3 — Interlobar Arteries:
Segmental arteries branch into interlobar arteries
Run between the renal pyramids of the medulla
Travel toward the corticomedullary junction
Step 4 — Arcuate Arteries:
Interlobar arteries branch into arcuate arteries
Arch over the base of each renal pyramid — hence the name "arcuate" (arc-shaped)
Sit at the corticomedullary junction
Run parallel to the kidney surface
Step 5 — Cortical Radiate Arteries (Interlobular Arteries):
Arcuate arteries branch into cortical radiate arteries (also called interlobular arteries)
Radiate outward from the arcuate arteries into the cortex
Run perpendicular to the kidney surface toward the outer cortex
Step 6 — Afferent Arterioles:
Cortical radiate arteries give off afferent arterioles
Each afferent arteriole delivers blood to one nephron's glomerulus
Constriction or dilation of the afferent arteriole directly controls blood flow into the glomerulus and therefore controls GFR
Step 7 — Glomerular Capillaries (Glomerulus):
The afferent arteriole feeds into the glomerulus — a tangled knot of capillaries sitting inside Bowman's capsule
This is the site of filtration — fluid is pushed out of the blood into Bowman's capsule
High hydrostatic pressure (~60 mmHg) drives filtration — much higher than typical systemic capillaries
Blood cells and large proteins remain in the blood; everything small is filtered out
The filtration membrane consists of three layers:
Fenestrations of glomerular endothelial cells
Basal lamina (basement membrane)
Filtration slits between podocyte pedicels
Step 8 — Efferent Arteriole:
Blood that was not filtered exits the glomerulus via the efferent arteriole
Still an arteriole (not a venule) — maintains pressure control
Because significant fluid was filtered out in the glomerulus, the blood in the efferent arteriole has:
Higher protein concentration (proteins were retained)
Higher colloid osmotic pressure than blood entering the afferent arteriole
This elevated osmotic pressure becomes the major driving force for reabsorption in the peritubular capillaries
Transition: Efferent Arteriole → Peritubular Capillaries
Step 9a — Peritubular Capillaries (Cortical Nephrons):
The efferent arteriole of cortical nephrons branches into peritubular capillaries
These capillaries surround the renal tubules in the cortex (PCT, DCT)
Low pressure, high osmotic pressure — ideal conditions for reabsorption of water and solutes from tubular fluid back into the blood
Also the site where secretion occurs — substances move from peritubular capillary blood into the tubular fluid
Step 9b — Vasa Recta (Juxtamedullary Nephrons):
The efferent arteriole of juxtamedullary nephrons (those with long loops of Henle) gives rise to vasa recta
Long, hairpin-shaped capillaries that dip deep into the medulla alongside the loop of Henle
Named separately only when in the medulla — they are still peritubular capillaries functionally
Their countercurrent arrangement (blood descends in one limb, ascends in the other) allows them to supply the medulla with nutrients and remove waste without washing out the medullary osmotic gradient
As blood descends into the concentrated medulla: water leaves, solutes enter
As blood ascends back toward cortex: the reverse occurs — net effect on gradient is minimal
Essential for maintaining the hyperosmotic medullary interstitium that enables urine concentration
Step-by-Step Venous Path (Deoxygenated Blood Out)
Step 10 — Cortical Radiate Veins (Interlobular Veins):
Peritubular capillaries and vasa recta drain into cortical radiate veins
Run back toward the arcuate veins
Step 11 — Arcuate Veins:
Cortical radiate veins drain into arcuate veins
Run along the corticomedullary junction, mirroring the arcuate arteries
Step 12 — Interlobar Veins:
Arcuate veins drain into interlobar veins
Run between the renal pyramids back toward the hilum
Step 13 — Renal Vein: ⚠ NO SEGMENTAL VEIN
Interlobar veins drain directly into the renal vein — skipping the segmental level entirely
There is no segmental vein — this asymmetry between the arterial and venous pathways is a critical and commonly tested point
The renal vein exits the kidney at the hilum
Drains into the inferior vena cava (IVC)
Complete Blood Flow Summary
Renal artery → Segmental artery → Interlobar artery → Arcuate artery → Cortical radiate artery → Afferent arteriole → Glomerular capillaries → Efferent arteriole → Peritubular capillaries / Vasa recta → Cortical radiate vein → Arcuate vein → Interlobar vein → Renal vein
Arterial Path | Venous Path |
|---|---|
Renal artery | Renal vein |
Segmental artery |
|
Interlobar artery | Interlobar vein |
Arcuate artery | Arcuate vein |
Cortical radiate artery | Cortical radiate vein |
Afferent arteriole | Peritubular capillaries / vasa recta |
Glomerular capillaries | — |
Efferent arteriole | — |
Part 2 — Path of a Drop of Urine: Renal Papilla → Ureter Overview
Urine is produced by the nephron through three processes: filtration, reabsorption, and secretion
By the time fluid reaches the renal papilla it is fully formed urine — all reabsorption and secretion is complete
The collecting system simply channels urine out of the kidney — no further modification occurs here
The pathway is a progressively widening funnel system
Step-by-Step Urine Flow
Step 1 — Renal Papilla:
The renal papilla is the pointed tip of each renal pyramid
This is where the collecting ducts open and release urine
Multiple collecting ducts converge and drain through tiny openings (called the area cribrosa) at the tip of the papilla
Urine drips from the papilla into the surrounding collecting structure
Step 2 — Minor Calyx:
Each renal papilla drips urine into a cup-shaped structure called a minor calyx
The minor calyx cups around the papilla like a hand cupped around a faucet
Each kidney has approximately 8–18 minor calyces
Function: collects urine from one renal papilla and channels it forward
Step 3 — Major Calyx:
Several minor calyces merge to form a major calyx
Each kidney typically has 2–3 major calyces
Function: collects urine from multiple minor calyces
Step 4 — Renal Pelvis:
The major calyces merge into the renal pelvis — a large, funnel-shaped basin
The renal pelvis is the central collecting reservoir of the kidney
Located at the hilum of the kidney
Function: collects all urine from the major calyces and funnels it into the ureter
Step 5 — Ureter:
The renal pelvis narrows and continues as the ureter
The ureter exits the kidney at the hilum
Two ureters (one per kidney) carry urine down to the urinary bladder
Urine is propelled through the ureter by peristaltic contractions of smooth muscle in the ureter wall — not by gravity alone
Important distinction: the ureter connects each kidney to the bladder (there are two); the urethra is the single tube that carries urine out of the body — these are commonly confused
Complete Urine Flow Summary
Collecting duct → Renal papilla (area cribrosa) → Minor calyx → Major calyx → Renal pelvis → Ureter → Urinary bladder → Urethra → External environment
For Completeness — Urine Formation Within the Nephron (Before Reaching the Papilla)
Nephron Segment | Key Process | What Happens |
|---|---|---|
Glomerulus / Bowman's capsule | Filtration | Protein-free plasma forced into tubule; ~125 mL/min produced |
Proximal convoluted tubule (PCT) | Bulk reabsorption + secretion | ~65% of filtrate reabsorbed; glucose, amino acids, Na⁺, water, HCO₃⁻ reclaimed; H⁺, drugs, creatinine secreted |
Descending limb of loop of Henle | Water reabsorption | Water leaves by osmosis; filtrate becomes concentrated |
Ascending limb of loop of Henle | Solute reabsorption | Na⁺, K⁺, Cl⁻ pumped out via NKCC; water cannot follow; filtrate becomes dilute |
Distal convoluted tubule (DCT) | Fine-tuning | Na⁺/Cl⁻ reabsorption; Ca²⁺ reabsorption regulated by PTH; aldosterone begins to act |
Collecting duct | Final concentration | ADH controls aquaporin-2 insertion → water reabsorption; aldosterone controls Na⁺/K⁺; urea recycling |
Renal papilla | Exit point | Fully formed urine drips into minor calyx |
Side-by-Side Comparison: Blood vs. Urine Pathways
Feature | Blood Pathway | Urine Pathway |
|---|---|---|
Entry/start point | Renal artery (at hilum) | Collecting duct (in medulla) |
First structure | Segmental artery | Renal papilla |
Functional capillary beds | Glomerulus + peritubular capillaries/vasa recta | N/A — purely collecting structures |
Key functional site | Glomerulus (filtration); peritubular capillaries (reabsorption/secretion) | No modification — purely drainage |
Collecting structures | Sinusoids → central vein → hepatic veins (within lobule) | Minor calyx → major calyx → renal pelvis |
Exit point | Renal vein (at hilum) | Ureter (at hilum) |
Notable asymmetry | No segmental vein on venous side | N/A |
Propulsion mechanism | Cardiac output / blood pressure | Peristalsis of ureter smooth muscle |
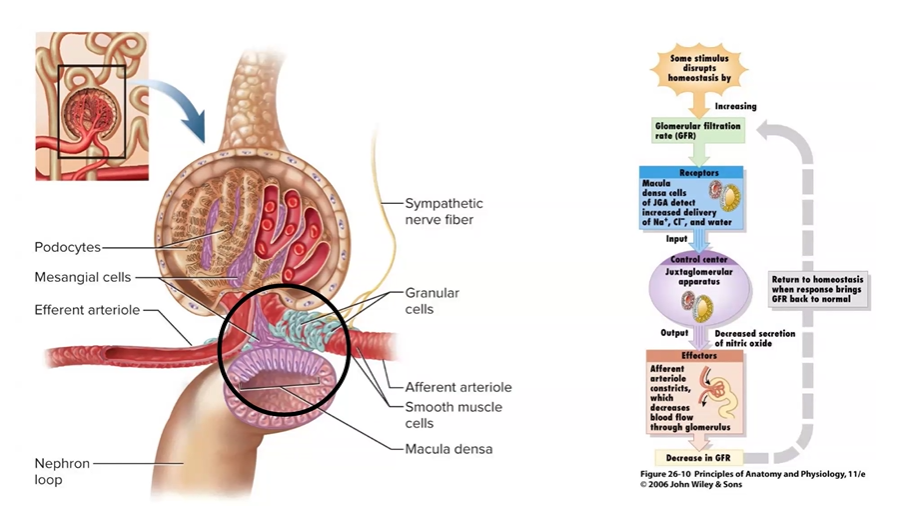
Describe filtration in the glomerulus. Identify the three forces that produce filtration. Describe autoregulatory, hormonal and neuronal mechanisms that regulate the rate of filtration.
Basic Processes of Urine Formation: Filtration, Reabsorption, and Secretion Overview
Urine formation involves three fundamental processes operating sequentially and simultaneously throughout the nephron
The formula: Filtered load − Reabsorbed + Secreted = Excreted in urine
General principle:
Filtration — blunt instrument; pushes everything small out of blood indiscriminately
Reabsorption — reclaims what the body needs
Secretion — adds what wasn't filtered but still needs to be eliminated
Process 1 — Filtration Overview
Passive, pressure-driven process — no ATP required directly
Occurs exclusively at the renal corpuscle (glomerulus + Bowman's capsule) in the renal cortex
Produces ~125 mL/min (~180 L/day) of protein-free plasma filtrate
Of this, ~99% is reabsorbed — only 1.5–2 L becomes urine
The Filtration Membrane — Three Layers
Every substance leaving the glomerular capillaries must cross three barriers:
Fenestrations of glomerular endothelial cells — pores block blood cells; first size-based barrier
Basal lamina — acellular molecular sieve; blocks large proteins; negatively charged → repels albumin electrostatically
Filtration slits between podocyte pedicels — interdigitate like zipper teeth; slit diaphragm blocks medium-sized proteins
Result: filtrate = protein-free plasma
Freely filtered: water, Na⁺, K⁺, Cl⁻, HCO₃⁻, glucose, amino acids, urea, creatinine, small drugs
Not filtered: RBCs, WBCs, platelets, albumin, globulins
Proteinuria = filtration membrane damage → kidney disease
Hematuria = severe membrane damage
The Three Forces — Net Filtration Pressure (NFP) NFP=GBHP−(BCOP+CHP)=60−(32+18)=+10 mmHgNFP = GBHP - (BCOP + CHP) = 60 - (32 + 18) = +10 \text{ mmHg}NFP=GBHP−(BCOP+CHP)=60−(32+18)=+10 mmHg
Force | Value | Direction | Effect |
|---|---|---|---|
GBHP (glomerular blood hydrostatic pressure) | 60 mmHg | Pushes fluid OUT | Promotes filtration — primary driving force |
BCOP (blood colloid osmotic pressure) | 32 mmHg | Pulls fluid IN | Opposes filtration — plasma proteins retained in blood pull fluid back |
CHP (capsular hydrostatic pressure) | 18 mmHg | Pushes back IN | Opposes filtration — fluid in Bowman's capsule resists more entering |
Positive NFP = filtration occurring
Zero or negative NFP = filtration ceases — unlike systemic capillaries, negative NFP at the glomerulus does NOT cause reabsorption back into capillaries; filtration simply stops
This is pathological — not normal physiology
Clinical Examples
Scenario | Change | Effect on GFR |
|---|---|---|
Elevated blood pressure | ↑ GBHP | ↑ GFR |
Liver cirrhosis / malnutrition | ↓ BCOP (fewer proteins) | ↑ GFR |
Kidney stone blocking ureter | ↑ CHP | ↓ GFR |
Severe hemorrhage / shock | ↓ GBHP | ↓ GFR → may cease |
GBHP drops to 40 mmHg | ↓ GBHP | NFP = −10 → filtration ceases |
Glomerular Filtration Rate (GFR) and Its Regulation
GFR = total filtrate produced by both kidneys per minute (~125 mL/min)
Kept remarkably stable across MAP ~80–160 mmHg by autoregulatory mechanisms
Estimated clinically via creatinine clearance — creatinine is freely filtered, minimally reabsorbed, and secreted in small amounts
Effect of Arteriole Diameter on GFR
Arteriole | Change | Effect on Glomerular Pressure | Effect on GFR |
|---|---|---|---|
Afferent | Constriction | ↓ less blood entering | ↓ GFR |
Afferent | Dilation | ↑ more blood entering | ↑ GFR |
Efferent | Constriction | ↑ blood backs up | ↑ GFR |
Efferent | Dilation | ↓ blood drains freely | ↓ GFR |
Autoregulation of GFR — Two Intrinsic Mechanisms
Both operate locally without brain or hormonal input and act primarily on the afferent arteriole
Myogenic Mechanism:
↑ BP → afferent arteriole wall stretched → voltage-gated Ca²⁺ channels open → Ca²⁺ influx → smooth muscle contracts → afferent arteriole constricts → ↓ glomerular pressure → GFR returns to normal
Reverse occurs with ↓ BP → less stretch → dilation → ↑ GFR back to normal
Rapid, second-to-second response
Chronic hypertension can overwhelm this → glomerular damage → progressive kidney disease
Tubuloglomerular Feedback:
The ascending limb / early DCT loops back to make direct physical contact with the afferent arteriole of its own nephron → forms the juxtaglomerular apparatus (JGA)
JGA contains three cell types:
Macula densa cells — detect Na⁺/Cl⁻ concentration in tubular fluid
Granular (JG) cells — modified smooth muscle in afferent arteriole wall; release renin
Mesangial cells — between capillary loops; adjust filtration surface area
↑ GFR → faster tubular flow → less time for Na⁺/Cl⁻ reabsorption → macula densa detects high Na⁺/Cl⁻ → paracrine signals → afferent arteriole constricts → ↓ GFR back to normal
↓ GFR → opposite occurs → afferent arteriole dilates → ↑ GFR back to normal
Classic negative feedback loop — solute concentration in tubular fluid used as proxy for filtration rate
Hormonal Regulation of GFR
RAAS (Renin-Angiotensin-Aldosterone System):
Trigger: ↓ BP/volume → granular cells release renin → angiotensinogen → angiotensin I → angiotensin II (via ACE in lungs)
ACE inhibitors block this conversion step — common antihypertensives
Angiotensin II effects:
Target | Effect | Result |
|---|---|---|
Systemic vessels | Vasoconstriction | ↑ peripheral resistance → ↑ BP |
Afferent + efferent arterioles | Constriction (greater on efferent) | Net ↓ GFR |
Mesangial cells | Contraction | ↓ filtration surface area → ↓ GFR |
Adrenal cortex | ↑ Aldosterone | ↑ Na⁺ reabsorption → ↑ blood volume |
Posterior pituitary | ↑ ADH | ↑ water reabsorption → ↑ blood volume |
Brain | Thirst | ↑ fluid intake → ↑ blood volume |
ANP (Atrial Natriuretic Peptide):
Trigger: ↑ blood volume → atrial stretch → ANP released
Physiological counterweight to RAAS
Effects: dilates afferent arteriole → ↑ GFR; relaxes mesangial cells → ↑ filtration surface; inhibits renin, aldosterone, ADH → ↑ Na⁺ and water loss in urine → ↓ BP
Feature | RAAS | ANP |
|---|---|---|
Trigger | ↓ BP/volume | ↑ BP/volume |
Afferent arteriole | Constricts → ↓ GFR | Dilates → ↑ GFR |
Aldosterone/ADH | ↑ both | ↓ both |
Net effect | ↑ BP; ↓ urine output | ↓ BP; ↑ urine output |
Neural Regulation of GFR
Kidney innervated exclusively by sympathetic division — no significant parasympathetic innervation
Sympathetic activation (fight-or-flight) → norepinephrine/epinephrine → constricts afferent arteriole → ↓ GFR + stimulates renin release → activates RAAS
Physiological sense: during stress, blood diverted to muscles/heart; urine production suppressed (SLUD = parasympathetic functions; sympathetic suppresses all)
Process 2 — Reabsorption Overview
Movement of substances FROM tubular fluid BACK INTO blood via peritubular capillaries
Called reabsorption — substances were originally in blood, filtered out, and recaptured (distinct from GI absorption where nutrients enter blood for the first time)
Occurs throughout the entire tubule and collecting duct — PCT handles the bulk (~65%)
~99% of 180 L/day filtrate reabsorbed → only 1.5–2 L excreted as urine
Routes Across the Tubular Epithelium
Paracellular — between cells through tight junctions; passive diffusion down gradients
Transcellular — through cells; crosses apical membrane (faces lumen) then basolateral membrane (faces blood); requires carriers, pumps, channels
The Master Pump — Na⁺/K⁺ ATPase
Located on basolateral membrane of all tubular cells throughout the entire nephron
Pumps 3 Na⁺ OUT and 2 K⁺ IN using ATP (primary active transport)
Maintains low intracellular Na⁺ → creates electrochemical gradient driving Na⁺ from lumen into cell
Powers virtually all secondary active transport — glucose, amino acids, HCO₃⁻ reabsorption all depend on this Na⁺ gradient
Most important transporter in the entire nephron
Reabsorption by Segment Proximal Convoluted Tubule (PCT) — The Workhorse
Handles ~65–70% of all reabsorption
Dense brush border (microvilli) on apical surface — massively increases surface area
Packed with mitochondria — reflects high energy demand
Substance | Apical Mechanism | Basolateral Exit | Notes |
|---|---|---|---|
Na⁺ | Na⁺/H⁺ antiporter; SGLT cotransporters | Na⁺/K⁺ ATPase | Drives reabsorption of nearly everything else |
Glucose | SGLT (secondary active — Na⁺ cotransport) | GLUT (facilitated diffusion) | 100% reabsorbed normally; glucosuria if Tm exceeded |
Amino acids | Na⁺-coupled cotransporters (secondary active) | Facilitated diffusion | Multiple carriers for different amino acid classes |
HCO₃⁻ | Na⁺/H⁺ antiporter → H⁺ + HCO₃⁻ → CO₂ (carbonic anhydrase) → diffuses in → reassembled as HCO₃⁻ | HCO₃⁻/Cl⁻ antiporter | Links H⁺ secretion to HCO₃⁻ reabsorption |
Water | AQP1 (aquaporin-1) — osmosis | AQP1 | Obligatory — always occurs regardless of hydration status; ~65% filtered water reabsorbed here |
Cl⁻, K⁺, Ca²⁺, Mg²⁺, phosphate | Various cotransporters and channels | Various | Follow electrochemical gradients established by Na⁺ reabsorption |
Urea, lipid-soluble substances | Simple diffusion | Simple diffusion | Passive; follow concentration gradient |
Loop of Henle
Segment | Water Permeability | Solute Transport | Mechanism | Effect on Tubular Fluid |
|---|---|---|---|---|
Descending limb | Freely permeable (AQP1) | Impermeable to most solutes | Osmosis — water exits into concentrated medullary interstitium | Fluid becomes progressively more concentrated (→ ~1200 mOsm/L at tip) |
Thick ascending limb | Impermeable — no aquaporins | Actively transports Na⁺, K⁺, Cl⁻ out | NKCC cotransporter + Na⁺/K⁺ ATPase; K⁺ leaks back via apical channels | Fluid becomes progressively more dilute (~100 mOsm/L at DCT); medullary gradient built |
Loop diuretics (furosemide/Lasix) block NKCC → prevent solute reabsorption → collapse medullary gradient → large dilute urine output
Distal Convoluted Tubule (DCT)
Segment | Substance | Mechanism | Regulated By |
|---|---|---|---|
Early DCT | Na⁺ + Cl⁻ | NCC symporter | — |
Early DCT | Water | Does NOT follow — impermeable | — |
DCT | Ca²⁺ | Ca²⁺ channels (apical) → Ca²⁺ ATPase (basolateral) | PTH — opens Ca²⁺ channels when blood Ca²⁺ falls |
Late DCT | Na⁺ | ENaC channels + Na⁺/K⁺ ATPase | Aldosterone |
Thiazide diuretics block NCC in early DCT → reduce Na⁺ reabsorption → moderate diuresis → used for hypertension
Collecting Duct — Fine-Tuned Hormonal Control
Principal cells:
Substance | Direction | Mechanism | Regulated By |
|---|---|---|---|
Na⁺ | Tubule → blood | ENaC channels (apical) + Na⁺/K⁺ ATPase (basolateral) | Aldosterone — ↑ ENaC + Na⁺/K⁺ ATPase expression |
K⁺ | Blood → tubule (secretion) | Apical K⁺ leak channels | Aldosterone — primary route of K⁺ excretion |
Water | Tubule → blood | AQP2 (apical — inserted by ADH); AQP3/4 (basolateral — constitutive) | ADH — inserts AQP2 only when present |
ADH present (dehydrated): AQP2 inserted → water reabsorbed at each level as collecting duct passes through hyperosmotic medullary gradient → small concentrated urine (~1200 mOsm/L)
ADH absent (well-hydrated): no AQP2 → collecting duct impermeable → large dilute urine (~65–100 mOsm/L)
Urea recycling:
Inner medullary collecting duct permeable to urea → urea diffuses out into medullary interstitium → contributes ~50% of deep medullary osmolarity → amplifies concentrating ability
Intercalated cells (pH regulation):
Type A (acidosis): secrete H⁺ via H⁺ ATPase; reabsorb HCO₃⁻ → blood pH rises
Type B (alkalosis): secrete HCO₃⁻; reabsorb H⁺ → blood pH falls
Transport Maximum (Tm)
All carrier-mediated reabsorption has a maximum rate — when all carriers are saturated, excess solute cannot be reabsorbed and appears in urine
Glucosuria in diabetes = plasma glucose exceeds Tm for SGLT → not because kidney is broken but because glucose load exceeds carrier capacity
Competition — two solutes sharing the same transporter reduce each other's effective Tm → clinically significant drug interactions
Process 3 — Secretion Overview
Movement of substances FROM blood (peritubular capillaries) INTO tubular fluid
Opposite direction of reabsorption
Allows elimination of substances not filtered at the glomerulus
Fine-tunes pH and ionic composition of urine
Occurs throughout the tubule — PCT is the major site for drugs, toxins, and H⁺; collecting duct fine-tunes K⁺ and H⁺
What Is Secreted and Where H⁺ — Throughout the Tubule
PCT: secreted via Na⁺/H⁺ antiporter (secondary active transport) — as Na⁺ moves in, H⁺ is pumped out
Secreted H⁺ combines with filtered HCO₃⁻ → H₂CO₃ → CO₂ + H₂O (carbonic anhydrase) → CO₂ diffuses into cell → reassembled as HCO₃⁻ → reabsorbed to blood
Links H⁺ secretion directly to HCO₃⁻ reabsorption — tightly couples pH regulation to ion transport
Collecting duct (Type A intercalated cells): secreted via H⁺ ATPase (primary active transport) — directly uses ATP
Kidney is the primary long-term regulator of blood pH — respiratory system makes fast adjustments; kidneys make slower sustained corrections
Same carbonic anhydrase reaction seen in respiratory gas transport and gastric HCl secretion
NH₄⁺ — PCT
Secreted via Na⁺/H⁺ antiporter — NH₄⁺ substitutes for H⁺ on the antiporter
Produced inside tubular cells from glutamine metabolism
Additional route for acid elimination — increases during acidosis
K⁺ — Collecting Duct (Primary Route of K⁺ Excretion)
Principal cells of late DCT and collecting duct
Na⁺/K⁺ ATPase pumps K⁺ INTO the cell → K⁺ exits through apical K⁺ leak channels into tubular fluid
Regulated by aldosterone — increases Na⁺/K⁺ ATPase activity and apical K⁺ channel expression
Hyperkalemia directly stimulates aldosterone → more K⁺ secretion → negative feedback
Excess aldosterone → hypokalemia (over-stimulated K⁺ secretion)
Creatinine — PCT
Secreted AND freely filtered
Because creatinine is filtered + secreted with minimal reabsorption → used clinically to estimate GFR
Rising blood creatinine = declining GFR and kidney function
Drugs and Toxins — PCT (Major Route)
Multiple organic acid and organic base secretory transporters in PCT actively secrete pharmaceuticals into tubular fluid
When two drugs share the same transporter → compete for the carrier → Tm for each effectively reduced → one or both drugs accumulate to toxic levels
Classic example: probenecid blocks the organic acid transporter that secretes penicillin → prolongs penicillin's action by reducing its renal clearance
Integration — How All Three Processes Work Together
Process | Site | Direction | Purpose |
|---|---|---|---|
Filtration | Renal corpuscle only | Blood → tubule | Indiscriminately pushes everything small out of blood |
Reabsorption | Entire tubule + collecting duct | Tubule → blood | Reclaims useful substances (glucose, amino acids, water, electrolytes) |
Secretion | Entire tubule + collecting duct | Blood → tubule | Adds waste, drugs, excess ions, and acids not captured by filtration |
Clinical Correlations Summary
Finding | Process Affected | Cause |
|---|---|---|
Proteinuria | Filtration | Filtration membrane damage — hallmark of kidney disease |
Hematuria | Filtration | Severe filtration membrane damage |
Glucosuria | Reabsorption | Tm for glucose exceeded — classic sign of diabetes mellitus |
Drug toxicity from interactions | Secretion | Competition for PCT transporters — one drug blocks clearance of another |
Massive dilute urine | Reabsorption | Loop diuretics blocking NKCC; or absent ADH (diabetes insipidus) |
Hypokalemia | Secretion | Excessive aldosterone → over-stimulation of K⁺ secretion |
Rising blood creatinine | Filtration + secretion | Declining GFR → reduced creatinine clearance → kidney disease |
Master Summary Table Three Forces Governing Filtration
Force | Normal Value | Direction | Effect on GFR |
|---|---|---|---|
GBHP | 60 mmHg | Pushes OUT | Promotes filtration |
BCOP | 32 mmHg | Pulls IN | Opposes filtration |
CHP | 18 mmHg | Pushes back IN | Opposes filtration |
NFP | +10 mmHg | Net outward | Filtration occurs |
Regulatory Mechanisms Summary
Mechanism | Type | Trigger | Afferent Arteriole | Effect on GFR |
|---|---|---|---|---|
Myogenic | Autoregulation | ↑ Stretch → ↑ BP | Constricts | ↓ Back to normal |
Tubuloglomerular feedback | Autoregulation | ↑ Na⁺/Cl⁻ at macula densa | Constricts | ↓ Back to normal |
RAAS (Angiotensin II) | Hormonal | ↓ BP / volume | Constricts | ↓ GFR; ↑ BP overall |
ANP | Hormonal | ↑ BP / volume | Dilates | ↑ GFR; ↓ BP overall |
Sympathetic NS | Neural | Stress / fight-or-flight | Constricts | ↓ GFR |
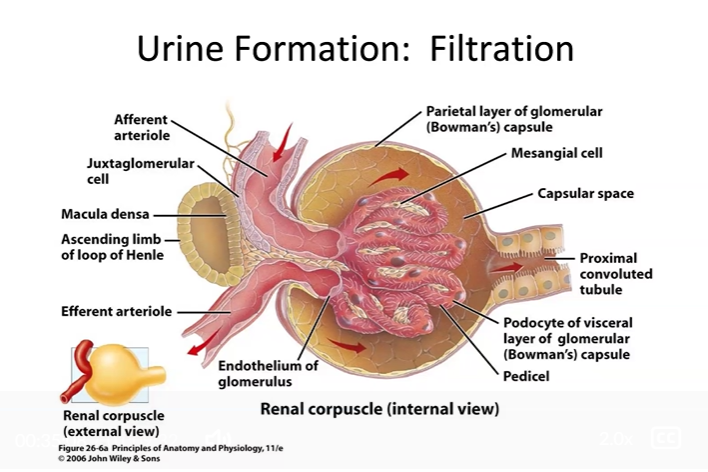
Name and describe the processes involved in the formation of urine. Be sure you can describe the movement of solutes and water (ie, from the kidney tubule to the body; from the body to the kidney tubule; etc.)
Formation of Urine: Filtration, Reabsorption, and Secretion Overview
Urine formation involves three fundamental processes that together determine the final composition and volume of urine
These three processes occur in sequence along the nephron:
Filtration — at the renal corpuscle only
Reabsorption — along the entire tubule and collecting duct
Secretion — along the entire tubule and collecting duct
The general principle:
Filtration is a blunt instrument — pushes almost everything out of the blood indiscriminately
Reabsorption reclaims what the body needs
Secretion adds what wasn't filtered but still needs to be eliminated
Direction conventions used throughout:
Tubule → blood = reabsorption
Blood → tubule = secretion
Process 1 — Filtration What It Is
A passive, pressure-driven process
Occurs exclusively at the renal corpuscle (glomerulus + Bowman's capsule)
Fluid is pushed from the glomerular capillaries into the capsular space of Bowman's capsule
Produces approximately 125 mL/min or 180 L/day of filtrate
Direction of Movement
Blood → tubule (glomerular capillaries → Bowman's capsule)
What Is Filtered
Freely filtered (pass through all three membrane layers):
Water, Na⁺, K⁺, Cl⁻, HCO₃⁻, glucose, amino acids, urea, creatinine, drugs, small toxins
Not filtered (too large):
Red blood cells, white blood cells, platelets
Large plasma proteins (albumin, globulins)
Result = protein-free plasma in the capsular space
The Three Forces (NFP = +10 mmHg)
GBHP (60 mmHg) — pushes fluid OUT → promotes filtration
BCOP (32 mmHg) — pulls fluid IN → opposes filtration
CHP (18 mmHg) — pushes back IN → opposes filtration
NFP = 60 − (32 + 18) = +10 mmHg → net filtration occurs
The Filtration Membrane (Three Layers)
Fenestrations of glomerular endothelial cells — blocks blood cells
Basal lamina — molecular sieve; blocks large proteins; negatively charged
Filtration slits between podocyte pedicels — slit diaphragm blocks medium proteins
Clinical Relevance
Proteinuria = damage to filtration membrane → proteins leak through → kidney disease
Hematuria = severe damage → blood cells appear in urine
If GBHP drops to 40 mmHg: NFP = 40 − (32+18) = −10 mmHg → filtration ceases entirely
Process 2 — Reabsorption What It Is
Movement of substances FROM the tubular fluid BACK INTO the blood (peritubular capillaries)
Occurs along the entire length of the renal tubule and collecting duct
The body reclaims useful materials that were filtered out
Called reabsorption (not absorption) because these substances were originally in the blood, filtered out, and are being recaptured — distinguishing it from absorption in the digestive system where nutrients enter blood for the first time
Direction of Movement
Tubule → peritubular capillaries → blood
Why the Peritubular Capillaries Favor Reabsorption
Because filtration in the glomerulus removes large volumes of fluid while retaining proteins, the blood in the efferent arteriole has:
Higher protein concentration than normal
Higher colloid osmotic pressure than blood entering the afferent arteriole
This elevated osmotic pressure in peritubular capillaries is a major driving force pulling reabsorbed fluid back into the blood
Routes of Transport Across the Tubular Epithelium
Paracellular route — substances move between epithelial cells through tight junctions; driven by concentration or electrochemical gradients; passive diffusion
Transcellular route — substances move through epithelial cells; crossing both:
Apical membrane (faces tubular lumen)
Basolateral membrane (faces interstitial fluid and peritubular capillaries)
Carrier proteins, pumps, and channels facilitate movement across each membrane
The Master Pump: Na⁺/K⁺ ATPase
Located on the basolateral membrane of virtually all tubular epithelial cells
Continuously pumps Na⁺ OUT of the cell and K⁺ IN using ATP (primary active transport)
Keeps intracellular Na⁺ concentration low
Creates a steep electrochemical gradient that drives Na⁺ from tubular fluid into the cell across the apical membrane
This Na⁺ gradient then powers the cotransport of glucose, amino acids, and other solutes via secondary active transport
The Na⁺/K⁺ ATPase is the engine that drives most reabsorption throughout the nephron
Reabsorption in the Proximal Convoluted Tubule (PCT)
Overview:
The workhorse of reabsorption — handles the bulk (~65%) of all reabsorption
Luminal (apical) surface is densely covered with microvilli (brush border) — massively increases surface area for transport
Both reabsorption and secretion occur here
Sodium (Na⁺)
Direction: Tubule → blood
Na⁺ enters the cell across the apical membrane via multiple cotransporters (with glucose, amino acids) and antiporters (Na⁺/H⁺)
Na⁺ is pumped out the basolateral membrane via Na⁺/K⁺ ATPase into the interstitium → diffuses into peritubular capillaries
Na⁺ reabsorption drives reabsorption of almost everything else
Glucose
Direction: Tubule → blood
Cotransported with Na⁺ across the apical membrane via SGLT (sodium-glucose cotransporters) — secondary active transport
Exits across the basolateral membrane via GLUT (facilitated diffusion transporters)
Enters the peritubular capillaries → mesenteric veins → portal vein → liver
Virtually all filtered glucose is reabsorbed under normal conditions
Transport maximum (Tm): SGLT transporters are present in finite numbers
When plasma glucose is very high (e.g., uncontrolled diabetes mellitus), filtrate glucose exceeds Tm
Excess glucose cannot be reabsorbed → remains in tubular fluid → glucosuria (glucose in urine)
This is NOT because the kidney is broken — it is because the Tm for glucose transporters is exceeded
Amino Acids
Direction: Tubule → blood
Cotransported with Na⁺ across the apical membrane via specific amino acid cotransporters
Exit through basolateral membrane via facilitated diffusion
Enter peritubular capillaries → portal circulation
Bicarbonate (HCO₃⁻)
Direction: Tubule → blood
Not directly transported — reclaimed through a carbonic anhydrase mechanism:
H⁺ is secreted into the tubule via Na⁺/H⁺ antiporter
H⁺ combines with filtered HCO₃⁻ in the lumen → H₂CO₃ → CO₂ + H₂O
CO₂ diffuses into the tubular cell → carbonic anhydrase → H₂CO₃ → H⁺ + HCO₃⁻
HCO₃⁻ exits the basolateral membrane into the blood
Net effect: HCO₃⁻ is reclaimed from the filtrate
Water
Direction: Tubule → blood (obligatory in PCT)
Follows solute reabsorption by osmosis through aquaporins (water channels)
Water reabsorption in the PCT is obligatory — always occurs regardless of body hydration status
As Na⁺, glucose, and other solutes are removed, the tubular fluid becomes slightly hypotonic
Water follows osmotically through aquaporins in the apical and basolateral membranes
Enters the peritubular capillaries
Other Ions
Cl⁻, K⁺, Ca²⁺, Mg²⁺, phosphate — reabsorbed via various cotransporters and channels
Follow the electrochemical gradients established by Na⁺ reabsorption
Lipid-Soluble Substances and Urea
Reabsorbed by simple diffusion across cell membranes
Urea is partially reabsorbed here (the rest is handled in the collecting duct)
Secretion in the PCT
H⁺ — secreted via Na⁺/H⁺ antiporter (as Na⁺ moves in, H⁺ is pumped out) → important for pH regulation
NH₄⁺ (ammonium ions) — secreted; another mechanism for acid excretion
Creatinine — secreted; used clinically to estimate GFR
Drugs and toxins — the PCT is a major route for clearing pharmaceuticals from the body
Direction for all secretion: blood → tubule
Reabsorption in the Loop of Henle
Overview:
The loop of Henle is essential for establishing the osmotic gradient in the renal medulla
This gradient is what makes it possible for the kidney to produce concentrated urine
The descending and ascending limbs have opposite permeability properties — this difference is the key to the entire system
Descending Limb
Freely permeable to WATER
Relatively impermeable to solutes
As filtrate descends into the increasingly concentrated medullary interstitium:
Water moves OUT by osmosis through aquaporins → enters the interstitium → picked up by vasa recta
Solutes remain → tubular fluid becomes progressively more concentrated
Reaches maximum concentration (~1200 mOsm/L) at the tip of the loop
Ascending Limb (Thick Portion)
Impermeable to WATER — no aquaporins
Actively transports solutes OUT
Contains the Na⁺-K⁺-2Cl⁻ (NKCC) cotransporter on the apical membrane:
Moves 1 Na⁺, 1 K⁺, and 2 Cl⁻ from tubular fluid into the cell simultaneously
Na⁺ then pumped out basally via Na⁺/K⁺ ATPase
K⁺ leaks back into the tubular lumen
Net: Na⁺, K⁺, Cl⁻ removed from tubular fluid without water following
Tubular fluid becomes progressively more dilute as it ascends
Medullary interstitium becomes progressively more concentrated
By the time filtrate reaches the DCT it is hypotonic (~100 mOsm/L)
Clinical relevance:
Loop diuretics (e.g., furosemide/Lasix) block the NKCC cotransporter
Prevents establishment of the medullary concentration gradient
Results in large volumes of dilute urine → used to treat fluid overload, hypertension, heart failure
Countercurrent Multiplier
The descending and ascending limbs run parallel with fluid flowing in opposite directions
Their different permeability properties create a positive feedback amplification:
Water leaving the descending limb concentrates the tubular fluid
Solutes pumped out of the ascending limb concentrate the interstitium
The more concentrated the interstitium, the more water is drawn from the descending limb
This progressively builds a steep osmotic gradient from cortex (~300 mOsm/L) to deep medulla (~1200 mOsm/L)
Vasa Recta — Countercurrent Exchanger
Long hairpin-shaped capillaries running alongside the loop of Henle
Their countercurrent arrangement preserves the medullary gradient:
As blood descends: water leaves, solutes enter
As blood ascends: water re-enters, solutes leave
Net effect on the gradient: minimal
Supply the medulla with nutrients and remove waste without washing out the osmotic gradient
Reabsorption in the Distal Convoluted Tubule (DCT) Early DCT
Na⁺ and Cl⁻ reabsorbed via Na⁺-Cl⁻ symporter (NCC transporter)
Direction: tubule → blood
Water does NOT follow — early DCT is impermeable to water
Tubular fluid continues to become more dilute
Calcium (Ca²⁺)
Direction: Tubule → blood
Reabsorption in the DCT is regulated by parathyroid hormone (PTH)
When blood calcium falls → PTH released → opens Ca²⁺ channels in DCT apical membrane
Ca²⁺ enters the tubular cell and exits through the basolateral membrane → enters blood
Net: promotes Ca²⁺ reabsorption when blood calcium is low
Reabsorption and Secretion in the Collecting Duct
Overview:
Site of fine-tuned, hormonally regulated reabsorption and secretion
Determines the final composition and volume of urine
Two major cell types:
Principal cells — handle Na⁺, K⁺, and water
Intercalated cells — handle acid-base balance (H⁺ and HCO₃⁻)
Principal Cells — Na⁺ Reabsorption
Direction: Tubule → blood
Na⁺ enters the cell from the tubular fluid through ENaC (epithelial sodium channels) on the apical membrane
Driven by low intracellular Na⁺ maintained by basolateral Na⁺/K⁺ ATPase
Na⁺ is pumped out the basolateral membrane by Na⁺/K⁺ ATPase → enters peritubular capillaries
Aldosterone dramatically increases this process:
Increases number and activity of ENaC channels on the apical membrane
Increases Na⁺/K⁺ ATPase activity on the basolateral membrane
Net: more Na⁺ reabsorbed → water follows → blood volume and pressure increase
Principal Cells — K⁺ Secretion
Direction: Blood → tubule (secretion)
As Na⁺ is pumped out by Na⁺/K⁺ ATPase, K⁺ is pumped into the cell
K⁺ then exits through apical leak channels into the tubular fluid → excreted in urine
This is the primary route of potassium excretion in the kidney
Aldosterone also promotes K⁺ secretion — which is why aldosterone excess causes hypokalemia (low blood K⁺)
Principal Cells — Water Reabsorption (ADH-Dependent)
Direction: Tubule → blood
Water reabsorption here is NOT obligatory — it depends entirely on ADH (antidiuretic hormone / vasopressin)
When body is dehydrated or blood osmolarity rises:
ADH released from the posterior pituitary
ADH acts on principal cells → triggers insertion of aquaporin-2 (AQP2) water channels into the apical membrane
Water moves by osmosis from tubular lumen → through the cell → into the hyperosmotic medullary interstitium → into the blood
Result: small volume of concentrated urine
When ADH is absent (well-hydrated state or diabetes insipidus):
No aquaporin-2 channels inserted
Collecting duct is impermeable to water
Water stays in the tubule → excreted
Result: large volume of dilute urine
Urea Recycling
The inner medullary collecting duct is permeable to urea
Urea diffuses out of the collecting duct → into the medullary interstitium
Contributes significantly to the high osmolarity of the deep medulla (~half of medullary osmolarity at the papilla)
This urea then enters the descending limb of the loop of Henle and recirculates — called urea recycling
Amplifies the medullary concentration gradient → enhances the kidney's ability to concentrate urine
Bicarbonate Reabsorption and H⁺ Secretion
Intercalated cells handle acid-base balance in the collecting duct
H⁺ secreted into tubular fluid → excreted in urine → lowers blood acidity
HCO₃⁻ reabsorbed → enters blood → raises blood pH
Important mechanism for long-term blood pH regulation
Process 3 — Formation of Dilute vs. Concentrated Urine Formation of Dilute Urine (ADH Absent / Low)
Segment | Event | Tubular Fluid Osmolarity |
|---|---|---|
PCT | Solutes and water reabsorbed proportionally | ~300 mOsm/L (iso-osmotic) |
Descending limb | Water leaves → fluid concentrates | Increases to ~1200 mOsm/L at tip |
Ascending limb | Na⁺/K⁺/Cl⁻ pumped out; water cannot follow | Decreases to ~100 mOsm/L |
Early DCT | Na⁺/Cl⁻ reabsorbed; water cannot follow | Further dilutes |
Collecting duct | No ADH → no aquaporins → water stays in tubule | Remains dilute → large volume excreted |
Formation of Concentrated Urine (ADH Present / High)
Segment | Event | Tubular Fluid Osmolarity |
|---|---|---|
PCT | Solutes and water reabsorbed proportionally | ~300 mOsm/L (iso-osmotic) |
Descending limb | Water leaves → fluid concentrates | Increases to ~1200 mOsm/L at tip |
Ascending limb | Na⁺/K⁺/Cl⁻ pumped out; water cannot follow | Decreases to ~100 mOsm/L |
DCT and collecting duct | ADH inserts AQP2 → water reabsorbed at each level as duct passes through hyperosmotic medulla | Equilibrates with medulla → up to ~1200 mOsm/L |
Final urine | Highly concentrated; small volume | ~1200 mOsm/L |
Complete Summary: Reabsorption and Secretion by Segment PCT Summary
Substance | Direction | Mechanism |
|---|---|---|
Na⁺ | Tubule → blood | Na⁺/K⁺ ATPase (basolateral); cotransporters and antiporters (apical) |
Glucose | Tubule → blood | SGLT (apical); GLUT (basolateral) — secondary active transport |
Amino acids | Tubule → blood | Na⁺-coupled cotransporters |
HCO₃⁻ | Tubule → blood | Carbonic anhydrase mechanism; Na⁺/H⁺ antiporter |
Water | Tubule → blood | Osmosis through aquaporins — obligatory |
Cl⁻, K⁺, Ca²⁺ | Tubule → blood | Cotransporters and channels |
Urea, lipid-soluble substances | Tubule → blood | Simple diffusion |
H⁺ | Blood → tubule | Na⁺/H⁺ antiporter — secretion |
NH₄⁺ | Blood → tubule | Secretion — acid elimination |
Creatinine, drugs, toxins | Blood → tubule | Secretion via specific transporters |
Loop of Henle Summary
Segment | Substance | Direction | Mechanism |
|---|---|---|---|
Descending limb | Water | Tubule → interstitium | Osmosis through aquaporins |
Descending limb | Solutes | Remain in tubule | Impermeable to most solutes |
Ascending limb (thick) | Na⁺, K⁺, Cl⁻ | Tubule → interstitium | NKCC cotransporter + Na⁺/K⁺ ATPase |
Ascending limb | Water | Does NOT move | Impermeable to water |
DCT Summary
Substance | Direction | Mechanism |
|---|---|---|
Na⁺, Cl⁻ | Tubule → blood | NCC symporter (early DCT) |
Ca²⁺ | Tubule → blood | PTH-regulated Ca²⁺ channels |
Water | Does NOT move in early DCT | Impermeable |
Collecting Duct Summary
Substance | Direction | Mechanism | Regulated By |
|---|---|---|---|
Na⁺ | Tubule → blood | ENaC channels + Na⁺/K⁺ ATPase | Aldosterone |
K⁺ | Blood → tubule | Apical leak channels | Aldosterone |
Water | Tubule → blood | Aquaporin-2 (AQP2) | ADH |
Urea | Tubule → interstitium | Diffusion (urea recycling) | — |
H⁺ | Blood → tubule | Intercalated cells | Acid-base status |
HCO₃⁻ | Tubule → blood | Intercalated cells | Acid-base status |
Hormonal Regulation Summary
Hormone | Stimulus | Site of Action | Effect |
|---|---|---|---|
ADH | ↑ Blood osmolarity; ↓ blood volume | Collecting duct principal cells | Inserts AQP2 → ↑ water reabsorption → concentrated urine |
Aldosterone | ↑ Angiotensin II; ↑ extracellular K⁺ | Late DCT + collecting duct principal cells | ↑ ENaC + Na⁺/K⁺ ATPase → ↑ Na⁺ reabsorption; ↑ K⁺ secretion |
Angiotensin II | ↓ Blood pressure/volume | PCT Na⁺/H⁺ antiporters; arterioles | ↑ Na⁺ reabsorption in PCT; constricts arterioles; stimulates aldosterone and ADH |
ANP | ↑ Blood volume (atrial stretch) | Collecting duct; arterioles | ↓ Na⁺ reabsorption; ↑ GFR; opposes RAAS |
PTH | ↓ Blood calcium | DCT Ca²⁺ channels | ↑ Ca²⁺ reabsorption |
Identify the types of transport mechanisms found along the nephron that are responsible for reabsorption and secretion
Transport Mechanisms Along the Nephron Overview
The nephron uses virtually every type of membrane transport mechanism known in physiology
Understanding which mechanism moves which substance where is the key to understanding how the kidney regulates the composition of urine and blood
All transport occurs across two membranes in sequence:
Apical membrane — faces the tubular lumen (filtrate side)
Basolateral membrane — faces the interstitial fluid and peritubular capillaries (blood side)
The Na⁺/K⁺ ATPase on the basolateral membrane is the master engine that directly or indirectly powers the majority of reabsorption throughout the entire nephron
Categories of Transport Mechanisms 1. Primary Active Transport
Definition: Directly uses ATP to move substances against their concentration or electrochemical gradient
Direction: Can move substances either into or out of the cell
Na⁺/K⁺ ATPase (Sodium-Potassium Pump)
Location: Basolateral membrane of virtually all tubular epithelial cells throughout the entire nephron
PCT, loop of Henle, DCT, collecting duct
Mechanism:
Uses ATP directly
Pumps 3 Na⁺ OUT of the cell (into the interstitium → peritubular capillaries)
Pumps 2 K⁺ IN to the cell (from the interstitium)
Maintains low intracellular Na⁺ concentration at all times
Why it matters:
Creates the electrochemical gradient that drives Na⁺ from the tubular lumen into the cell across the apical membrane
This Na⁺ gradient then powers virtually all secondary active transport throughout the nephron
Without Na⁺/K⁺ ATPase, the entire reabsorption machinery would fail
It is the single most important transport protein in the kidney
Substance moved: Na⁺ (out), K⁺ (in)
Net effect: Reabsorption of Na⁺ from tubule to blood
H⁺/K⁺ ATPase (Proton Pump)
Location: Apical membrane of intercalated cells of the collecting duct
Mechanism:
Uses ATP directly
Pumps H⁺ OUT of the cell into the tubular lumen
Pumps K⁺ IN from the tubular lumen
Why it matters:
Primary mechanism for acid secretion into the urine in the collecting duct
Allows kidney to eliminate excess H⁺ and regulate blood pH
Same type of pump as the gastric H⁺/K⁺ ATPase in parietal cells — same principle, different location
Substance moved: H⁺ (secreted into tubule), K⁺ (reabsorbed)
Net effect: Acid elimination; pH regulation
Ca²⁺ ATPase
Location: Basolateral membrane of DCT cells
Mechanism:
Uses ATP to pump Ca²⁺ OUT of the tubular cell into the interstitium → peritubular capillaries
Why it matters:
Works in conjunction with PTH-regulated apical Ca²⁺ channels
Maintains low intracellular Ca²⁺ so that Ca²⁺ can continue to enter from the tubular lumen
Substance moved: Ca²⁺ (reabsorbed)
Regulated by: PTH
2. Secondary Active Transport
Definition: Uses the electrochemical gradient of Na⁺ (created by Na⁺/K⁺ ATPase) to move another substance against its own gradient — no direct ATP use
Two subtypes:
Symporters (cotransporters) — Na⁺ and another substance move in the same direction
Antiporters (exchangers) — Na⁺ and another substance move in opposite directions
Symporters (Cotransporters) — Na⁺ and Substance Move Together INTO the Cell SGLT — Sodium-Glucose Linked Transporter
Location: Apical membrane of PCT cells
Mechanism:
Na⁺ moves DOWN its electrochemical gradient into the cell
Glucose is carried ALONG (against its own gradient) — piggybacks on Na⁺ entry
Substance moved: Na⁺ and glucose both move from tubule → cell
Net effect: Glucose reabsorption (tubule → blood)
Transport Maximum (Tm):
Finite number of SGLT carriers
When plasma glucose is very high → filtrate glucose exceeds Tm → excess glucose not reabsorbed → glucosuria
Basis of glucose in urine in uncontrolled diabetes mellitus
Also the target of SGLT2 inhibitors (e.g., empagliflozin) — a class of diabetes drugs that deliberately block this transporter to increase glucose excretion
Amino Acid Cotransporters
Location: Apical membrane of PCT cells
Mechanism:
Na⁺ moves down its gradient; amino acids carried along
Substance moved: Na⁺ and amino acids from tubule → cell
Net effect: Amino acid reabsorption (tubule → blood)
NKCC — Na⁺-K⁺-2Cl⁻ Cotransporter
Location: Apical membrane of thick ascending limb of loop of Henle
Mechanism:
Simultaneously transports 1 Na⁺, 1 K⁺, and 2 Cl⁻ from the tubular lumen into the cell
Driven by Na⁺ gradient created by basolateral Na⁺/K⁺ ATPase
K⁺ then leaks back into the tubular lumen through apical K⁺ channels
Na⁺ and Cl⁻ exit at the basolateral membrane
Critical feature: The ascending limb is impermeable to water — so solutes leave without water following
Net effect:
Tubular fluid becomes progressively more dilute as it ascends
Medullary interstitium becomes progressively more concentrated
Establishes the medullary osmotic gradient essential for urine concentration
Clinical relevance:
Loop diuretics (furosemide/Lasix) block NKCC
Prevents solute reabsorption → water follows solutes into urine → large volume of dilute urine
Used for fluid overload, hypertension, heart failure, edema
NCC — Na⁺-Cl⁻ Cotransporter
Location: Apical membrane of early DCT cells
Mechanism:
Na⁺ moves down its gradient; Cl⁻ carried along
Water does NOT follow — early DCT is impermeable to water
Substance moved: Na⁺ and Cl⁻ from tubule → cell → blood
Net effect: Na⁺ and Cl⁻ reabsorption; tubular fluid continues to dilute
Clinical relevance:
Thiazide diuretics (e.g., hydrochlorothiazide) block NCC
Reduce Na⁺ reabsorption in the DCT → more Na⁺ and water excreted → lower blood pressure
Commonly used for hypertension
Phosphate Cotransporter
Location: Apical membrane of PCT cells
Mechanism: Na⁺-coupled transport of phosphate into the cell
Net effect: Phosphate reabsorption (tubule → blood)
Antiporters (Exchangers) — Na⁺ and Substance Move in Opposite Directions Na⁺/H⁺ Antiporter (NHE — Na⁺/H⁺ Exchanger)
Location: Apical membrane of PCT cells and also stimulated by angiotensin II
Mechanism:
Na⁺ moves DOWN its gradient into the cell
H⁺ is simultaneously pumped OUT of the cell into the tubular lumen (against its gradient)
Na⁺ entry powers H⁺ secretion
Substance moved:
Na⁺: tubule → cell (reabsorption)
H⁺: cell → tubule (secretion)
Net effects:
Drives Na⁺ reabsorption
Secretes H⁺ into urine → important for blood pH regulation
Also drives HCO₃⁻ reabsorption indirectly (see bicarbonate mechanism below)
Regulated by: Angiotensin II — stimulates NHE activity → more Na⁺ reabsorption and H⁺ secretion
HCO₃⁻/Cl⁻ Antiporter
Location: Basolateral membrane of PCT cells and intercalated cells of collecting duct
Mechanism:
HCO₃⁻ exits the cell (into the interstitium → blood) in exchange for Cl⁻ entering the cell
Net effect: HCO₃⁻ reabsorption (tubule → blood); contributes to blood pH buffering
3. Facilitated Diffusion
Definition: Passive transport — moves substances down their concentration gradient through specific carrier proteins or channels
No ATP required; no coupling to Na⁺ gradient
Rate limited by number of available carrier proteins
GLUT Transporters (Glucose Transporters)
Location: Basolateral membrane of PCT cells
Mechanism:
Glucose moves down its concentration gradient from inside the cell (where it has accumulated via SGLT) out to the interstitium → peritubular capillaries
Substance moved: Glucose (cell → blood)
Net effect: Completes glucose reabsorption — the exit step after SGLT entry
Amino Acid Transporters (Basolateral)
Location: Basolateral membrane of PCT cells
Mechanism: Facilitated diffusion of amino acids from cell → interstitium → blood
Net effect: Completes amino acid reabsorption
Urea Transporters
Location:
Descending thin limb of loop of Henle — some urea entry
Inner medullary collecting duct — primary site
Mechanism:
Urea moves passively down its concentration gradient out of the inner medullary collecting duct
Regulated by ADH — ADH increases urea permeability of the inner medullary collecting duct
Net effect:
Urea diffuses into the medullary interstitium → urea recycling
Contributes approximately half of the medullary osmolarity at the deepest papilla
Amplifies the medullary concentration gradient → enhances ability to concentrate urine
K⁺ Leak Channels
Location:
Apical membrane of principal cells of the collecting duct
Apical membrane of thick ascending limb cells (K⁺ recycling)
Mechanism:
K⁺ that was pumped into the cell by Na⁺/K⁺ ATPase leaks back out through channels down its concentration gradient
Net effect:
In collecting duct: K⁺ secretion into tubular fluid → primary route of potassium excretion
In ascending limb: K⁺ recycling back into lumen maintains NKCC function
Regulated by: Aldosterone — increases number of apical K⁺ channels → more K⁺ secretion
Ca²⁺ Channels (Apical)
Location: Apical membrane of DCT cells
Mechanism:
Ca²⁺ moves passively down its electrochemical gradient from tubular lumen into the cell
Net effect: First step of Ca²⁺ reabsorption (tubule → cell)
Regulated by: PTH — opens Ca²⁺ channels → increases Ca²⁺ reabsorption when blood calcium is low
4. Osmosis (Water Transport via Aquaporins)
Definition: Passive movement of water down its osmotic gradient through specific aquaporin (AQP) water channels
Water always follows solute — when solutes are reabsorbed, water follows osmotically
Aquaporin-1 (AQP1) — Constitutive, Always Present
Location:
Apical AND basolateral membranes of PCT cells
Apical AND basolateral membranes of descending limb of loop of Henle
Mechanism:
Always present — not regulated by hormones
Water moves freely and obligatorily whenever osmotic gradient exists
Net effect:
PCT: ~65% of filtered water reabsorbed here — obligatory water reabsorption
Descending limb: water leaves as tubular fluid enters increasingly concentrated medulla
Key point: AQP1-mediated water reabsorption is always on regardless of hydration status
Aquaporin-2 (AQP2) — Regulated by ADH
Location: Apical membrane of principal cells of the collecting duct (and late DCT)
Mechanism:
NOT present in the apical membrane unless ADH is present
When ADH is released (dehydration, high blood osmolarity):
ADH binds to V2 receptors on principal cells
Triggers insertion of AQP2-containing vesicles into the apical membrane
Water channel is now open → water moves by osmosis from lumen into cell
When ADH is absent (well-hydrated):
AQP2 vesicles are retrieved from the apical membrane
Collecting duct becomes impermeable to water
Water stays in tubule → excreted as dilute urine
Net effect:
ADH present → AQP2 inserted → water reabsorbed → small volume concentrated urine
ADH absent → AQP2 removed → water not reabsorbed → large volume dilute urine
Clinical relevance:
Diabetes insipidus — either no ADH produced (central DI) or principal cells don't respond to ADH (nephrogenic DI)
Result: AQP2 not inserted → massive volumes of dilute urine (up to 20 L/day)
Aquaporin-3 and Aquaporin-4 (AQP3/AQP4) — Constitutive
Location: Basolateral membrane of principal cells of collecting duct
Mechanism: Always present; water exits the cell into the interstitium after entering via AQP2
Net effect: Completes the transcellular water reabsorption pathway in the collecting duct
5. Simple Diffusion
Definition: Passive movement of substances directly through the lipid bilayer down their concentration gradient
No carrier proteins or channels needed
Limited to lipid-soluble (hydrophobic) substances and very small molecules
Substances Reabsorbed by Simple Diffusion in the PCT:
Urea — partially reabsorbed by diffusion as water reabsorption concentrates urea in the tubule
Lipid-soluble drugs and toxins — diffuse back across tubular membranes; this is why some drugs are difficult to eliminate
CO₂ — diffuses freely across all membranes; relevant to bicarbonate handling
Substances Secreted by Simple Diffusion:
Some lipid-soluble waste products move from peritubular capillaries into the tubular fluid by diffusion
6. ENaC — Epithelial Sodium Channels
Technically facilitated diffusion but important enough to discuss separately
Location: Apical membrane of principal cells of the late DCT and collecting duct
Mechanism:
Na⁺ leaks passively from the tubular lumen into the cell through ENaC channels
Driven by the low intracellular Na⁺ maintained by basolateral Na⁺/K⁺ ATPase
Na⁺ entry is passive (downhill) but dependent on channel availability
Regulated by Aldosterone:
Aldosterone increases transcription of ENaC channel proteins → more channels inserted into the apical membrane
Also increases Na⁺/K⁺ ATPase activity → maintains the Na⁺ gradient driving ENaC
Net: dramatically increases Na⁺ reabsorption → water follows → blood volume and pressure increase
Clinical relevance:
Spironolactone (aldosterone antagonist / potassium-sparing diuretic) blocks aldosterone receptors → fewer ENaC channels expressed → less Na⁺ reabsorption → more Na⁺ and water excreted
Does NOT cause K⁺ loss (unlike loop and thiazide diuretics) because it also blocks aldosterone-stimulated K⁺ secretion
7. Transcytosis / Endocytosis
Definition: Vesicle-mediated uptake of substances too large for channels or carriers
Location: Apical membrane of PCT cells
Mechanism:
Small proteins that manage to pass through the filtration membrane (filtered despite its selectivity) are taken up by receptor-mediated endocytosis
Internalized into vesicles → degraded by lysosomes inside the cell → amino acid components returned to blood
Substance moved: Small filtered proteins (tubule → cell → broken down)
Net effect: Prevents protein loss in urine; recovers amino acids
Summary Table: Transport Mechanisms by Segment PCT
Substance | Membrane | Mechanism | Type | Direction |
|---|---|---|---|---|
Na⁺ | Apical | Cotransporters, antiporters | Secondary active | Tubule → cell |
Na⁺ | Basolateral | Na⁺/K⁺ ATPase | Primary active | Cell → blood |
Glucose | Apical | SGLT (Na⁺ cotransport) | Secondary active | Tubule → cell |
Glucose | Basolateral | GLUT | Facilitated diffusion | Cell → blood |
Amino acids | Apical | Na⁺ cotransporters | Secondary active | Tubule → cell |
Amino acids | Basolateral | Facilitated diffusion transporters | Facilitated diffusion | Cell → blood |
H⁺ | Apical | Na⁺/H⁺ antiporter | Secondary active | Cell → tubule (secretion) |
HCO₃⁻ | Basolateral | HCO₃⁻/Cl⁻ antiporter | Secondary active | Cell → blood |
Water | Apical + basolateral | AQP1 | Osmosis | Tubule → blood (obligatory) |
Urea, lipid-soluble | Both | Simple diffusion | Passive | Tubule → blood |
Small proteins | Apical | Endocytosis | Transcytosis | Tubule → cell (degraded) |
Drugs/toxins | Apical | Specific secretory transporters | Secondary active | Blood → tubule (secretion) |
Creatinine | Apical | Secretory transporters | Secondary active | Blood → tubule (secretion) |
Loop of Henle
Substance | Segment | Membrane | Mechanism | Type | Direction |
|---|---|---|---|---|---|
Water | Descending limb | Apical + basolateral | AQP1 — osmosis | Passive | Tubule → interstitium |
Na⁺, K⁺, Cl⁻ | Thick ascending limb (apical) | NKCC cotransporter | Secondary active | Tubule → cell | |
Na⁺ | Thick ascending limb (basolateral) | Na⁺/K⁺ ATPase | Primary active | Cell → interstitium | |
K⁺ | Thick ascending limb (apical) | K⁺ leak channels | Facilitated diffusion | Cell → tubule (recycling) | |
Water | Ascending limb | — | NONE — impermeable | — |
DCT
Substance | Membrane | Mechanism | Type | Direction |
|---|---|---|---|---|
Na⁺, Cl⁻ | Apical | NCC cotransporter | Secondary active | Tubule → cell |
Na⁺ | Basolateral | Na⁺/K⁺ ATPase | Primary active | Cell → blood |
Ca²⁺ | Apical | Ca²⁺ channels (PTH-regulated) | Facilitated diffusion | Tubule → cell |
Ca²⁺ | Basolateral | Ca²⁺ ATPase | Primary active | Cell → blood |
Water | — | NONE in early DCT | — | Impermeable |
Collecting Duct
Substance | Membrane | Mechanism | Type | Direction | Regulated By |
|---|---|---|---|---|---|
Na⁺ | Apical | ENaC channels | Facilitated diffusion | Tubule → cell | Aldosterone |
Na⁺ | Basolateral | Na⁺/K⁺ ATPase | Primary active | Cell → blood | Aldosterone |
K⁺ | Apical | K⁺ leak channels | Facilitated diffusion | Cell → tubule (secretion) | Aldosterone |
H⁺ | Apical | H⁺/K⁺ ATPase | Primary active | Cell → tubule (secretion) | Acid-base status |
Water | Apical | AQP2 (inserted by ADH) | Osmosis | Tubule → cell | ADH |
Water | Basolateral | AQP3/AQP4 (constitutive) | Osmosis | Cell → interstitium | Constitutive |
Urea | Both | Urea transporters | Facilitated diffusion | Tubule → interstitium (recycling) | ADH |
HCO₃⁻ | Basolateral | HCO₃⁻/Cl⁻ antiporter | Secondary active | Cell → blood | Acid-base status |
Master Concept Map: How It All Connects
Na⁺/K⁺ ATPase (primary active) → maintains low intracellular Na⁺
Powers SGLT (glucose reabsorption)
Powers amino acid cotransporters
Powers NKCC (loop of Henle)
Powers NCC (early DCT)
Powers Na⁺/H⁺ antiporter (H⁺ secretion + HCO₃⁻ recovery)
Powers ENaC (collecting duct Na⁺ entry)
AQP1 (constitutive osmosis) → obligatory water reabsorption in PCT and descending limb
AQP2 (ADH-regulated osmosis) → facultative water reabsorption in collecting duct
NKCC (ascending limb) → builds medullary osmotic gradient → enables AQP2-driven concentration
ENaC + Na⁺/K⁺ ATPase → Na⁺ reabsorption in collecting duct → regulated by aldosterone → controls blood volume and pressure
Describe the countercurrent mechanism and explain how it relates to the concentration gradient in the medulla
The Countercurrent Mechanism and Medullary Concentration Gradient Overview
The kidney can produce urine ranging from very dilute (~65–100 mOsm/L) to very concentrated (~1200 mOsm/L) depending on the body's hydration needs
This remarkable range is made possible by the countercurrent mechanism
The countercurrent mechanism refers to two related but distinct systems:
Countercurrent Multiplier — the loop of Henle; builds and maintains the medullary osmotic gradient
Countercurrent Exchanger — the vasa recta; preserves the medullary osmotic gradient without washing it away
A third contributor — urea recycling — amplifies the deep medullary gradient
Without this system, the kidney could only produce urine iso-osmotic to plasma (~300 mOsm/L) — it could never concentrate urine
Part 1 — The Medullary Osmotic Gradient What It Is
The medullary interstitium maintains a steep, progressive osmotic gradient
Osmolarity increases from the cortex → outer medulla → inner medulla → papilla:
Location | Osmolarity |
|---|---|
Cortex (corticomedullary junction) | ~300 mOsm/L |
Outer medulla | ~600 mOsm/L |
Inner medulla | ~900 mOsm/L |
Deep papilla (tip of pyramid) | ~1200 mOsm/L |
What Creates It
The gradient is established and maintained by two main contributors:
NaCl pumped out of the thick ascending limb — accounts for roughly half of medullary osmolarity
Urea recycled from the inner medullary collecting duct — accounts for roughly the other half at the deepest levels
The gradient is continuously maintained — it would dissipate quickly without ongoing active transport
Why It Matters
The medullary osmotic gradient is the driving force for water reabsorption in the collecting duct
When ADH is present and aquaporins are inserted, water is drawn osmotically out of the collecting duct at each successive level as it passes through the increasingly concentrated medulla
The deeper the collecting duct descends, the more concentrated the surrounding interstitium → the more water is pulled out
Without this gradient, ADH would have nothing to drive water reabsorption against
Part 2 — The Countercurrent Multiplier (Loop of Henle) What "Countercurrent" Means
The descending and ascending limbs of the loop of Henle run parallel to each other
Fluid flows in opposite directions in the two limbs:
Descending limb: fluid flows DOWN toward the medulla
Ascending limb: fluid flows UP toward the cortex
This opposing flow arrangement is the countercurrent arrangement
The term "multiplier" refers to the fact that this arrangement progressively amplifies the osmotic gradient — each cycle of the loop makes the gradient steeper
The Key: Opposite Permeability Properties
The descending and ascending limbs have completely opposite permeability properties — this difference is the entire basis of the system:
Property | Descending Limb | Ascending Limb (Thick) |
|---|---|---|
Permeable to water? | YES — freely permeable (AQP1) | NO — completely impermeable |
Permeable to solutes? | Relatively impermeable | YES — actively transports NaCl out |
Transport mechanism | Passive osmosis (AQP1) | NKCC cotransporter + Na⁺/K⁺ ATPase |
Effect on tubular fluid | Becomes more concentrated as it descends | Becomes more dilute as it ascends |
Effect on interstitium | Receives water → interstitium concentrates | Receives NaCl → interstitium concentrates |
Step-by-Step Mechanism of the Countercurrent Multiplier Starting Conditions
Filtrate entering the descending limb from the PCT is iso-osmotic to plasma (~300 mOsm/L)
The medullary interstitium is already slightly concentrated from previous cycles
Step 1 — Descending Limb: Water Leaves
Filtrate enters the descending limb from the cortex at ~300 mOsm/L
As filtrate descends deeper into the medulla, it encounters an increasingly concentrated interstitium
The descending limb is freely permeable to water (AQP1 channels)
Water moves OUT of the tubular fluid by osmosis into the interstitium
Solutes remain in the tubular fluid (descending limb is impermeable to most solutes)
As more water leaves, the tubular fluid becomes progressively more concentrated
At the tip of the loop (deepest point in the medulla), tubular fluid reaches its maximum concentration — approximately ~1200 mOsm/L — equilibrating with the surrounding interstitium
Step 2 — Ascending Limb: Solutes Leave, Water Cannot
Tubular fluid turns the corner and begins ascending in the thick ascending limb
The ascending limb is completely impermeable to water — no aquaporins
The NKCC cotransporter on the apical membrane actively pumps Na⁺, K⁺, and 2Cl⁻ OUT of the tubular fluid into the interstitium
Na⁺ is then pumped out the basolateral side by Na⁺/K⁺ ATPase
K⁺ leaks back into the tubular lumen through apical K⁺ channels (recycling)
Because water cannot follow the solutes being pumped out:
Tubular fluid becomes progressively more dilute as it ascends
By the time fluid reaches the DCT it is hypotonic (~100 mOsm/L)
Simultaneously, NaCl being deposited into the interstitium concentrates the medullary interstitium
Step 3 — The Multiplier Effect
The NaCl deposited into the interstitium by the ascending limb raises the osmolarity of the interstitium
This higher interstitial osmolarity draws more water out of the descending limb in the next cycle
More water leaving the descending limb → tubular fluid at the tip becomes even more concentrated
More concentrated fluid at the tip → more NaCl pumped out by the ascending limb on the next pass
This is a positive feedback amplification — each cycle builds on the previous one
The result: a progressively steeper gradient from cortex (300) to deep medulla (1200)
This is why it is called a "multiplier" — the countercurrent arrangement multiplies the effect of the pump
Summary of Countercurrent Multiplier Effect on Tubular Fluid
Location | Tubular Fluid Osmolarity | Key Event |
|---|---|---|
PCT → entering descending limb | ~300 mOsm/L | Iso-osmotic to plasma |
Descending limb (cortex → medulla) | 300 → 1200 mOsm/L | Water leaves via AQP1 |
Tip of loop (deepest medulla) | ~1200 mOsm/L | Maximum concentration |
Ascending limb (medulla → cortex) | 1200 → 100 mOsm/L | NaCl pumped out; water cannot follow |
Early DCT | ~100 mOsm/L | Hypotonic to plasma |
Part 3 — Urea Recycling What It Is
Urea is a metabolic waste product of protein metabolism
It is freely filtered at the glomerulus and mostly stays in the tubular fluid through the PCT and loop of Henle
At the inner medullary collecting duct, urea is able to diffuse out — and this contributes significantly to the medullary gradient
Mechanism
As the collecting duct descends through the medulla with ADH present:
Water is reabsorbed via AQP2 → tubular fluid becomes increasingly concentrated
Urea concentration inside the collecting duct rises dramatically
The inner medullary collecting duct is permeable to urea (especially when ADH is present — ADH also increases urea permeability)
Urea diffuses OUT of the collecting duct into the deep medullary interstitium down its concentration gradient
This urea then enters the descending thin limb of the loop of Henle and recirculates through the nephron — called urea recycling
Why It Matters
Urea deposited in the deep medullary interstitium contributes ~50% of the osmolarity at the deepest papilla
Without urea recycling, the medullary gradient would only reach approximately 600 mOsm/L at the papilla — not 1200 mOsm/L
Urea recycling is therefore essential for the kidney's ability to produce maximally concentrated urine
This is also why high-protein diets enhance urine concentrating ability — more protein metabolism = more urea = stronger medullary gradient
Urea Recycling Summary
Step | Location | Event |
|---|---|---|
1 | Collecting duct (inner medulla) | Water reabsorbed → urea concentrates |
2 | Inner medullary collecting duct | Urea diffuses out into medullary interstitium |
3 | Medullary interstitium | Urea raises osmolarity (contributes ~50% at papilla) |
4 | Descending thin limb | Urea enters tubular fluid and recirculates |
5 | Net effect | Deep medullary gradient amplified → maximally concentrated urine possible |
Part 4 — The Countercurrent Exchanger (Vasa Recta) The Problem It Solves
The medulla must be supplied with oxygen and nutrients and have metabolic waste removed
However, if blood simply flowed straight through the medulla in ordinary capillaries, it would:
Pick up the NaCl and urea concentrated in the interstitium as it entered the medulla
Carry all of it away as it exited → washing out the gradient
Without a solution to this problem, the medullary gradient could never be maintained
What the Vasa Recta Are
Long, hairpin-shaped capillaries that run parallel to and alongside the loop of Henle
Arise from the efferent arterioles of juxtamedullary nephrons (nephrons with long loops)
Run deep into the medulla and loop back up — just like the loop of Henle
Their walls are highly permeable to water and solutes
The Countercurrent Exchange Mechanism Descending Limb of Vasa Recta (Blood Flowing DOWN into Medulla)
As blood descends into the increasingly concentrated medullary interstitium:
Interstitial osmolarity > blood osmolarity
Water leaves the blood (osmosis) → blood becomes more concentrated
Solutes (NaCl, urea) enter the blood from the interstitium (diffusion)
Blood osmolarity rises progressively as it descends
At the deepest point: blood osmolarity ≈ interstitial osmolarity ≈ ~1200 mOsm/L
Ascending Limb of Vasa Recta (Blood Flowing UP toward Cortex)
As blood ascends back toward the cortex through less concentrated interstitium:
Blood osmolarity > interstitial osmolarity (blood is now hyperosmotic relative to the surrounding tissue)
Water re-enters the blood from the interstitium
Solutes (NaCl, urea) leave the blood back into the interstitium
Blood osmolarity decreases progressively as it ascends
At the top: blood osmolarity returns to approximately ~300 mOsm/L
Why This Preserves the Gradient
Because blood descends and ascends in parallel, adjacent channels with flow in opposite directions:
Solutes and water that leave the blood on the way down are essentially recaptured on the way up
The net movement of solutes OUT of the medulla is minimal
The gradient is preserved because what the descending limb picks up, the ascending limb releases back
Analogy: imagine carrying a bucket of water down into a warm basement (water evaporates) and then carrying the bucket back up (water condenses back in) — the net change in water is minimal compared to simply leaving the bucket down there
What the Vasa Recta Actually Remove
The vasa recta do still remove a small net amount of water and solutes from the medulla — just enough to:
Remove the water that was reabsorbed from the descending limb of the loop and the collecting duct
Remove metabolic waste products generated by medullary cells
Supply oxygen and nutrients to medullary tissue
The key is that this removal is slow and minimal compared to what would occur with straight-through capillaries
Summary of Vasa Recta Countercurrent Exchange
Location | Water Movement | Solute Movement | Blood Osmolarity |
|---|---|---|---|
Entering medulla (descending) | Water LEAVES blood | NaCl + urea ENTER blood | Rises: 300 → 1200 mOsm/L |
Tip of vasa recta | Equilibrates with interstitium | Equilibrates with interstitium | ~1200 mOsm/L |
Exiting medulla (ascending) | Water RE-ENTERS blood | NaCl + urea LEAVE blood | Falls: 1200 → ~300 mOsm/L |
Net effect on gradient | Minimal disruption | Minimal net removal | Gradient preserved |
Part 5 — Putting It All Together: Concentrated vs. Dilute Urine Formation of Concentrated Urine (ADH Present — Dehydrated State)
Trigger: Dehydration → ↑ blood osmolarity → posterior pituitary releases ADH
ADH inserts AQP2 channels into the apical membrane of collecting duct principal cells
Segment | Key Event | Result |
|---|---|---|
PCT | Na⁺, solutes, and water reabsorbed proportionally (AQP1) | Fluid remains ~300 mOsm/L (iso-osmotic) |
Descending limb | Water leaves via AQP1 into concentrated interstitium | Fluid concentrates: 300 → 1200 mOsm/L |
Ascending limb | NKCC pumps NaCl out; water cannot follow | Fluid dilutes: 1200 → 100 mOsm/L; interstitium concentrates |
Early DCT | NCC removes NaCl; water impermeable | Fluid remains ~100 mOsm/L or slightly lower |
Collecting duct (cortex) | ADH → AQP2 inserted → water reabsorbed | Fluid begins to concentrate |
Collecting duct (outer medulla) | More water reabsorbed as surrounding interstitium is 600 mOsm/L | Fluid concentrates further |
Collecting duct (inner medulla) | Water reabsorbed into 1200 mOsm/L interstitium; urea diffuses out | Fluid reaches ~1200 mOsm/L |
Final urine | Highly concentrated; small volume | ~1200 mOsm/L |
Formation of Dilute Urine (ADH Absent — Well-Hydrated State)
Trigger: Excess water intake → ↓ blood osmolarity → posterior pituitary suppresses ADH release
Segment | Key Event | Result |
|---|---|---|
PCT | Na⁺, solutes, and water reabsorbed proportionally | Fluid remains ~300 mOsm/L |
Descending limb | Water leaves via AQP1 | Fluid concentrates: 300 → 1200 mOsm/L |
Ascending limb | NKCC pumps NaCl out; water cannot follow | Fluid dilutes: 1200 → 100 mOsm/L |
DCT | NaCl reabsorbed; water cannot follow | Fluid further dilutes |
Collecting duct | No ADH → no AQP2 inserted → collecting duct impermeable to water | Water stays in tubule |
Final urine | Large volume; very dilute | ~65–100 mOsm/L |
Key insight: The countercurrent multiplier still runs and still builds the medullary gradient even when ADH is absent — the gradient is always maintained
What changes is whether the collecting duct can USE that gradient — which depends entirely on ADH and AQP2 insertion
Complete Summary Table
Component | Structure | Mechanism | Function |
|---|---|---|---|
Countercurrent Multiplier | Loop of Henle | Descending limb loses water (AQP1); ascending limb pumps out NaCl (NKCC); opposite flow directions amplify gradient | Builds medullary osmotic gradient (300 → 1200 mOsm/L) |
Urea Recycling | Inner medullary collecting duct + loop | Urea diffuses out of inner collecting duct into interstitium; recirculates via thin descending limb | Amplifies deep medullary gradient; contributes ~50% of papillary osmolarity |
Countercurrent Exchanger | Vasa recta | Blood descends (gains solutes, loses water) and ascends (loses solutes, gains water) in parallel countercurrent flow | Preserves medullary gradient; supplies medulla without washing out gradient |
ADH + AQP2 | Collecting duct | ADH inserts AQP2 → water reabsorbed osmotically at each level as duct passes through gradient | Uses the gradient to concentrate urine when body is dehydrated |
Describe how ADH and aldosterone influence the production of dilute or concentrated urine
ADH and Aldosterone: Influence on Urine Concentration Overview
The final composition and volume of urine is determined largely by two hormones acting on the distal nephron (late DCT and collecting duct)
ADH (antidiuretic hormone / vasopressin) — controls water reabsorption
Aldosterone — controls sodium reabsorption and potassium secretion
Although both hormones act on overlapping segments of the nephron, they regulate different substances through different mechanisms and are released in response to different stimuli
Together they fine-tune the balance between water and sodium in the blood — and therefore regulate blood volume, blood pressure, and urine concentration
Part 1 — ADH (Antidiuretic Hormone / Vasopressin) What It Is
A peptide hormone produced in the hypothalamus and stored in and released from the posterior pituitary gland
Name encodes its function: anti (against) + diuretic (urine production) = hormone that opposes urine production by promoting water reabsorption
Also called vasopressin because at very high concentrations it causes vasoconstriction — but its primary physiological role is water regulation
Stimuli for ADH Release
↑ Blood osmolarity (most important trigger):
Detected by osmoreceptors in the hypothalamus
When blood becomes too concentrated (e.g., dehydration, not drinking enough water), osmoreceptors shrink slightly → triggers ADH release
Normal blood osmolarity = ~285–295 mOsm/L; rises above this → ADH released
↓ Blood volume / ↓ blood pressure:
Detected by baroreceptors in the atria and large vessels
Even if osmolarity is normal, significant volume loss (hemorrhage, severe dehydration) triggers ADH
Angiotensin II:
Released during RAAS activation (low blood pressure/volume)
Stimulates ADH release from the posterior pituitary — coordinates volume restoration
Other stimuli:
Pain, nausea, stress — all stimulate ADH release
Alcohol — inhibits ADH release → increased urine output → dehydration (explains why alcohol causes frequent urination and hangover thirst)
Site of Action
Principal cells of the:
Late DCT
Collecting duct (cortical and medullary portions)
Mechanism of Action — Detailed Step 1 — ADH Binds to V2 Receptors
ADH is released into the bloodstream and travels to the kidney
Binds to V2 receptors on the basolateral membrane of principal cells
V2 receptors are G-protein coupled receptors (GPCRs)
Step 2 — cAMP Second Messenger Cascade
V2 receptor activation → activates adenylyl cyclase
Adenylyl cyclase converts ATP → cAMP (cyclic AMP)
cAMP activates protein kinase A (PKA)
Step 3 — AQP2 Insertion into Apical Membrane
PKA phosphorylates aquaporin-2 (AQP2)-containing vesicles stored inside the cell
Phosphorylation triggers these vesicles to fuse with the apical membrane
AQP2 water channels are inserted into the apical (luminal) membrane
The cell is now permeable to water on the luminal side
Step 4 — Water Reabsorption
With AQP2 now in the apical membrane:
Water moves by osmosis from the tubular lumen → through AQP2 → into the cell
Water exits the cell across the basolateral membrane via AQP3 and AQP4 (always present — constitutive)
Water enters the medullary interstitium → picked up by peritubular capillaries → returned to blood
The medullary osmotic gradient (300–1200 mOsm/L built by the countercurrent multiplier) provides the osmotic driving force
The deeper the collecting duct descends through the medulla, the stronger the gradient and the more water is pulled out
Step 5 — When ADH Is Withdrawn
When blood osmolarity returns to normal → ADH secretion stops
AQP2 channels are retrieved from the apical membrane back into cytoplasmic vesicles
Collecting duct becomes impermeable to water again
Water stays in the tubule → excreted as dilute urine
ADH and Urea Permeability
ADH also increases the permeability of the inner medullary collecting duct to urea
Urea diffuses out into the medullary interstitium → contributes to the osmotic gradient
This amplifies the concentrating ability of the kidney — another way ADH enhances water conservation
Effect of ADH on Urine: Two Scenarios Scenario 1 — High ADH (Dehydration / High Blood Osmolarity)
Segment | Event |
|---|---|
Late DCT | AQP2 inserted → water reabsorbed |
Cortical collecting duct | AQP2 inserted → water reabsorbed into ~300 mOsm/L cortical interstitium |
Outer medullary collecting duct | AQP2 inserted → water reabsorbed into ~600 mOsm/L interstitium |
Inner medullary collecting duct | AQP2 inserted → water reabsorbed into ~1200 mOsm/L interstitium; urea also exits |
Result | Small volume, highly concentrated urine (~1200 mOsm/L) |
Water is maximally conserved
Blood osmolarity decreases back toward normal → negative feedback → ADH release slows
Scenario 2 — Low/No ADH (Well-Hydrated / Low Blood Osmolarity)
Segment | Event |
|---|---|
Late DCT | No AQP2 → water stays in tubule |
Collecting duct (all levels) | Impermeable to water → water not reabsorbed |
Tubular fluid | Remains dilute (~100 mOsm/L or lower) from ascending limb |
Result | Large volume, very dilute urine (~65–100 mOsm/L) |
Excess water is excreted
Blood osmolarity increases back toward normal → negative feedback → ADH remains suppressed
Clinical Conditions Related to ADH Diabetes Insipidus (DI)
Central DI:
Posterior pituitary fails to produce or release adequate ADH
Collecting duct remains impermeable to water regardless of hydration status
Result: massive volumes of very dilute urine (up to 20 L/day)
Treated with synthetic ADH (desmopressin)
Nephrogenic DI:
ADH is produced normally but principal cells fail to respond to it
V2 receptors or AQP2 channels are dysfunctional
Same result: large volumes of dilute urine despite high circulating ADH
Treated by addressing underlying cause; paradoxically treated with thiazide diuretics
SIADH — Syndrome of Inappropriate ADH Secretion
ADH is released continuously and inappropriately regardless of blood osmolarity
Collecting duct constantly reabsorbs water
Blood becomes abnormally dilute → hyponatremia (low blood sodium)
Urine is inappropriately concentrated
Causes: certain medications, brain injury, lung tumors (ectopic ADH production)
Alcohol and ADH
Alcohol inhibits ADH release from the posterior pituitary
Collecting duct becomes impermeable to water
Large volumes of dilute urine produced → dehydration
Explains frequent urination during alcohol consumption and the intense thirst and headache of a hangover
Part 2 — Aldosterone What It Is
A steroid hormone produced by the zona glomerulosa of the adrenal cortex (adrenal glands sit atop the kidneys)
Because it is a steroid, it is lipid-soluble — crosses cell membranes freely and acts on intracellular receptors
Acts on nuclear receptors → changes gene transcription → takes hours to produce full effect (unlike ADH which acts within minutes)
Primary role: regulates Na⁺ reabsorption and K⁺ secretion → indirectly controls water reabsorption, blood volume, and blood pressure
Stimuli for Aldosterone Release
↑ Angiotensin II (most important trigger):
Produced during RAAS activation when blood pressure/volume falls
Angiotensin II travels to the adrenal cortex → stimulates aldosterone release
This links low blood pressure → RAAS → aldosterone → Na⁺ retention → water retention → ↑ blood volume → ↑ blood pressure
↑ Extracellular K⁺:
When blood potassium rises (hyperkalemia), the zona glomerulosa is directly stimulated → aldosterone released
Aldosterone then promotes K⁺ secretion into the tubule → K⁺ excreted → blood K⁺ returns to normal
This is a direct feedback loop — aldosterone is a key regulator of potassium balance
ACTH (adrenocorticotropic hormone):
From the anterior pituitary; plays a minor role in aldosterone release under stress conditions
Site of Action
Principal cells of the:
Late DCT
Collecting duct
Same cells targeted by ADH — but different receptors and different effects
Mechanism of Action — Detailed Step 1 — Aldosterone Enters the Cell
Aldosterone is a steroid (lipid-soluble) → diffuses freely across the cell membrane
No receptor on the cell surface needed — crosses directly into the cytoplasm
Step 2 — Binds to Intracellular Mineralocorticoid Receptor
Aldosterone binds to the mineralocorticoid receptor (MR) in the cytoplasm
Aldosterone-receptor complex forms
Step 3 — Nuclear Translocation and Gene Transcription
The aldosterone-MR complex moves into the nucleus
Binds to specific hormone response elements (HREs) on DNA
Activates transcription of specific genes
New mRNA is produced → translated into new proteins
Step 4 — New Proteins Increase Na⁺/K⁺ Transport
The proteins produced include:
More ENaC (epithelial sodium channels) — inserted into the apical membrane
More Na⁺ channels → more Na⁺ enters the cell from the tubular lumen
More Na⁺/K⁺ ATPase pumps — inserted into the basolateral membrane
More pumps → more Na⁺ pumped out to blood; more K⁺ pumped into the cell
More apical K⁺ leak channels
K⁺ pumped into the cell by Na⁺/K⁺ ATPase exits through apical channels → secreted into tubular fluid → excreted
Step 5 — Consequences of Na⁺ Reabsorption
As Na⁺ is reabsorbed into the blood:
Blood osmolarity rises slightly
This triggers osmoreceptors → stimulates ADH release
ADH inserts AQP2 → water follows Na⁺ by osmosis
Blood volume increases
Blood pressure increases
Aldosterone Effects Summary
Target Protein | Membrane | Effect | Result |
|---|---|---|---|
ENaC channels | Apical | More Na⁺ enters cell from tubular lumen | ↑ Na⁺ reabsorption |
Na⁺/K⁺ ATPase | Basolateral | More Na⁺ pumped to blood; more K⁺ pumped into cell | ↑ Na⁺ reabsorption; sets up K⁺ secretion |
K⁺ leak channels | Apical | K⁺ exits cell into tubular fluid | ↑ K⁺ secretion → K⁺ excreted |
Water (indirect) | Via ADH | Na⁺ reabsorption raises osmolarity → triggers ADH → AQP2 inserted | ↑ Water reabsorption follows Na⁺ |
Effect of Aldosterone on Urine: Two Scenarios Scenario 1 — High Aldosterone (Low Blood Pressure/Volume or High K⁺)
Event | Result |
|---|---|
↑ ENaC channels in apical membrane | More Na⁺ reabsorbed from tubular fluid |
↑ Na⁺/K⁺ ATPase in basolateral membrane | Na⁺ efficiently moved to blood; K⁺ accumulates in cell |
↑ K⁺ leak channels in apical membrane | K⁺ secreted into tubular fluid → excreted |
↑ Blood osmolarity (from Na⁺ retention) | Triggers ADH → AQP2 inserted → water reabsorbed |
Result | Low Na⁺ in urine; high K⁺ in urine; small concentrated urine volume; ↑ blood volume and pressure |
Scenario 2 — Low Aldosterone (High Blood Pressure/Volume or Low K⁺)
Event | Result |
|---|---|
↓ ENaC channels | Less Na⁺ reabsorbed |
↓ Na⁺/K⁺ ATPase activity | Less Na⁺ moved to blood |
↓ K⁺ secretion | K⁺ retained in blood |
Less water follows Na⁺ | Less water reabsorbed |
Result | High Na⁺ in urine; low K⁺ in urine; more dilute and higher volume urine; ↓ blood volume and pressure |
Clinical Conditions Related to Aldosterone Hyperaldosteronism (Conn's Syndrome)
Adrenal gland produces excess aldosterone (often due to adrenal adenoma)
Excessive Na⁺ reabsorption → excessive water retention
Consequences:
Hypertension (↑ blood volume)
Hypokalemia (excessive K⁺ secretion → low blood K⁺)
Muscle weakness, fatigue, cardiac arrhythmias (from low K⁺)
Urine is low volume and concentrated with high K⁺
Hypoaldosteronism / Addison's Disease
Adrenal cortex fails to produce adequate aldosterone (and cortisol)
Insufficient Na⁺ reabsorption
Consequences:
Hypotension (↓ blood volume)
Hyperkalemia (K⁺ not secreted → accumulates in blood)
Na⁺ wasting in urine
Potentially life-threatening if untreated
Treated with mineralocorticoid replacement (fludrocortisone)
Aldosterone Antagonists — Spironolactone
Blocks the mineralocorticoid receptor → aldosterone cannot bind
Results in:
↓ ENaC channels → less Na⁺ reabsorption → more Na⁺ and water excreted → ↓ blood pressure
↓ K⁺ secretion → K⁺ retained in blood → potassium-sparing diuretic
Used for hypertension, heart failure, hyperaldosteronism
Unlike loop and thiazide diuretics, does NOT cause hypokalemia — actually prevents it
ACE Inhibitors and Aldosterone
Block conversion of angiotensin I → angiotensin II
Less angiotensin II → less aldosterone stimulation
Less Na⁺ and water retention → ↓ blood pressure
Also reduce K⁺ secretion (less aldosterone) → can cause hyperkalemia as a side effect
Part 3 — ADH vs. Aldosterone: Key Comparisons
Feature | ADH | Aldosterone |
|---|---|---|
Chemical nature | Peptide hormone | Steroid hormone |
Produced by | Hypothalamus | Adrenal cortex (zona glomerulosa) |
Released from | Posterior pituitary | Adrenal cortex directly |
Primary stimulus | ↑ Blood osmolarity; ↓ blood volume | ↑ Angiotensin II; ↑ blood K⁺ |
Receptor location | Basolateral membrane (V2 GPCR) | Intracellular (mineralocorticoid receptor) |
Mechanism | cAMP → PKA → AQP2 vesicle fusion | Gene transcription → new ENaC + Na⁺/K⁺ ATPase proteins |
Speed of action | Minutes (vesicle fusion is fast) | Hours (gene transcription takes time) |
Primary substance regulated | Water | Sodium (and potassium) |
Site of action | Principal cells of late DCT + collecting duct | Principal cells of late DCT + collecting duct |
Effect on water | Direct — AQP2 insertion | Indirect — water follows Na⁺ (via ADH) |
Effect on Na⁺ | Indirect — retains water around existing Na⁺ | Direct — increases Na⁺ reabsorption |
Effect on K⁺ | Minimal direct effect | Direct — increases K⁺ secretion |
Effect on urine volume | ↑ ADH → ↓ urine volume | ↑ Aldosterone → ↓ urine volume (indirectly) |
Effect on urine concentration | ↑ ADH → ↑ urine concentration | ↑ Aldosterone → ↑ urine concentration (indirectly) |
Clinical excess | SIADH → hyponatremia | Conn's syndrome → hypertension + hypokalemia |
Clinical deficiency | Diabetes insipidus → massive dilute urine | Addison's disease → hypotension + hyperkalemia |
Part 4 — How ADH and Aldosterone Work Together
Although they regulate different substances, ADH and aldosterone are functionally linked and work in a coordinated fashion:
The Link: Na⁺ Retention Drives Water Retention
Aldosterone increases Na⁺ reabsorption → blood Na⁺ concentration rises → blood osmolarity rises
↑ Blood osmolarity detected by hypothalamic osmoreceptors → triggers ADH release
ADH inserts AQP2 → water reabsorbed → blood volume restored
Net: aldosterone drives Na⁺ reabsorption; the resulting osmolarity change then recruits ADH to bring water along
The RAAS Activates Both
Low blood pressure/volume → RAAS activated → angiotensin II produced
Angiotensin II:
Directly stimulates aldosterone release → Na⁺ reabsorption
Directly stimulates ADH release from posterior pituitary → water reabsorption
Both hormones are activated simultaneously → coordinated volume restoration
Summary of Coordinated Response to Dehydration
Step | Hormone | Event | Result |
|---|---|---|---|
1 | — | Dehydration → ↓ blood volume + ↑ blood osmolarity | Triggers both RAAS and osmoreceptors |
2 | Angiotensin II | Stimulates adrenal cortex | Aldosterone released |
3 | Aldosterone | ↑ ENaC + Na⁺/K⁺ ATPase in collecting duct | Na⁺ reabsorbed → blood osmolarity ↑ |
4 | ADH | Released by posterior pituitary (from osmoreceptors + angiotensin II) | AQP2 inserted → water reabsorbed |
5 | Both | Na⁺ and water retained | ↑ Blood volume; ↑ blood pressure; small concentrated urine |
6 | Negative feedback | Blood volume and osmolarity restored | Aldosterone and ADH release suppressed |