CHEM 241L
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
PPE Eye Protection
General Lab
1.Safety goggles- worn over regular glasses2. Outside bought goggles must be approved
PPE Skin Protection
1. Approved Lab Coat
2. Clothing that protect's the body from the shoulders to the ankles: avoid loose sleeves and cuffs, and bracelets
3. Footwear that entires foot: no open-toed, open-heeled, high heels, sandals, flip flops, flats, etc.
4.Gloves must be worn at all times while handling glassware and/or chemicals: remove before leaving the lab- place in proper recycling bin or hazardous waste
5. Long hair must be tied back
Laboratory Conduct
1. you are not permitted to enter or work in the lab without TA supervision
2. no bags, coats, or umbrellas are allowed by the laboratory bench
3.Report any accidents, injuries, or hazardous spills, no matter how minor, to your TA or instructor at once
4. Do not sit on the floors in the hallways or lab spaces
AAA
Always Add Acids (to water) never water to acids
Waste Disposal
Proper disposition of a discarded or discharged material in accordance with local environmental guidelines or laws
Safety Equipment
1. Fire Extinguishers
2.Shower- near corridor
3.Eye Wash station- at the front sink
4. First aid kit- mounted on side of front hood
5. Telephone
Late Lab Reports
1. 24 hours late= 10% deduction
2. between 24 and 48 hours late= 20% deduction
3. Later than 48= 0
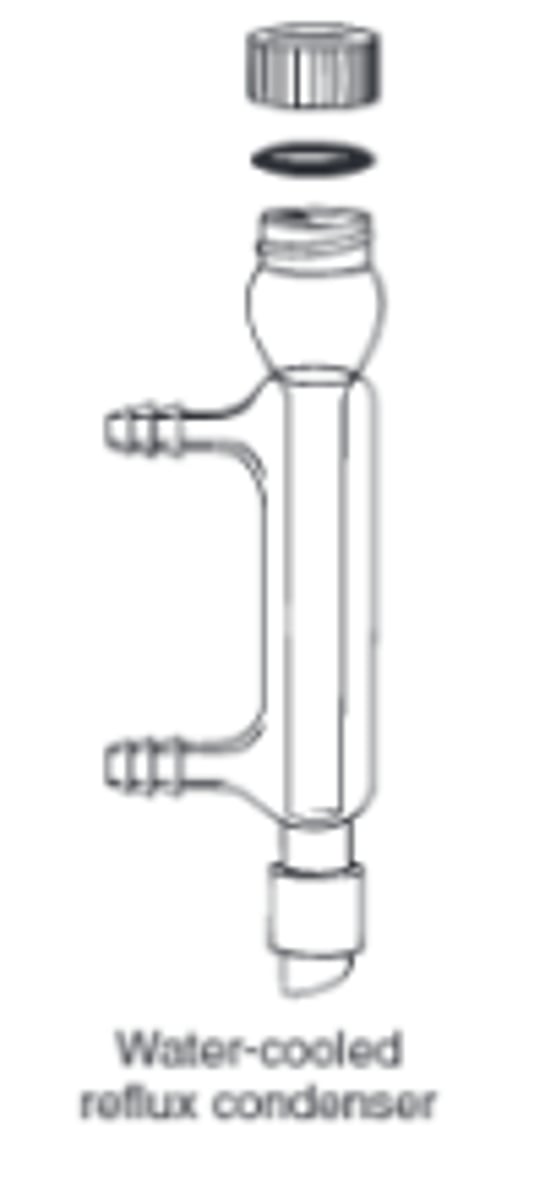
reflux condenser
Fitted vertically in a flask to prevent vapour escaping while a liquid is being heated. Vapour from the boiling reaction mixture condenses and flows back into the flask
Buchner Funnel

Ceramic Hirsch Funnel

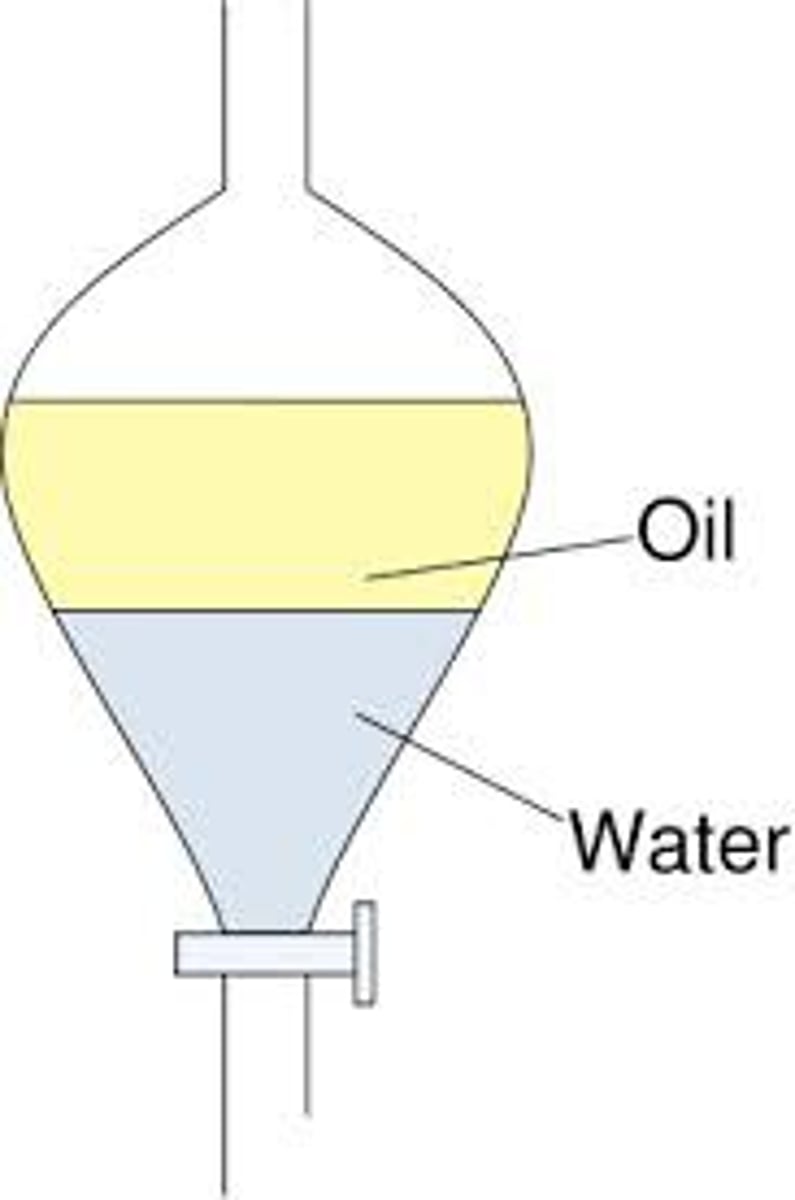
separatory funnel
Used to portion off components of mixture between two immiscible (doesn't mix) liquids of varying densities.
NMR tubes
Filter flask
used in conjunction with a vacuum connection to a water faucet to speed up filtration

Watchglasses

Distillation Head

Connecting adaptor
how to use a separatory funnel
Ensure the funnel is clean and dry before use.
Add the mixture of liquids to the funnel, filling it no more than two-thirds full.
Securely close the stopcock at the bottom to prevent leakage.
Invert the funnel gently and swirl to mix the liquids, then allow them to settle.
Open the stopcock to release the denser liquid from the bottom layer into a separate container.
Close the stopcock once the bottom layer is fully drained, then remove the top layer if needed.
Experiment 4 Key Objectives
Calibrate laboratory equipment with a focus on volume-measuring equipment and the proper use of volumetric glassware. Create a calibration curve to correct volume measurements. Use a micropipette, a volumetric flask, a balance, and a pH probe. Make a buffer solution to a given pH. Analyze buffer capacity and to understand its significance
Calibration
process of relating an actual physical quantity to the quantity indicated on the scale of an instrument
Increases the accuracy of a measurement
Correction Factor
adjustment that is necessary
for physical quantity to equal quantity indicated
on the scale of an instrument
Vmeasured - Vexpected
Tolerance
Accepted variance in precision
Gravimetric Analysis
a type of quantitative analysis in which the amount of a species in a material is determined by converting the species to a product that can be isolated completely and weighed
Calibrated a pipette, increased volume after each addition
Buffer
Solution resistant to a change in pH, even when a strong base or strong acid is added
Buffered Solution chemically made
chemically, a buffered solution contains substantial amounts of both a weak acid and its conjugate base- there needs to be enough of both present to react with either an acid or a base Ex: Formic acid will have a small amount of dissociation in water, producing a very small amount of its conjugate base (formate)
Henderson Hasselbach
this equation is used when working with buffer problems: OFTEN USED TO DETERMINE THE PH OF A BUFFERED SOLUTION
pH= pKa + Log ([Conjugate base]/[weak acid])
or
pH=pKa +Log ([A-]/[HA])
pKa is for the weak acid
Beer’s Law
A= measured absorbance at a given wavelength
E= molar absorptivity (M^-1 cm^-1)
b= path length of the cuvette (cm)
C= concentration (M)
MOLAR ABSORPTIVITY IS CONSTANT FOR EVERY MOLECULE AND PROVIDES INFORMATION ON HOW MUCH LIGHT THAT MOLECULE CAN ABSORB AT A PARTICULAR WAVELENGTH
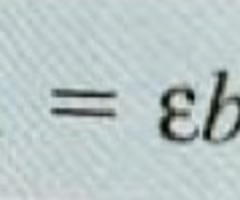
Buffer Capacity
the number of moles needed of a strong base or acid to change pH of 1L buffered solution by 1 pH unit
First derivative of the titration curve
d = change

In this lab, you calibrated a micropipette and determined the correction factor. How would you apply the correction factor if the volume delivered was between two points on your calibration curve?
Find the correction factor and also find “in between value” based on this pattern

Stocks of HCl and NaOH
1.0 M HCl and NaOH
For 1.4, 0.100 M HCl and NaOH in addition to 0.050 M prepared stocks
Formal Concentration
moles of strong base or acid added / Corrected Volume + Starting in beaker
Vctv+Vin
Picking an acid/base pair
Pick the weak acid whose pKa is closest to your target pH.
Calculating appropriate amount of solution to make buffer
Use Henderson Hasselbach (ratio of base concentration over acid concentration)
R/ 1 + R Concentration Total = Base 1/ R +1 C total = Acid
Multiply by V final to get moles
Divide moles by concentration
Practical Buffer
Measure out the weak acid and/or conjugate base solutions (often approximate amounts).
Combine them in a beaker or flask.
Insert a calibrated pH meter.
Add small amounts of strong acid (HCl) or strong base (NaOH) dropwise.
Stir and monitor until the solution reaches the target pH.
Dilute to the final volume
Easier, fast, does not require calculations but it might be less accurate, can overshoot the pH, buffer capacity might be lower or uneven, broader capacity
Theoretical Buffer
Choose an acid/base pair with pKa close to the target pH.
Use Henderson–Hasselbalch to calculate the required ratio:
[base][acid]=10pH−pKa
Decide on a total buffer concentration.
Calculate the exact moles of acid and base needed.
Measure those amounts precisely (mass or volumetric).
Mix and dilute to the final volume
Highly reproducible, exact acid base ratio, predictable buffer capacity, no titrant contamination (BUT requires accurate calculations, you cannot “fix it with titrant”
Experiment 3
Determine the pKa of
bromothymol blue.
2. Construct absorption spectra
using a UV-VIS spectrophotometer
3. Use Beer’s Law to draw
conclusions about
concentrations.
4. Perform a
solvent-solvent
extraction to
qualitatively analyze
solubility
determine the pKa of bromothymol blue, a common acid-base indicator
do this by determining the maximum wavelength for the basic and the acid
Absorbance
The absorption of light by molecules and materials is how we see colors
A=Log(Io/I)
-Io = irradiance of the light measured in the absence of an absorbing solution
-I = irradiance of light measured after it passes through a solution of interest
-typically measured between 0 and 2 since above that, only a small portion of the light is being transmitted to the detector
-Concentration is DIRECTLY proportional to absorbance
Transmittance
Wavelengths of light that pass through sample
A = log(Io/I) -log(T)
Solvent Solvent Extractions
Solutes prefer aq(polar, deprotanoted) or org form, move between phases until system reaches solute equilibrium
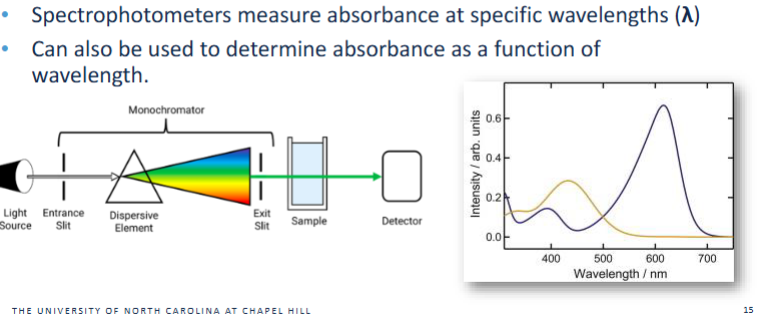
Spectrophotometers
Measure absorbance at specific wavelengths
Experiment 3 Technique
-determine the pKa of bromothymol blue
-photometric mode
-set spec ti max wavelength of basic (In-)
-measure absorbance at max for 8 buffers
-use max for basic form and Beer's Law to solve the concentration of In- at each pH
-fromt his construct table and plot to find pKa
Color Indicators
class of dye molecules designed such that the acidic (HIn) yellow and basic (In-) blue forms of the dye molecules are different colors
-allows you to visually observe the change from acid to base and vice versa
-commonly used to detect the endpoints in acid-base titrations
Color Change
occurs because of the structural reorganization of the bonds in bromothymol blue when it loses or accepts a proton
- the acidic proton (the proton lost to water) - converted to conjugate base (anion)- the acidity comes from the ability to stabilize this
Bromothymol loses proton
When bromothymol blue loses its proton (In-), the negative charge does not reside on the oxygen atom. The bonds in the ring reorganize to delocalize the negative charge
Cause of color change
the five-member ring that contains the sulfur atom is broken in this structural rearrangement process, thus accounting for the dramatic color change from yellow to blue
Bromothymol solubilities
the two forms of bromothymol blue have varying polarities due to their difference in molecular charge- this leads to difference in solubility in aqueous and organic solvents (need solvent-solvent extraction)
Experiment 3 Additional Information
pH used: 5, 6, 7, 7.3, 7.6, 8, 9, 10
Draw from aqueous layer
UV Vis and how Spectrophotometers work
Reason 1 — Bromothymol blue has different absorbance spectra in acidic vs. basic forms
Acidic form (HIn): λmax ≈ 430 nm, appears yellow
Basic form (In⁻): λmax ≈ 600 nm, appears blue
As pH changes, the relative amounts of these two species change — and UV–Vis directly measures that shift.
Reason 2 — UV–Vis allows quantitative determination of pKa
The Henderson–Hasselbalch relationship links absorbance to the ratio of acidic/basic forms.
By measuring absorbance across a pH series, you can determine the pH where the two forms are equal → pKa.
For the analysis of the solvent-solvent extraction, you were required to use a glass cuvette instead of a plastic one. Why was this necessary?
Organic solvents attack plastic
Your extraction used acetone and an organic layer (often hexane, benzene, or similar).
Most plastic cuvettes (polystyrene, PMMA) are not compatible with organic solvents:
They swell
They dissolve
They become cloudy
They leach plasticizers into the sample
Any of these would distort the UV–Vis signal.
Glass (especially quartz or optical glass) is chemically resistant to organic solvents, so it stays clear and intact.
2. Plastic absorbs strongly in the UV region
Plastic cuvettes often absorb below ~300–350 nm.
Your pigment extract and indicator forms have absorbance features that extend into the UV.
Using plastic would:
Increase baseline absorbance
Distort peak shapes
Reduce accuracy of λmax measurements
Glass has much lower UV absorbance, giving clean spectra.
3. Organic solvents can permanently etch or fog plastic
Even brief exposure can leave:
Micro‑scratches
Haze
Residue
Experiment 1
Prepare a set
of standard
EtOH solutions
in 1-Pentanol
2%, 4%, and
6%
Analyze standard
solutions via gas-
liquid
chromatography
Construct a calibration
plot using data
collected from GC
analysis of standard
solutions
Ethanol peak area vs.
% ethanol
Determine the
partition
coefficient of
ethanol
between water
and 1-Pentanol
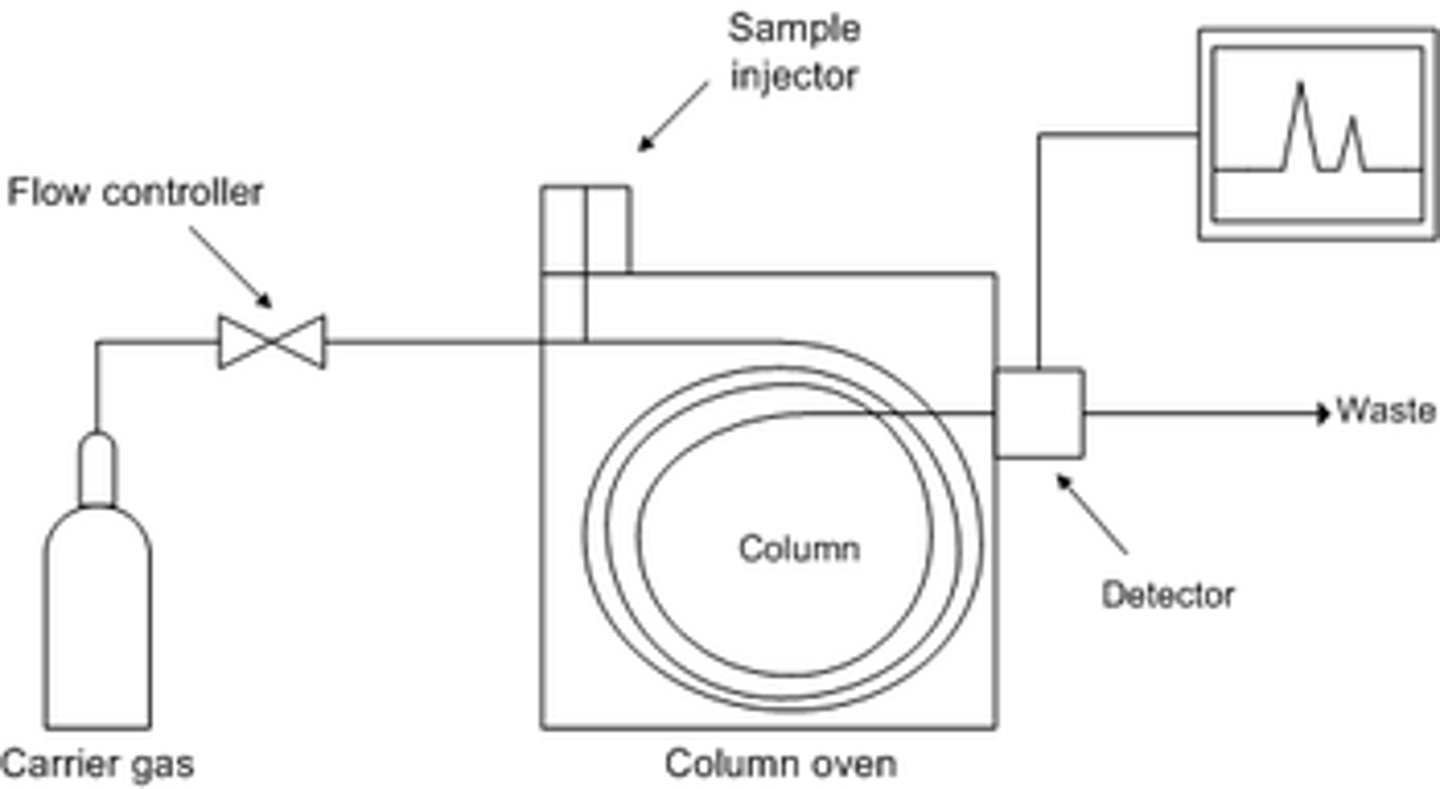
Gas Chromatography
Gas Chromatography-
separation technique based
on the differential
partitioning of chemical
species into the liquid
stationary phase versus the
gaseous mobile phase.
• Used to separate volatile
organic compounds
Making an Injection
Clean the syringe
1. Draw 2.5 μL of 100% 1-pentanol and dispense to waste. Repeat once.
2. Repeat with your sample to be injected
3. Check that the needle is expelling liquid – if not, the needle is clogged, and
you should check with your TA
2. Wet the syringe barrel (remove air bubbles)
1. Immerse the clean needle into the sample you wish to inject
2. Quickly pull up and down on the plunger
3. Draw up 2.5 μL of sample.
4. Make the injection following lab manual instructions
Solvent-solvent extraction
The analyte is transferred to a new solvent in which only the analyte is soluble- relies on the solubility of the analyte in different liquids (best when the analyte has a higher solubility than it does in the original solvent and the other compounds have low solubility)