7.Carboxylic acids/Carbonic acids
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Carbonic acids
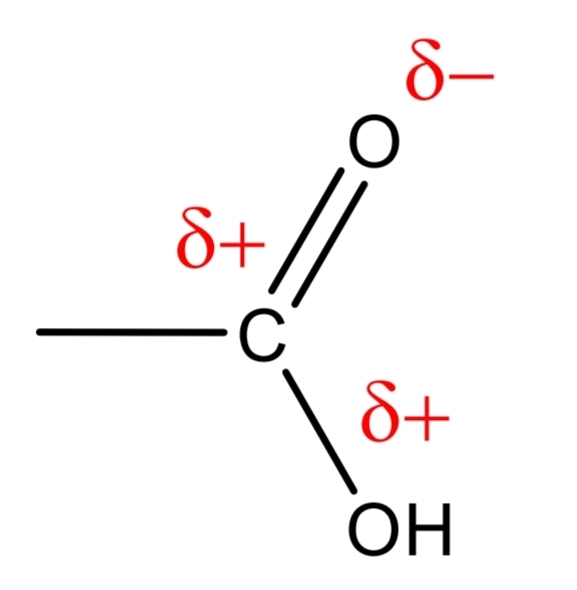
=O and –OH group influences each other:
• =O it does not show the properties typical of aldehydes
• -OH it dissociates much more
Carbonic acids: Numbering
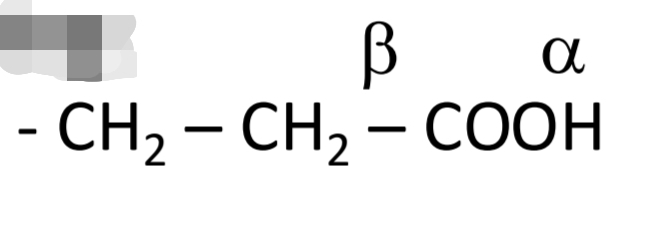
Reactions of carbonic acids
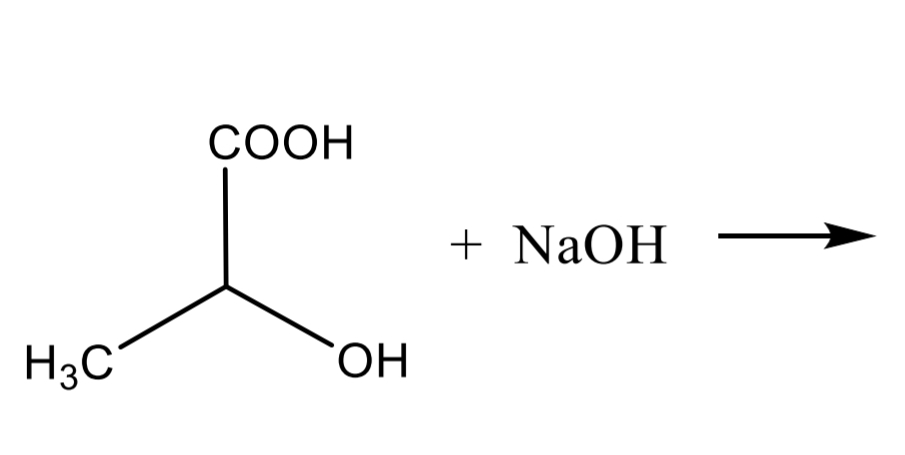
Salt formation: NaOH, Na2CO3, Na
Ester formation
Decarboxylation: under the influence of heat
Strongest acid
Formic acid
Acid Strength
+ alkil-group – (e-sending prop.) → acid strength decreases
+ e- attractive group (Cl, Br) → acid strength increases
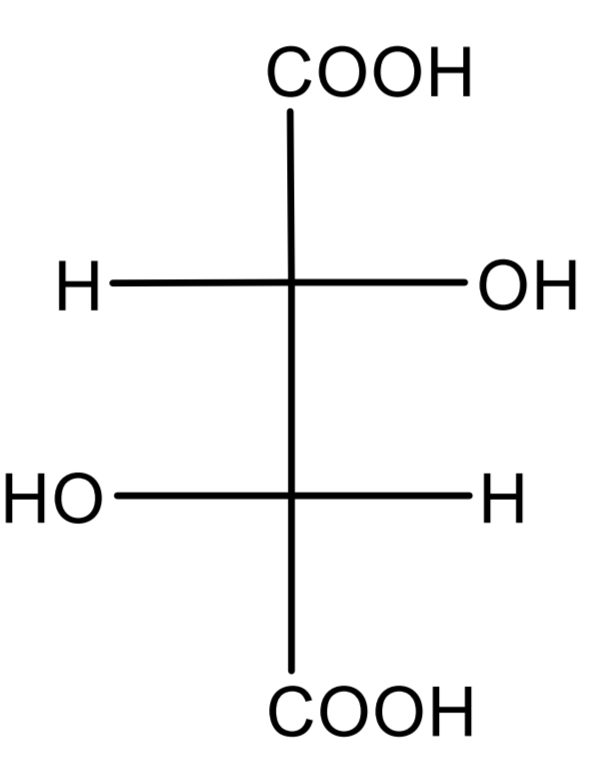
Dicarboxylic acids
one is a strong acid, the other a weak acid
Aromatic acids
Stronger
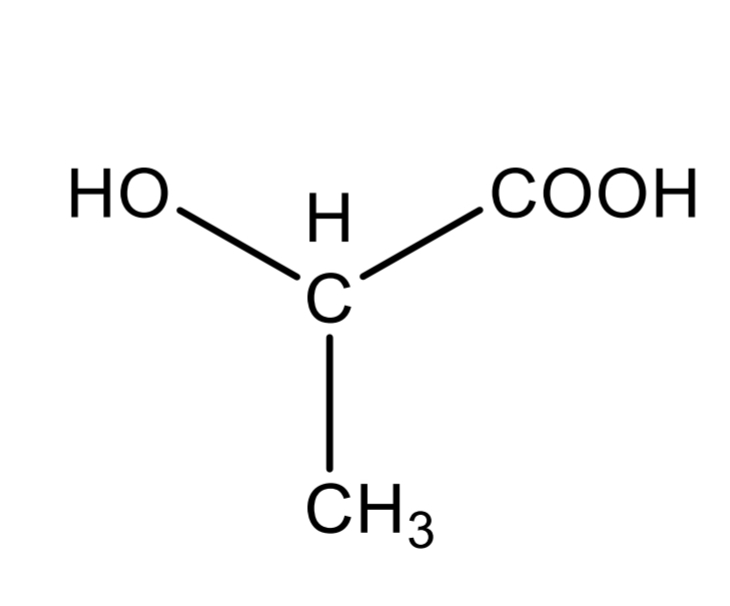
Lactic acid
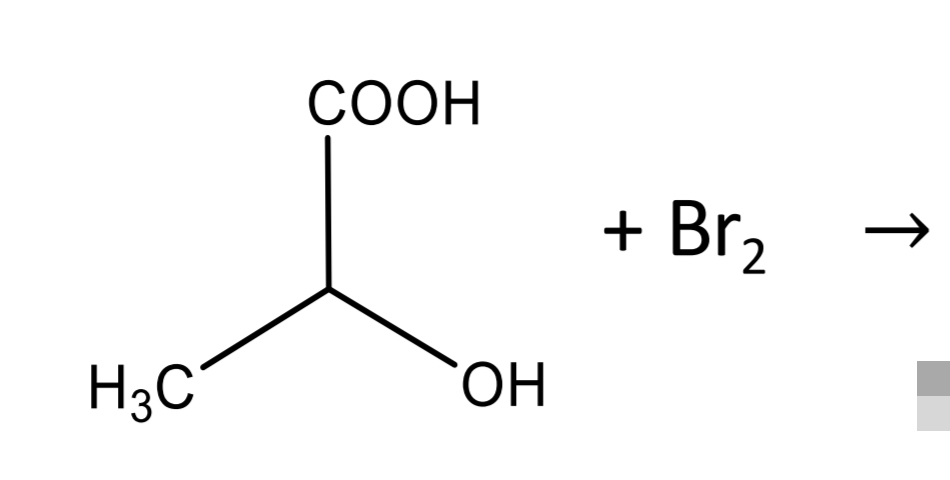
Lactic acid identification: Legal reaction_Step 1
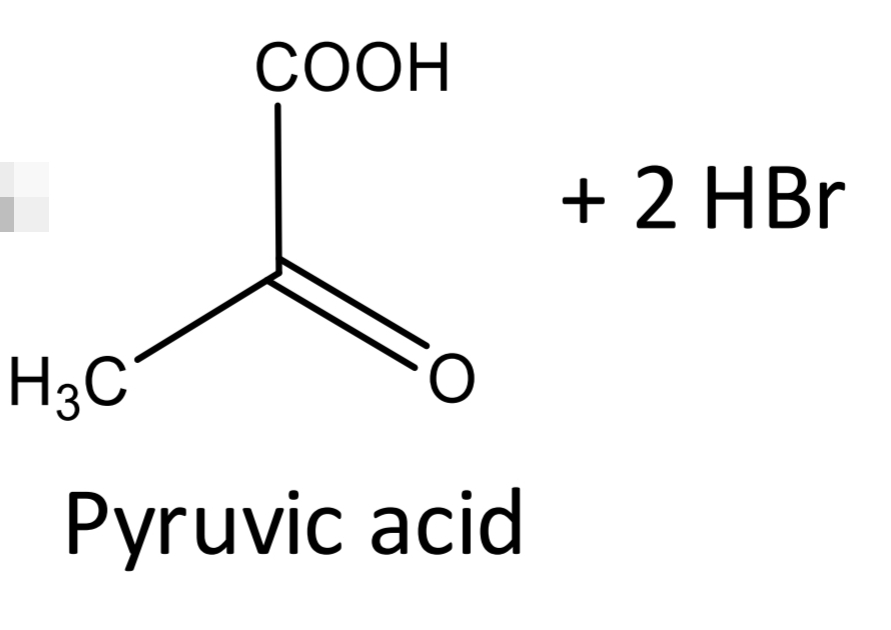
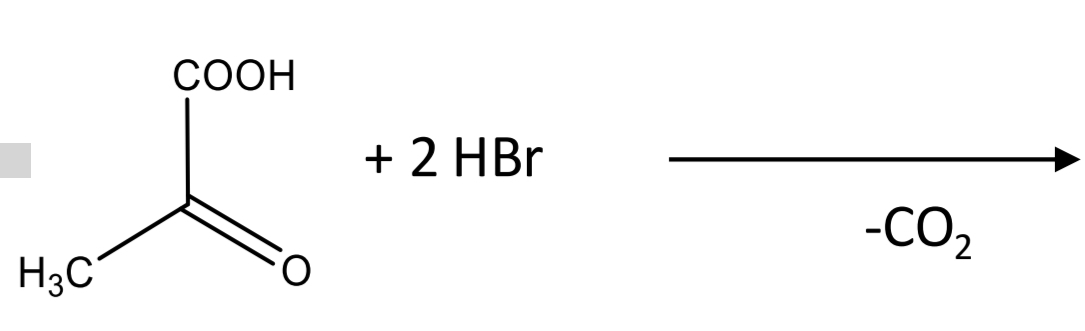
Lactic acid identification: Legal reaction_Step 2
acetaldehyde
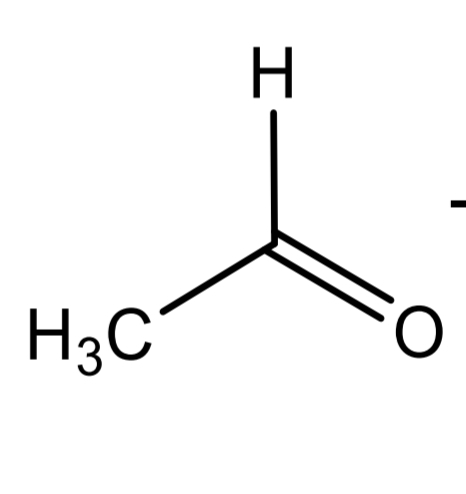
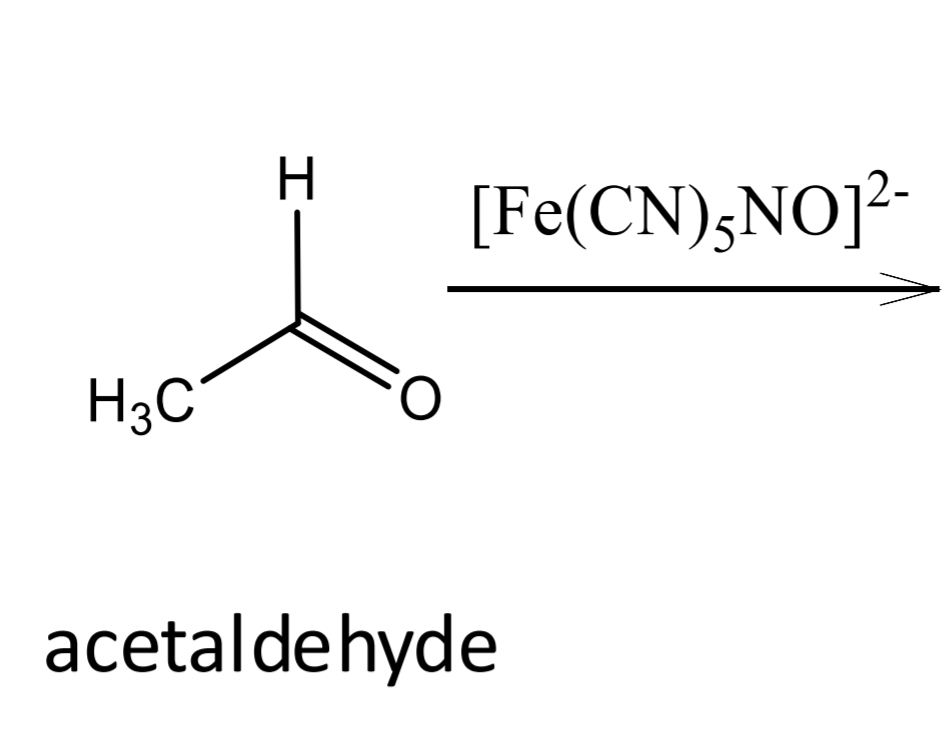
Lactic acid identification: Legal reaction_Step 3
Sodium nitroprusside: [Fe(CN)5 NO]2-
![<p>Sodium nitroprusside: [Fe(CN)5 NO]2-</p>](https://assets.knowt.com/user-attachments/f3ef4bda-771d-4148-a0d7-2a9daf078d4a.jpg)
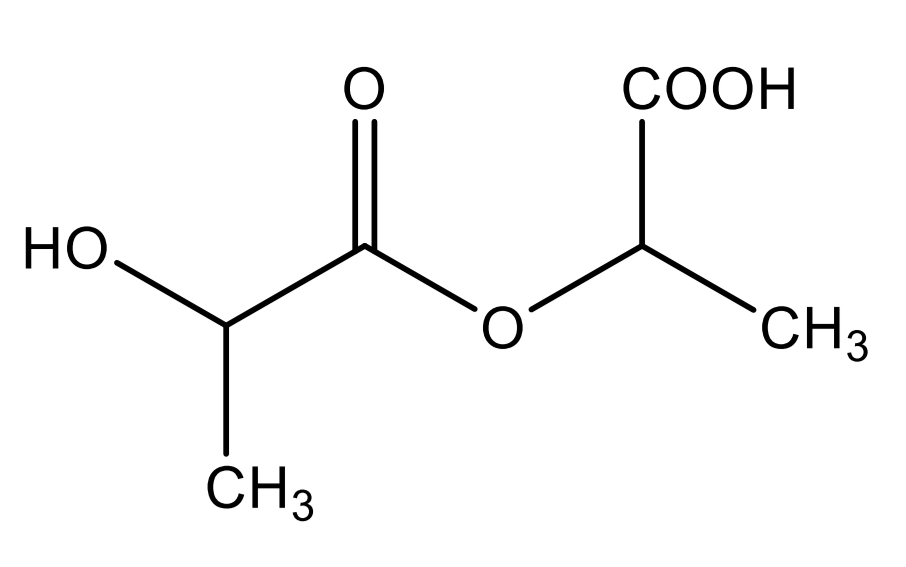
Lactil lactate
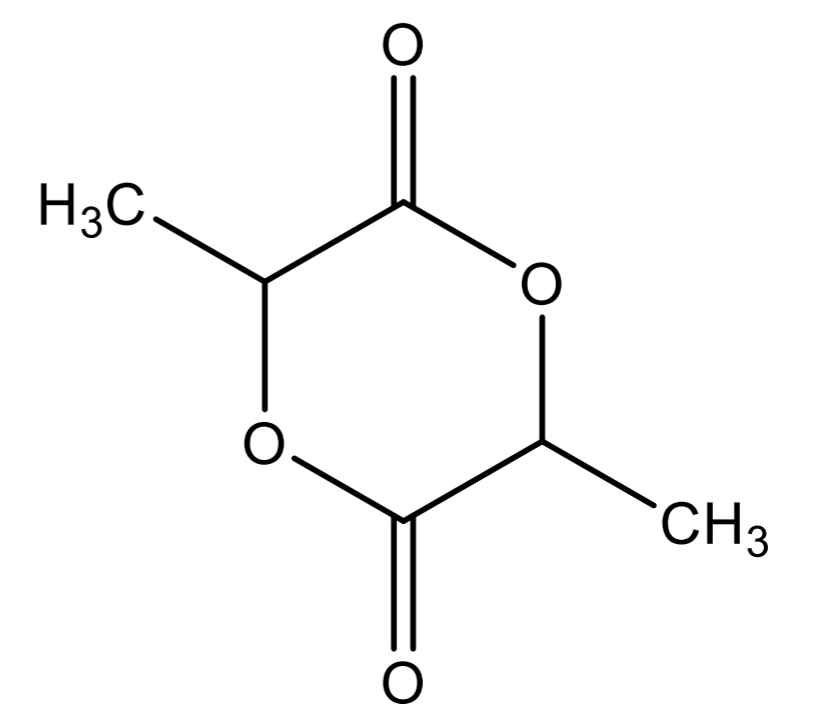
Dilactone
Lactic Acid Assay Step 1
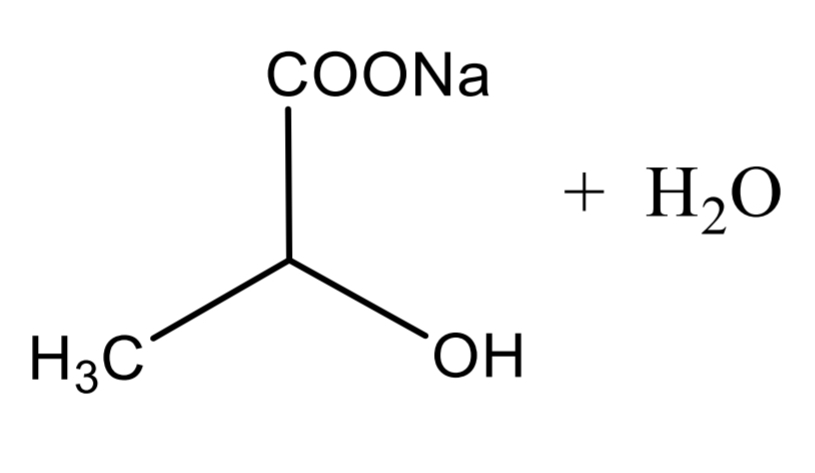
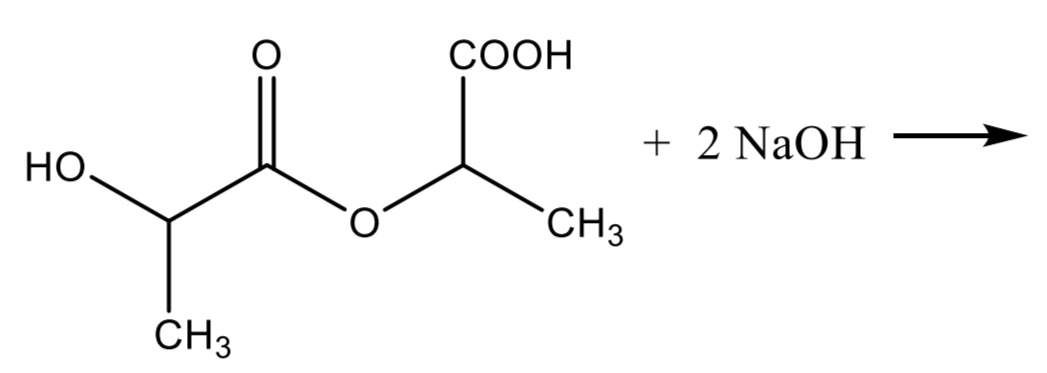
Lactic Acid Assay Step 2
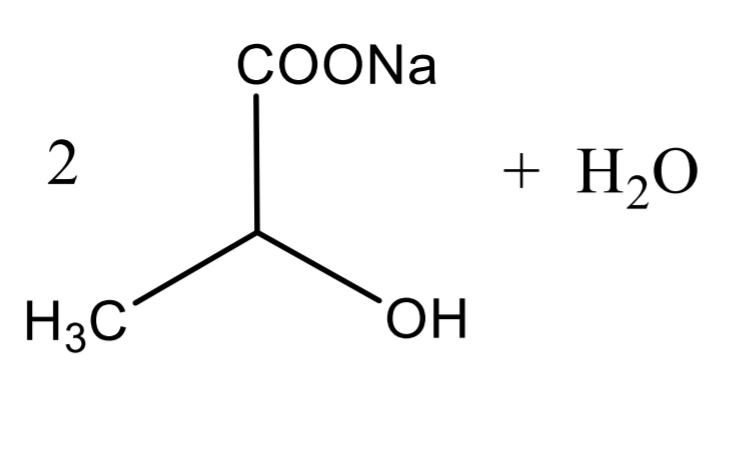
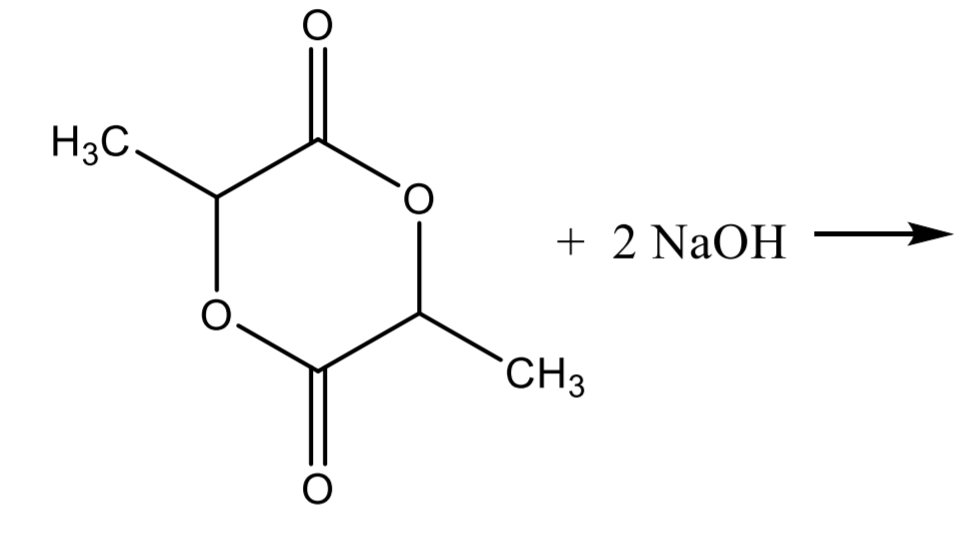
Lactic Acid Assay Step 3
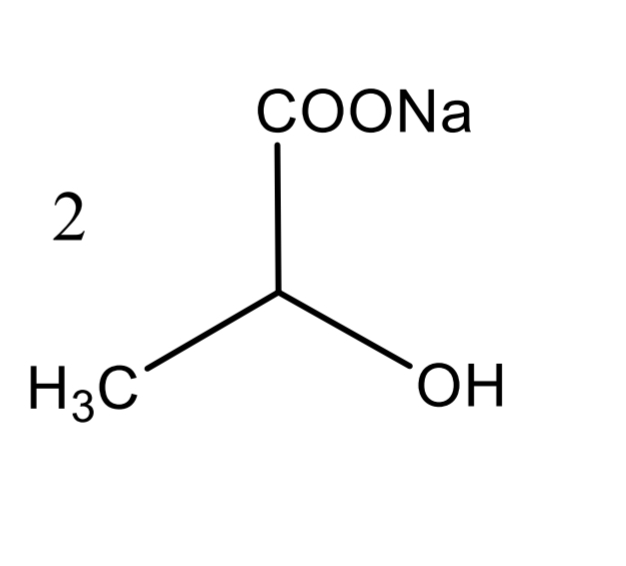
Lactic Acid Assay Calculation
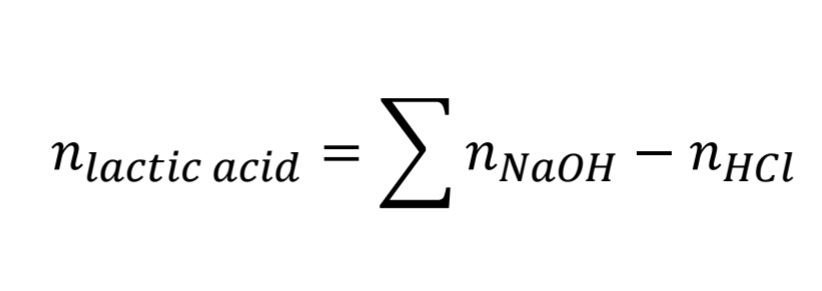
Tartaric acid
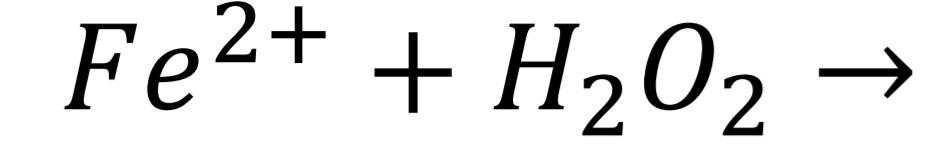
Tartaric acid Identification: Fenton reaction Step 1
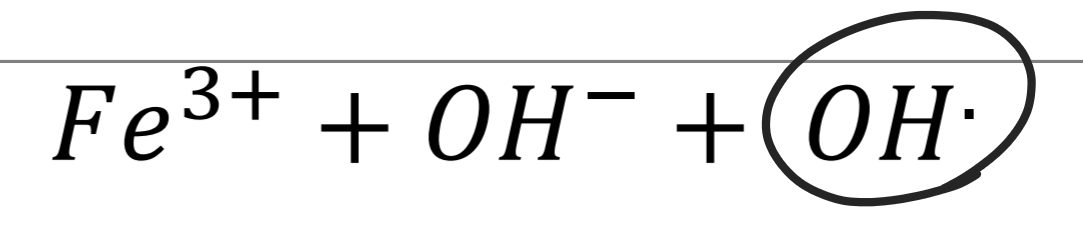
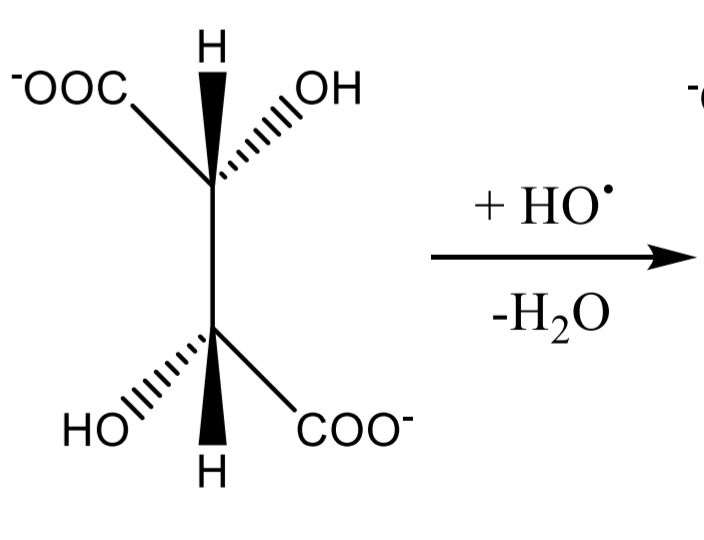
Tartaric acid Identification: Fenton reaction Step 2
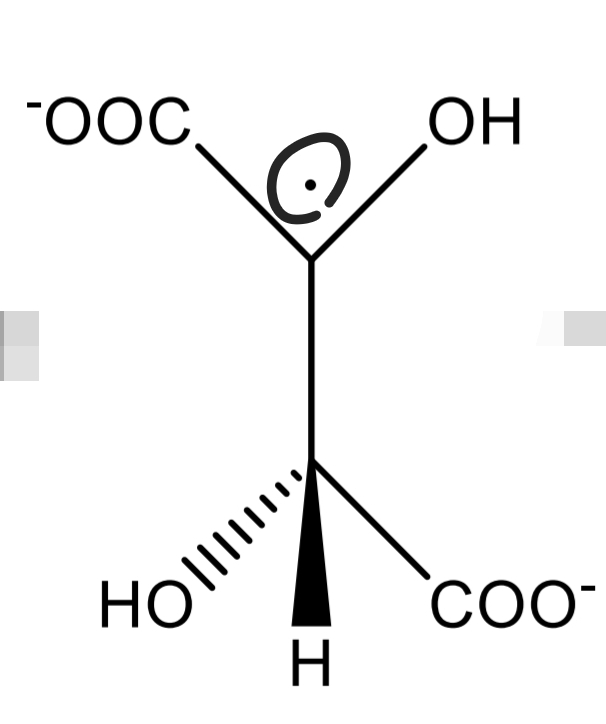
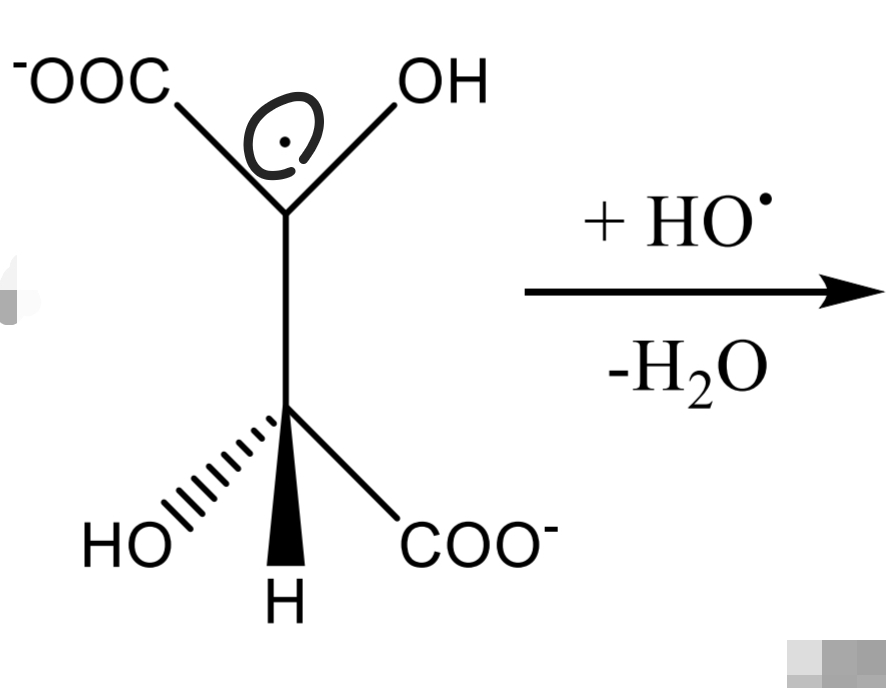
Tartaric acid Identification: Fenton reaction Step 3
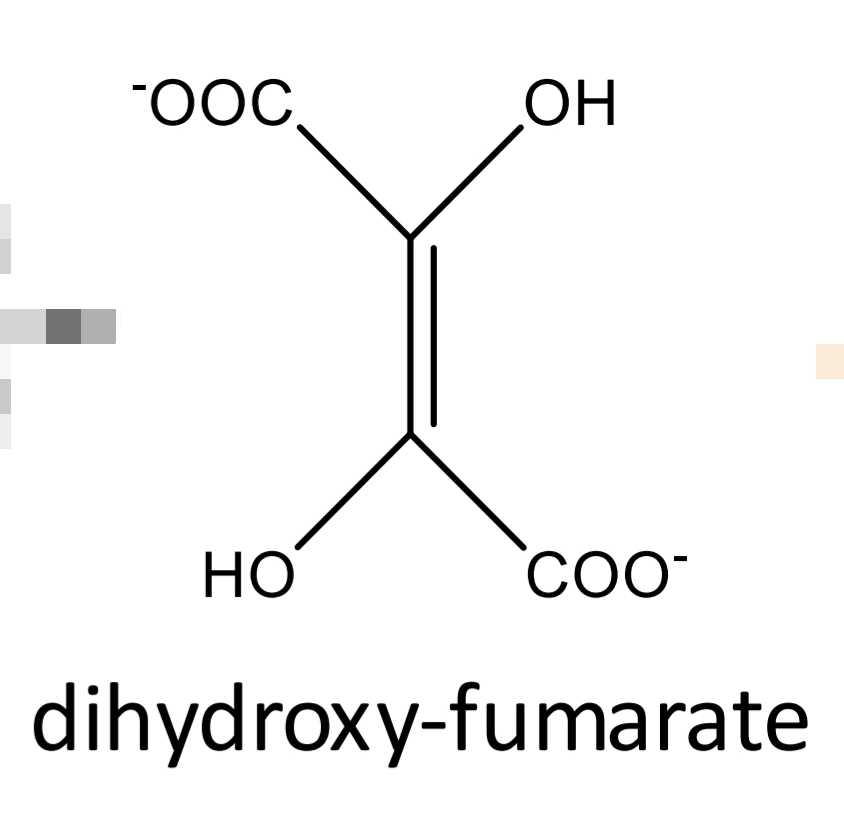
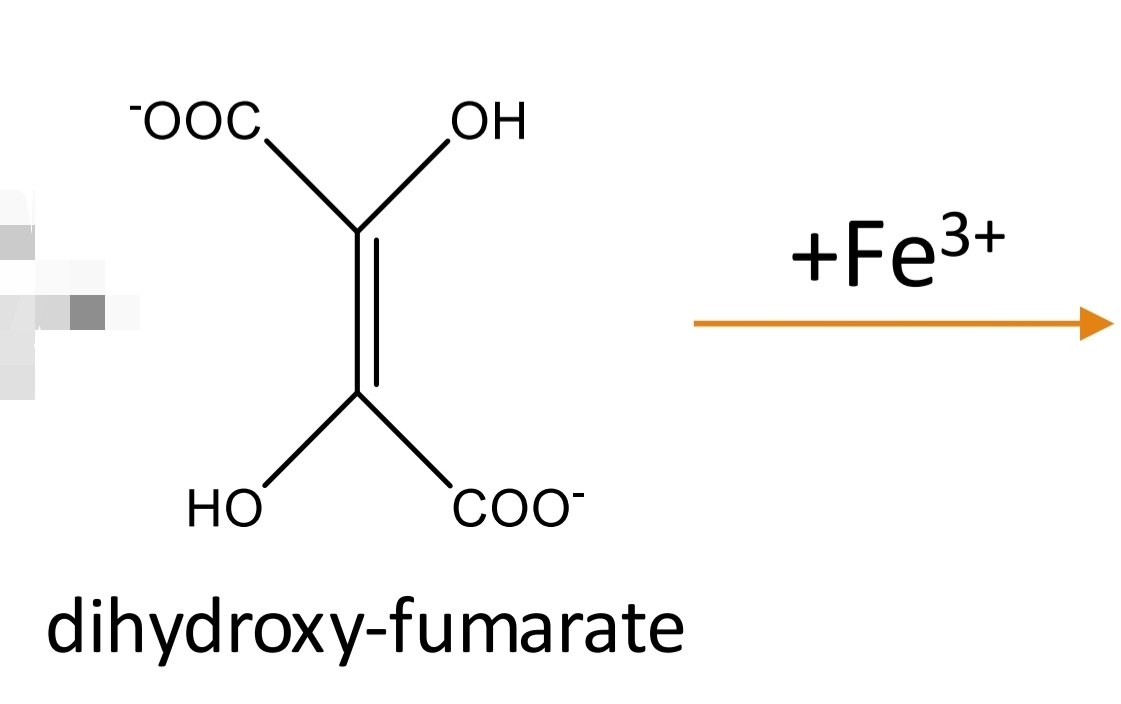
Tartaric acid Identification: Fenton reaction Step 4

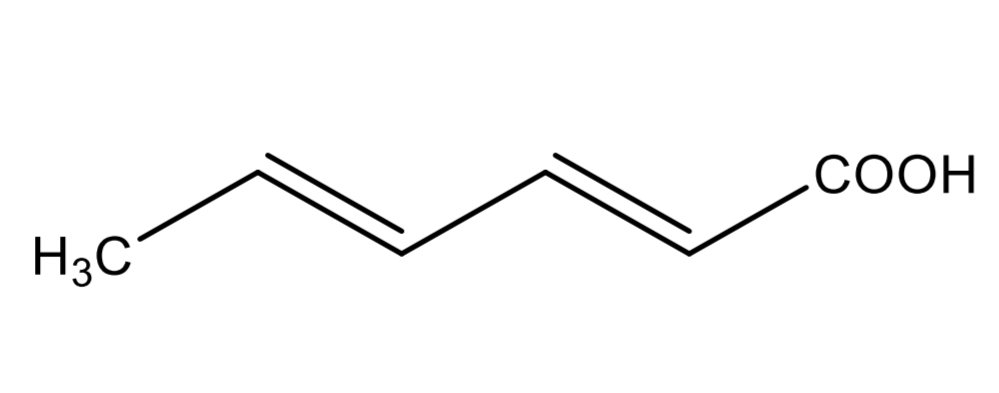
Sorbic acid
Citric acid
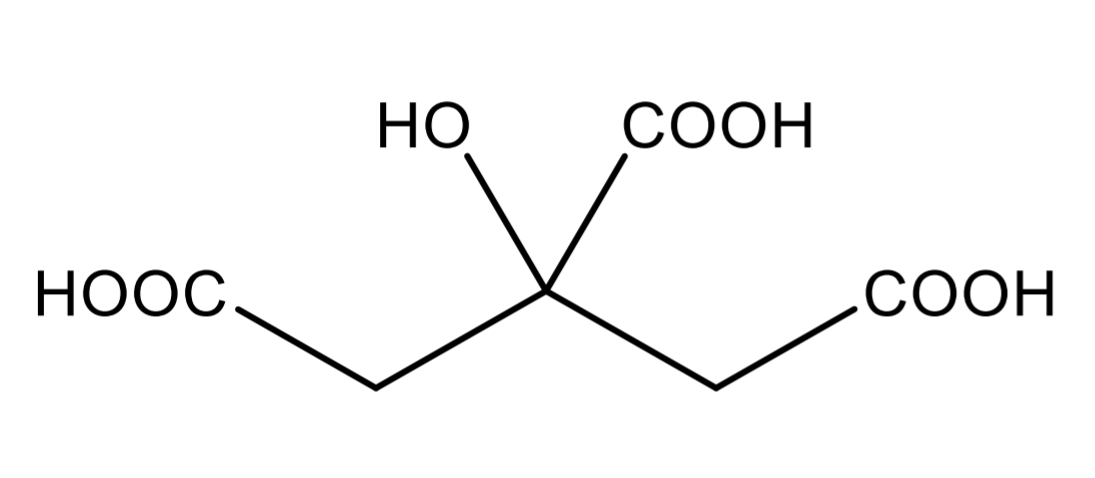
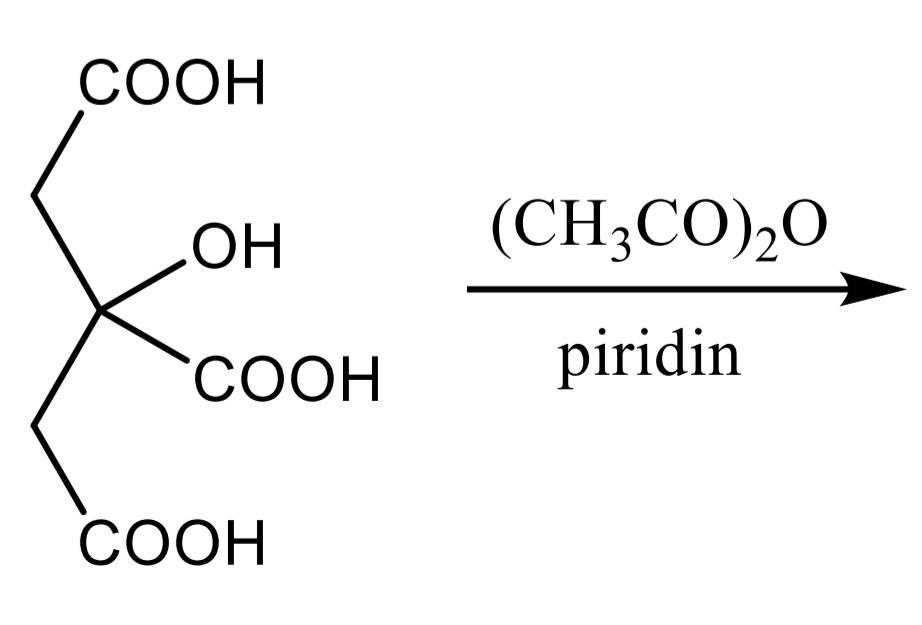
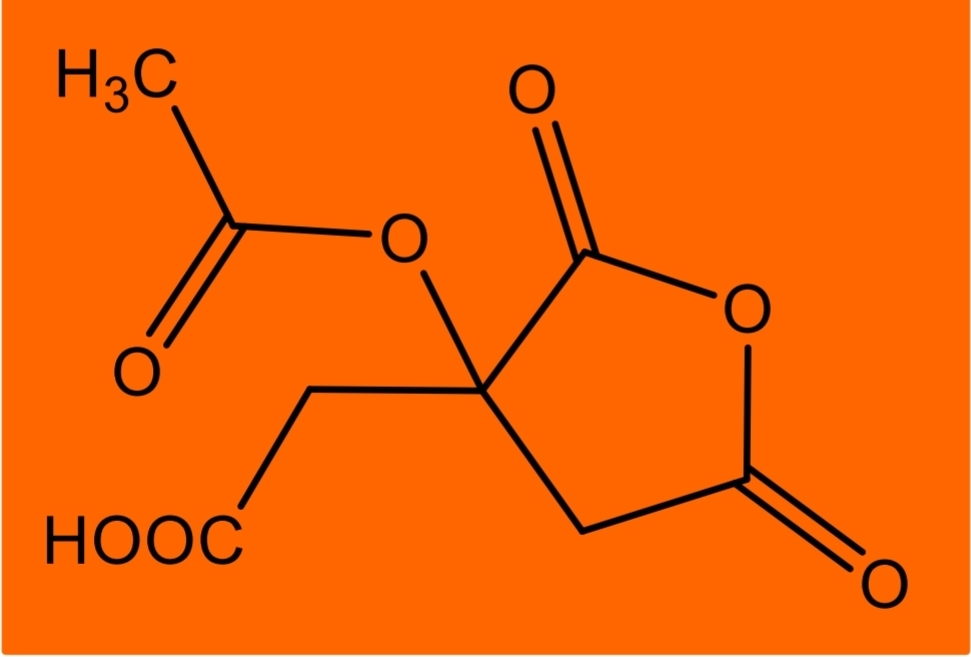
Citric acid Identification: dissolve in acetic anhydride + pyridine mixture
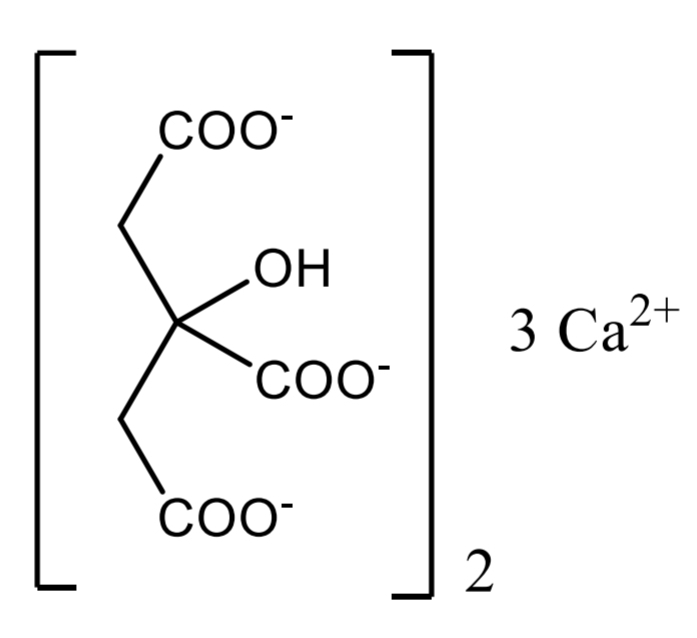
Citric acid Identification: compound + water + NaOH solution + calcium-chloride-solution → boiling → white ppt
White ppt.
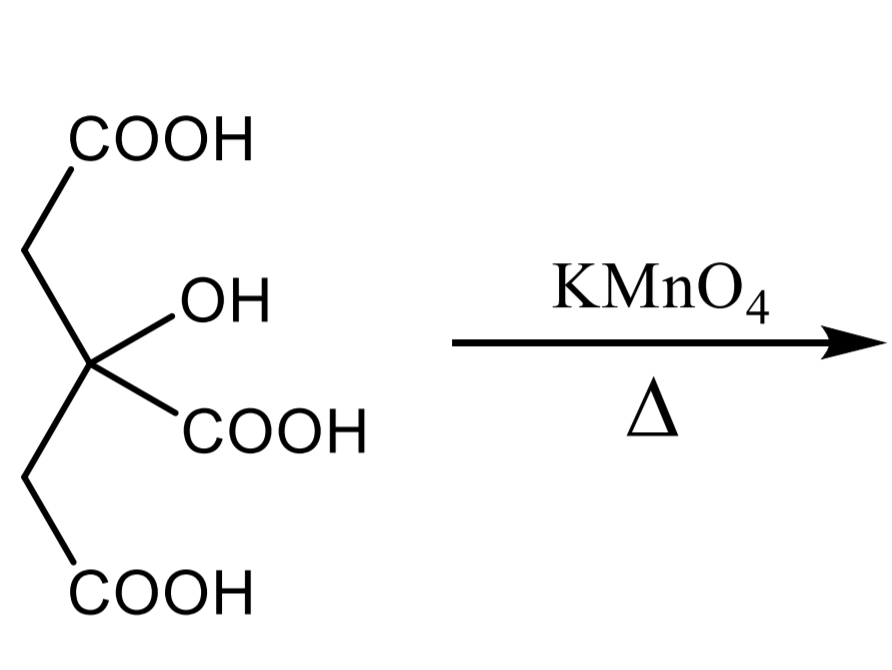
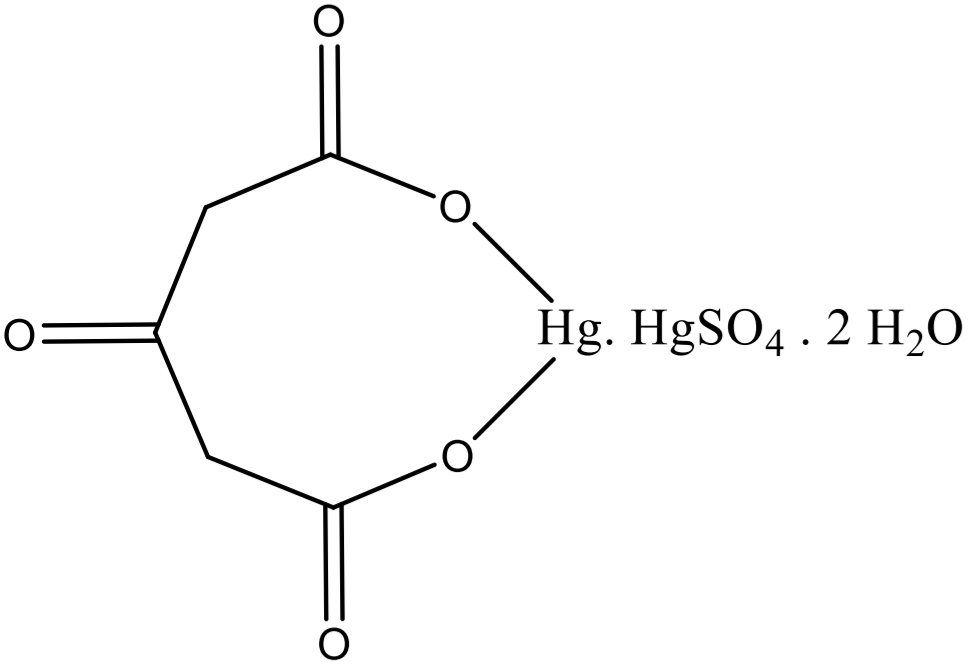
DENIGES-REACTION: Citric acid Step 1
compound + water + mercury (II) sulphate solution → boiling → + 3 drops potassium permanganate → white ppt
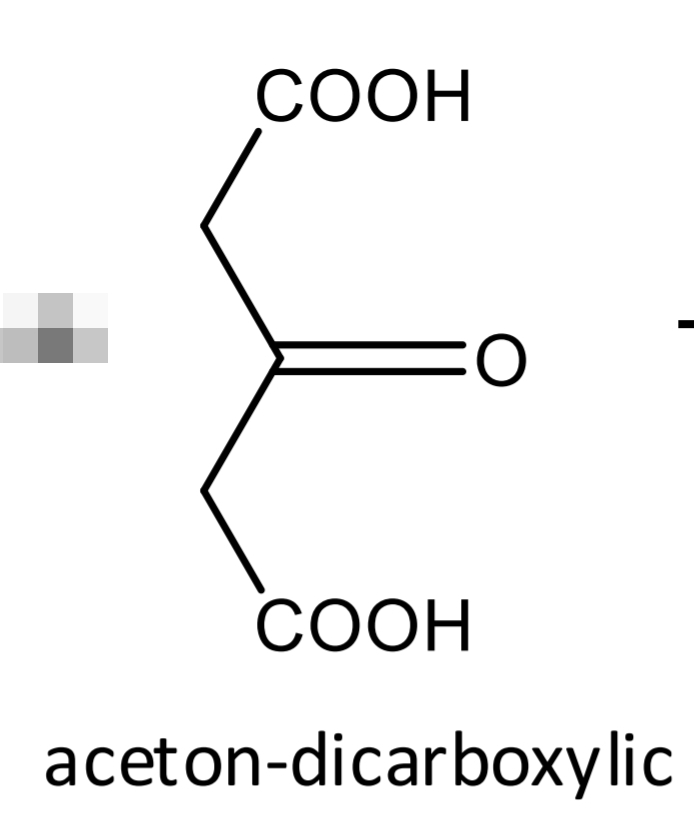
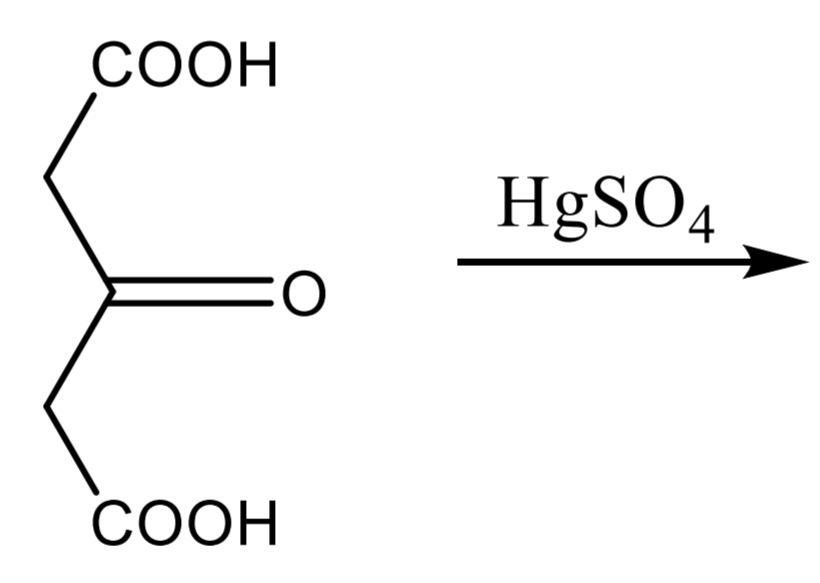
DENIGES-REACTION: Citric acid Step 2
White ppt