. ݁₊ ⊹ bio lab ; practical study guide (new one)
1/62
Earn XP
Description and Tags
bio lab practical
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
☆ what are the steps of the scientific method?
observation/problem → background info → hypothesis → prediction → experiment → data analysis → conclusion
revise the hypothesis if results do not support it
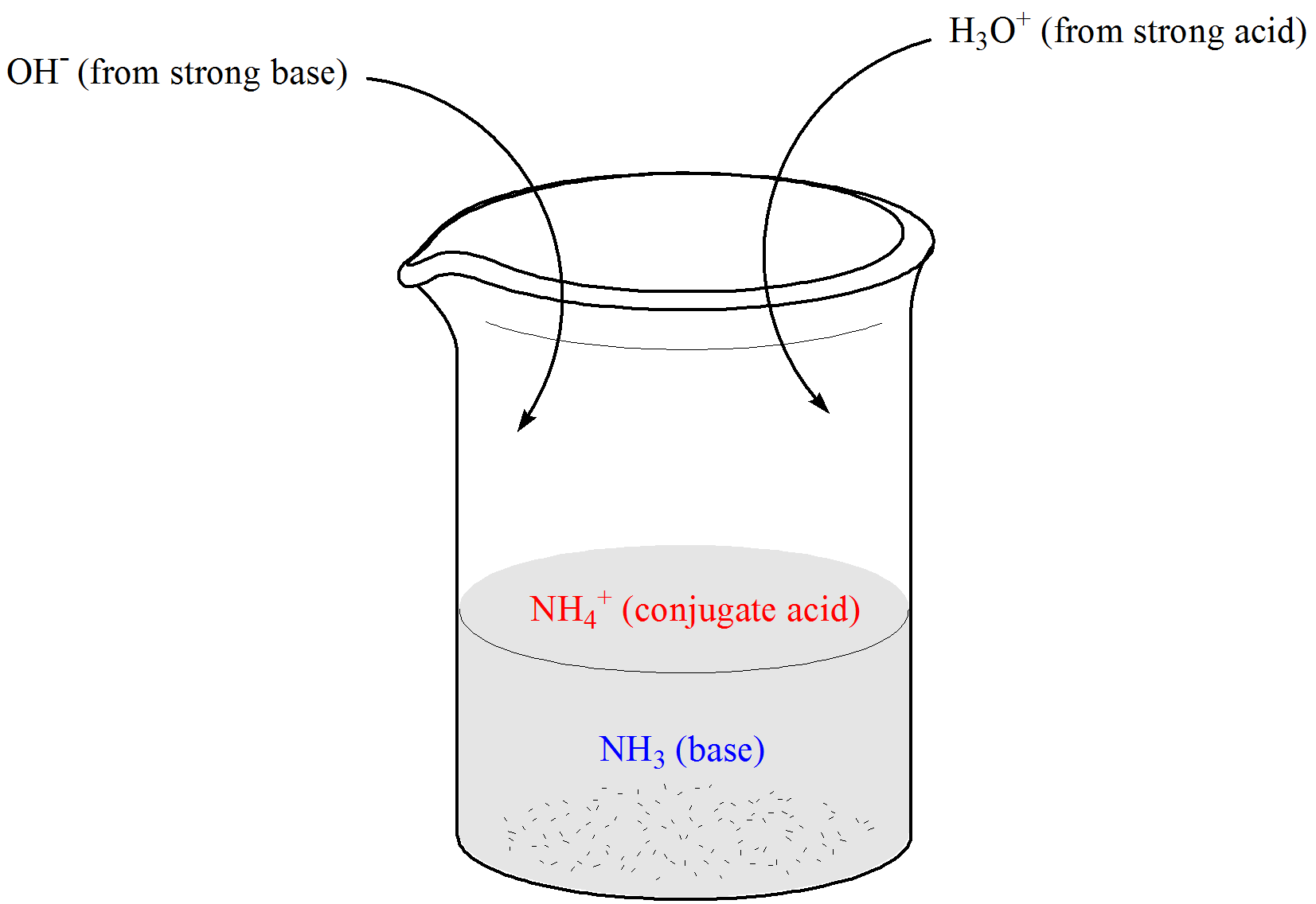
☆ what is a buffer vs. a stock solution?
a buffer keeps conditions stable, often pH; a stock solution is a concentrated starting solution used to make dilutions
☆ what equation is used for making a dilution?
c1v1 = c2v2
- c1 = concentration of stock buffer
- v1 = volume of stock buffer
- c2 = concentration of dilute buffer
- v2 = volume of dilute buffer
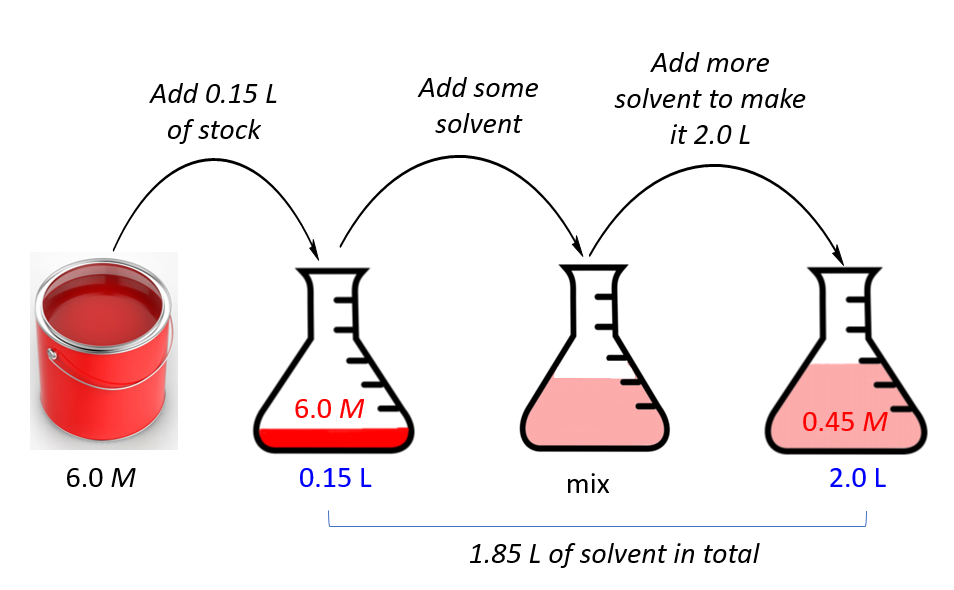
☆ how do you make a dilution from a stock?
1. write c1, c2, v2
2. solve for v1 using c1v1 = c2v2
3. measure the stock volume
4. add water/buffer until final volume is reached
☆ what is an assay?
a quantitative test for one specific component in a sample

☆ what are the most important lab safety rules?
1. wear proper clothing (long pants / skirt, shirt covering shoulders, etc.) and goggles
2. no food or drink
3. tie hair back
4. report spills/accidents
5. use the correct waste container
chemical waste never goes in the sink unless directed
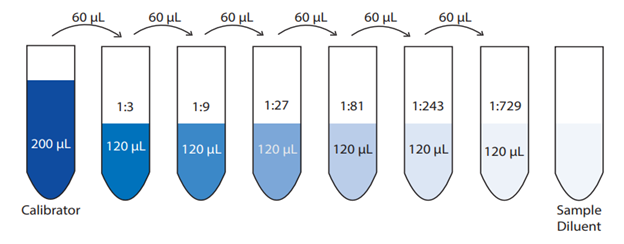
☆ what is a serial dilution?
a stepwise dilution where each new tube/cuvette is made from the previous one
used when the original sample is too concentrated to measure directly
☆ what is the difference between step df and total df?
step df = one dilution step; total df = overall dilution from the original stock (= to df x df x df….)
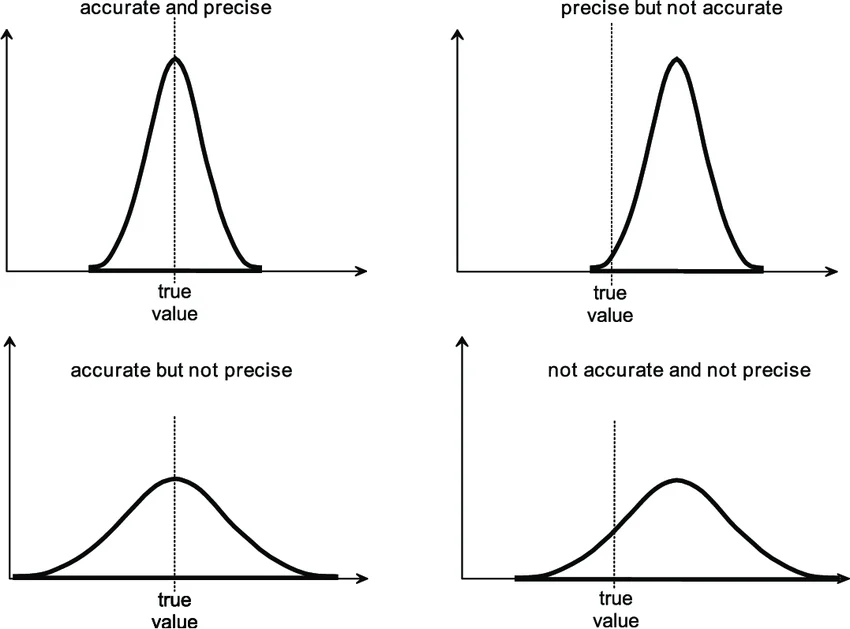
☆ what is the difference between accuracy and precision?
accuracy = closeness to the true value; precision = consistency across trials (how close data points are to each other)
☆ what is percent error?
|theoretical - measured| / theoretical × 100%
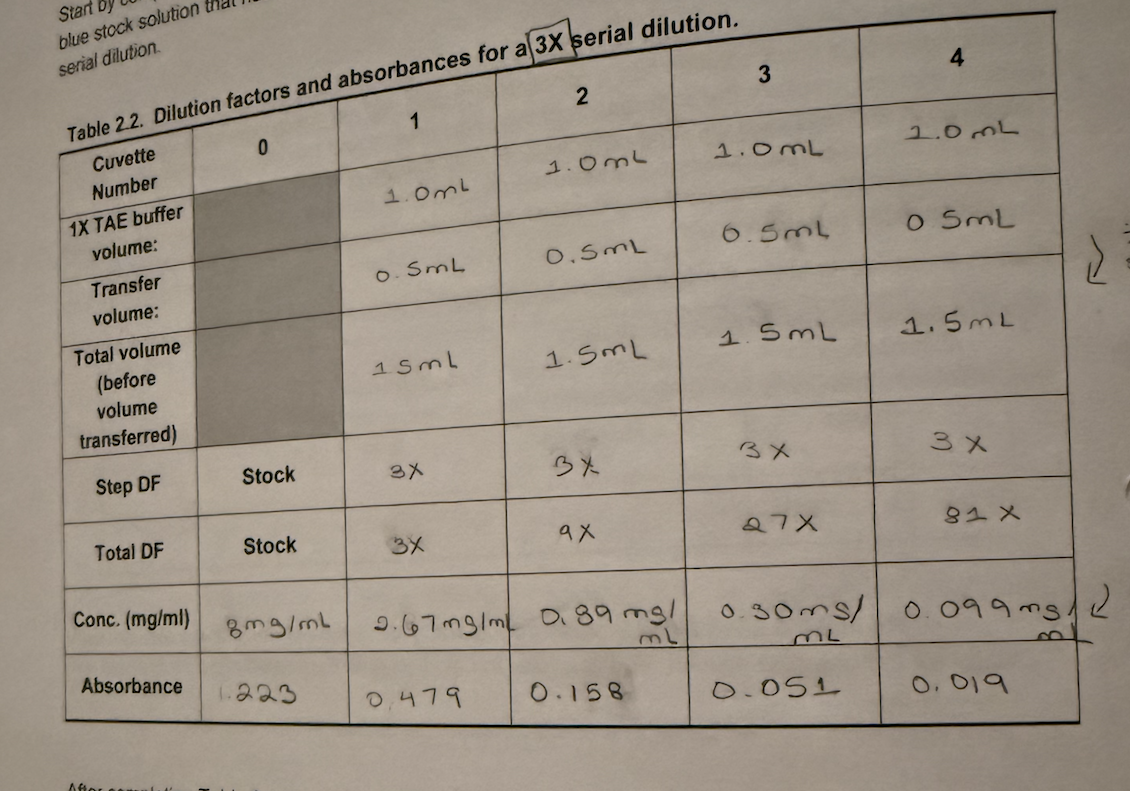
☆ how do you set up a serial dilution?
1. choose the step df and total volume (e.g. 2X serial dilution of 2 mL of phosphate standard [starting solution], using distilled water as diluent).
2. calculate transfer volume (v1) = total volume (v2) / step df
v1 = v2/df
if you use c1v1 = c2v2, make sure to use columns next to each other.
3. calculate diluent volume = total volume - transfer volume
diluent = substance used to dilute something (e.g. water volume).
4. label all tubes/cuvettes
5. add diluent first
6. add stock/standard to tube 0 / first tube
7. transfer from one tube to the next
8. mix after every transfer (vortex)
never pour diluted sample back into the stock bottle
☆ how do concentrations change in 2x, 3x, and 7x serial dilutions?
half each step; 3x = one-third each step; 7x = one-seventh each step
concentration = initial / df.
☆ what is absorbance?
how much light a sample absorbs
relationship between absorbance & concentration — they are directly proportional
higher concentration = higher absorbance
☆ what is a blank?
solvent/reagents without analyte (e.g. just stock), used to set absorbance baseline to zero.

☆ how do you use the spectrovis and cuvettes correctly?
1. connect spectrovis to labquest
2. let it warm up (~ 90 secs)
3. use the blank to calibrate
4. choose the correct wavelength (550 nm)
5. use time-based mode for one wavelength
6. fill cuvette with at least about 1 ml
7. wipe cuvette with kimwipe
8. hold only the top
9. align cuvette arrow with the light-path arrow
☆ how do you find the concentration of an unknown?
either ratio method or standard curve
ratio: cu = (au × cs) / as
- cs = concentration of stock solution
- as = absorbance of stock solution
- cu = concentration of unknown solution
- au = absorbance of unknown solution
☆ what does r² tell you?
how well the line fits the data
closer to 1 = better fit
☆ what is the wqi?
the water quality index, an overall water-quality score based on multiple tests
calculation:
test result → q-value → q-value × weighting factor → add all subtotals
test parameter | weighting factor |
change in temperature | 0.11 |
pH | 0.11 |
dissolved oxygen | 0.17 |
BOD | 0.11 |
total solids | 0.07 |
total phosphate | 0.10 |
nitrates | 0.10 |
turbidity | 0.08 |
fecal coliform | 0.16 |
☆ what are the 9 wqi tests?
temperature, pH, dissolved oxygen, bod, total solids, total phosphate, nitrates, turbidity, fecal coliform
☆ which wqi factors have the highest weighting factors?
dissolved oxygen (DO) first, then fecal coliform
do = 0.17; fecal coliform = 0.16

☆ what is lentic vs. lotic?
lentic = standing water; lotic = moving water
☆ how does temperature affect other wqi factors?
warmer water decreases do (it evaporates), increases bod, can increase biological activity (more total solids & turbidity), and can worsen eutrophication (more nutrients — phosphates, nitrates, etc.; and more nutrients — lower pH).
☆ what pH range is best for most aquatic life?
about 6.5–8.2
☆ how do you measure pH?
NO CALIBRATION
1. plug the probe into labquest
2. gently mix the sample
3. rinse the probe tip with distilled water
4. blot gently with kimwipe
5. place the tip into the sample
6. wait for stabilization
7. collect data (for 10 secs)
8. use statistics (graphing feature) to record the average
rinse between readings to prevent contamin
☆ what is dissolved oxygen (do)?
oxygen dissolved in water
comes mainly from photosynthesis and diffusion (so it peaks during the middle of the day)
☆ how do you measure dissolved oxygen?
NO CALIBRATION
1. plug in the do probe
2. remove the protective case
3. rinse and gently dry the tip
4. set the probe to % saturation for the regular do test
5. submerge the tip so contacts are under water
6. keep it off the bottom
7. avoid air bubbles
8. wait for stabilization/temperature compensation
9. collect data and record the average
☆ what is bod?
biochemical oxygen demand, the amount of oxygen used by microorganisms over time
regular do test = % saturation, bod uses mg/l because you subtract initial and final do
☆ what does high bod mean compared with high do?
high bod usually means poorer water quality; high do usually means healthier water
they are inversely proportional: higher do, lower bod—and vice versa.
☆ how are turbidity and total solids related?
both reflect particles in water
more particles can block light, reduce photosynthesis, raise temperature, increase turbidity (cloudiness of water), and lower do
☆ how do you measure turbidity?
1. connect the turbidity sensor to labquest
2. use the macro plastic cuvette
3. label near the top
4. fill with sample using serological pipette + bulb
5. cap and gently invert
6. avoid bubbles
7. wipe outside with kimwipe
8. place in sensor
9. read immediately
☆ what are nitrates and why do they matter?
nitrates are a usable form of nitrogen needed for amino acids and proteins
too much nitrate contributes to eutrophication.
☆ how do you measure nitrates with the nitrate ise?
TWO POINT CALIBRATION
if the meter has a reading of 1.0 mg/L and the sensor is not in a 1.0 mg/L solution, you need to calibrate!
1. mount the probe vertically on the stand
2. rinse and blot gently
3. soak in the high standard
4. calibrate with the high and low standards
5. wait for live voltage to stabilize at each point
6. rinse again
7. place probe in sample
8. keep it off the bottom
9. keep white reference contacts immersed
10. avoid trapped bubbles
11. let it stabilize
12. collect data and record the average
☆ what is fecal coliform?
a biotic indicator of fecal contamination
common example: e. coli
☆ why is the fecal coliform test a presumptive test?
because it infers contamination from gas production (CO2), not direct identification of every bacterium.
☆ how is fecal coliform analyzed?
1. gather the incubated tubes
2. look for gas in the durham tubes
3. count tubes with 10% gas or more as positive
4. record positives at each volume
5. use the table to find mpn
do not dump the contents

☆ what is eutrophication?
nutrient enrichment that causes excess algal growth
it can increase turbidity, decrease light penetration, kill plants, and lower do through bacterial decomposition.
☆ what is total phosphate?
all forms of phosphorus present in the sample
can be plentiful in lentic samples — high movement of water releases inorganic phosphate from rocks
☆ how do you do the total phosphate procedure?
1. label the flask
2. measure sample into the flask
3. add digestion reagents
4. heat in the hood
5. cool the sample
6. add the color-forming reagent
7. swirl for the required time (5 mins)
8. prepare the phosphate standards by serial dilution
9. calibrate the spectrovis with the blank
10. read standards, then the sample
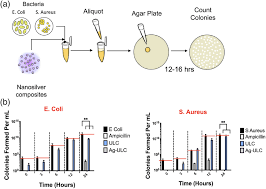
☆ what is a cfu?
a colony forming unit, one viable cell that can grow into a colony
☆ how do you do the cfu assay?
1. label microfuge tubes
2. add sterile water for the dilution plan
3. mix the original water sample
4. add sample to tube 0
5. transfer stepwise through the dilution series
6. mix between transfers
7. label the agar plate on the bottom
8. spot plate each dilution
9. start with the most diluted sample
10. let the spots dry
11. incubate plate bottom side up
12. count colonies from a usable dilution
use a fresh tip between transfers
☆ how do you calculate cfu/ml?
number of colonies × dilution factor) / volume plated

☆ what is the main goal of dna barcoding?
identify bacteria using a specific dna marker
the bacterial marker is the 16s rrna gene
☆ what is the 16s rrna gene?
a bacterial barcode gene in the small ribosomal subunit
conserved regions allow primer binding; variable regions allow identification
☆ what is pcr?
polymerase chain reaction, a method used to make many copies of a specific dna sequence
☆ what are the 3 steps of pcr?
denaturation (dna heated & two strands separate; ~95ºC for 5 mins), annealing (temperature lowered two primers bind to target dna — ~51ºC for 30 sec), extension (taq polymerase builds new dna strand — ~72ºC).
repeated 30 times
use thermocycler
because pcr depends on repeating different temperatures in the correct order.
☆ how do you set up pcr samples?
1. prepare the master mix (makes pcr setup faster, reduces pipetting, and lowers experimental error).
taq polymerase
heat-stable enzyme that builds new dna strands
primers
short dna pieces that bind the target sequence and tell polymerase where to start
dntps / nucleotides
the dna building blocks added into the new strand
buffer
keeps the reaction at the correct chemical conditions for the enzyme
nuclease-free water
brings the reaction to the correct volume without breaking down dna/reagents
2. set up the negative control with no dna
3. set up the sample with your diluted bacterial culture
4. set up the positive control with known bacterial dna
5. place tubes in the thermocycler
negative control checks contamination; positive control checks whether pcr worked
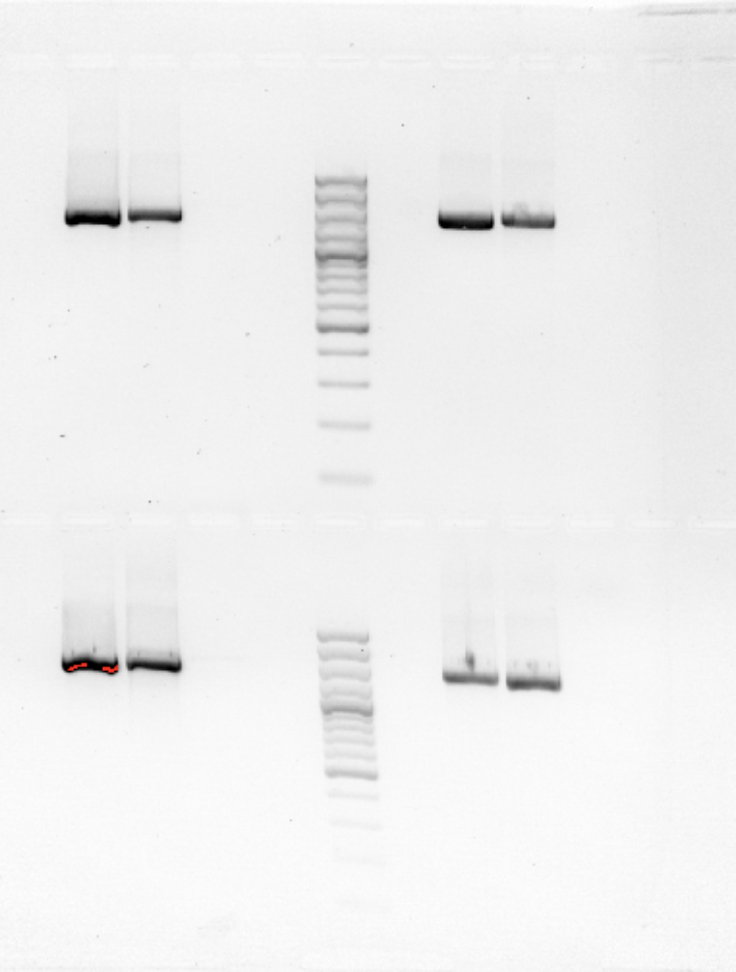
☆ what is gel electrophoresis?
a method that separates dna fragments by size using an electric field (dna = negatively charged so needs current to move from negative [anode] to positive [cathode).
☆ how do you run gel electrophoresis?
1. make the agarose gel with buffer and stain (ethidium bromide (etbr) — binds nucleic acids so dna bands can be seen under uv light)
2. place the gel in the chamber
3. add running buffer
4. load ladder [contains dna fragments of known sizes and is used as the size reference] and samples into wells
loading dye contains glycerol, which makes the sample dense enough to sink into the wells + tracking dyes
bromophenol blue helps you see movement during the run
- xylene cyanol is another tracking dye
5. run the gel
6. visualize the bands
7. photograph the gel
☆ what should the gel results look like?
sample = band if amplification worked; positive control = band; negative control = no band
a band in negative control = contamination.
no bands visible = no dye added.
no band in positive control = pcr didnt work (issue w/ master mix).
☆ what is the difference between descriptive and inferential statistics?
descriptive summarizes data (mean, median, mode, range); inferential tests whether differences matter (t-test, p-value, confidence interval, etc.)
☆ what does a small standard deviation mean?
values are close to the mean
higher precision!!!
☆ what does a t-test do?
compares the means of two groups
☆ what does p < 0.05 mean?
the result is statistically significant
☆ how do lotic and lentic systems often differ in dissolved oxygen?
lotic water often has higher do because movement increases aeration; lentic water is more likely to stratify and hold less do in some layers
☆ how do lotic and lentic systems often differ in turbidity/solids?
lotic systems may stay cloudy from constant movement; lentic systems may allow more settling
but runoff can raise turbidity in either one
☆ how do lotic and lentic systems often differ in eutrophication risk?
lentic systems often show eutrophication more easily because nutrients can build up in standing water; loticsystems flush nutrients downstream more quickly
☆ how do lotic and lentic systems differ in temperature effects?
lentic water is more likely to stratify; lotic water is more mixed
stratification can affect do and nutrient distribution
☆ compare high do, high bod, and high nutrients
high do usually means healthier water; high bod and high nutrients usually point toward poorer water quality
high nutrients can drive eutrophication, which can later lower do
☆ compare turbidity vs. cfu vs. fecal coliform
turbidity measures suspended particles; fecal coliform measures contamination by indicator bacteria; cfu estimates the number of viable culturable bacteria
one is abiotic, the other two are biotic
☆ where does the waste go for nitrates, phosphates, stock solution, pipetting solution, turbidity, and do labs?
put phosphate & nitrate waste in the designated liquid chemical waste container.
put used pipette tips in the designated disposable/medical waste
leftover stock solutions/standards discarded in rinsate beaker (liquid waste).
rinse the probes with distilled/di water, blot gently, and store it properly.
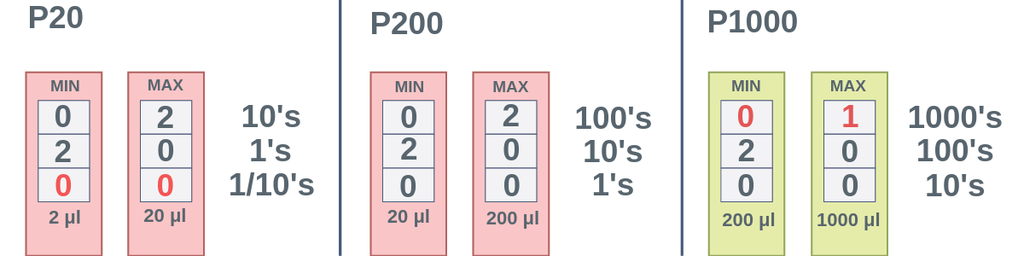
☆ when do you use the p-1000 micropipette?
use the p-1000 for the largest microliter volumes
good for bigger transfers like solution setup and serial dilution
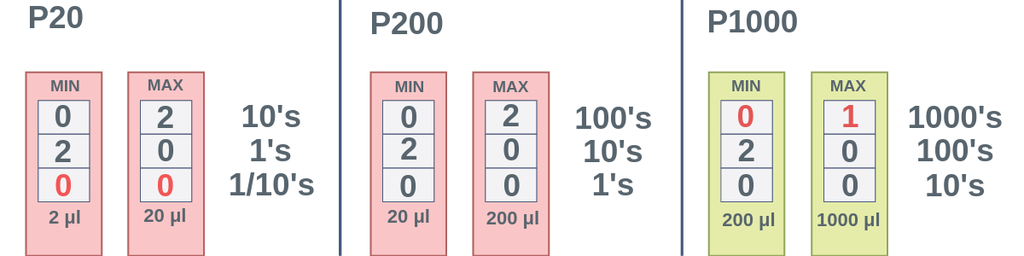
☆ when do you use the p-200 micropipette?
use the p-200 for medium microliter volumes
good when the volume is too small for the p-1000 but too large for the p-20
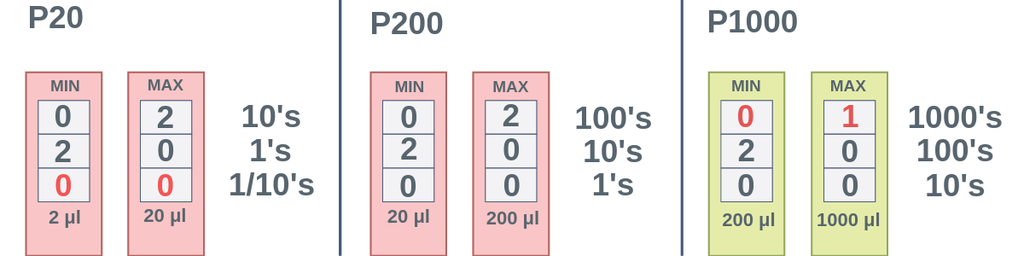
☆ when do you use the p-20 micropipette?
use the p-20 for the smallest microliter volumes
commonly used for very small transfers like pcr setup or spot plating