Chem Lab Quiz 6
1/26
Earn XP
Description and Tags
Experiment 6, 7 & 8
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
What temperature units are used for calculations related to the Charles’ Law?
Kelvins (K)
Name at least 3 gas laws you will study in today’s experiment?
Boyle’s Charles’, Avogadro’s Laws
Sketch Boyle’s Law Graph, which is # of books vs. 1/V. Could the graph cross the x-axis i.e. could 1/V=0?
No, because it’s only true for V=infinity
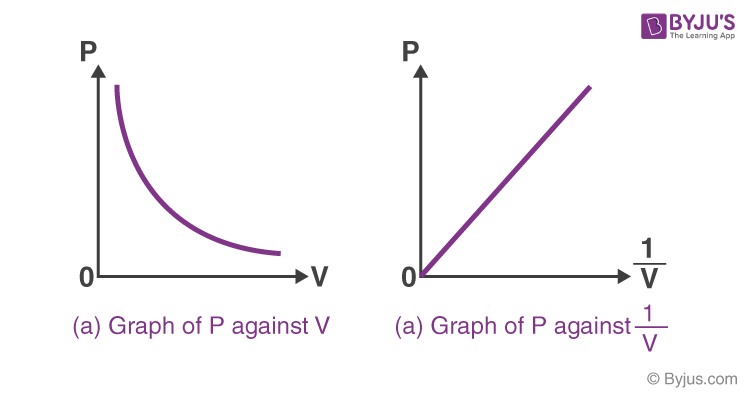
For Boyle’s Law calculations, # of books vs. 1/V, why is the mass of the book not important?
We only need to prove linearity of changing volume vs. # of books
Avogadro’s Law describes the relationship volume of gas vs. mass of reagent: T of F
F, it’s vs. mols, not vs. mass
Charles’ Law concerns relationship between volume and temperature, T of F?
F
What coolant is used in experiment 6, to get temperature below C?
dry ice - isopropanol
What thermometer would you choose to measure temperature below -40C?
alcohol-filled
Graham’s Law of diffusion states that the diffusion rate is inversely proportional to the molecular weight of gas, T of F?
T
Gas occupies all the volume available, T of F?
T
Graham’s Law experiment allows us to calculate:
ratio of molecular weights of gases involved
For the Ideal Gas Law equation (PV=nRT), what are the units for P, V, n, and T?
P = atm, V = L, n = mol, T = K
For the Ideal Law equation PV=nRT, define “n”
n = number of moles
For the Ideal Gas Law equation PV=nRT, what parameter is not measurable in experiment 7?
R
Units for molar mass are?
g/mol
For molecular mass measurement, the manual instructs you to take about 3mL of unknown liquid. How will molecular mass will change if 10mL of liquid will be used in the experiment?
Will not change: excess of vapors will escape from flask
Given that 0.88g of certain gas occupies volume at 140 mL at 97 C at 1.0 atm, what is the molecular mass of gas?
gas=167 g/mol or ~170 g/mol after rounding up to 2 sig figs
Gaussian distribution is:
distribution of numbers
Why the real scientist does not base any conclusion on just one result?
In science and statistics, a single result can be a “fluke” or an outlier. Scientists require multiple trials to ensure their findings are reliable and valid.
What the First Law of Thermodynamics states?
Energy may be transferred but never created or destroyed
Where goes the energy gained by a process in thermochemistry lab experiment?
It’s consumed by surrounding
Define specific heat. What are the units for specific heat?
Specific heat is the amount of energy required to raise the temperature of one gram of a substance by one degree Celsius. Units are cal/g*C
Express the heat change in terms of mass, specific heat and temperature change.
q=mc*deltaT
What is the surrounding in the thermochemistry experiment?
Water
Express heat change of process in terms of water and heat change of calorimeter?
Heat change Qprocess = -(Qwater + heat absorbed by calorimeter)
What is used as a calorimeter for experiment 8?
coffee cup
For calorimeter heat capacity, why mass of calorimeter is not measured?
Since mc is OK for the purpose, we don’t need to know “c” for calorimeter