Unit 14 Translation
1/155
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
156 Terms
What does each ribosomal subunit consist of?
A major rRNA and a number of small =proteins
What is required for the activity of the active centers within ribosomal subunits?
Direct participation by rRNA
How many tRNA-binding sites does each ribosome have?
three
What are the three tRNA-binding sites?
An aminoacyl-tRNA enters the A site
Peptidyl-tRNA is bound in the P site
Deacylated tRNA exists via the E site
Which component constitutes the largest portion of a ribosome’s mass?
rRNAs
What do most ribosomal proteins physically contact and what do they serve as?
they contact a rRNA and it serves as a backbone of each subunit
How do eukaryotic ribosomal subunits compare in size to a bacterial subunit?
Eukaryotic subunits are larger
Are mitochondrial and chloroplast ribosomes identical to cytoplasmic ones?
No, they are different and take varied forms
What term describes ribosomes because of their catalytic properties?
Ribozymes
Where is the catalytic site for peptide bond formation located?
Is far from amino acids but close to RNAs
What contributes to catalytic site structure and participates in catalysis?
23S rRNA and P site tRNA
What are the three stages of translation
Initiation
Elongation
Termination
Explain Initiation
Small subunit on mRNA binding site is joined by large subunit and aminoacyl-tRNA binds
What is initiation in relation to translation
Are the steps of translation up to the entrance of the first aminoacyl-tRNA into the A site
Explain Elongation and its relation to translation
The ribosome moves along mRNA, extending protein by transfer from peptidyl-tRN to aminoacyl-tRNA. They are repeated rounds of polypeptide chain extension and the addition of one amino acid at a time to the growing polypeptide chain.
Explain Termination and its relation to translation
The polypeptide chain is released from the tRNA, and the ribosome dissociates from the mRNA. There is a separate reaction that ends translation by stopping the addition of subunits and stimulating disassembly of the apparatus
Why is tRNA considered the “bridge” between nucleic acids and proteins?
It translates the genetic code of mRNA (nucleic acid) into a specific sequence of amino acids.
What is the typical length of a tRNA molecule?
betwene 75-90 nt in length
What sequence is found at the 3’ terminus of all tRNAs
The 5”-CCA=3” which serves as the amino acid acceptor stem
How are the “unusual bases” in tRNA structure created?
They are all created post-transcriptionally from standard ribonucleotides (ex: uridine, pseudouridine, dihydrouridine)
Name the five characteristics and conserved patterns of singel and double-stranded regions of a tRNA cloverlead structure
Amino acid acceptor arm
Pseudouridine loop
Dihydrouridine (D) loop
Anticodon loop
Variable loop
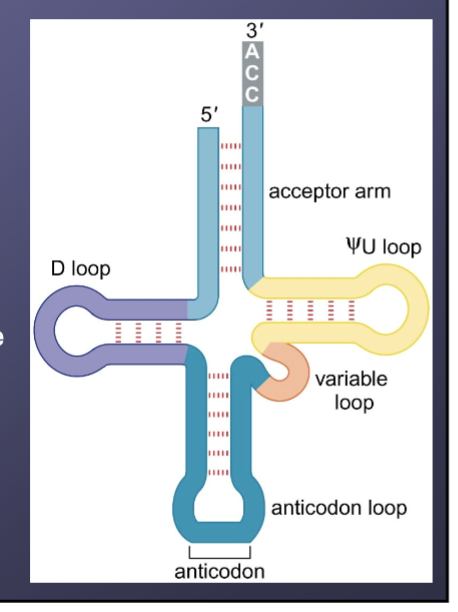
What is the represented and actual 3D shape of a tRNA molecule?
represented as a cloverleaf structure but is actuallty an inverted L-shape
What are aminoacyl-tRNA synthetases?
The family of enzymes that load tRNAs with the correct amino acid.
What is the first step of the tRNA charging reaction?
An amino acid reacts with ATP to form an aminoacyl adenylate intermediate, which releases pyrophosphate
What provides the energy for the charging reaction?
The hydrolysis of ATP, which is trapped in the mixed anhydride linkage of the adenylate.
What is step 2 of the tRNA charging reaction?
The 2'‘ OH or 3’ OH of the terminal 3’ nucleotide in the tRNA attacks the carbonyl carbon of the adenylate
What is step 3 of the tRNA charging reaction?
An aminoacyl-tRNA and AMP is formed
What should each tRNA synthetase be selected for?
For a specific group of tRNAs and a specific amino acid
When selecting, how do they detect tRNA differences?
All tRNAs share the same general tertiary structure, but differ at the nucleotide positions of the four arms.
there are changes in the nucleotide sequences and subtle differences between the shape of the L-shaped arms
What are the two ways tRNA synthetases discriminate between tRNAs?
They use both direct (nucleotide differences( and indirect (phosphodiester) methods
What are the most common discriminator regions for tRNA synthetases?
The anticodon loop and the amino acid acceptor arm
When selecting, how do they detect amino acid differences?
The primary discriminator is the shape of the different amino acids, but since amino acids are very small, some are very similar in structure. Those that are similar have different binding efficiences and free energies.
What do the two detection methods work together to produce?
The optimal induced fit between enzyme, tRNA and amino acid
There are ________ distoinct families of tRNA synthetases divided into two classes
23

Explain Class I tRNA synthetases
Primarily monomeric
Aminoacylates tRNA at the 2’-OH attachment site
Binds tRNA in the minor groove of the amino acid acceptor stem and requires hairpin formation
Reaction rate is limited by rate of aminoacyl-tRNA release.

Explain Class II tRNA synthetases
Primarily dimeric
Aminoacylates tRNA at the 3’-OH attachment site
Binds tRNA in the major groove of the amino acid acceptor stem
Reaction rate is limited by previous chemical steps or active site rearrangemnet
What is kinetic proofreading?
an active site in an enzyme and things that are the correct shape stay long enough to catalyze, but ones that are incorrect only stick for a brief time but doe’t stay long enough for catalytic activity reaction
What determines the "alignment” of a tRNA in the synthetase active site during kinetic proofreading?
Whether the tRNA matches the specific nucleotide sequence combination required by that synthetase
What part of the tRNA must align with ATP and the amino acid tin the active site to trigger a reaction?
The amino acid acceptor stem
What is triggered quickly when kinetic proofing happens
Triggers the aminoacylation reaction
What is the physical result of an incorrect tRNA entering the active site?
The acceptor stem is misaligned, preventing the aminoacylation reaction from being triggered quickly
Why do incorrect tRNAs fail to be charged during the kinetic phase?
They dissociate from the enzyme much faster than the time required for the chemical reaction to occur
What is chemical proofreading
A process where the enzyme corrects errors after the incorrect amino acid has bound, similar to DNA polymerase proofreading
Why is shape alone sometimes insufficient for amino acid discrimination in chemical proofreading?
Some amino acids (like Isoleucine and Valine) are so similar in structure that the binding site cannot effectively distinguish them.
What specific error occurs if Isoleucyl-tRNA synthetase fails to proofread?
It is unable to prevent significant levels of valine-tRNA^lle being synthesized
How many different tRNA synthetase families are able to proofread and correct errors once an incorrect amino acid has bound to the enzyme.
nine
To what DNA replication process is chemical proofreading in translation analogous?
The 3’ —> 5’ exonuclease proofreading function of DNA polymrases
What is the overall accuracy rate of the tRNA charging reaction?
Approximately 1 error for every 40,000 charging reactions.
What are the two forms of chemical proofreading?
Pre-transfer editing and post-transfer editing
What is pre-transfer editing?
The incorrect aminoacyl-AMP is hydrolyzed after tRNA binding but before charging has occurred
What is post-transfer editing
The amino acid is hydrolyzed from the aminoacyl-tRNA after the charging reaction has already occured
Where does post-transfer editing take place within the synthetase enzyme?
In a separate editing active site that is distinct from the synthetic/loading active site.
What is the integrated double-sieve model in tRNA charging?
A post-transfer editing pathway based on the relative sizes of the synthetic and editing site of the tRNA synthetase.
Which site in the double-sieve model is larger?
The synthetic site is larger than the editing site
Which site acts as the “first sieve”?
The synthetic site
What happens during the first sieve?
Amino acids that are larger than the correct amino acid will be excluded from the synthetic site, and loading will not occur.
What us the second sieve called?
the editing site
How does the second sieve handle amino acids that are too small?
Amino acids that are smaller and incorrect will enter the synthetic site and the editing site and then hydrolyze and remove them in the editing site from the tRNA.
The correct amino acid can fit into the __________ site, but not the ___________ site
synthetic; editing
What happens when the correct amino acid fits into the synthetic site?
It will be correctly charged and retained in an aminoacyl-tRNA
Where are the synthetic and editing sites located relative to each other?
They are located a considerable distance apart across the enzyme from eachother
How does the amino acid reach the editing site?
The amino acid is covalently bonded to the tRNA in the aminoacyl-tRNA, but it forms an extended structure that can “reach” over to the editing site
What structure does the tRNA form before it is charged?
It forms a hairpin structure that is unable to reach the editing site because it is too compact to reach the editing site.
___________ editing cannot use the editing site
pre-transfer
What percentage of ribosomes are actively participating in translation in growing bacteria?
80%
What kind of equilibrium is in the cytoplasmic pool between intact ribosomes and subunits?
A dynamic equilibrium
What is initiation a function of?
It is a function of individual subunits, but not intact ribosomes (specifically the 30S subunit)
What two sequences on bacterial mRNA are required for initiation?
The start codon and the Shrine-Dalgarno sequence
Describe the Shine-Delgarno sequence
Is a polypurine-hexamer (5’-AGGAGG-3’) approximately 10 nt upstream of the start codon. This is complementary to a portion of the 16S rRNA in the small (30S) subunit
30S subunit binds to mRNA first, which is aided by __________ _______
initiation factors
What is the primary role of IF-3 during initiation?
It stabilizes the free 30S subunit and must eventually be released to allow the 50S subunit to join the 30S-mRNA complex
What is formed when a 30S subunit carrying several initiation factors binds to an initiation site on mRNA?
It forms an initiation complex
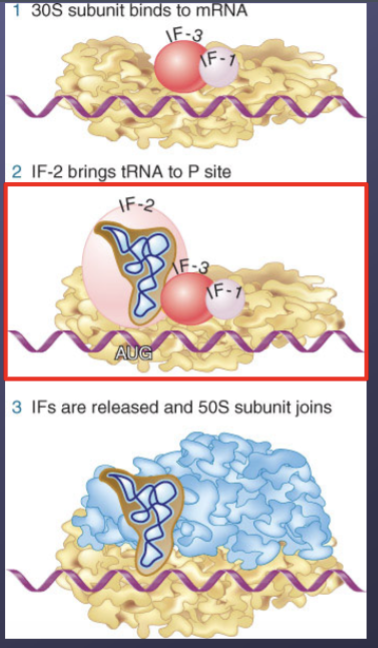
What is the role of IF-3?
It also helps the 30S subunit bind to the initiation sites on the mRNA
What is the role of IF-2?
It aids binding of the initiator tRNA to the complex
What is the role of IF-1
It binds to the 30S subunit at the A site and prevents aminoacyl-tRNAs from binding prematurely
When are all the initiation factors released?
Just before the 50S subunit joins to form the full (70S) ribosomal structure. The rRNA interacts between the subunits
What does bacterial translation usually begin with?
With a methionine amino acid, which is usually encoded by the AUG codon.
What are different methionine tRNAs involved in?
Initiation and Elongation
What is the specific initiator tRNA in bacteria called?
N-formyl-methionyl-tRNA (tRNA^fMet)
Explain the N-formyl-methionyl-tRNA
It is the aminoacyl-tRNa that initiates bacterial polypeptide translation and is where the amino group of the methionine is formylated
What is tRNA^mMet?
The bacterial tRNA that adds methionine at internal AUG codons
When is the methionine formylated?
After it has been loaded onto the initiator tRNA
How is the initiator tRNA unique
It has unique structural features that distinguish it from all other tRNAs
Why are 3 G-C base pairs important in the initiator tRNA?
They are needed for the tRNA to enter the P site
What is the only initiation complex component that can bind the mRNA?
The small subunit
Explain the way the mRNA binds the small subunit
It places the start codon in the partial P site
The initiator tRNA is the only tRNA able to do what?
Able to bind to the partial P site contained within the 30S subunit
What is required when tRNA binds to the partial P site
It requires an IF-2,
Can the initiator tRNA bind to a full A site?
No, it is unable to enter and bind the full A site on a complete ribosome
What is ensured when IF-2 binds the initiator fMet-tRNA
it ensures that only it can enter the partial P site on the 30S subunit
How does IF-3 also participate in specificity?
they participate by stabilizing the binding of the anticodon of the initiator tRNA with the second and third bases of the start codon.
What does the small ribosomal subunit in eukaryotes recognize?
It recognizes the 5’ cap of the mRNA and moves it to the initiation site.
Do eukaryotes have a Shine-Dalgarno sequence?
no
What does the small subunit bind to when scanning the eukaryotic initiation model?
It binds to the 5’ cap and begins to move 5’—3’ down the mRNA
As the 5’—>3’ moves, what can the small subunit do?
It can melt some secondary structures of the mRNA
When does the small subunit stop scanning?
Stops when it recognizes the start codon and flanking sequences at -4 and +1
What sequence does the eukaryotic ribosome recognize to stop scanning
The Kozak sequence
What is laky scanning?
When a weak Kozak consensus allows the ribosome to bypass the first start codon
There is a special initiator tRNA in eukaryotes, but the _________ is not formylated
methionine
What structural feature is unique to the eukaryotic initiator tRNA
phosphorylation of 2’-OH on nucleotide 64