LEAVING CERT HL CHEMISTRY
1/251
Earn XP
Description and Tags
Q4, Q5, Q6, Q,7/9 (water, acids, bases, pH and indicators, chemical equilibrium)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
252 Terms
define electronegativity
the relative attraction an atom has for the shared pair of electrions in a covalent bond
what contribution did Dobereiner make to the systematic arrangement of the elements?
Dobereiner placed elements with similar chemical properties into groups of three, called triads. In order of atomic weight, the weight of the middle element was approx. halfway between the other two
distinguish between an atomic orbital and a sub level
an atomic orbital is a region in space, arouund the nucleus, where there is a high probability of finding an electron. a sub level makes up part of the main energy levels in an atom
how could you confirm the presence of nitrate ions in a solution?
add freshly prepared, cold, saturated iron(II) sulfate solution, then pour conc sulfuric acid carefully down side of slanting test tube. bron ring at junction of liquids
define relative atomic mass
the average mass of the isotopes of that element, taking their abundances into account compared with the carbon 12 isotope
account for the difference in shapes of the ammonia (NH3) and boron trifluoride (BF3) molecules
BF3 - triangular planar
NH3 - pyramidal
state charles’ law
the volume of a fixed mass of gas is directly proportional to its temperature, at constant pressure
how could the presence of sulphite ions in aqueous solution be detected?
white precipitate with barium chloride solution which dissolves in dilute hydrochloric acid
how many electrons and neutrons has 1735 Cl-?
neutrons: 18
electrons: 18
how many electrons are there in 2.3g sodium metal, Na?
(calculations) 6.6 × 1023
on what principle is the analytical technique mass spectroscopy based?
mass spectroscopy is based on the principle that positive ios are accelerated through a magnetic field and separated according to their mass
list the types of radiation in order of increasing penetrating power
gamma
beta
alpha
state two ways in which Mendeleev’s periodic table of elements differ from that of Moseley
Mendeleev’s periodic table was organised in order of atomic weight. Moseley’s peiodic table is organised in order of atomic number. certain elements in Mendeleev’s table needed to be reversed to fit their groups, but this isn’t the case in Moseley’s
what are isotopes?
atoms of the same element (same atomic number) with different mass numbers due to different numbers of neutrons in the nuclei
write the electronic configuration of a neutral copper atom
1s2 2s2 2p6 3s2 3p6 4s1 3d10
state boyle’s law
the volume of a fixed mass of gas is inversely proportional to its pressure, at constant temperature
name the two reagents used to detect the presence of phosphate ions in solution
ammonium molybate
nitric acid
how many atoms are present in two moles of ammonia?
(calculations) 2.04 × 1025
how many protons and neutrons are present in the ion Al+3?
neutrons: 14
protons: 13
what type of bonding occurs between (i) hydrogren and oxygen in a molecule of water, and (ii) molecules of water in a molecule of ice?
i) intermolecular
ii) intramolecular
give two common uses of radioisotopes
americium-241 used in smoke detectors (alpha)
carbon-14 used to date artifacts containing carbon (beta)
calculate the relative atomic mass of the element X, whose composition was found to be 70% 20Ne and 30% 22Ne
(calculations) 20.6
write the electron configuration of (i) a sulphur atom and (ii) the chloride ion
i) sulphur: 1s2 2s2 2p6 3s2 3p4
ii) chloride ion: 1s2 2s2 2p6 3s2 3p6
state the shape of the following two molecules; (i) CO2 and (ii) NH3
i) v-shaped
ii) pyramidal
why does HF have a higher boiling poin than HCl?
HF is made with hydrogen bonds, the strongest type of bond, thus having the highest boilng point. HCl has Dipole-Dipole bonding, weaker bonding than hydrogen bonding, and thus a lower boiling point than HF
what is meant by the term “half-life” of an element?
the time taken for half of the nuclei in a sample to decay
how many atoms are there in 250cm3 of methane at STP?
(calculations) 5.376 × 1025
how many grams of NaOH would be required to make 250cm3 of a 0.1M solution?
(calculations) 40g
name the colour that would be produced in fireworks by salts containing (i) copper metal and (ii) potassium metal?
i) copper: blue-green
ii) potassium: lilac
write the equation of state for an ideal gas
PV=nRT
who is credited with the discovery of the electron?
JJ Thomson
what charge is associated with alpha particles?
double positive charge
what is the trend in the size of atomic radii across the periodic table?
decrease in atomic radius
state three characteristic properties of ionic substances
ionic compounds are:
composed of a network of ions in a crystal
hard and brittle - high melting and boiling points - solid at room temperature
conduct electricity when dissolved in water or molten - free to move - can carry a current
what is the shape of the methane molecule?
tetrahedral
what is diffusion?
the spontaneous spreading out of a substance due to the natural movement of its particles
greek philosophers contributions
all matter is made of tiny particles called atoms
no experiments
four elements (earth, fire, air, water)
atomos - indivisible
john dalton contributions
experiments on gases
atomic theory
william crookes contributions
investigated cathode rays in a vacuum tube
the maltese cross experiment
the paddle wheel experiment
george stoney contributions
named electrons
jj thomson contributions
investigated cathode rays
plum pudding model
robert millikan contributions
measure charge of electron
oil drop experiment
ernest rutherford contributions
gold foil experiment
james chadwick contributions
bombarded beryllium wutg alpha particles, observed a chargeless radiationemitted from the nucleus - neutrons
state dalton’s atomic theory
all matter is made of atoms
atoms are indivisible and small
atoms cannot be created or destroyed
elements are made of one kind of atom
atoms can join together to form compounds
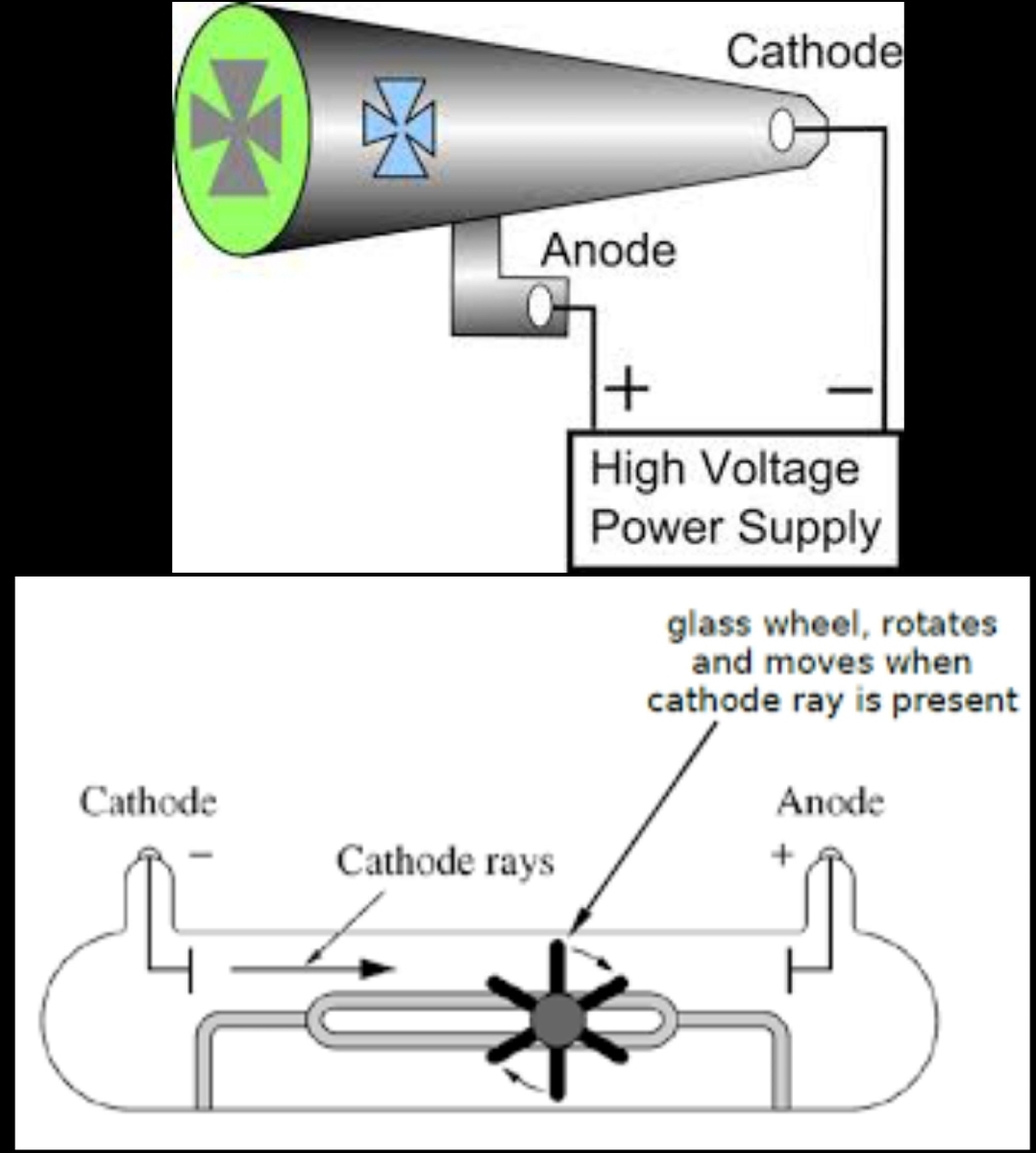
outline the experiment performed by crookes to discover cathode rays
maltese cross: saw that something was hitting the back of the glass. investigating the negative terminal of a battery in a vacuum tube (empty space)
paddle wheel: established cathode rays
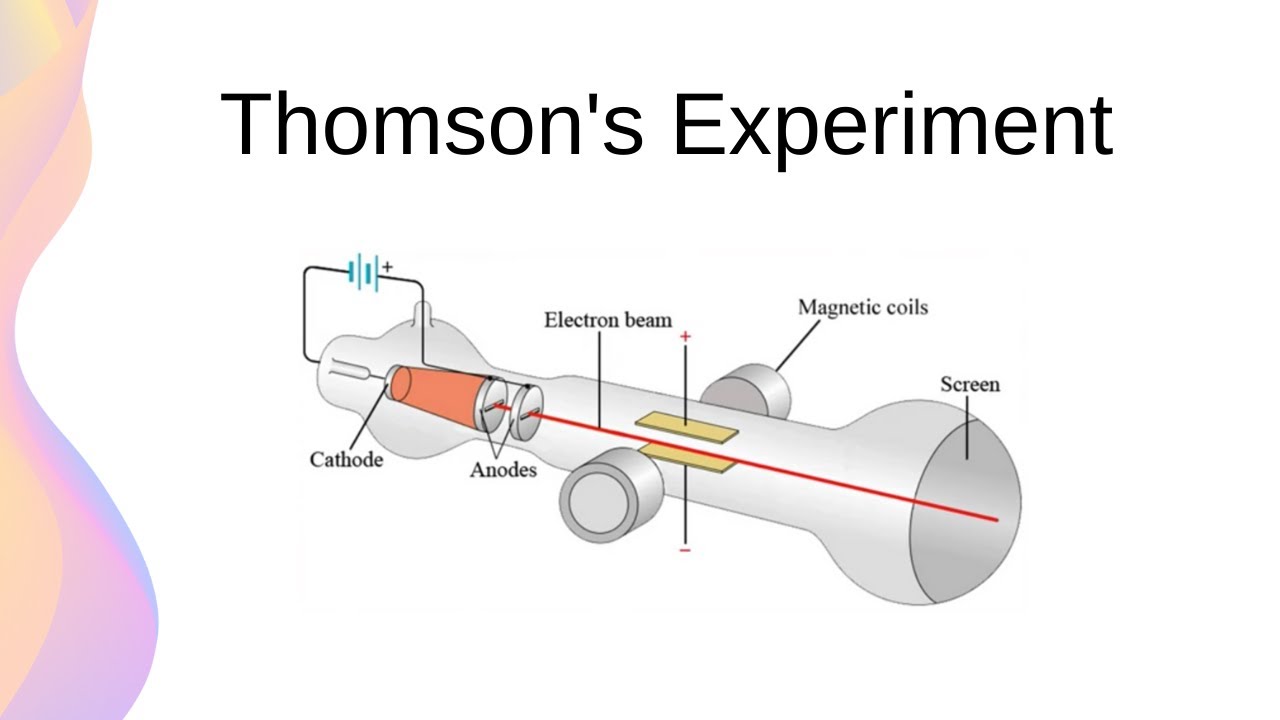
outine the experiment performed by jj thomson to discover the charge on cathode rays
investigated if cathode rays consisted of charged particles
observed cathode rays attracted to positive plate
concluded cathode rays consist of negatively charged particles - called these electrons
what are cathode rays?
streams of negatively charged electrons which travel from the cathode to the anode
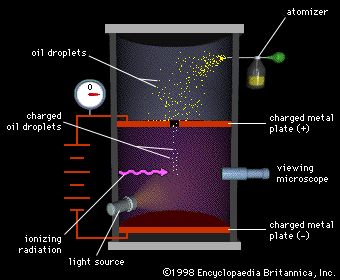
outline the experiment performed by millikan to measure the charge on an electron
oil drop experiment
investigated the size of a charge on an electron
charged oil drop suspended between two charged plates
e/m calculated (charge/mass)

outline thomson’s plum pudding model of the atom
sphere of positive charge with negatively charged electrons embedded in it
state three observations that rutherford made during his gold foil experiment
most alpha particles passed through the gold foil
some were deflected at large angles
a very small amount were deflected back along their own path - hit the nucleus head on
what is the relative charge, relative mass, and location of (i) protons, (ii) neutrons, (iii) electrons?
i) protons: +1, 1, nucleus
ii) neutron: 0, 1, nucleus
iii) electron: -1, 1/1860, space outside the nucleus
outline bohrs atomic theory basd on the hydrogen emission spectrum
electrons revolve around the nucleus in fixed paths called energy levels
while in a particular energy level, the energy of the electron is fixed (or quantised)
electrons normally occupy the lowest available energy level. they are said to be in their ground state
when an atom absorbs energy, electrons jump rom a lower energy level to a higher energy level. they are then said to be in an excited state, which is temporary and unstable
state the colour given by lithium in the flame test
crimson
state the colour given by potassium in the flame test
lilac
state the colour given by barium in the flame test
green
state the colour given by strontium in the flame test
red
state the colour given by copper in the flame test
blue-green
state the colour given by sodium in the flame test
yellow
define energy level
a region of definite energy within the atom that electrons can occupy
what is meant by the ground state of an atom?
the lowest energy state for an electron
what is meant by the excited state of an atom?
when an electron occupies a higher energy state than the ground state by absorbing enough energy
define atomic orbital
a region in the space of an tom where the probability of finding an electron is relatively high
name the series of lines in the emission spectrum of hydrogen where electron transitions are from higher energy levels to n=2 and give rise to lines in the visible spectrum
balmer series
name the series of lines in the emission spectrum of hydrogen where electron transitions are from higher energy levels to n=1 and give rise to lines in the ultraviolet region of the spectrum
lyman series
name the series of lines in the emission spectrum of hydrogen where electron transitions are from higher energy levels to n=3 and give rise to lines in the infrared region of the spectrum
paschen series
how is an atomic absorption spectrum produced?
created when electrons transition from a low energy to a high energy since they absorb energy
what is a sublevel?
a subdivision of an energy level containing one or more atomic orbitals, all of equal energy
state heisenberg’s uncertainty principle
it is not possible to measure both the position and velocity of an electron, since doing one affects the other
state two limitations of bohrs theory
only worked for hydrogen
subsequent discoveries: energy sublevels, wave nature of the electron, uncertainty principle, orbitals
define an element
a simple substance that cannot be broken into anything simpler
what method did humphry davy use to discover new elements?
passed electric current through a number of compounds. compounds split into their component elements
what contribution did dobereiner make to the systematic arrangement of the elements?
triads - placed elements with similar chemical properties into groups of threes. the atomic weight of the middle one was approx. halfway between that of the other two
what contribution did newlands make to the systematic arrangement of the elements?
the law of octaves - arranged elements in order of atomic weights, noticed every eighth element repeated propertiesw
what is mendeleev’s periodic law?
when elements are arranged in order of atomic weight, their properties vary periodically
outline mendeleev’s work to construct a periodic table of elements
prioritised chemical properties over atomic weight
left gaps to ensure elements fit into the correct columns
predicted the propertie of undiscovered elements
revesed the order of some elements to make them fit their column
state two differences between mendeleev’s periodic table and the modern periodic table of elements
mendeleev - in order of atomic weight. modern - in order of atomic number
mendeleev - gaps for undiscovered elements. modern - no gaps
what contribution did henry moseley make to the systematic arrangement of the elements?
used x-rays to discover the number of protons in the nuclei of atoms (atomic number)
placed elements in order of increasing atomic number - no reversing
define atomic number
number of protons in an atom of that element
define mass number
average mass of the isotopes of the element
define relative atomic mass
the average mass of the isotopes of that element, taking their abundances into account, compared with the carbon-12 isotope
what is an isotope?
atoms of the same element (atomic number) with different mass numbers due to different numbers of neutrons in the nuclei
name the five processes that occur in a mass spectrometer
vapourisation
ionisation
acceleration
separation
detection
describe the five processes that occur in a mass spectrometer
vapourisation: the sample is turned into a vapour or gas
ionisation: elecrons are removed from the atoms to form positive ions
acceleration: through a magnetic field
separation: the ions are separated according to their mass
detection: both the type and the abundance of each type of ion is detected
state the principle of mass spectrometry
positive ions are accelerated through a magnetic field and separated according to their mass
state the aufbau principle
electrons always occupy the lowest available energy level first
state hund’s rule of maximum multiplicity
where two or more orbitals are available, electrons will occupy them singly before filling them in pairs
state the pauli exclusion principle
no more than two electrons may occupy an orbital and they must have opposite spin
define atomic radius
half the distance between the nuclei of two atoms of the same element that are joined together by a single covalent bond
give two reasons why the values of atomic radius increase as you go down a group in the periodic table
addition of a new level
screenng effect of inner electrons
give two reasons why the values of atomic radius decrease as you go across a period in the periodic table
increase in nuclear charge
no increase in screening effect
define first ionisation energy
the minimum energy required to removed the most loosely bound electron from a neutral gaseous atom in its ground state
give two reasons why the values of first ionisation energy decreases as you go own a group in the periodic table
increasing atomic radius
screening effect of inner electrons
give two reasons why the values of first ionisation energy increase as you go across a period in the periodic table
increasing nuclear charge
decreasing atomic radius
explain why there are some exceptions to the general trend in first ionisation energy as you go across a period in the periodic table
a completely filled outer sublevel or half filled outer sublevel gives an atom extra stability. thus, it makes it more difficult to remove the most loosely bound electron, resulting in higher ionisation energy
define second ionisation energy
the energy required to remove the most loosely bound electron from a positive ion
represent the second ionisation energy as an equation
Be+ → Be2+ + e-
describe the trend in the reactivity of alkali metal as you go down the group
increase:
increase in atomic radius
increase in shielding
decrease in ionisation energy
describe the trend in the reactivity of halogens as you go down the group
decrease:
increase in atomic radius
increase in shielding
define electronegativity
a measure of the relative attraction an atom of an element has for the shared pair of electrons in a single covalent bond