gen chem 1 final
1/53
Earn XP
Description and Tags
georgia southern, threatt
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
An atom of 31P (phosphorus-31) contains how many neutrons
16
The atomic mass of chlorine is 35.45 amu. This value is best explained
Cl is a mixture of isotopes of different mass
Which of the following is a correct ground-state electron configuration
1s22s 22p 63s 23p 5 (Cl)
An element in period 3 with 6 valence electrons i
S
how to calculate the wavelength of a photon
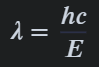
According to the Bohr model, light is emitted when an electro
Moves from a higher to a lower energy level
The Balmer series of hydrogen emission lines (when we see photons of specific colors emitted) appears in which region of the electromagnetic spectrum
visible
Cr (Z = 24) has the ground-state configuration [Ar]3d54s 1 rather than [Ar]3d44s 2 because:
A half-filled d subshell provides extra stability
how to find molar mass
locate the atomic mass of each element on the periodic table and multiply by the number of atoms
A compound is 75.0% C and 25.0% H by mass. Its empirical formula is
CH3