Alkane reactions
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
Why are alkanes generally unreactive with most reagents?
Two reasons: firstly, C–C and C–H bonds have high bond enthalpies meaning a large input of energy is required to break them.
Secondly, C–H and C–C bonds have very low polarity because carbon and hydrogen have similar electronegativities. With no significant permanent dipole, alkanes do not attract polar reagents or ions, so most potential reactants simply cannot interact effectively with alkane molecules.
Write a general equation for the complete combustion of an alkane, and give a specific example.
Complete combustion occurs in excess oxygen and produces only carbon dioxide and water. The general equation for an alkane CₙH₂ₙ₊₂ is: CₙH₂ₙ₊₂ + (3n+1)/2 O₂ → n CO₂ + (n+1) H₂O
Example for methane: CH₄ + 2O₂ → CO₂ + 2H₂O
Complete combustion is strongly exothermic and is the basis of alkanes' use as fuels (natural gas, petrol, diesel).
What happens during incomplete combustion of alkanes, and why is it dangerous?
When oxygen supply is limited, alkanes undergo incomplete combustion, producing carbon monoxide (CO) and/or carbon (soot) instead of CO₂. Carbon monoxide is a colourless, odourless toxic gas.
It is dangerous because it binds very strongly and irreversibly to haemoglobin in red blood cells, forming carboxyhaemoglobin. This prevents haemoglobin from transporting oxygen around the body, which can be fatal even in small concentrations.
Why do longer-chain alkanes release more energy per mole when burned?
Longer alkane molecules contain more C–H and C–C bonds. Combustion involves breaking these bonds (endothermic) and forming C=O and O–H bonds in CO₂ and H₂O (exothermic). More bonds being formed overall means a greater net energy release per mole of fuel.
What conditions are required for alkanes to react with chlorine or bromine?
Ultraviolet (UV) light is required. Without UV light no reaction occurs, The reaction does not occur in the dark.
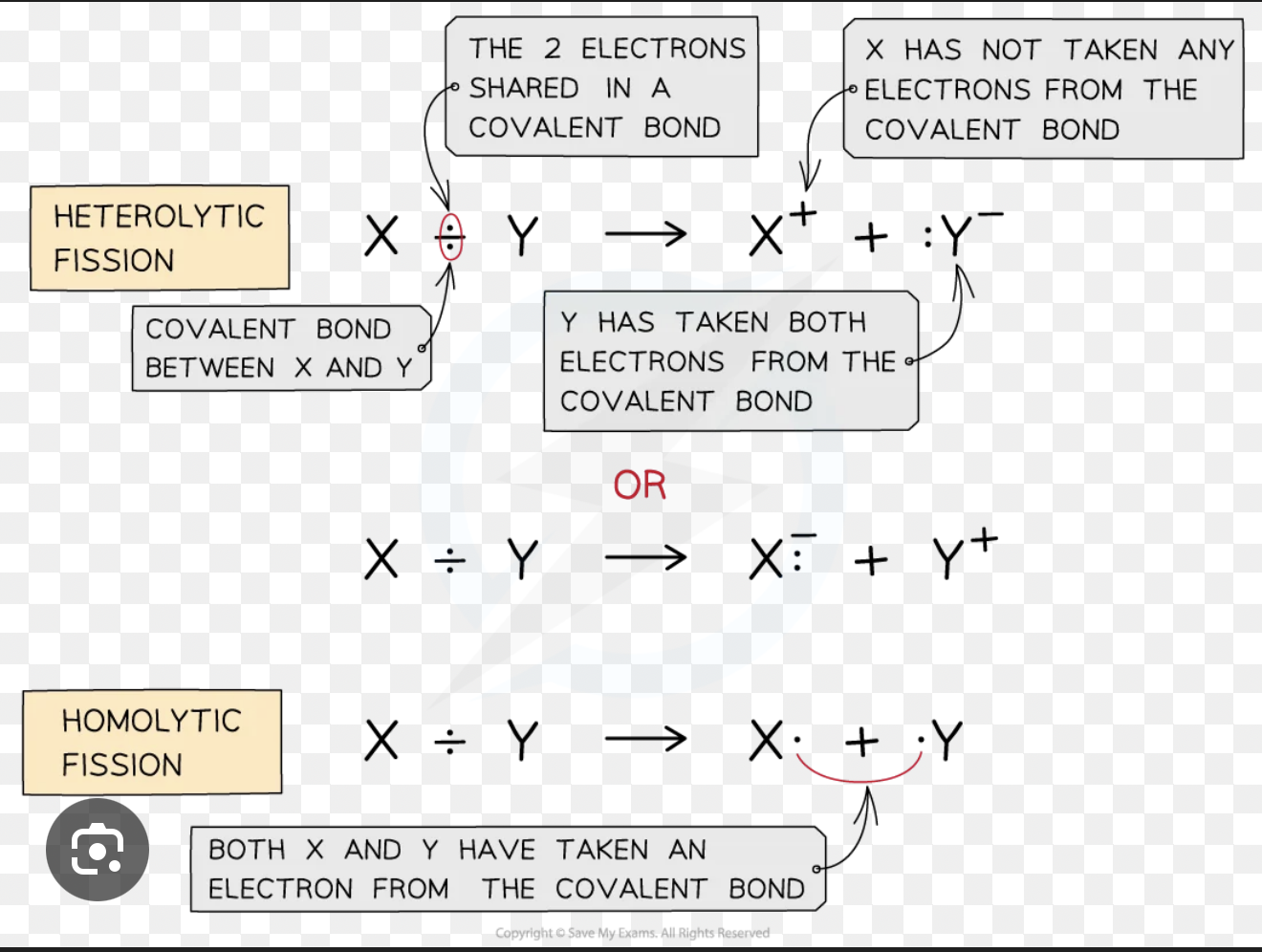
What is homolytic fission, and how does it differ from heterolytic fission?
In homolytic fission, a covalent bond breaks so that one electron from the bonding pair goes to each atom, producing two species each with an unpaired electron — called radicals (shown with a dot, e.g. Cl•).
In heterolytic fission, both electrons from the bonding pair go to one atom, producing a cation and an anion. Radical substitution involves homolytic fission; electrophilic addition involves heterolytic fission.
Describe the three stages of the radical substitution mechanism for the reaction of methane with chlorine, giving equations for each stage.
The three stages are initiation, propagation, and termination.
Initiation — UV light provides the energy to break the Cl–Cl bond homolytically, producing two chlorine radicals: Cl₂ → 2Cl•
Propagation — two steps that repeat in a chain, regenerating a radical each time:
Step 1: Cl• + CH₄ → HCl + CH₃•
Step 2: CH₃• + Cl₂ → CH₃Cl + Cl•
Termination — two radicals collide and combine, using up their unpaired electrons and ending the chain: Cl• + Cl• → Cl₂ CH₃• + Cl• → CH₃Cl. CH₃• + CH₃• → C₂H₆
Why is radical substitution a poor method for making a single, pure haloalkane product?
Further Substitution as the product formed from propagation can react with another halogen radical for example CH3Br into CH2Br2 because of the reaction with another bromine radical and this can continue until CBr4
Substitution can occur at different positions in the chain, giving a mixture of structural isomers (e.g. 1-chloropropane and 2-chloropropane). Separating these mixtures is expensive and wasteful, making radical substitution impractical for pure product synthesis.