L16 Pentose Phosphate Pathway
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
what is the pentose phosphate pathway
part of glucose metabolism and a potential fate of glucose (through G-6-P) - this pathway produces NADPH and pentose phosphate
why is NADPH production important in PPP
it’s needed for fatty acid synthesis, important in redox biology, and levels of NADP+ control the rate of PPP
why is pentose (ribose-5-phosphate) production important for PPP
the production of ribose is needed for nucleotide synthesis
what intermediates does PPP and glycolytic pathway share
glyceraldehyde-3-phosphate and fructose-6-phosphate - intermediate are how PPP interacts with other metabolic pathways
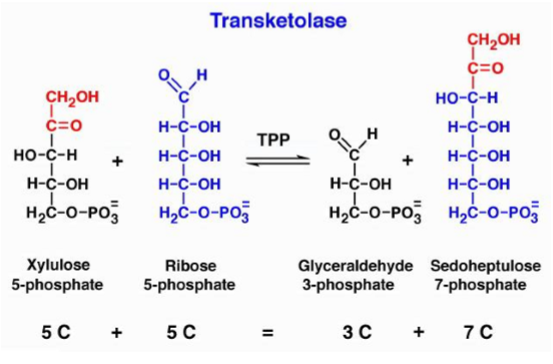
what is transketolase
reversible reaction that links PPP with glycolysis - enzyme that moves 2 carbon fragments from one molecule to another - transfer 2 carbon molecules from a ketone to an aldose and requires TPP as a cofactor/vitamin B1 which means it can diagnose thiamine deficiencies
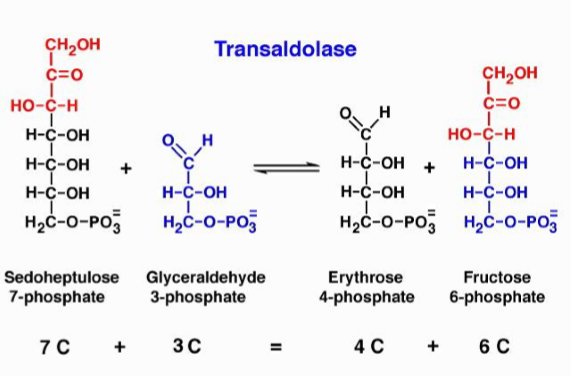
what is transaldolase
reversible reaction that moves 3 carbon fragments - an enzyme that catalyzes the transfer of a 3-carbon group (dihydroxyacetone) - does not use a cofactor like TPP but instead forms a temporary covalent bond (Schiff base) with its own lysine residue to hold the carbons
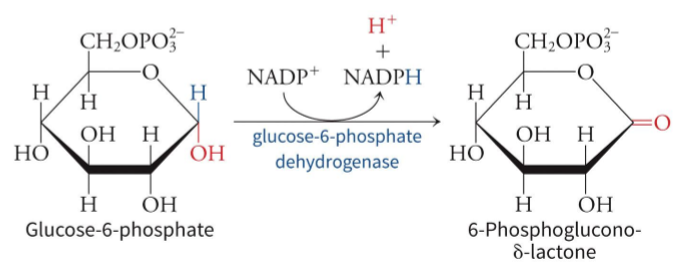
what does the oxidative phase require
glucose-6-phosphate dehydrogenase which is allosterically regulated
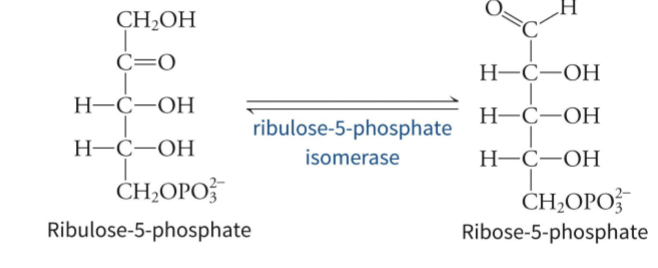
what are the two major phases of the PPP
oxidative phase where NADPH and ribose-5-phosphate are produced, and non-oxidative phase where ribulose-5-phosphate is converted to various carbon intermediates
what happens in the oxidative phase
three steps where G6P is converted into ribulose-5-phosphate through 3 main irreversible steps - this phase is primarily to generate NADPH
what is the rate limiting step of PPP and how is it regulated
the first reaction, catalyzed by Glucose-6-phosphate dehydrogenase (G6PD) - Glucose-6-phosphate dehydrogenase (G6PD). This step converts glucose-6-phosphate (G6P) into 6-phosphogluconolactone, generating NADPH, and is regulated primarily by the concentration of its substrate, NADP+, and the feedback inhibition of its product, NADPH
why is the production of NADPH important
used for nucleotide synthesis, fatty acid and cholesterol synthesis, and ROS neutralization
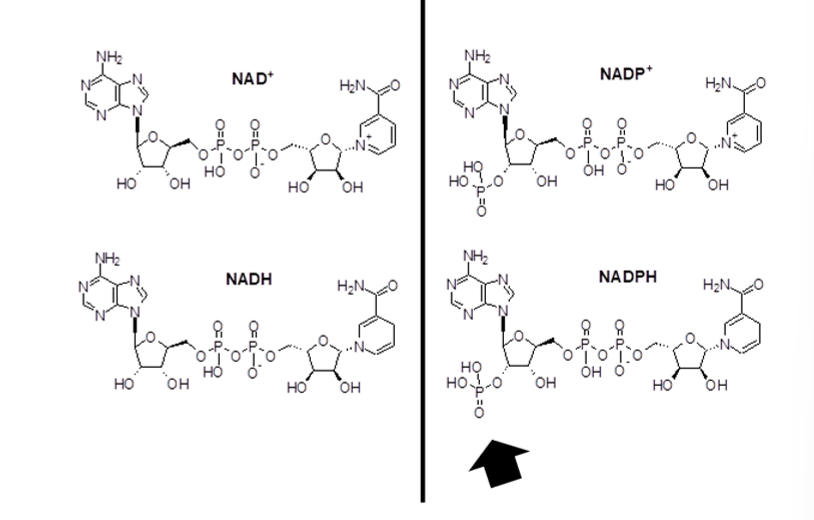
what are the similarities between NADH and NADPH
NADP+ and NAD+ are both oxidizing agents that accept electrons, but NADH feeds hydrogens/electrons to respiratory chain (oxidative phosphorylation/generates ATP)
what is the reducing agent in ribonucleotide reductase reaction for nucleotide synthesis
NADPH - it reduces ribose to deoxyribose which is important for rapidly dividing cells because it directly powers synthesis of dNTPs which are required for DNA replication and repair
why is ribose-5-phosphate generated
PPP interacts with other metabolic pathways and ribose-5-phosphate is the primary product as well as an intermediate that feeds back into PPP to generate more NADPH, feeds back into glycolysis to generate ATP, and is a starting product for nucleotide synthesis (outcome and pathway determined by metabolic need)
what do transketolase and transaldolase perform
non-oxidative phase - molecular rearrangements to produce F6P and G3P, and generate a variety of backbones
how is PPP regulated
the rate-limiting step, glucose-6-phosphate dehydrogenase, which is inhibited by high levels of NADPH - Ribose-5-phosphate levels DO NOT control PPP
where is NADPH usage high
in rapidly dividing cells that need lots of NADPH and nucleotides to make DNA, and all cells that need to defend their molecules against oxidative damage from oxidative stress (reduction of glutathione)
what are the main functions of NADPH
fatty acid, cholesterol synthesis and nucleotide synthesis, however, the biggest function of NADPH is in redox biology as the ultimate source of reducing equivalents keeping our biomolecules reduced in order to inactivate ROS via an antioxidant enzyme cycle that requires NADPH (ultimate reducing agent)
what are ROS/free radicals
extremely reactive molecules with unpaired electrons that come in and oxidize paired electrons and steal them which then makes those healthy molecules in the cell unstable - oxidative stress promotes DNA mutations, protein aggregation, and lipid membrane changes
where do ROS come from
external sources (radiation/UV rays, pollution), non-mitochondrial sources (enzymatic production of ROS like NADPH oxidase), and mitochondrial sources (ETC from electron leak from complex I and III, and O2 can pick up an electron while forming the superoxide anion O-2)
what are the types of ROS
superoxide, hydrogen peroxide and the hydroxy radical
what do ROS do to cells
acts on protein, lipids, and DNA to drive oxidative damage like DNA mutations, loss of membrane structure, and protein aggregation - steals an electron from these molecules which creates a new radical/unstable species - however, ROS is a required component of the acute immune response (needs to be controlled in small amounts)
what are some of the consequences of lipid or DNA oxidation
structural changes in the membrane like altered fluidity, ion channels, membrane-bound signaling proteins, and increase permeability
form lipid oxidation products adducts and crosslink with non-lipids like proteins and DNA
cause direct toxicity (4-HNE)
DNA damage and mutagenesis
what are the consequences of amino acid oxidation
no meth in the chapel, it makes it unstable - cysteine and methionine are highly susceptible to oxidative damage leading to protein instability and structural changes
what is superoxide dismutase
an anti-oxidant enzyme that neutralizes ROS by catalyzing the conversion of the highly reactive superoxide anion into the less reactive hydrogen peroxide
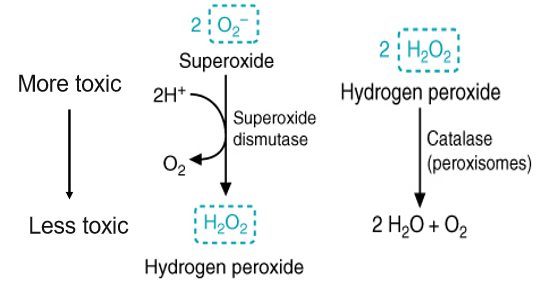
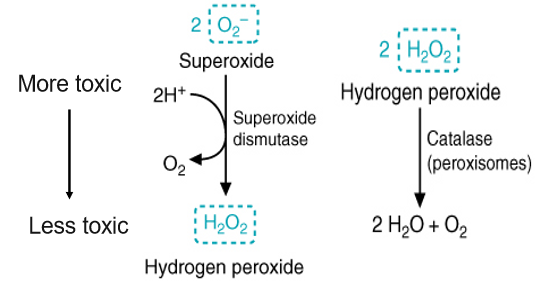
what is catalase - goose loves water
an anti-oxidant enzyme that neutralizes ROS by breaking down hydrogen peroxide into water and oxygen - second step after superoxide dismutase
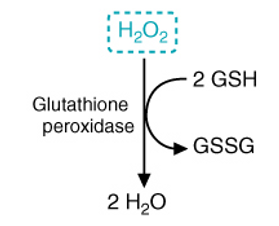
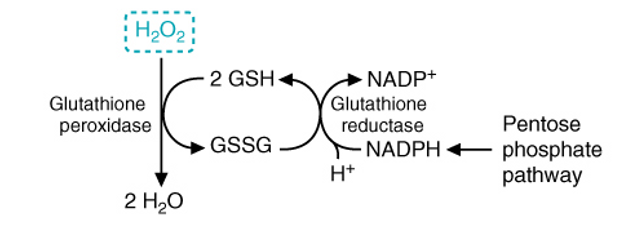
what does the enzyme glutathione peroxidase require
glutathione (GSH) as a cofactor that is can oxidize it to GGSH in order to neutralize hydrogen peroxide - GSSG is the reduced form where 2 glutathione tripeptides become linked through a disulfide bond between their Cys residues
why does the oxidized glutathione (GGSH) have to be reduced by reductase
to maintain cellular antioxidant defense and prevent toxicity - PPP provides NADPH which acts as the reducing power, and glutathione reductase consumes vast amounts of NADPH in order to reduce oxidized GSH back to reduced GSH - this cycle continuously operates to keep hydrogen peroxide levels in check
what can increasing levels of ROS/declining anti-oxidant levels do to the body in terms of disease
can contribute to diseases like Alzheimer’s, Parkinson’s, cancer, psoriasis, atherosclerosis, hypertension, and type II diabetes