L10 - Radiogenic Isotope Geochemistry
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Define an Isotope
Same number of protons (Z) and different neutrons (N)
Example: 12C and 13C
Define an Isobars
Same mass number
Example: 87Rb and 87Sr or 14C and 14N
Define an isotone
Same number of neutrons (N) and different number of protons (Z)
Example: 2H and 3He
Define a stable isotope
An isotope that is time invariant
Define a radioactive isotope
An isotope that decays with time
Define a radiogenic Isotope
An isotope that is produced (at least in part) by decay from a radioactive isotope
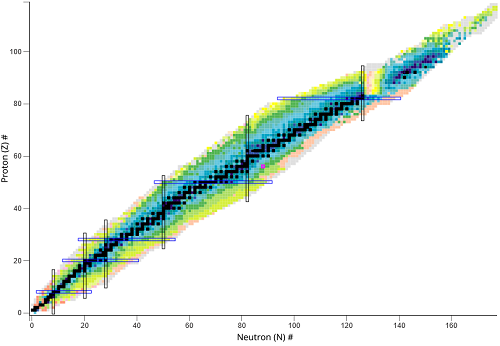
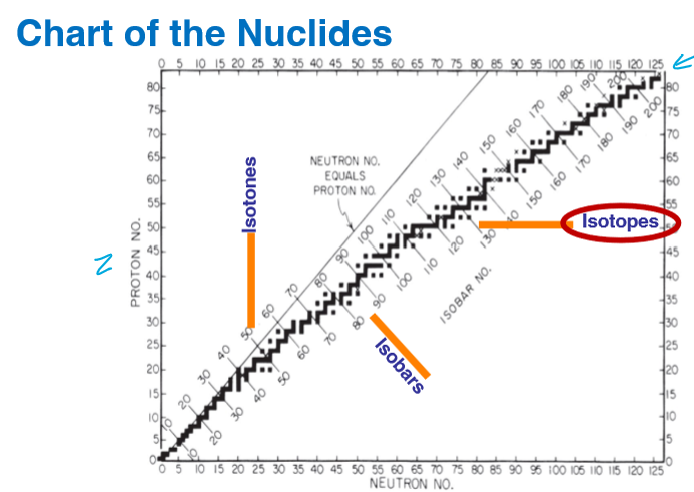
What is this chart called?
Chart of nuclides
What type of nuclear species represents a vertical change?
Isotones - different number of protons
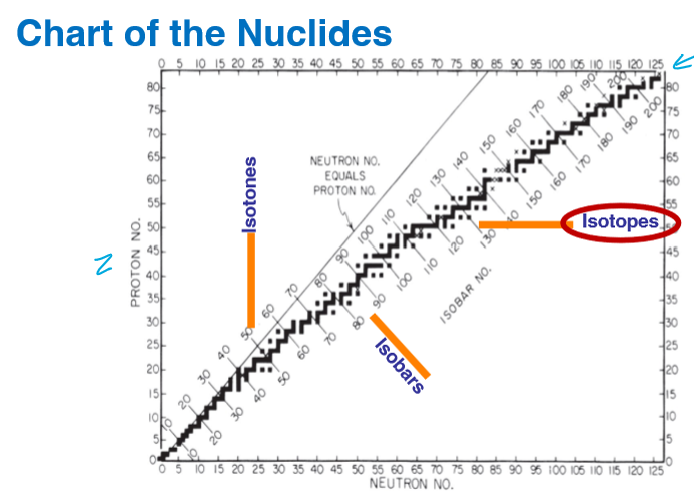
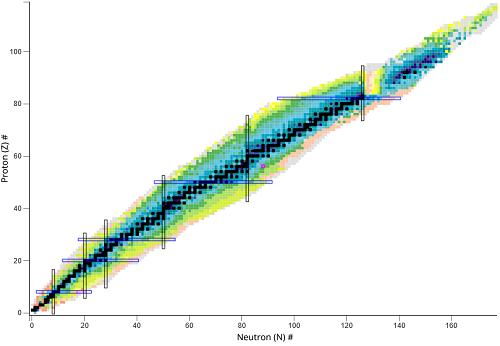
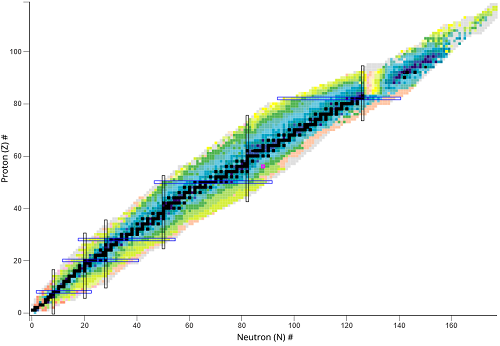
What type of nuclear species represents a horizontal change?
Isotopes different number of neutrons
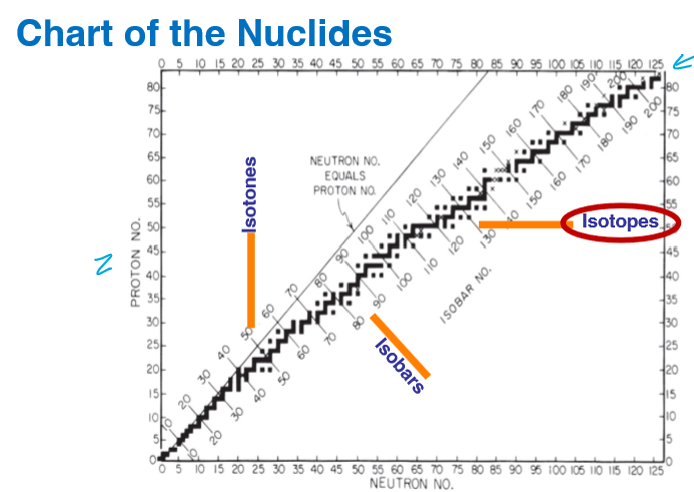
What type of nuclear species represents a diagonal change?
Isobars - same mass number (A = Z + N)
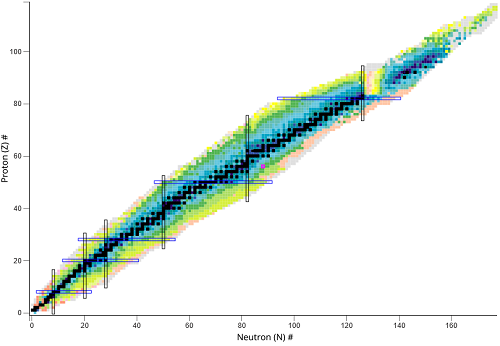
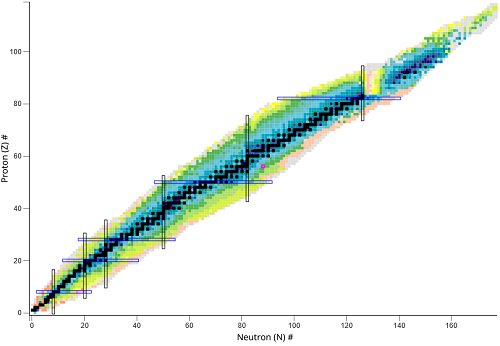
What does the black line represent?
The stable isotopes
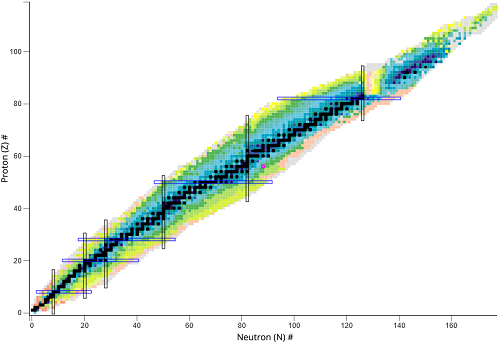
What do the colored areas represent?
Unstable nuclides
Where is the black line located of the graph?
Where protons(Z) = neutrons(N)
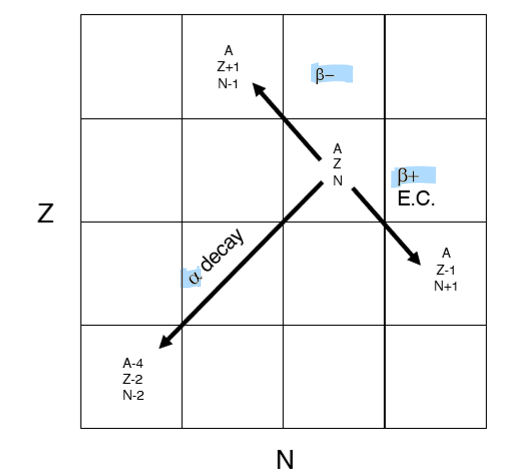
Define alpha decay
When an unstable isotope emits an alpha particle
Alpha particle = 2 protons and 2 neutrons
When does alpha decay usually occur?
Occurs in heavy nuclei
Define beta decay and its two main types
Beta decay occurs through the emission of a B- or B+ particle
B+ = add 1 proton, and subtract 1 neutron
B- = subtract 1 proton, and add 1 neutron
How does the mass change with the occurrence of beta decay?
Atomic mass remains the same
A = Z + N (they always equate out)
Define Gamma Decay
When a excited nucleus is brought to a relaxed state through the loss of high energy photons
What can stop the different decay particles?
Alpha Particle
Beta particle
Gamma Particle
Alpha Particle - paper
Beta particle - Aluminium
Gamma Particle - Lead
Where do the Radioactive decay schemes plot on the chart of nuclides?
B- = above the stability line (Z+1, N-1)
B+ = below the stability line (Z-1, N+1)
alpha = diagonal down and left (Z-2 , N-2)
How does age terminology change in both absolute and relative age?
absolute = Ka, Ma, Ga
Relative = kyr, myr, gyr
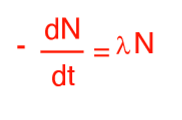
What equation is this?
The law of radioactive decay
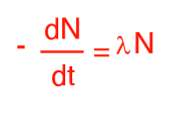
What does the N-value refer to?
The number of particles
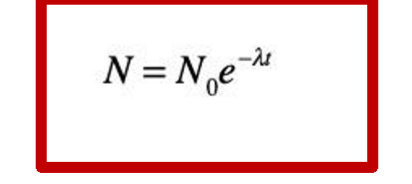
What is this equation called?
the familiar decay equation
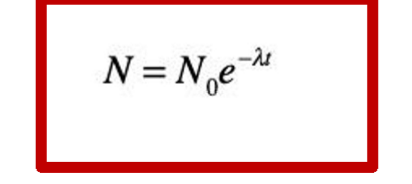
What do No and N represent?
No = the number of parent isotopes at time 0
N = the number of parent isotopes at time t
What is a half-life?
the time it takes for half of the parent nuclides to decay
defined by this formula
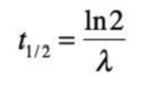
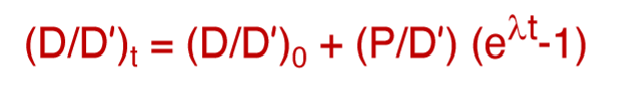
What is this equation called?
The isochron equation and initial ratio
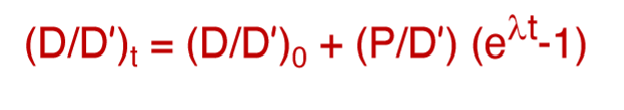
What do the following represent?
D
D’
P
t
0
D = radiogenic isotope of the daughter element
D’ = the stable isotope of the daughter element
P = isotope of parent element that decays into daughter
t = time elapsed
0 = time 0
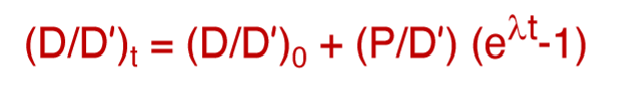
What do the following represent?
(D/D’)t
(D/D’)0
P/D’
(D/D’)t = the ratio of the system
(D/D’)0 = the initial isotope ratio
P/D’ = the parent-daughter ratio of the system
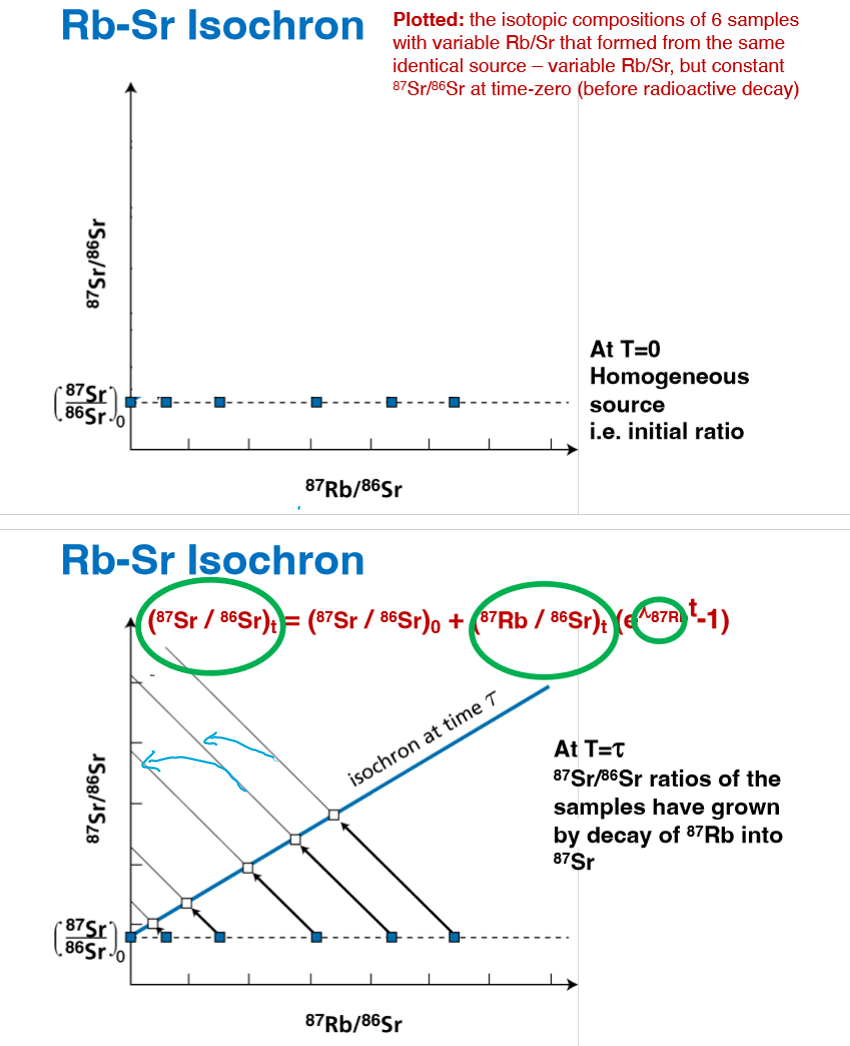
Why is the initial isotope ratio important?
Key to understanding the origin and evolution of the system under consideration
What type of plot is this? What is plotted on it?
An Isochron Plot
plot contains geologic materials of the same initial isotope ratio and age
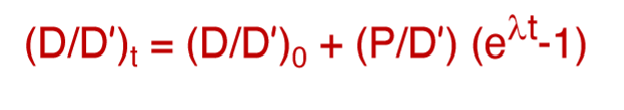
What part of this equation represents the following on an isochron plot?
Y
b
x
m
Y = (D/D’)t
b = (D/D)0
x = (P/D’)t
m = e^(lamdat*)-1
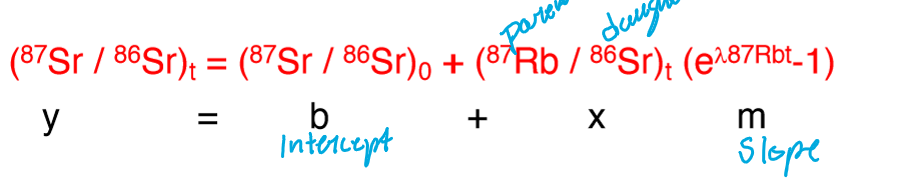
How do the sample ratios look on an isochron at t=0 and t = t?
note the flat to diagonal lines
What is Elemental Fractionation
When different elements arrange themselves to achieve the most stable configuration (elements separate)
different element = separate
What is isotopic homogenization?
Since isotopes have similar properties (same element) the isotopes are arranged randomly among the available sites
same element and different isotopes = homogenous/mixed
Define the decay constant
The probability that a radioactive nuclide will decay per unit of time
specific to the radioactive nuclide under consideration