Structural Characterisation of Solids
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Describe the difference between crystalline and amorphous solids.
Crystalline: long-range, ordered atomic arrangement. Often show distinct diffraction patterns and sharp melting points.
Amorphous: no long-range atomic arrangement
Describe basic crystallographic concepts (lattice, basis, unit cell and crystal structure).
Lattice – an infinite array of points in space where each point has identical surroundings
Basis – a motif associated with each lattice point. Can be an atom, a molecule or part of, an ion etc.
Crystal structure – the periodic arrangement of atoms in a solid.
Unit cell – the smallest component from which the entire crystal can be built up, with purely translational displacements. Parameters: a, b, y.
Describe the crystallographic symmetry elements: screw axis, glide plane.
Screw axis: Np = (360deg/N rotation, then p/N translation parallel to axis).
Glide plane: reflection in m (mirror plane) + translation along the plan ½ the repeat distance. Can be a, b, or c, depending on the translation plane (a b or c).
What does the first letter of the space group show (P, I, F, A, B, C)?
P = Primitive, absence of unit cell centring, lattice points only at the corners of the unit cell.
I = Body-centred, one lattice point in centre of cell.
F = Face-centred, lattice points at the centres of all faces.
A, B, C = Base-centred, lattice points at one pair of opposite faces (e.g. A = bc faces).
What is the difference between molecular and crystallographic symmetry?
Molecular symmetry focuses on individual molecules and individual molecules symmetry elements-> point groups. Importance in spectroscopy.
Crystallographic symmetry focuses on the entire crystal lattice and includes molecular symmetry and translational symmetry -> space groups. Importance in diffraction patterns and X-ray crystallography
Why are not all point groups compatible with 3D periodic arrays?
Not all n-fold rotation axis of point groups are retained as they cannot fill space periodically. Only 2, 3, 4 and 6-fold axes retained. 5-fold axes unit cells cannot fit together without gaps.
Linear point groups (C∞v and D∞h) are not retained as they involve infinite symmetry elements which are not compatible with periodic nature of crystals – crystallography only allow discrete rotational symmetries
Explain the concept of Bravais lattices and crystal systems
Bravais lattice = unit cell. Characterised by lattice parameters (length and angles), and its unit cell centring. 5 for 2D and 14 for 3D.
Crystal systems = symmetry + unit cell geometry.
Define the concept of space groups
Describes the symmetry of a crystal structure – all symmetry operations that can be applied to a crystal such that the crystal remains unchanged.
Explain the difference between general and special positions
General position = a location in the unit cell that is not constrained by any symmetry element. Often high multiplicity. (x, y, z) can be any number between 0 and 1.
Special position = a location in the unit cell that lies on a symmetry element. (low multiplicity, high site symmetry). (x, y, z) do not have free parameters, must be fixed numbers.
Explain what is shown by: site symmetry, multiplicity and Wyckoff letter.
Site symmetry: local symmetry around an atomic position. 1 = Identity and Bar 1 = inversion centre (symmetry elements lie on inversion centre).
Multiplicity: number of equivalent atomic positions generated by applying the space group's symmetry operations// number of different coordinates.
Wyckoff letter: corresponds to a specific site symmetry and multiplicity.
Explain the concept of the asymmetric unit.
The smallest unique portion of the structure which must be specified before space group symmetry (+ symmetry operations) can define the unit cell and the entire structure.
What are the basic requirements for incident radiation/particles for a diffraction experiment
comparable wavelengths to interatomic distances, incident beam is in phase
Give the equation linking interplanar d-spacing and the reflection indices.
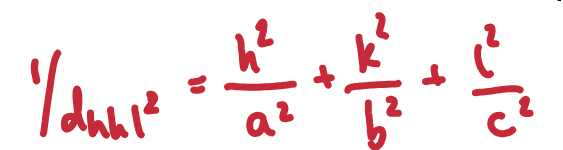
Give the Bragg equation

What is the atomic scattering factor? How does it change with atomic number and with theta?
= f. Measure of how strongly X-rays interact with individual atoms.
When sin(theta)/λ = 0, f = Z (atomic number).
f increases with atomic factor → more electrons.
As theta increases, f decreases, Destructive interference more pronounced at higher scattering angles.
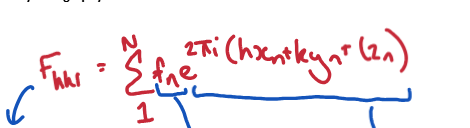
Explain the different parts of the structure factor equation
Structure factor represents the amplitude and phase of the wave diffracted from crystal lattice planes. Made up of the electronic property (1) and the structural property (2)
What is the structure factor proportional to?

What is the phase problem? (and solution)
In diffraction experiments, we measure the intensity of scattered waves, which gives us the amplitude of the structure factor. However we lose the phase information when determining structure factor.
To overcome, we use approximate atomic positions to calculate the model structure factors, including both the amplitude and phase, and combined we experimentally measure amplitudes. The model is then refined to locate atoms more precisely.
What are systematic absences? How do they arise?
Missing diffraction spots not observed in the diffraction pattern. Arise as certain symmetry elements cause destructive interference in scattered waves, cancelling out contributions to the structure factor (F=0).
Give the reflection conditions for each cell centring.
Note: do not occur for P. Note: can also occur from translational symmetry elements.
How are X-rays generated in an X-ray tube? How can more intense, higher resolution X-rays be produced?
Evacuated enclosure of glass and metal which produces electrons by passing an electrical current through a wire filament and accelerated into a metal target. Electrons KE generates X-rays by interaction with metal atoms (Cu or Mo) → Continuous spectrum.
Use of a Synchotron to get more intense X-rays.
What are characteristic X-rays?
Sharp peaks caused by electron transitions within the atoms.
Explain the differences between the variations of the X-ray and neutron scattering powers of elements across the Periodic Table, and the reasons for these differences.
There is no regular trend for neutron scattering power across the periodic table. Neutrons interact with atomic nuclei, and the strength of this interaction is described by the neutron scattering length, which varies across the periodic table.
For X-rays, elements with high electron densities (so going across the periodic table/increasing atomic number), resulting in higher scattering powers due to stronger interactions with X-rays hence easier to detect in diffraction.
What is the difference between X-ray and neutron diffraction?
Where X-ray scattering is due to interactions with the electron density, Neutrons are scattered by the nuclei.
What are the advantages and disadvantages of neutron diffraction (compared to X-ray)?
Advantages of neutron diffraction:
Scattering power does not fall off at higher scattering angles, whereas this does in X-rays due to atomic vibrations.
Can distinguish between isoelectronic species or species with very similar number of electrons (almost impossible in X-ray) e.g. H vs D.
Neutrons have a magnetic moment so can study magnetic structure/ordering in materials.
Disadvantages of neutron diffraction:
Much more difficult and expensive.
total intensity of diffraction is much lower than X-ray. Hence requires larger crystals.
Compare single crystal diffraction vs powder diffraction
Single crystal diffraction uses a single, well-formed crystal with specific orientations.
Precise determination of atomic positions.
Requires a high quality single crystal.
Powder diffraction uses a powdered sample of tiny crystallites in random orientation. Measures diffraction rings rather than discrete spots.
Produces 1D diffraction patterns (graph of intensity vs angle).
Suitable for phase identification and unit cell determination.
Faster use and easier sample preparation, however leads to problems with peak overlap – compresses the full 3D diffraction pattern into 1D pattern (variable = theta). Might only be able to determine structure for simple structures with few reflections.
What is SEM? What is some of the key applications of SEM?
Uses a focused beam of electrons to scan the sample's surface.
Produces 3 types of primary signal.
→ Backscattered electrons: chemical constrast (heavier elements have increasing backscattering, appear brighter in imaging).
→ Secondary electrons: topographical contrast, more secondary electrons = brighter image areas.
→ X-rays: Allows for elemental analysis, where the energy of the X-ray is characteristic of the element it came from (different energies).
What is TEM? What is some of its key applications?
Transmits electrons through a thin specimen to form an image.
Produces 2 types of primary signals: image and diffraction pattern
→ Imaging (HREM).
→ Electron diffraction: Used for crystal structure determination and unit cell determination.
Give the advantages and disadvantages of SEM and TEM.
Advantages of SEM:
Detailed surface imaging, easier sample preparation, very versatile.
Disadvantages of SEM:
Lower resolution, can only see surface samples.
Advantages of TEM:
High resolution, information about internal structure, electron diffraction enables phase identification.
Disadvantages of TEM:
Sample prep is more complex.
Expensive