CHE 2C - Main Group + a little Ochem
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
Alkali Metals
- Flame colors (ns1 goes to np1 when excited), use = fireworks
- Low density, low MP, soft b/c weak bonding
- Easily oxidized b/c ns1 configuration. Spontaneous oxidation
- +1 oxidation state, large E standard ox potential
- forms oxides (oxide, peroxide, superoxide)
- Used to make soaps (rxn with fat and NaOH/lye)
Hydrogen
- 1s1 (one proton and one electron)
- nonmetal, colorless, odorless, tasteless
- VERY abundant
- VERY light, escapes atmosphere easily
- liquid hydrogen = good jet fuel
Alkaline Earth Metals
- oxidation state of +2
- low MP, low BP, low density
- insoluble don't decompose with heat
- CaO is in concrete
- Chalk, limestone = CaCO3
- Acid rain
- Hard water (ion exchange column used to get rid of minerals)
Beryllium
Different from other Alkaline Earth Metals b/c:
- Harder
- Higher MP
- not reactive w/ air and water
- high ionization energy
- covalent bonds instead of ionic
- smaller size
- Be2+ formation limited
- unique structure w/ BeCl (see wikipedia)

Icosagens (Group 13, Al and down)
- ox states: +1 and +3
- soft metals
- relatively reactive
- good conductors
- forms dimers
Aluminum Facts
- most abundant metal in Earth's crust
- formed via electrolysis
- forms alumina (Al2O3)
-amphotheric
- Reducing agent

Gallium
- smelting to obtain
- liquid state is high density
- diffuses in crystal lattices to attack other metals
- Uses: solar cells, LEDs, semiconductors, etc.

Indium
- squeals when bent
- strong reducing or oxidizing agent
- Uses: LCD electrodes, alloys, solders, coatings, transistors, etc.

Thallium
- means "twig" in Greek
- TOXIC (can be utilized as poison)
- Uses: superconductors, lamps, pharmaceuticals, glass, etc.

Tetragens (Group 14)
- roasting then reduction to form
- ox states are +2 and +4, commonly
- Focus on Sn, Pb

Tin (Sn)
- pewter
- food storage (tin cans)
- solders

Lead (Pb)
- pipes/plumbing (hence why symbol is Pb, PlumBing)
- batteries
- ammunition
- lead poisoning (interferes w/ heme in blood)
- Used to be used for pencils, paint, gas, solders but not anymore b/c poisoning (esp with children and pencils, eating paint)

Noble Gases
- LDF forces (low)
- mostly unreactive
- Low MP, BP
- near ideal at STP
- monatomic, colorless, odorless (filled e- shells)

Helium
- can be trapped but mostly escapes atmosphere
- diluent for fuel (to lessen flammability risk)
- superfluidity (no viscosity)
- has several possible liquid phases

Neon
- fun lights
- emits light under current
- "neon" = "new one" in Greek

Argon
- small amount in atmosphere (third most common)
- Greek for "lazy"
- Uses in welding, radioactive dating, light bulbs

Krypton
- Product of nuclear fission; can be used to measure nuclear activity throughout world
- "Hidden one" in Greek
- Hcp packing structure

Xenon
- readily oxidized, more than other noble gases
- flash tubes
- halogen lamps
- CAN BE REACTED

Radon
- hot springs
- Lung cancer!
- made from deep-Earth radioactive processes

Halogens (Group 17)
- VERY reactive
- conventionally denoted by X
- electrolysis/oxidation to produce
- Iodine least reactive, Fluorine most
- Applications: HF in glass etching, oxy acids (put H on oxygen in lewis structures)
- Makes compounds like Teflon, PVC, Freon (more details in reader)
Chalcogens
Group 16
oxygen
O2 and O3 (ozone) elemental forms
Common ox states: 0, -1, -2

Sulfur
- elemental forms: S8 (most common), S, S2, S6, Sn (allotropes)
- makes oxides
- chemical weapons, fertilizers, skunk smell, vulcanized rubber, waste processing
- smells like eggs and generally not good
- S-S bonds responsible for hair texture. Also in hair products.

Selenium, Tellurium
Glass coloring, semiconductors, alloys

POLONIUM (my personal favorite)
one gram can kill 50 MILLION PEOPLE
Also:
- named after Poland
- discovered by Marie Curie
- radioactive, so nuclear weapons
- In tobacco, maybe carcinogen
- only element with simple cube packing structure

Pnictogens (group 15)
- several ox states
- electron config: ns2np3
Nitrogen
- Ability to be very nonreactive, or very reactive
- N2 inert, but TNT (Tri-Nitro-Toluene) and nitroglycerine are explosive
- Biology: DNA, nitrogen fixation
- NH3, aqua regia, laughing gas (nitrous oxide)

Phosphorus
- 3 allotropes: white, red, black phosphorus
- oxide formation
- P4 and P2O5 common compounds
- ATP and ADP
- fertilizer

Arsenic
- POISON, toxic
- metalloid, many allotropes
- alloy strengthening
- semiconductor
- in ground water
- pesticides, herbicides, insecticides

Antimony
Mined in China and used in solders, bullets, fire retardants, bearings

Bismuth
- Pepto bismol
- white, silver-pinkish color
- highest stable atomic mass
- low toxicity as a heavy metal; lead stand-in
- pigments, cosmetics
- pretty crystals

Tetragens
group 14

Carbon
- the element of life! essential to life.
- entire branch of chemistry based on C (organic chem)
- forms strong bonds to itself and other atoms
- cyclic and acyclic structure formation
- single and multiple bonds
- Allotropes: diamonds, graphite, buckminsterfullerenes (AKA bucky balls), nanotubes, graphene
- highest sublimation point of all elements

Silicon
- oxides = silicates, and there's many
- zeolites (hard water treatment, petroleum catalysts)
- Glass
- silicone (rubber, oils, implants, lubricants, etc.)
- dust, sand
- Silicon Valley namesake

Germanium
Similar so Si and Sn, semiconductor in transistors, solar cells, fiber optics, mined from sphalerite

Boron
- can have incomplete octet (6 e-)
- metalloid
- Dimers, allotropes w/ covalent bonds (like Borane)
- most common ox state is +3
- hard, brittle, generally unreactive
- similar to silicon in behavior
- semiconductor dopant
- Glassware applications: in Pyrex (i.e. lab glassware and glass tupperware/kitchen vessels) for thermal shock resistance

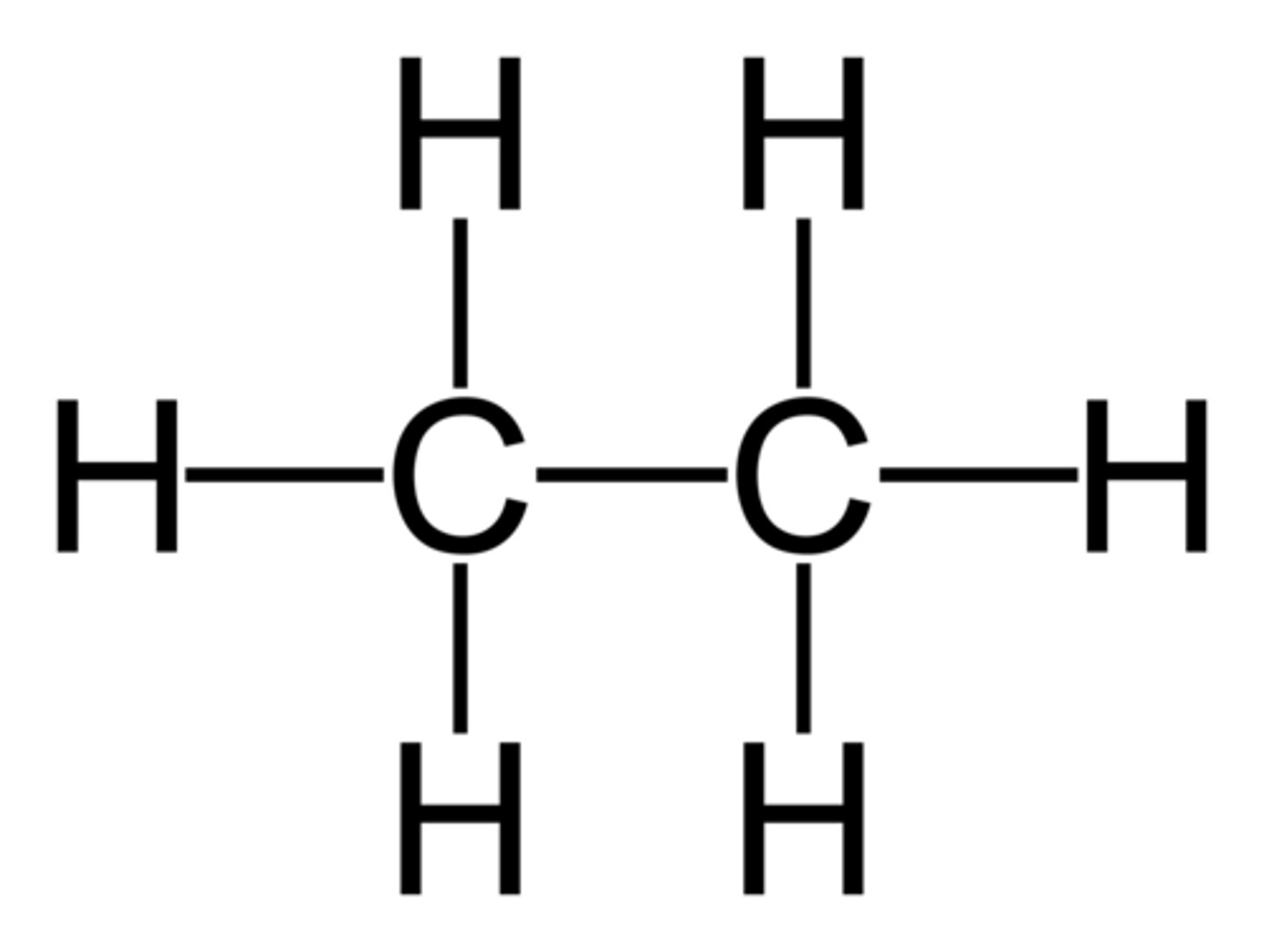
Alkanes
Alkenes
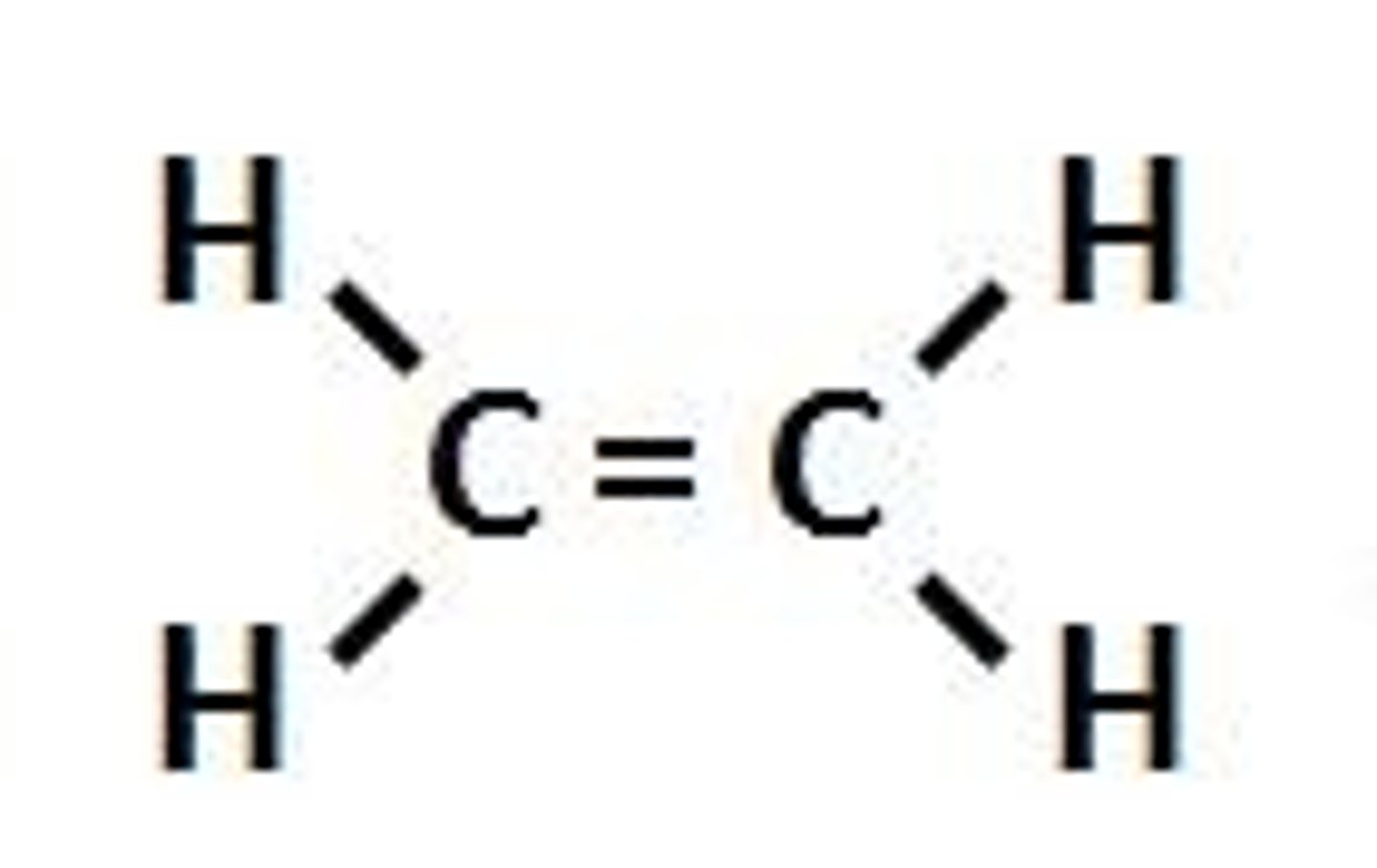
Alkynes
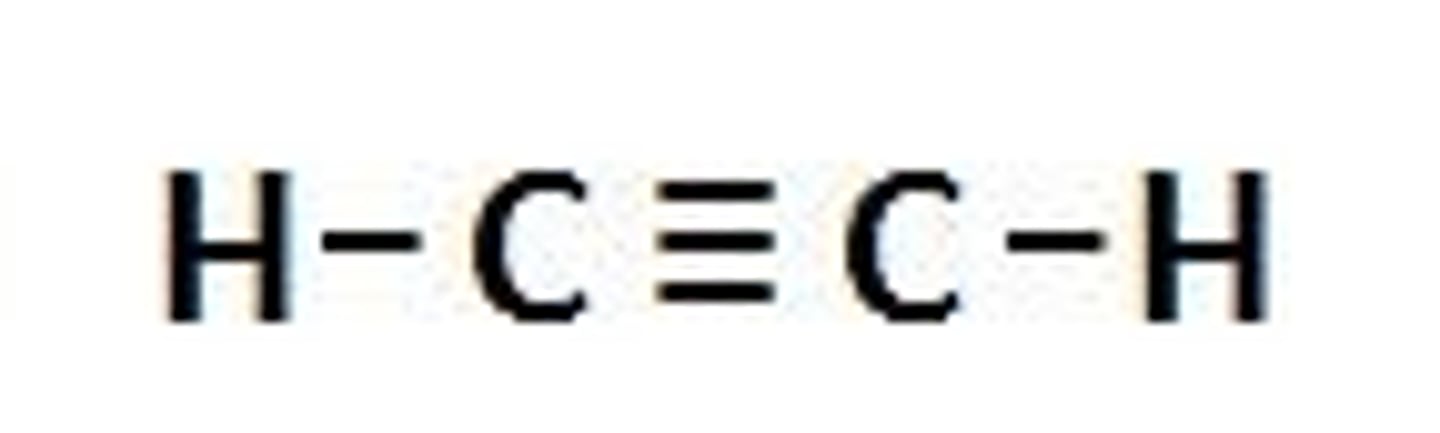
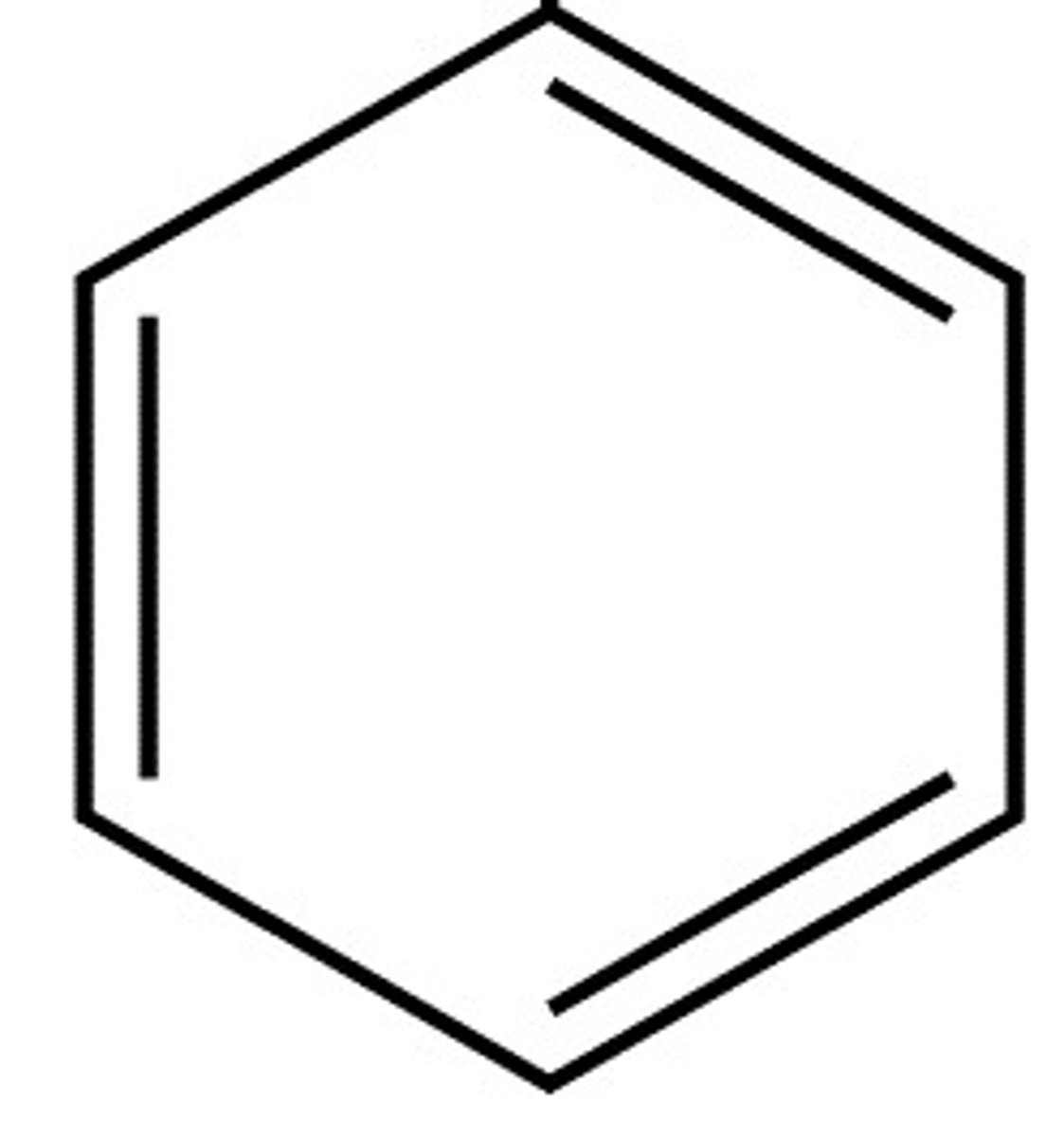
Arenes/Aromatics
Alkyl Halides
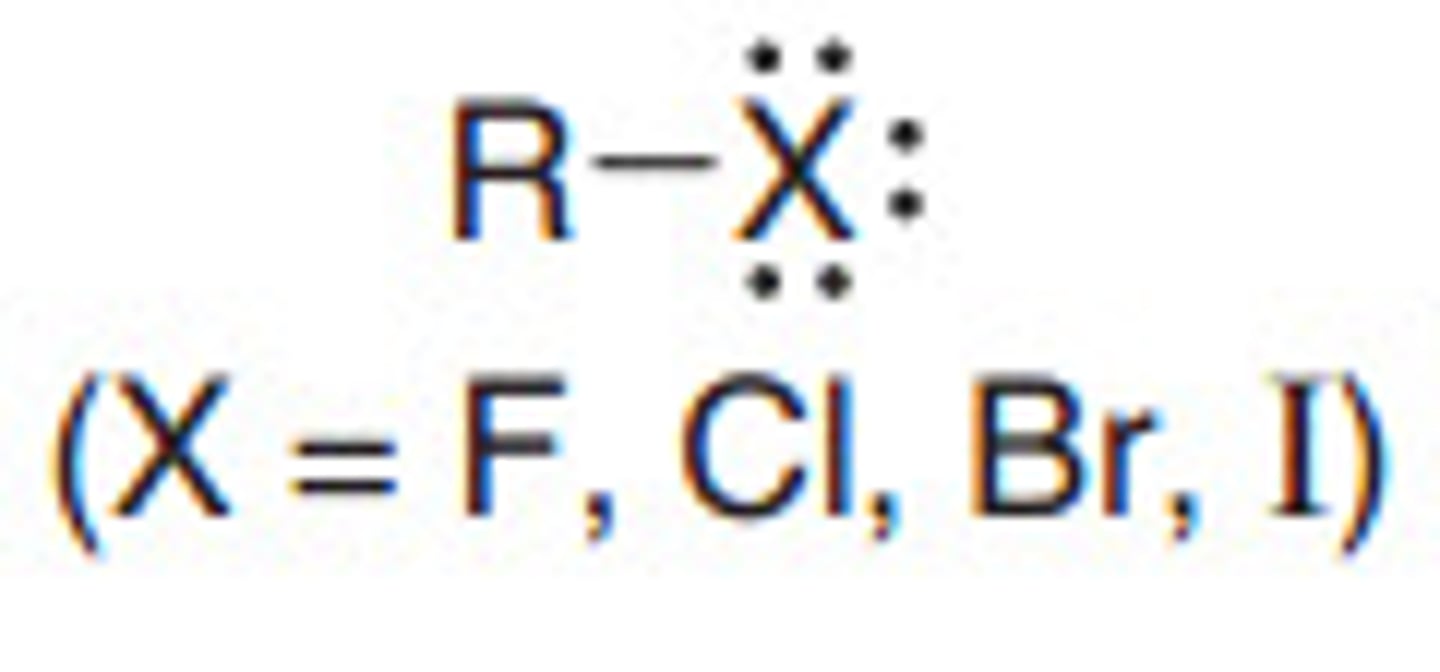
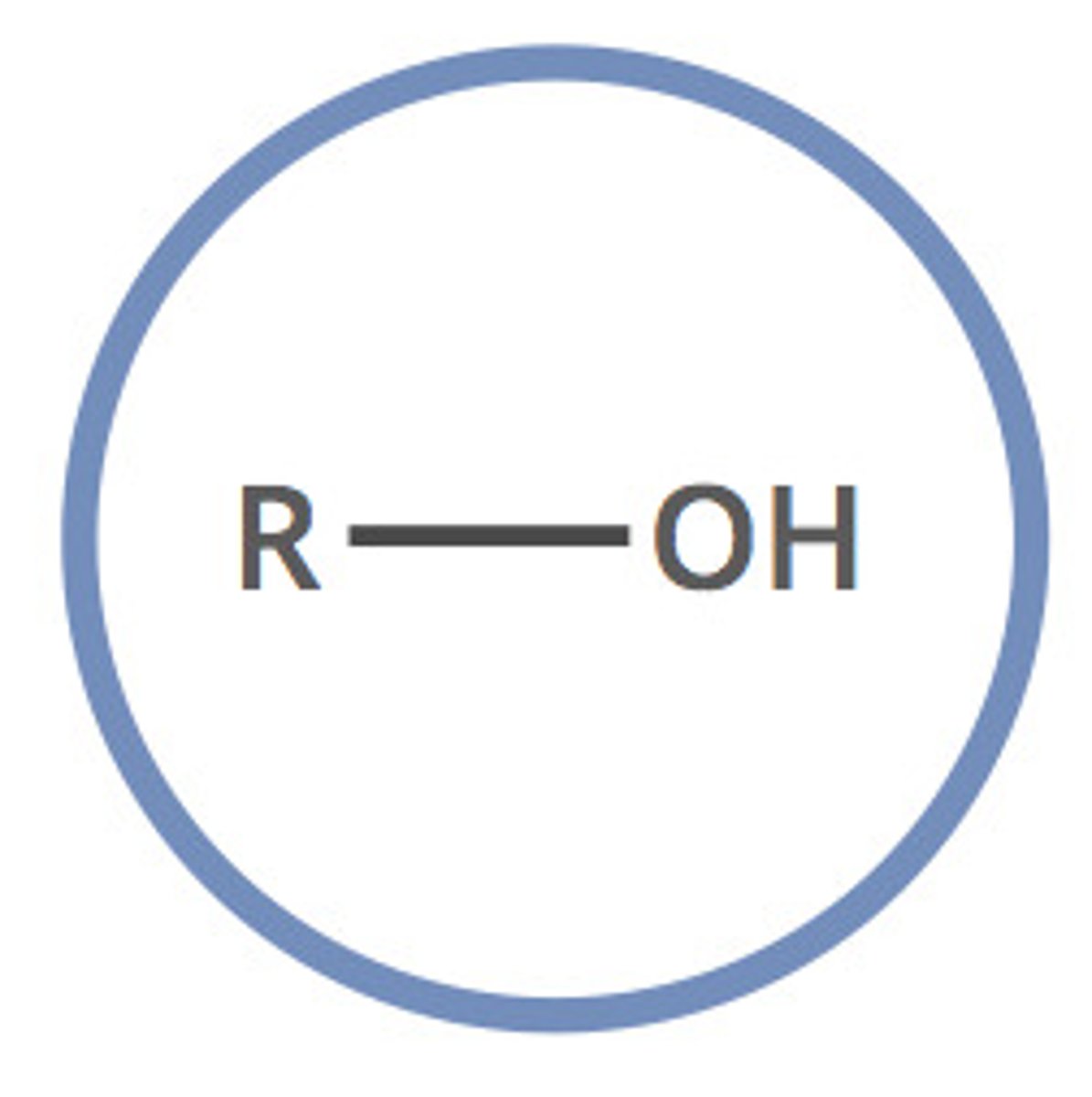
Alcohols
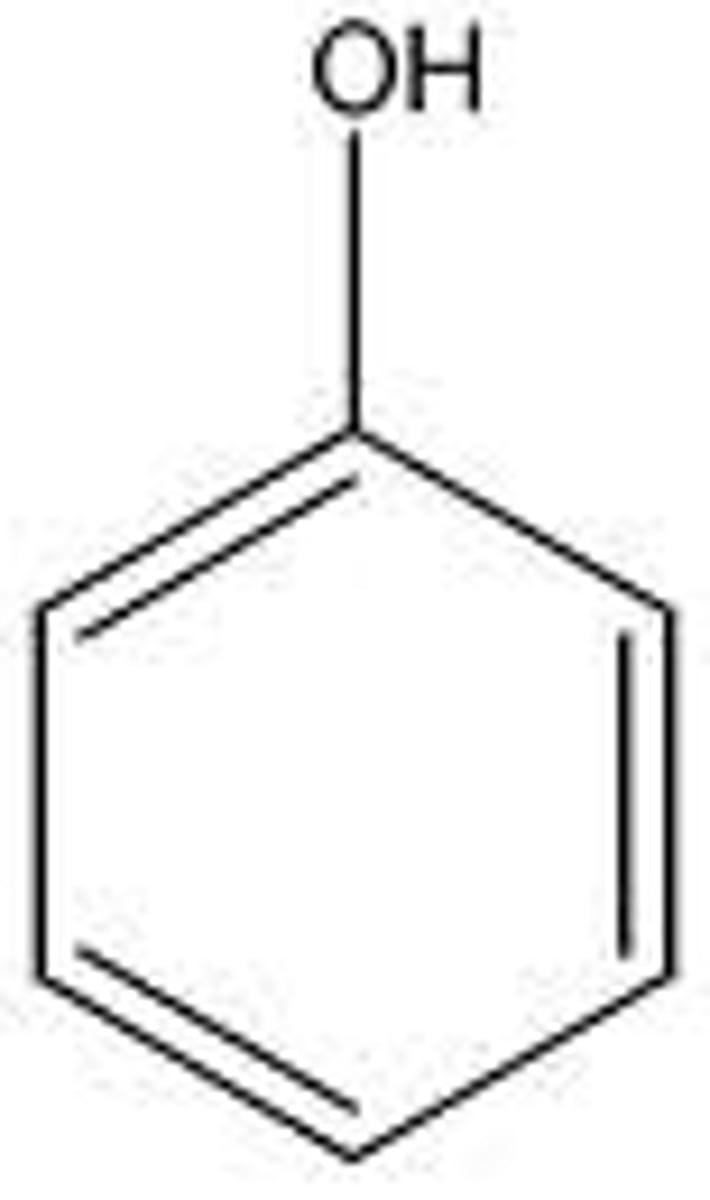
Phenols
Ar-OH
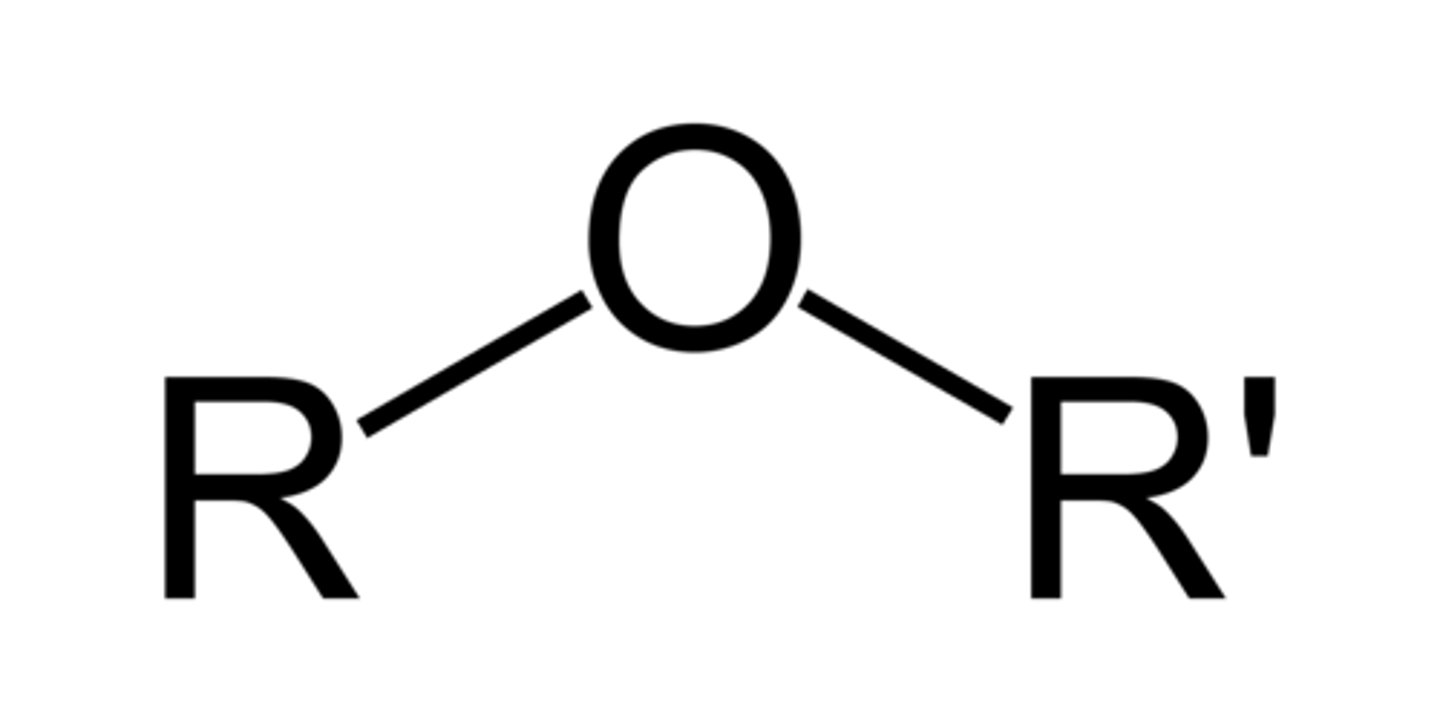
Ethers
R can be swapped for Ar too
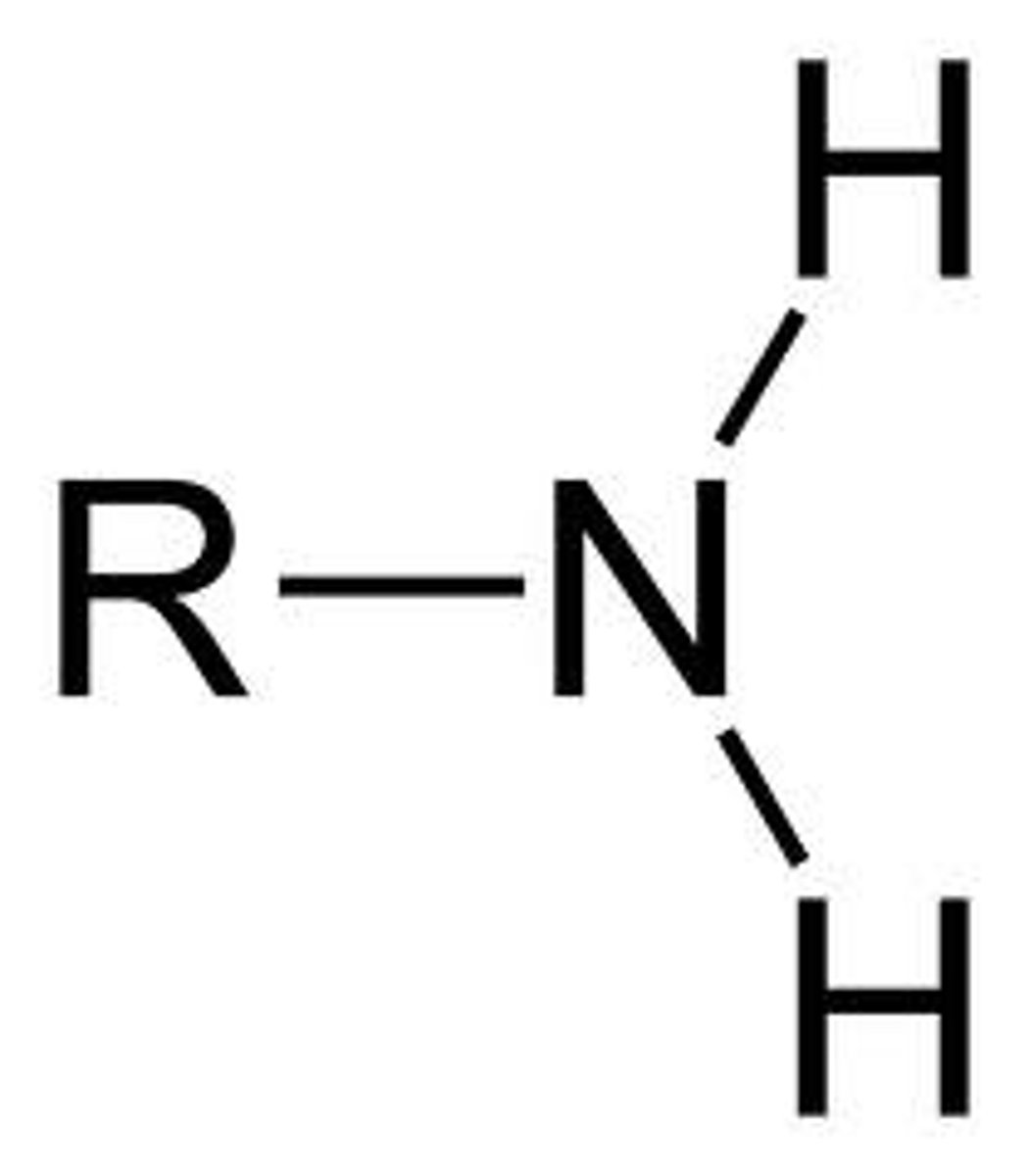
Amines
- R can also be Ar
- The two Hs can also be swapped for R/Ar groups
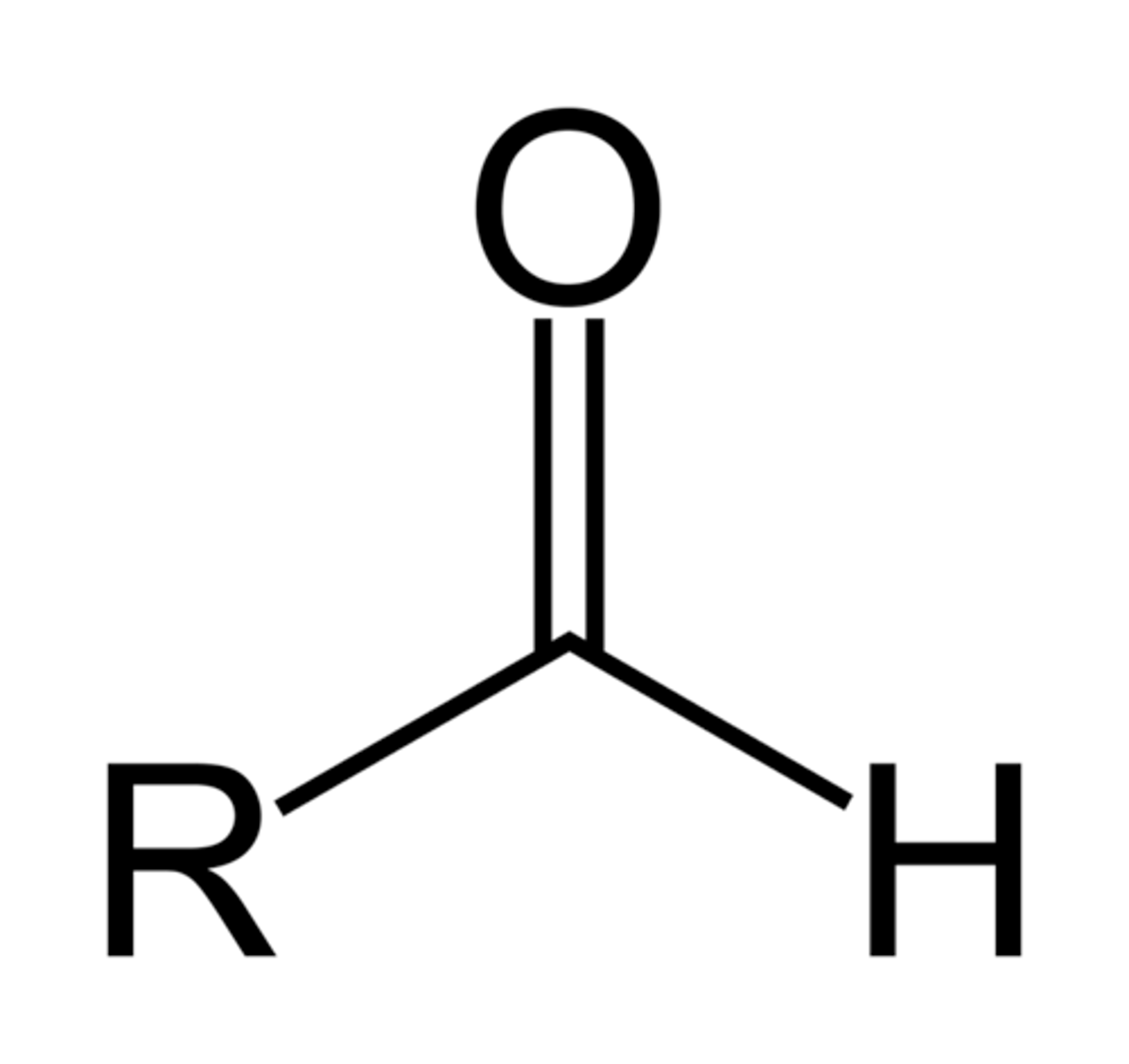
Aldehydes
R can also be Ar
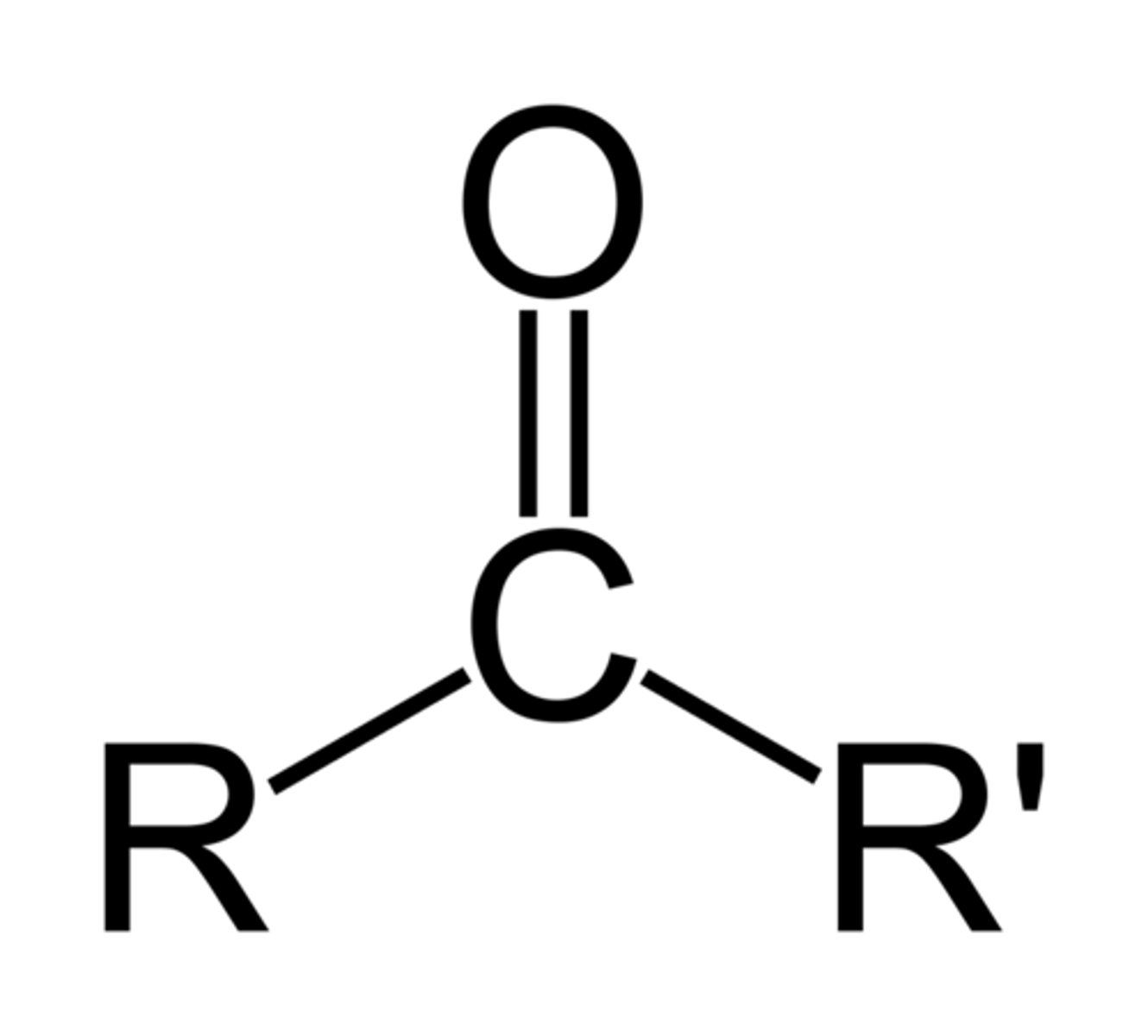
Ketones
R can also be Ar
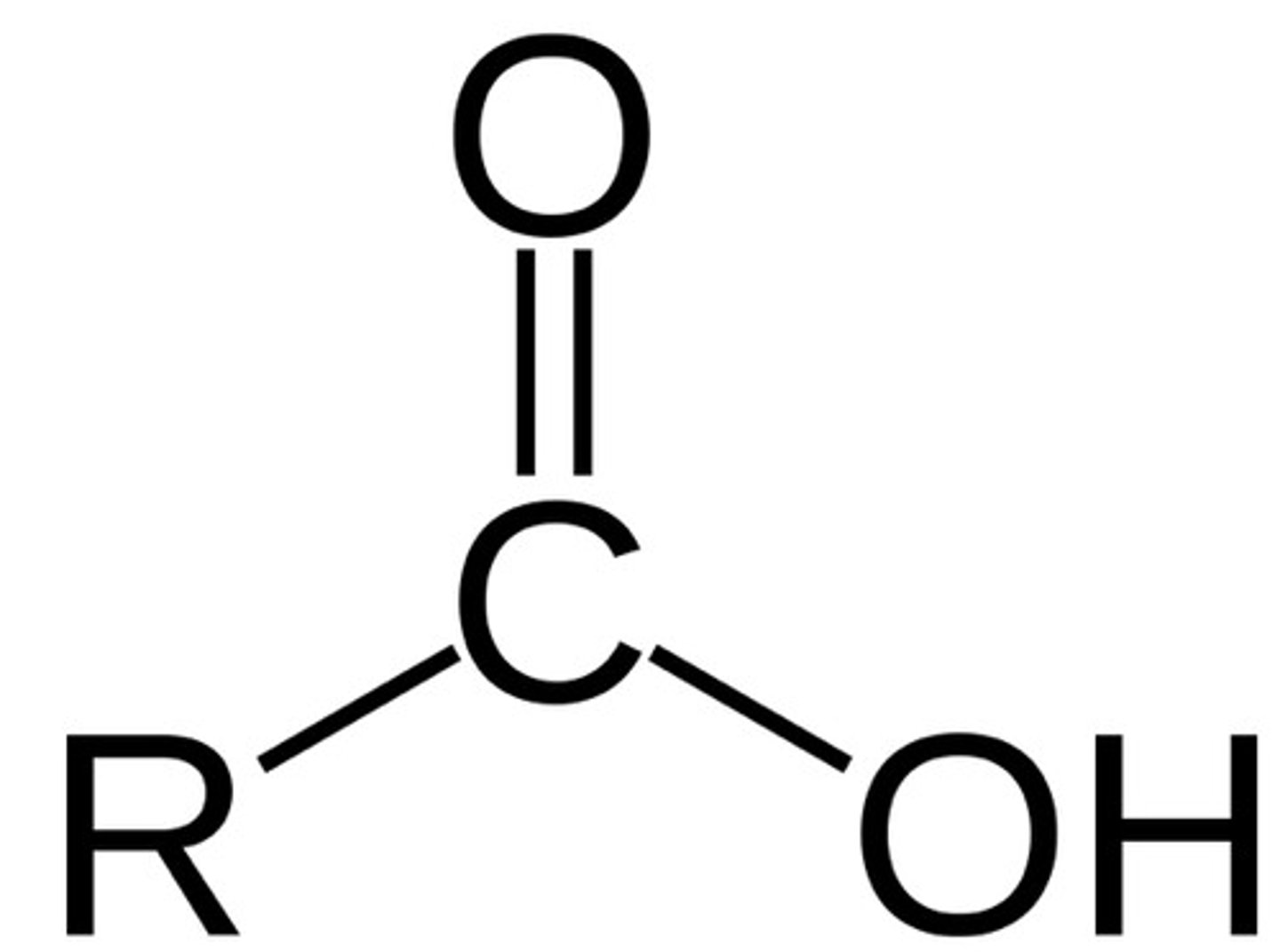
Carboxylic Acids
R can also be Ar
Esters
R can also be Ar
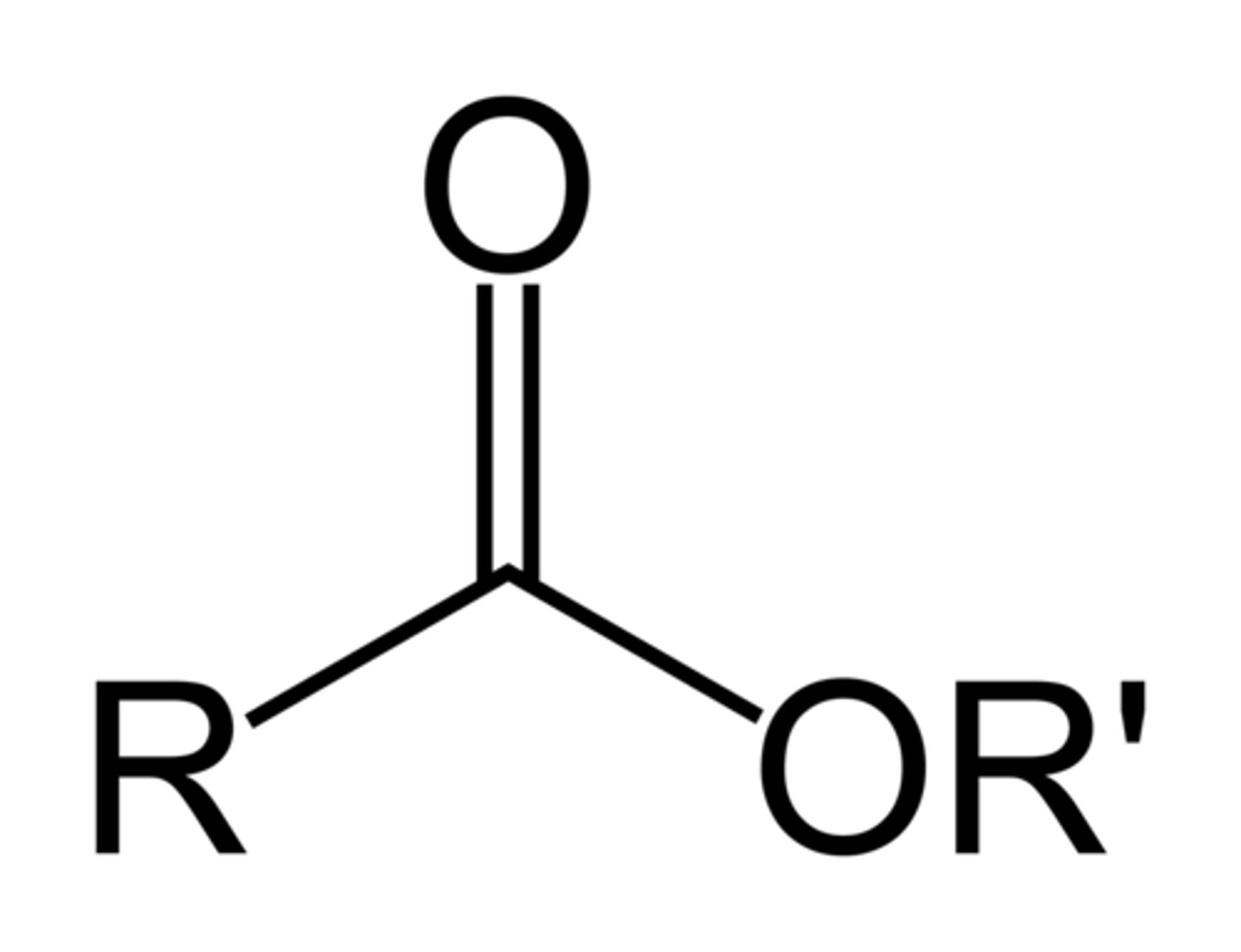
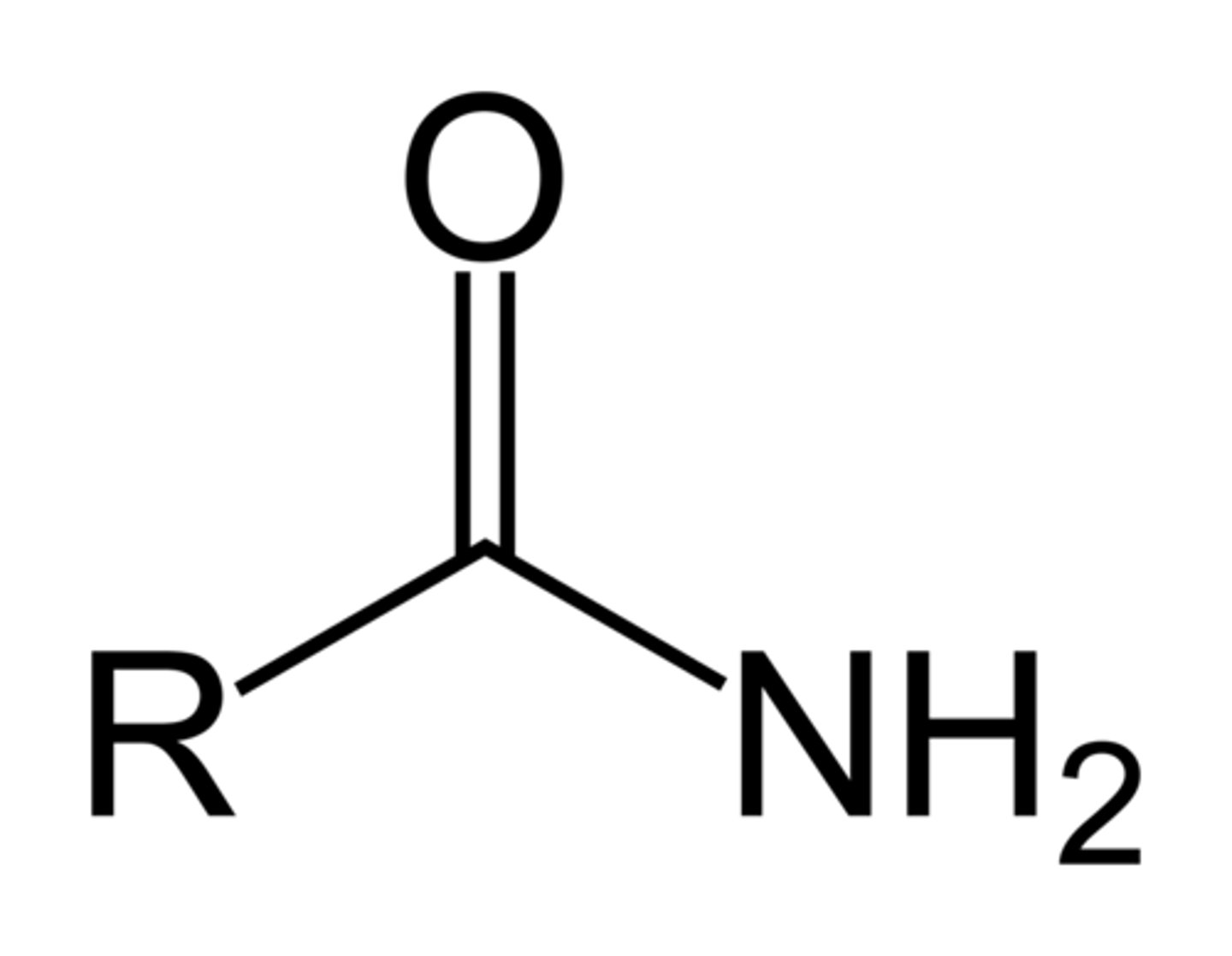
Amides
- R can also be Ar
- The two Hs can also be swapped for R/Ar groups
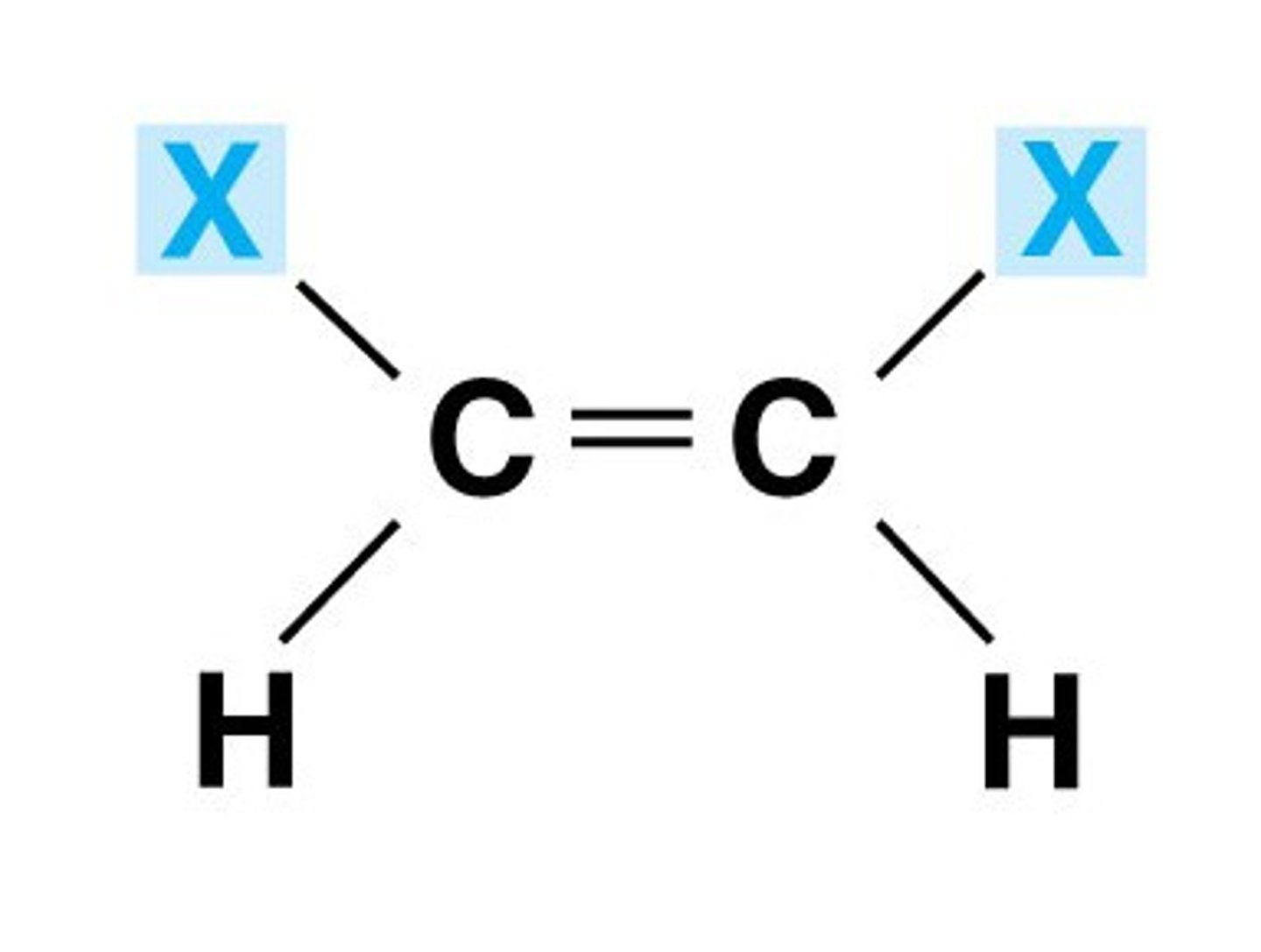
Cis double bond
Substituents are on same side
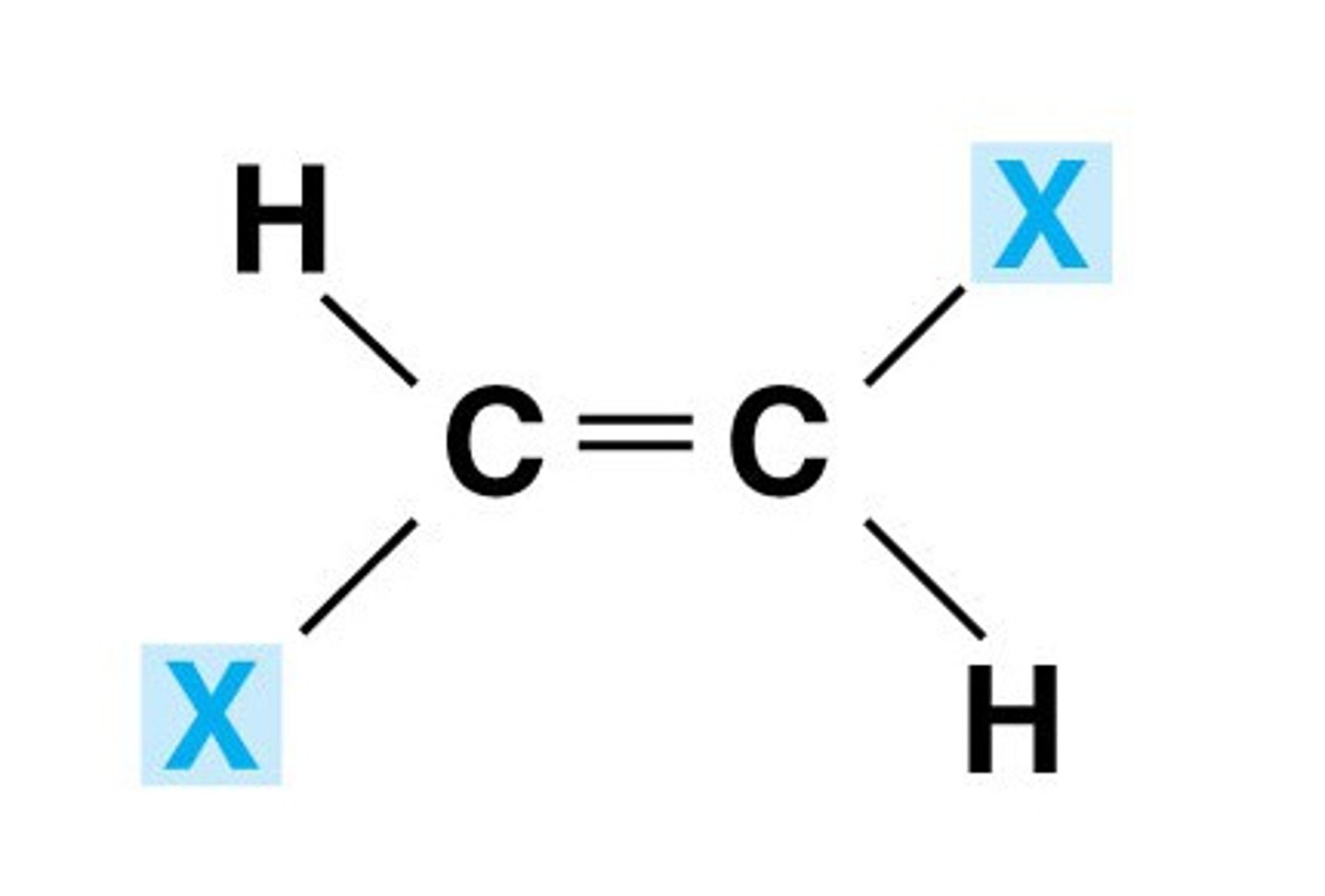
Trans double bond
Substituents are on opposite sides
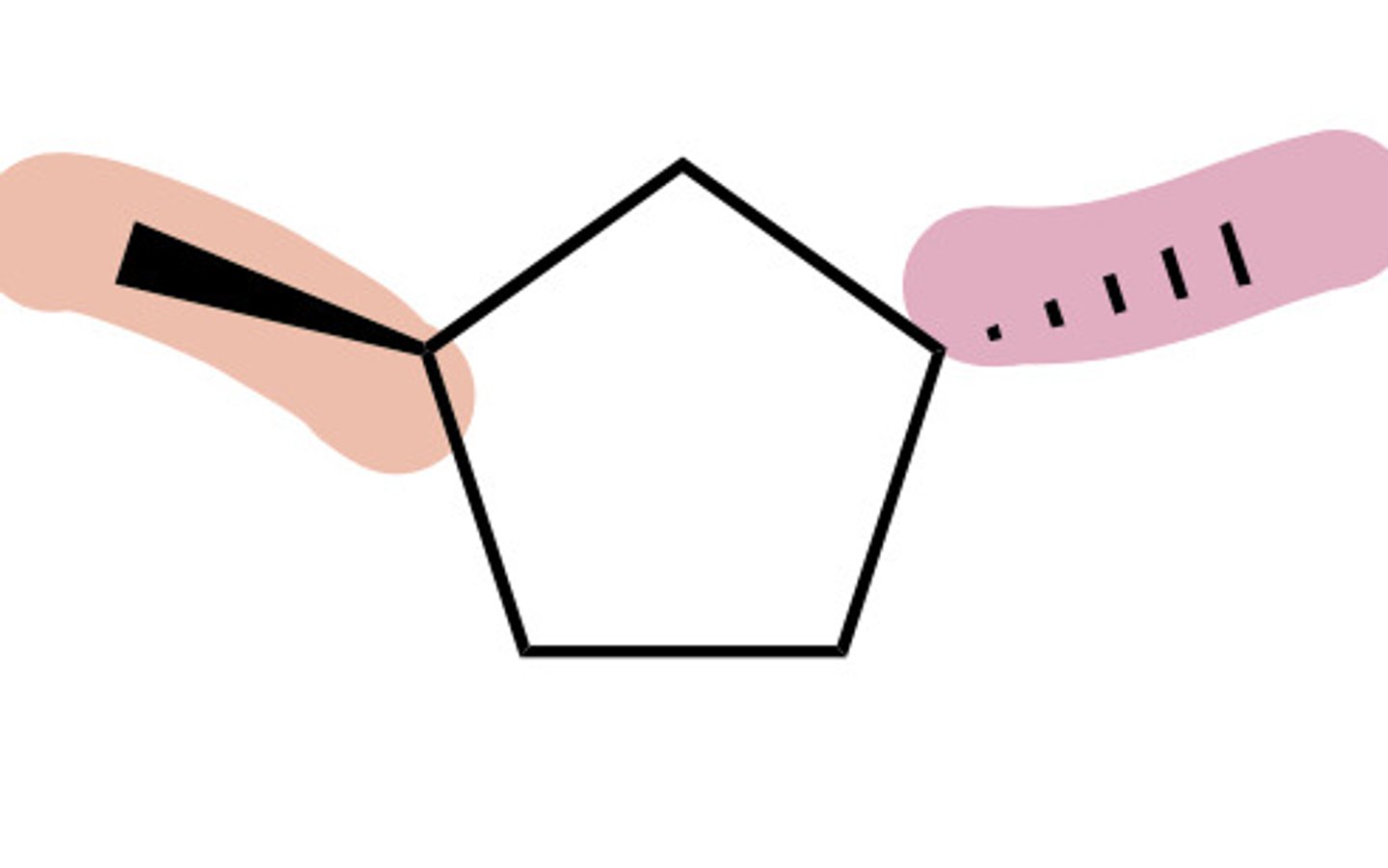
Cis ring
Substituents BOTH either dashed OR wedged
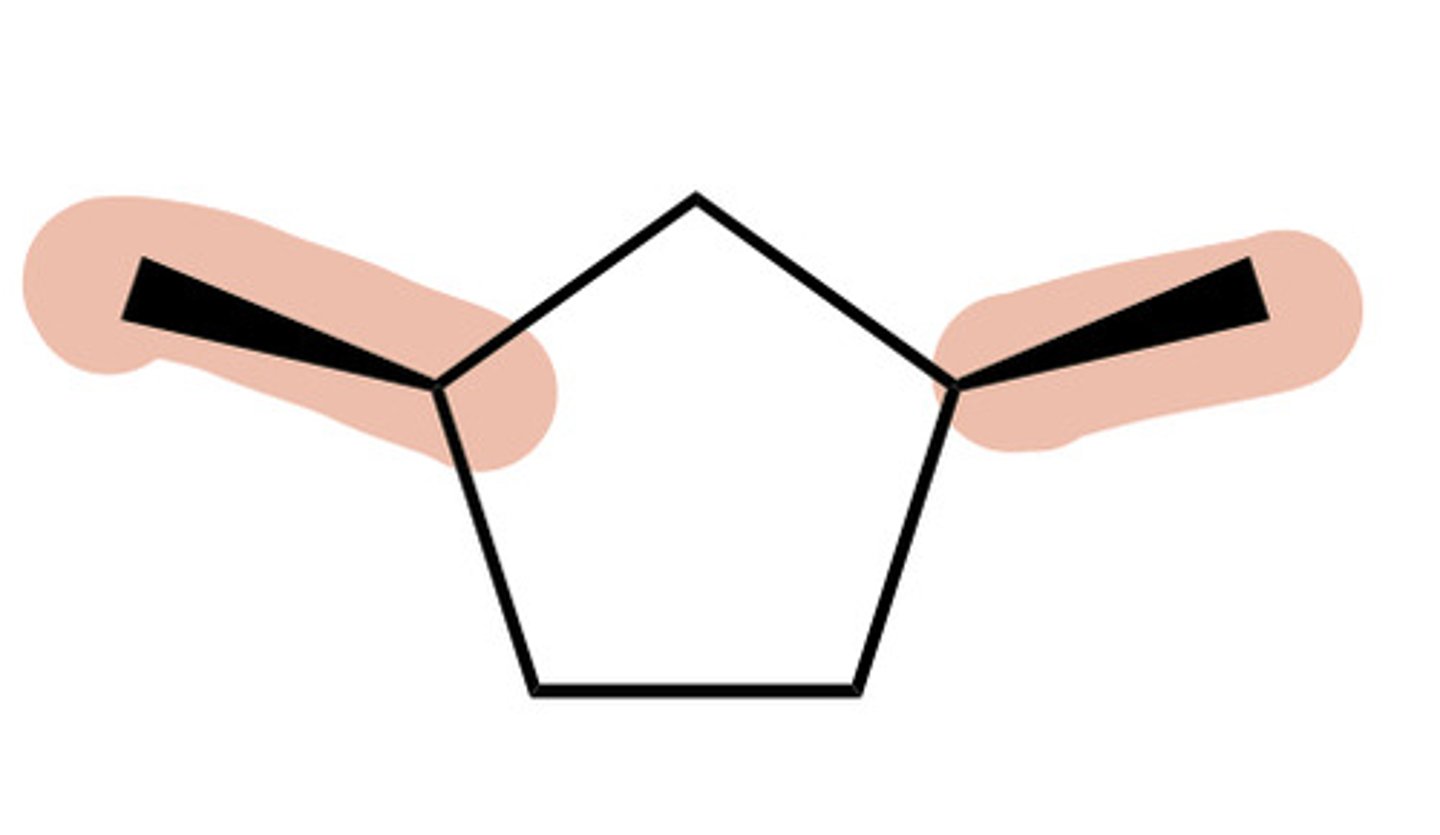
Trans ring
one substituent dashed AND the other is wedged