3.1.5 Kinetics
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What is activation energy?
Minimum energy needed to start a reaction
What is rate of reaction?
Change in concentration of a reactant or product over time (moldm-3s-1)
What is a catalyst?
A substance which speeds up the rate of a reaction without being used up in the process
What are the 3 things that are required for a reaction to occur?
Collision between the reactants
Particles must have E ≥ Ea
The particles must have the correct orientation
What is the units of rate?
moldm-3s-1
Explain the usually process of how a rate of a reaction goes?

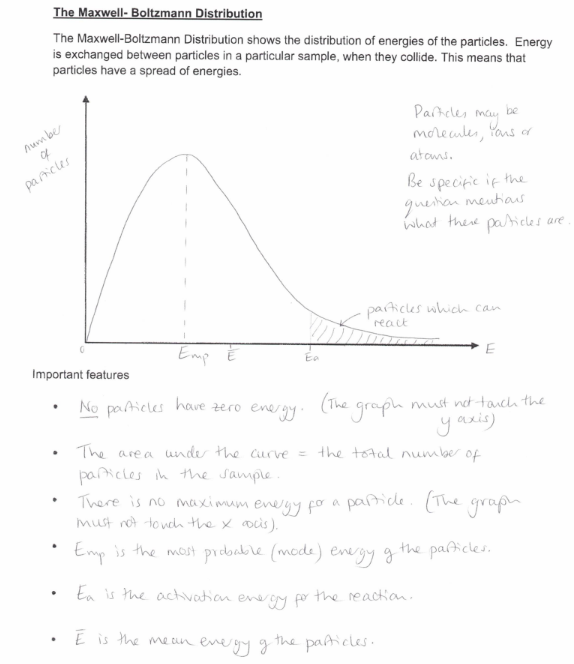
What is the Maxwell-Boltzmann distribution?
What are the factors affecting rate of reaction?
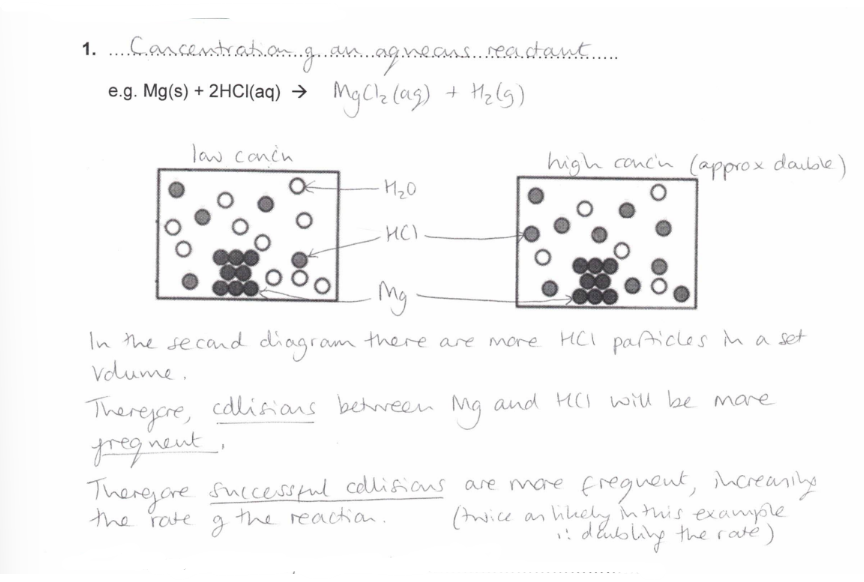
Concentration of an aqueous reactant
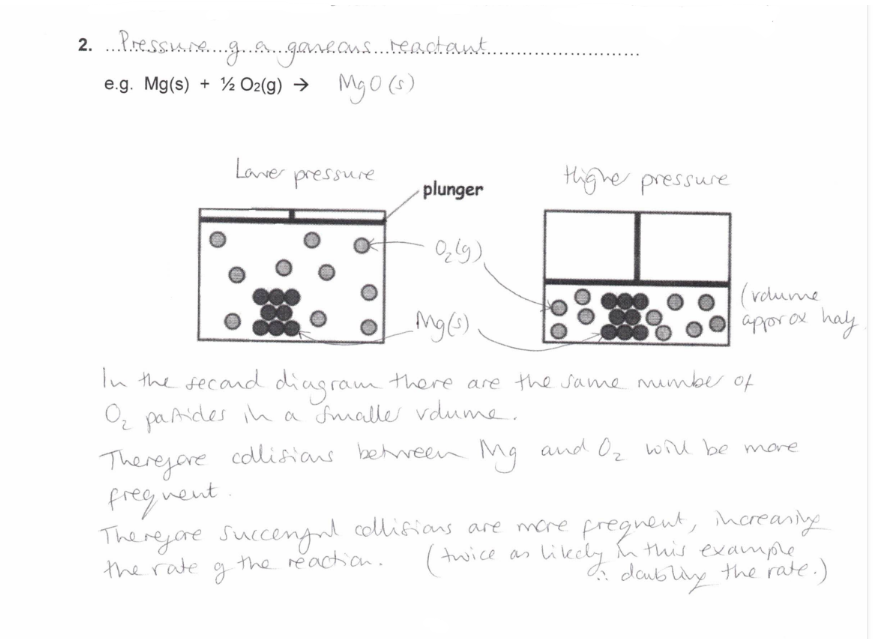
Pressure of a gaseous reactant
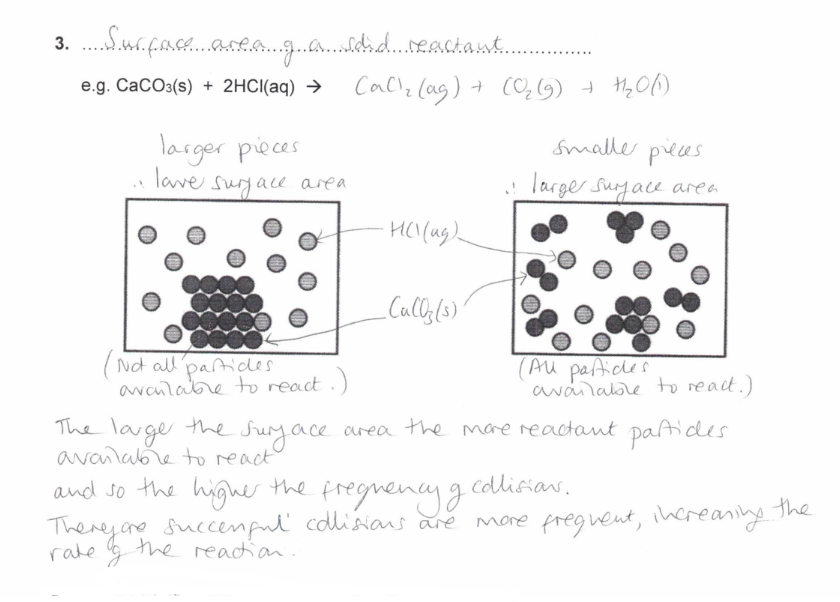
Surface area of a solid reactant
Temperature of the reactants
Addition of a catalyst
How does the Concentration of an aqueous reactant affect the rate of reaction?
How does the Pressure of a gaseous reactant affect the rate of reaction?
How does the Surface area of a solid reactant affect the rate of reaction?
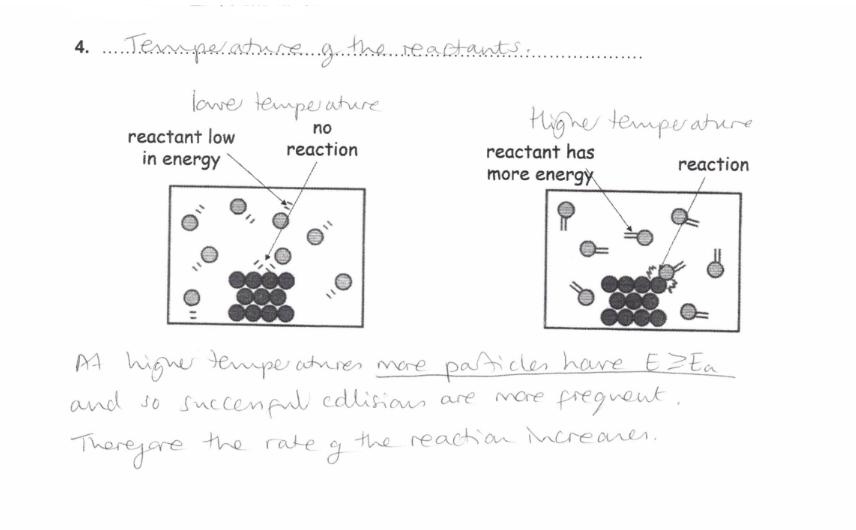
How does the Temperature of the reactants affect the rate of reaction?
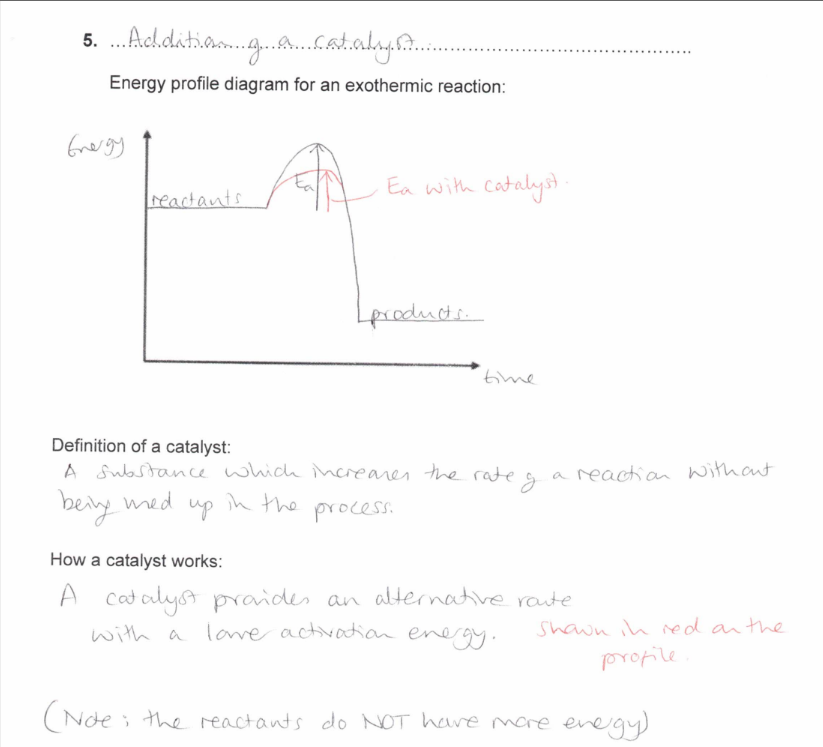
How does the Addition of a catalyst affect the rate of reaction?
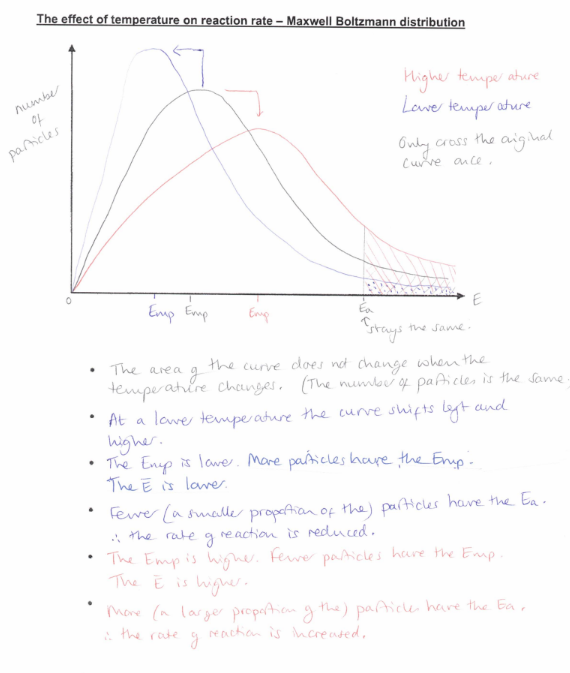
Show how the Maxwell Boltzmann distribution graph is affected by temperature.
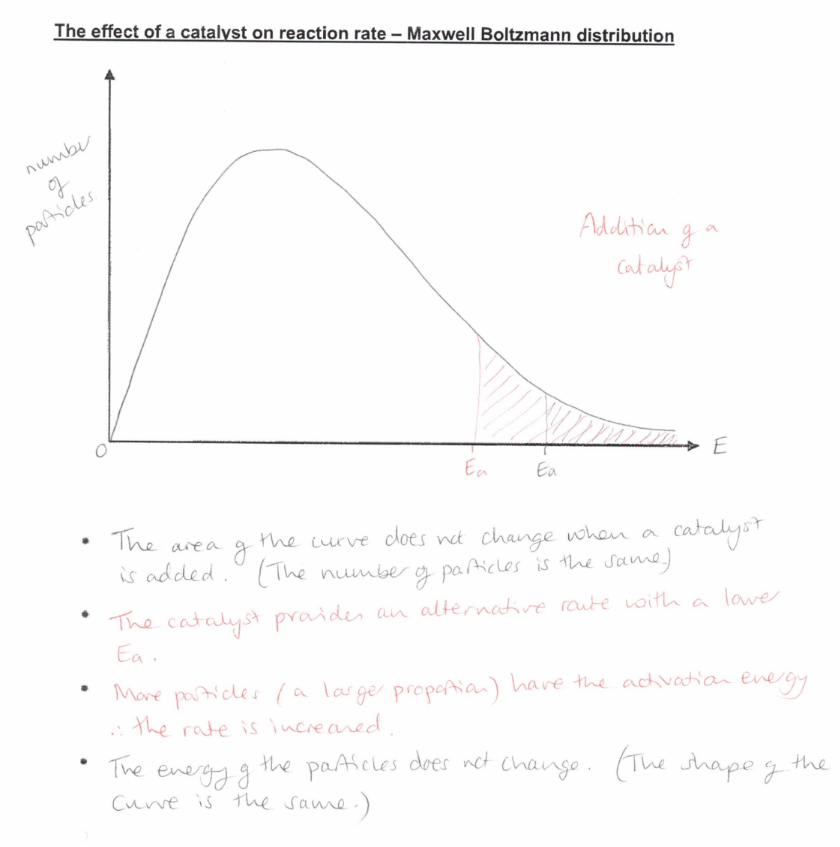
Show how the Maxwell Boltzmann distribution graph is affected by a catalyst.
Study the image about questions of maxwell-boltzmann distribution graph.
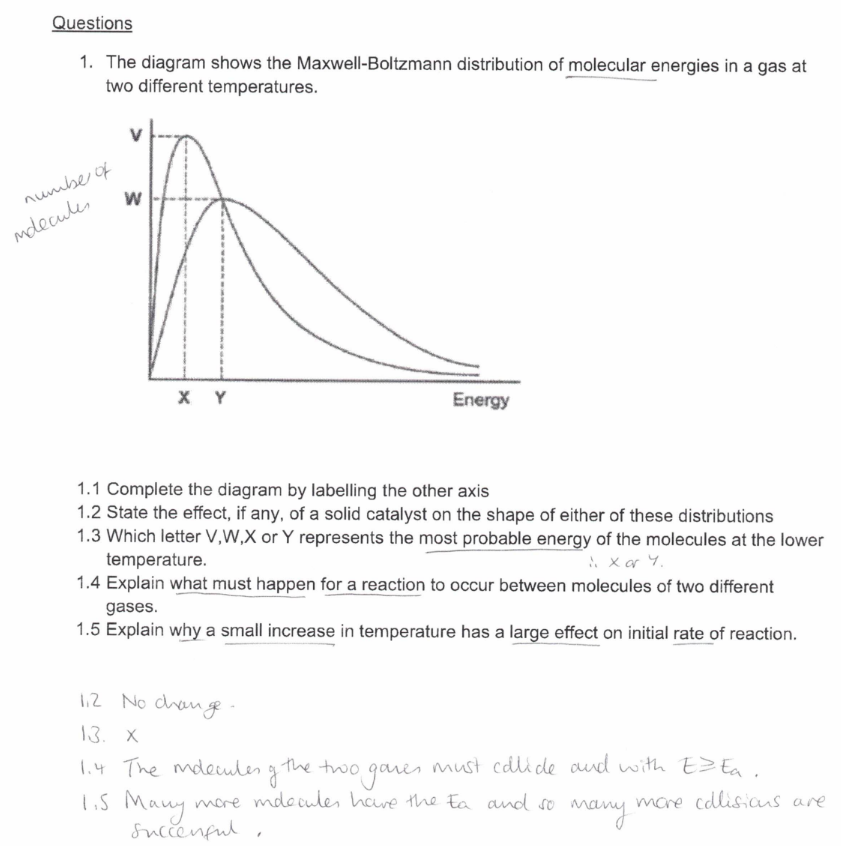
What are the 3 ways of measuring rates of reactions?
Change in mass of the reaction mixture
Change in volume of a gaseous product
Change in the amount of a solid product
How do you measure the rate of a reaction by observing change in mass of the reaction mixture?

How do you measure the rate of a reaction by observing change in volume of a gaseous product?
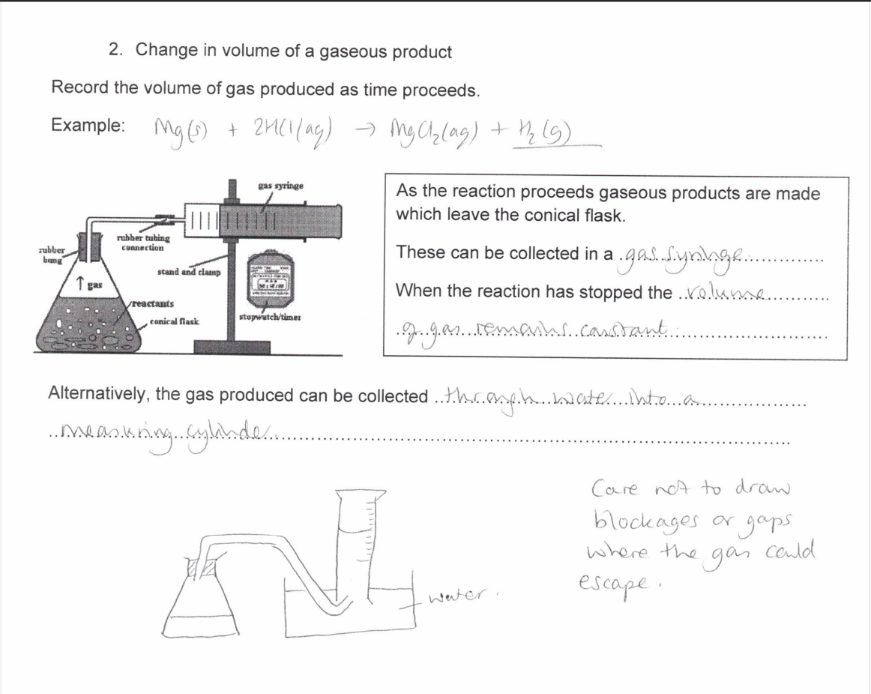
How do you measure the rate of a reaction by observing change in the amount of a solid product?

Why is rate = 1/time when measuring rate of reaction?
The change in concentration is always the same and so is simplified to 1
What is the relationship between rate and temperature? Explain your answer using collision theory and the Maxwell-Boltzmann distribution curve.
As temperature increases the rate of reaction increases
This is because the reactant particles have more energy and more have E ≥ Ea
Collisions are more frequent and successful collisions are more frequent
Therefore the rate increases
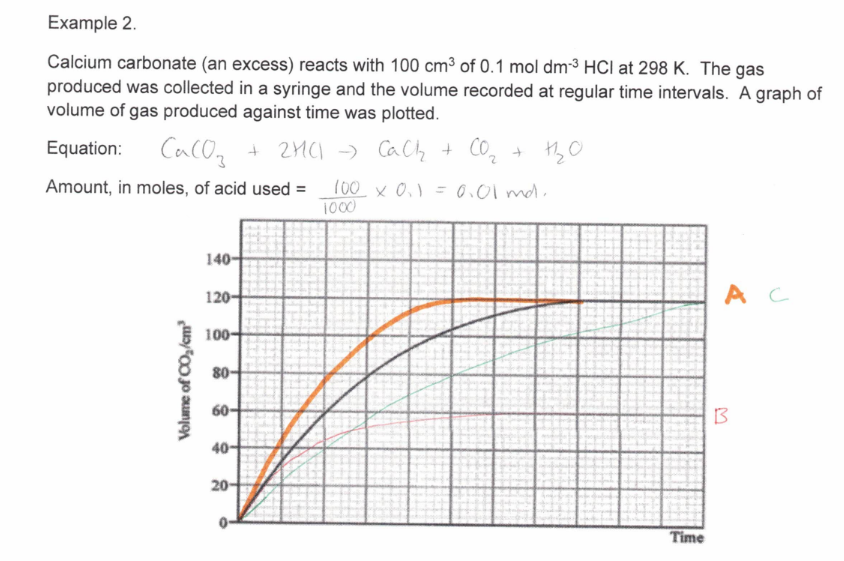
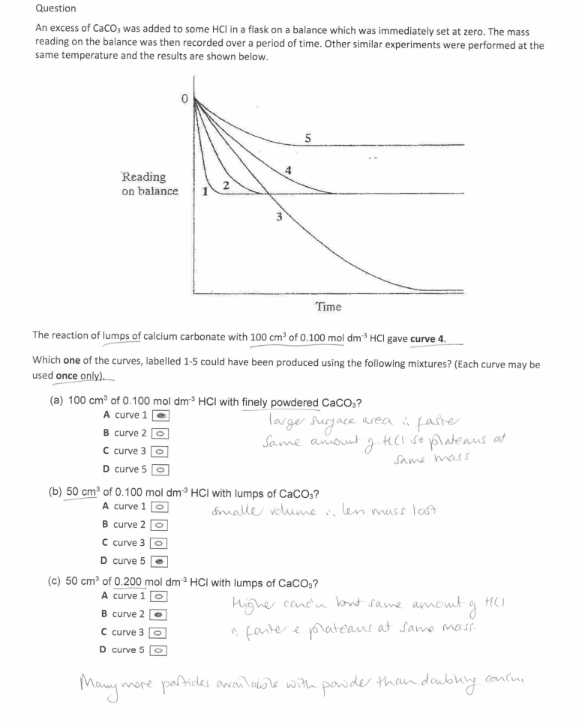
50cm3 0.200 mol dm-3 HCl with an excess of a CaCO3 at the same temperature as the original experiment
Study the image about maxwell boltzmann distribution graph.
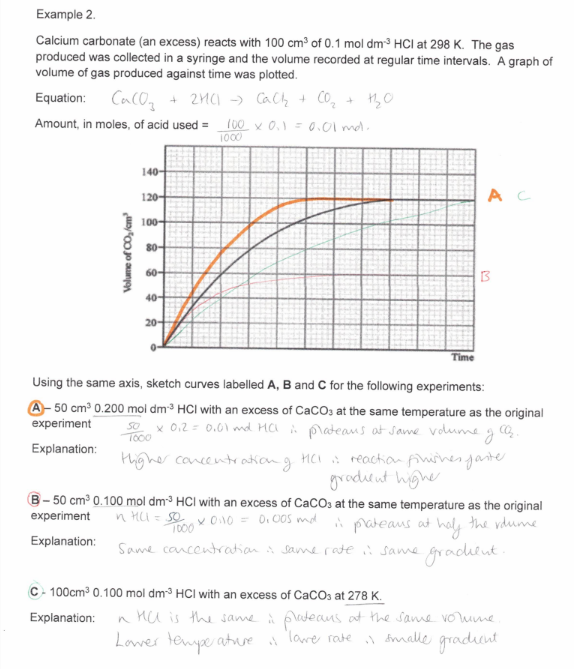
Study the image about the rate of reaction graph.
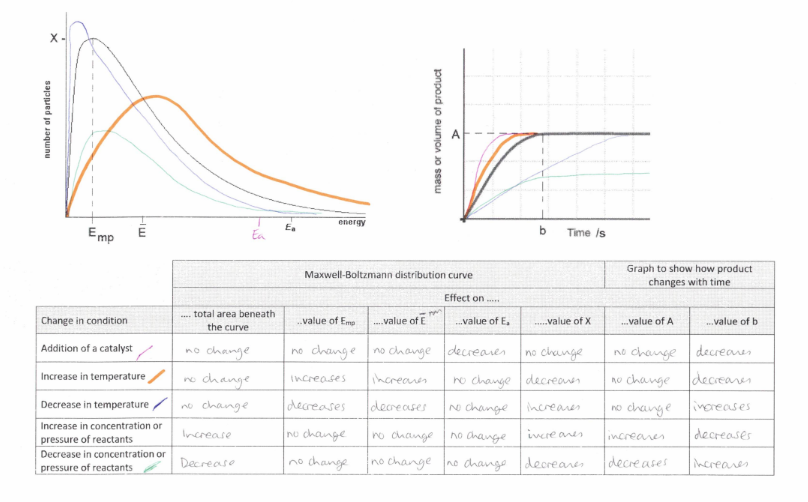
Study the image about the summary of how a maxwell-boltzmann distribution graph and a rate of reaction graph is affected by factors.

(exam) (6)


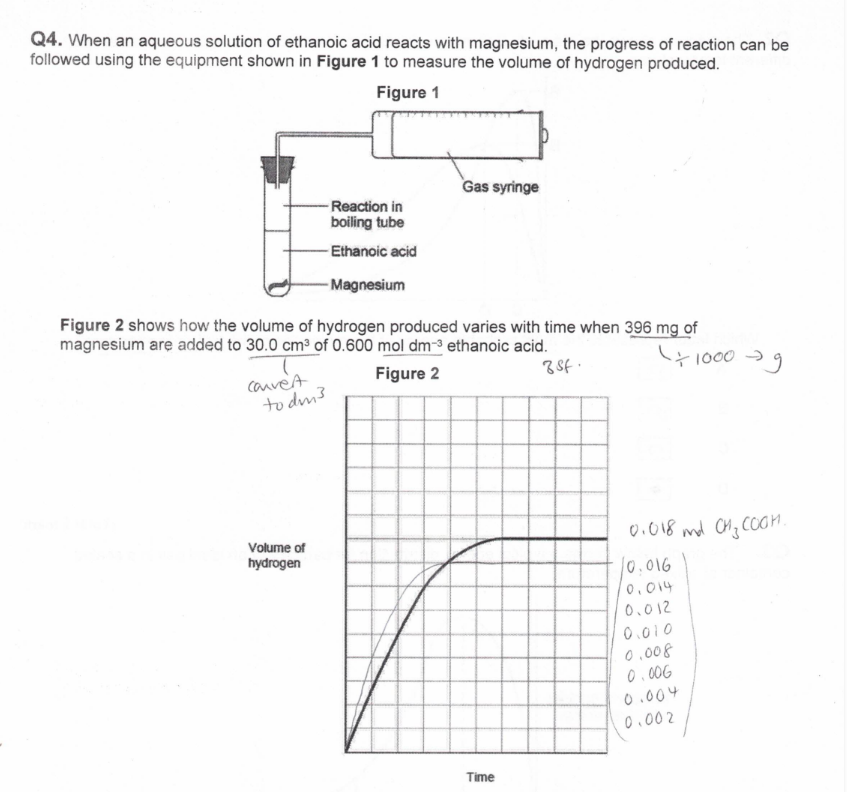
(exam)
What happens to EMP and mean energy at higher temperatures compared to low?
more molecules will have mean energy
Less molecules will have EMP
(exam) Explain the effect that loweing the temperature would have on the rate of reaction. (2)
Fewer particles have E ≥ Ea
Less frequent successful collisions
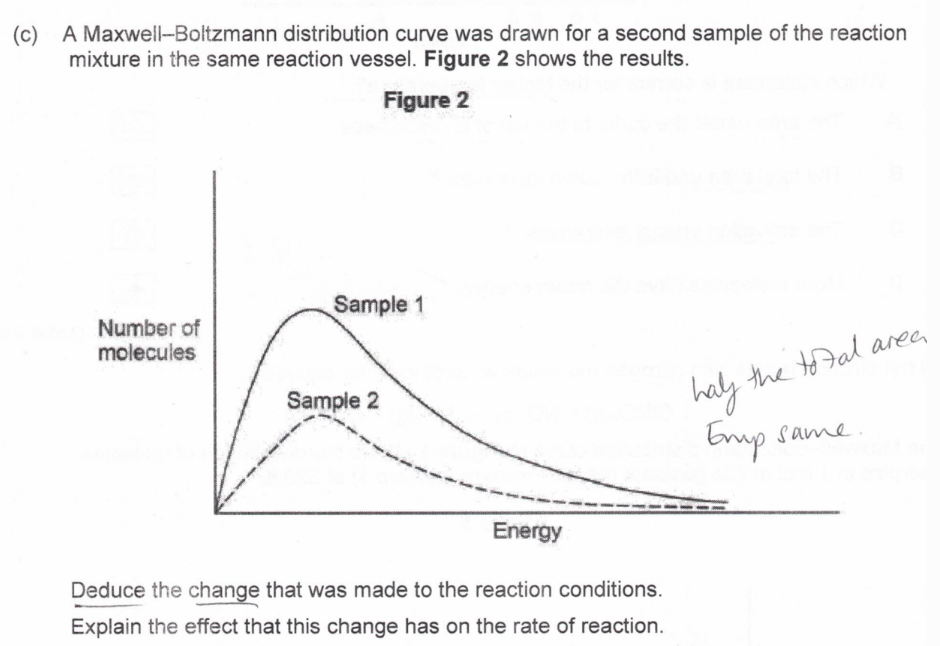
(exam) (3)
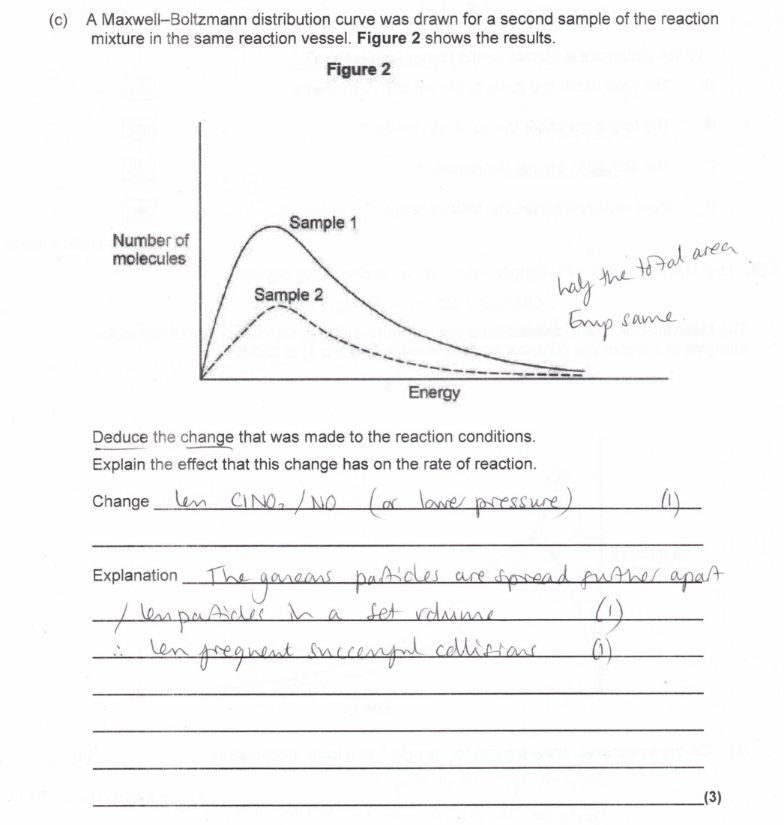
(exam) Explain, in general terms, how a catalyst increases the rate of a reaction (2)
A catalyst provides an alternative route
with a lower activation energy
(exam) Explain in terms of collision theory the effect of increasing the concentration of hydrogen peroxide on the rate of reaction. (2)
There are more H2O2 particles in a set volume
Therefore collisions and so successful collisions are more frequent
(exam) Explain why an increase in temperature increases the rate at which a gas decomposes. (2)
There are more molecules with E ≥ Ea
Successful collisions are more frequent