Biochem Ch. 13.4: Redox Reactions
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
Mechanical work can be driven by ___, as well as cellular processes.
electrons
What are three examples of cellular transducers
Mitochondria, flagellum, transport system
Chemical, mechanical, osmotic
What is pumped out as e- pass through the ETC?
Protons
What enzyme makes the majority of our ATP?
ATP synthase
__ organic compounds serve as fuels from which electrons can be stripped off during oxidation
Reduced
What’s the most oxidized compound?
CO2
Oxidation and reduction def
Oxidation = loss of e-, gain of O, loss of H
Reduction= Gain of e-, loss of O, gain of H
The thing being oxidized is the ___ agent and the thing being reduced is the __ agent
Reducing agent, oxidizing agent
Ferrous iron=… Ferric iron =…
Fe+2, Fe+3
Half reactions are always written as…
REDUCTIONS
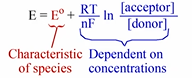
What is the Nernst equation?
Remember we use pH=7 for standard calculations
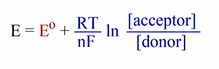
The half-rxn with a __ reduction potential will proceed as…
MORE +
Electrons move towards the ___ reduction potential
more positive
How were std red potentials determined?
By reference to the standard hydrogen electrode. Everything 1M BUT pH=7, std T and P
Equation relating reduction potential and free energy
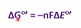
How to find the change in reduction potential when given two reduction potentials
Acceptor-donor

For a - change in G, we need a __ change in reduction potential
Positive- indicated spontaneous rxn under std conditions
How to find the actual reduction potential in a given system
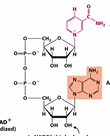
NAD and NADP are…
Nicotinamide (“pyridine”) nucleotides
FMN and FAD are…
Flavin nucleotides
NAD is used … and NADP is used…
For breaking things down, for building things up
NAD and NADP are redox ___
Cofactors
NAD and NADP move e- around in the form of a __
Hydride (two e-)
NAD and NADP are derived from…
The vitamin niacin
Usually, a hydride form an __ is transferred to NAD+ to give __
alcohol, NADH
On what part of NAD or NADP does the hydride come on and off?
Nicotinamide ring
In NADP, there is a phosphate group on…
the 2’ C
Draw NAD
Why do we need NAD and NADP?
Used for different things
Enzymes specifically recognize one or the other
Can keep them at different ratios in different areas of the cell
Both NAD and NADP act as… meaning they are __ bound
Substrates, loosely
NAD is usually found in the …
Mitochondria
NADP is mainly found in the…
Cytosol
NADH/NAD ratio is kept
LOW
NADPH/NADP ratio is kept
HIGH
NAD+ reduction involves 2 __ and 2 __
Protons and electrons though only one H is put onto the NAD
Reduction of NAD is ___, based on the ___. It can be put on as…
Stereospecific, enzyme. Pro-R or Pro-S
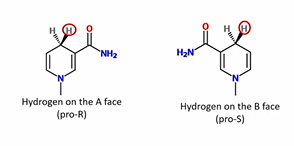
NADH absorbs at ___ nm
340 nm
Formation of NADH can be monitored by what method
UV-Spectrophotometry
What kind of enzyme typically uses NAD as a cofactor?
Dehydrogenases
Write out the two mechanisms by which NAD is either oxidized or reduced
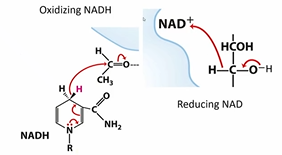
Why do we produce lactate in our muscle cells when we exercise?
We run out of oxygen locally, and we can’t do aerobic respiration, we only can do glycolysis. But now we aren’t regenerating NAD, and that’s bad
We turn pyruvate into lactate by adding two electrons
Rxn is catalyzed by lactate dehydrogenase (converts the carbonyl of pyruvate to an alcohol group
That’s why we do anaerobic respiration to do something with this lactate we produce and we get lactic acid buildup
What rxn does alcohol dehydrogenase catalyze
Ethanol to acetaldehyde with the reduction of NAD+ to NADH and vice versa
How can we measure the rate of the ethanol to acetaldehyde rxn
Absorbance at 340 nm to measure NADH production per unit og time
The reduction of acetaldehyde to ethanol is very __ under std conditions
Favorable!
So if reduction of acetaldehyde is very favorable, why aren’t we always drunk?
Acetaldehyde is rapidly removed from the body as CO2, so it skews equilibrium
Ratio of NADH to NAD+ is very LOW in the liver, so it changes the free energy change of the rxn
What is the 6th step in glycolysis and what cofactor does it use? What enzyme does it use?
Conversion of glyceraldehyde 3-phosphate to 1,3-bisphosphoglycerate (adds a phosphate onto the carbonyl C)
Uses NAD+ and reduces it to NADH
Glyceraldehyde 3-phosphate dehydrogenase (GAPDH)
The active site of GAPDH comes loaded with…
NAD+
What important AA reside is in the active site of GAPDH?
A cysteine, but it is ionized bc the surrounding environment dropped its pKa. It’s now a good nucleophile. It becomes covalently attached to GAPDH
Mech of GAPDH
Cys attacks carbonyl of GAPDH and becomes covalently linked
- tetrahedral int reforms a carbonyl (oxidation) and a hydrogen is pushed onto NAD+, forming NADH
NADH leaves active site and is replaced by another molecule of NAD+.
A free phosphate attacks the carbonyl of the covalent int, which generated 1,1-bisphosphoglycerate and the Cys residue
What is the disease pellagra caused by?
Niacin deficiency
Can we make niacin?
We need to take it in by our diet
What happens with pellagra?
Level of NADH is reduced bc we don’t have niacin to make the nicotinamide ring
All dehydrogenases are inhibited
Corn has very low__
Niacin, makes it easier to get pellagra
Now, certain foods are fortified with __ to reduce prevalence of pellagra
Niacin
Flavin cofactors allow __ e- transfers
Single (can do e- transfer stepwise)
Flavin cofactors are__ bound to proteins, unlike NAD and NADP
Tightly, sometimes covalently
Flavoproteins can __ flavin reduction potential
tune
FAD can be reduced to form…
Alkenes from alkanes
Flavins are derived from what vitamin
Vitamin B2
A flow of e-s occurs when two half cells have different __ for e-s. The difference in e- __ results in an __
Affinity. Difference in e- affinity results in an electromotive force (emf)
What are the 4 ways that e- can be transferred from a donor to an acceptor
Directly as e-
As protons
As hydrides
Direct combination with oxygen
T or F: the oxidation of glucose occurs all in one step
False! Happens step-wise to control the release of energy
Coenzymes function __and therefore are always __
Catalytically, recycled
Since FAD and FMN are bound so tightly to their corresponding flavoprotein, they are actually considered __ __
prosthetic groups