Salts
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
How is a salt formed
A salt is a substance formed when a metal ion or ammonium ion replaces all or some of replaceable H+ ions in an acid
What are the two parts of a salt
Metal: cation (comes from the base)
Non-metal or radical: anion (comes from the acid)
What are the different types of salts
Acid salts
Normal salts
What are acid salts
Acid salts still contain some replaceable H+ ions from the acid. These have a pH below 7
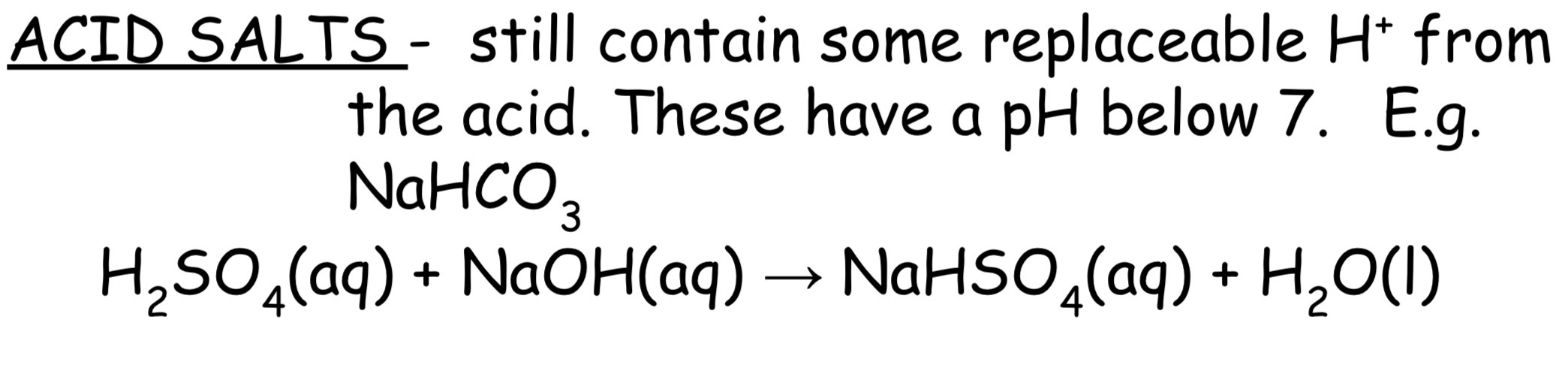
What are normal salts
Normal salts contain no replaceable H+ ions. These have a pH of 7 or neutral

What happens when dibasic and tribasic acids are partially neutralized
Dibasic and tribasic acids form acid salts if they are partially neutralized
Explain water of crystallization
This is when some salts contain water within their crystal structure which are called HYDRATED SALTS
This water can be driven off by gentle heating to give ANHYDROUS OR DRY SALTS
The salt can be rehydrated by adding water
Solubility of Salts

When heating salts what can they undergo
Thermal decomposition and thermal dissociation
What is Thermal Decomposition
When some salts are heated then break down and decompose. Some salts decompose faster than others due to the position of the metal in the reactivity series
Metals higher up in the reactivity series form salts form salts that are THERMAL STABLE, while those lower down form salts that decompose more easily when heated
General Thermal Decomposition

What is Thermal Dissociation
Thermal dissociation is when a compound decomposes on heating but the products rejoin on cooling

What is Double Decomposition
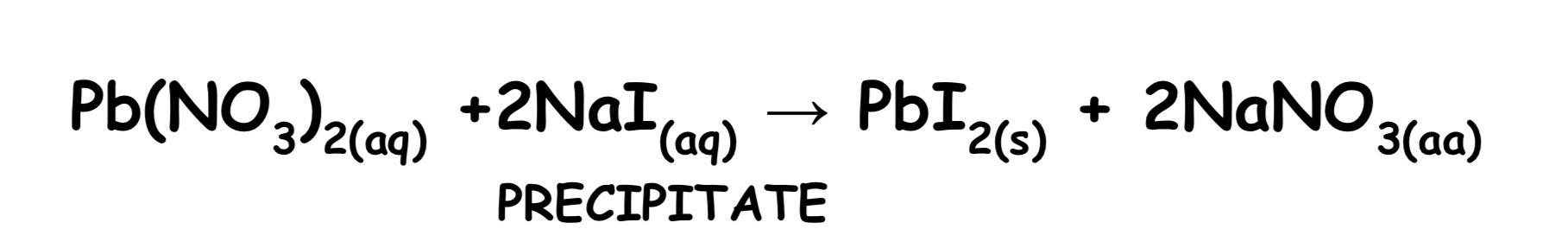
Double decomposition occurs when two salt solutions are added together and exchange ions
If an insoluble salt forms during double decomposition, a precipitate forms. These reaction is called PRECIPITATION REACTION