Chemistry Organic
1/370
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
371 Terms
define repeat unit
the specific arrangement of atoms in the polymer molecule that repeats over and over again
molecular formula of propene
C3H6
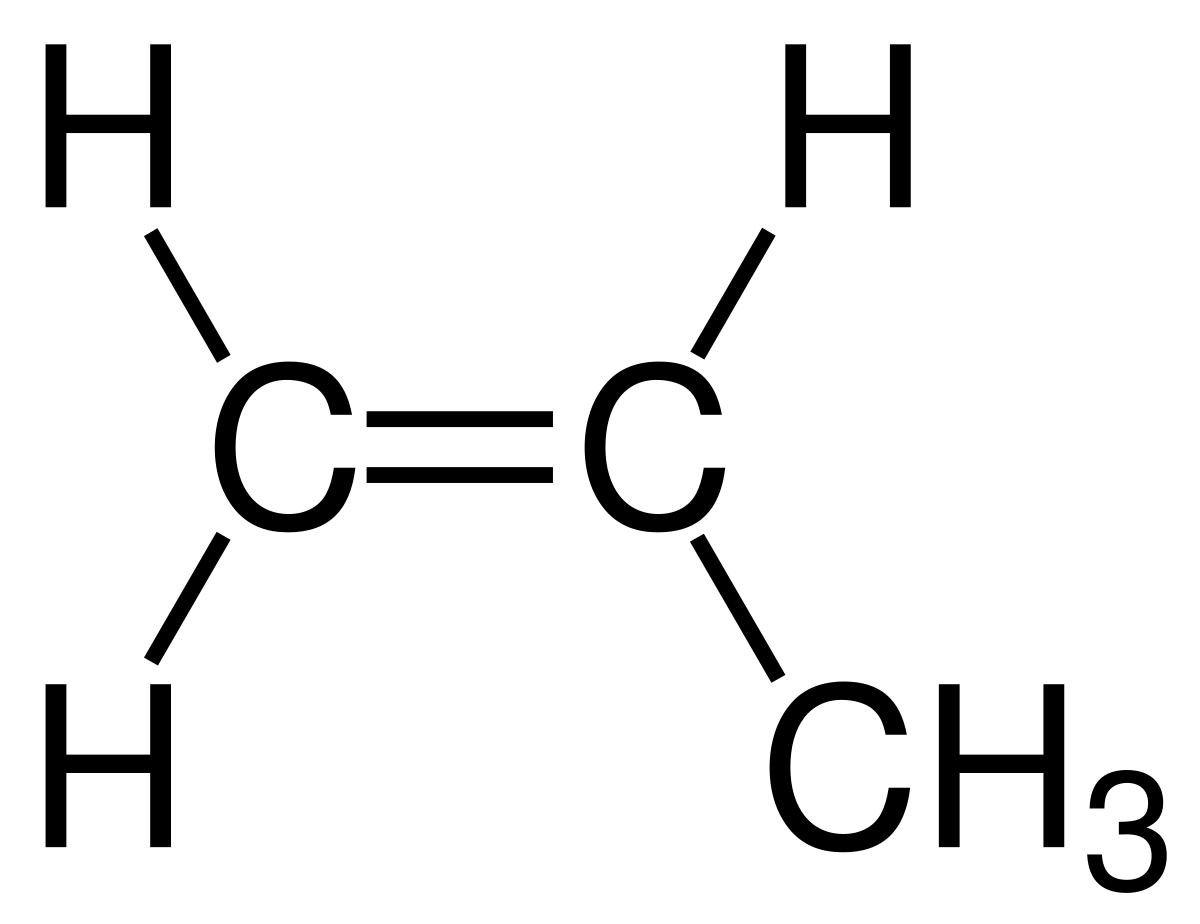
uses of propene
children’s toys, packing crates, guttering, uPVC windows, fibre for ropes
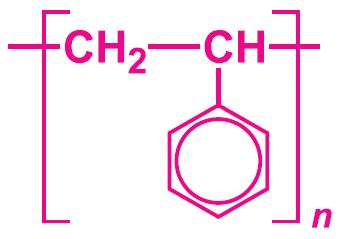
molecular formula of phenylethenestyrene
C8H8
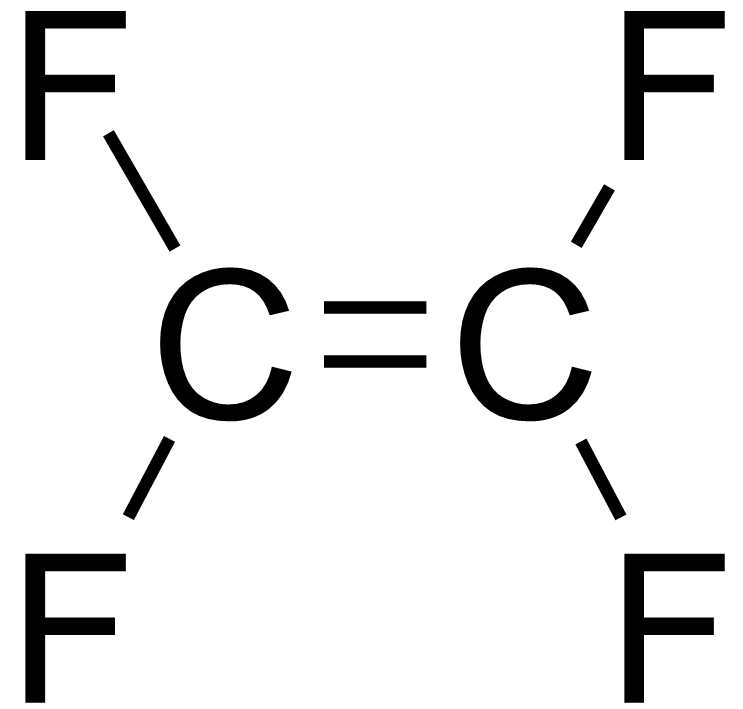
molecular formula of tetrafluoroethene
C2F4
name an environmental issue with the use of alkene based polymers
non-biodegradable
disadvantage of recycling polymers
discarded polymers have to be sorted by type
disadvantage of PVC recycling
PVC releases hydrogen chloride when burnt which is a corrosive gas
why are some polymers difficult to recycle
some polymers are derived from petroleum or natural gas which means it has a high stored energy value
steps of using waste polymers as fuel
1) waste polymers are incinerated
2) this produces heat
3) this heat generates steam
4) this steam drives a turbine which produces electricity
define feedstock recycling
the chemical and thermal processes that can reclaim monomers, gases, or oil from waste polymers
advantage of feedstock recycling
able to handle unsorted and unwashed polymers
define bioplastics
plastics produced from plant starch, cellulose, plant oils, and proteins that offer a renewable and sustainable alternative to oil based products
define compostable polymers
polymers based on poly(lactic acid) which degrade and leave no visible or toxic residues
define photodegradable polymer
polymer which degrades by absorbing UV
2bp 0lp
linear 180
3bp 0lp
trigonal planar 120
2bp 1lp
bent 119
4bp 0lp
tetrahedral 109.5
3bp 1lp
trigonal pyramidal 107
2bp 2lp
angular 104.5
5bp 0lp
trigonal bipyramdidal 90
4bp 1lp
seesaw 119, 89
why are alkenes more reactive than alkanes
1) alkenes have a pi bond 2) pi electron density is concentrated above and below the plane of the sigma bond 3) pi electrons are more exposed than the electrons in the sigma bond therefore the pi bond readily breaks
name 4 substances alkenes have addition reactions with
1) hydrogen in the presence of a nickel catalyst 2) halogens 3) hydrogen halides 4) steam in the presence of an acid catalyst
describe hydrogenation of alkenes
when an alkene is mixed with hydrogen and passed over a nickel catalyst at 423K an addition reaction takes place to form an alkane
describe halogenation of alkenes
alkenes react with the halogens chlorine or bromine at room temperature to form haloalkanes
describe a positive test for unsaturation
bromine water (orange) + alkene = haloalkane (colourless)
describe addition reactions of alkenes with hydrogen halides
alkenes react with gaseous hydrogen halides at room temperature to form multiple haloalkanes
how do addition reactions of alkenes with hydrogen halides occur when the alkene is a gas
reaction takes place when the two gases are mixed
how do addition reactions of alkenes with hydrogen halides take place when the alkene is a liquid
the hydrogen halide is bubbled through it
describe hydration reactions of alkenes
alkenes react with steam in the presence of a phosphoric acid catalyst H3PO4 to form alcohols
diagram of formation of a pi bond
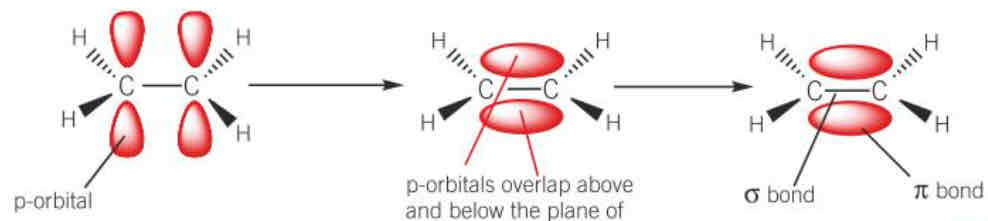
define homolytic fission
each of the bonded atoms takes one of the shared pair of electrons from the bond
define heterolytic fission
one of the bonded atoms takes both of the electrons from the bond
define reaction mechanism
how a reaction takes place
define addition
two reactants join together to form one product. eg. double bonds are broken to form single bonds
define substitution
an atom or group of atoms is replaced by a different atom or group of atoms
define elimination
the removal of a small molecule from a larger one so one reactant forms two products.
define nucleophile
an atom or compound with a negative or partial negative charge which is able to form a covalent bond by donating a lone pair of electrons
define electrophile
an atom or group of atoms that is attracted to a region of high electron density and can accept a pair of electrons
define free radical
an atom or group of atoms which have a single unpaired electron
functional group of aldehyde
-CHO
functional group of ketone
-CO-
why do aldehydes and ketones undergo nuceophilic addition
contain C=O bond
C=O is polar because oxygen is more electronegative than carbon
nucleophiles are attracted to and attack the delta positive C atom
aldehyde = primary alcohol
NaBH4 / H2O
reduction
ketone = secondary alcohol
NaBH4 / H2O
reduction
reduction of an aldehyde
Aldehyde + 2[H] → Primary alcohol
reduction of a ketone
Ketone + [H] → Secondary Alcohol
what chemicals are used in place of HCN
NaCN/ H2SO4
diagram of reaction of aldehyde with HCN
aldehyde/ ketone = hydroxynitrile
HCN (or NaCN/ H2SO4)
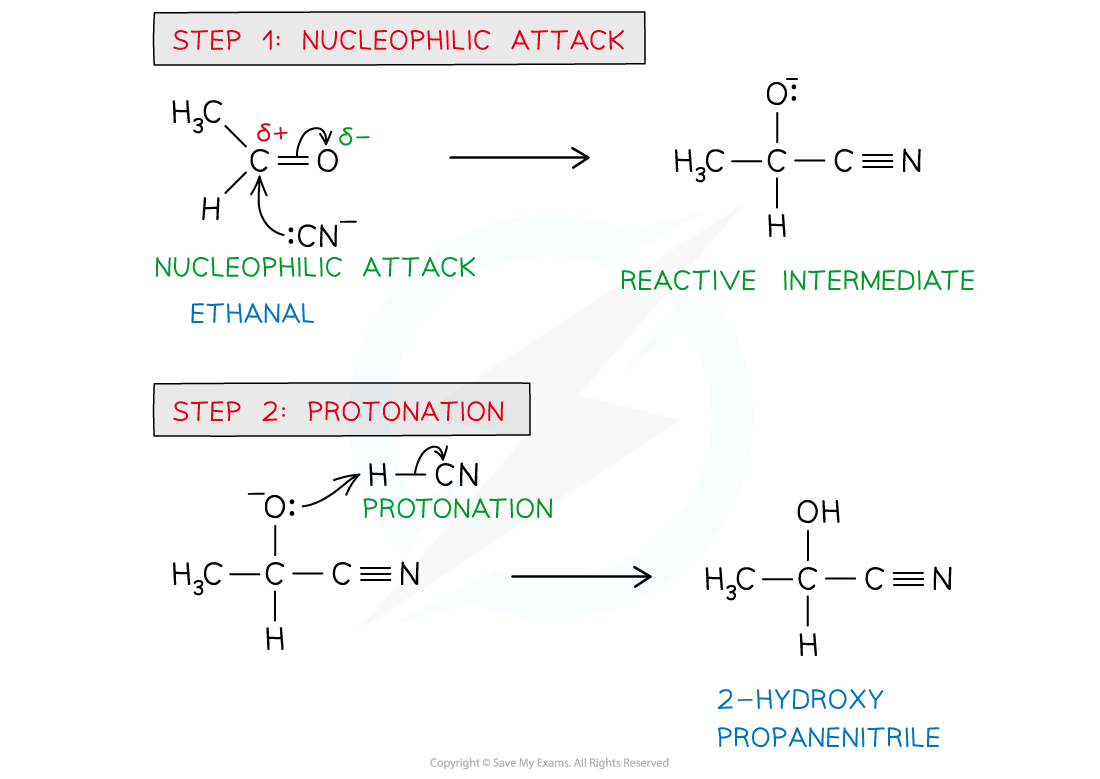
diagram of mechanism for the nucleophilic addition of a carbonyl compound
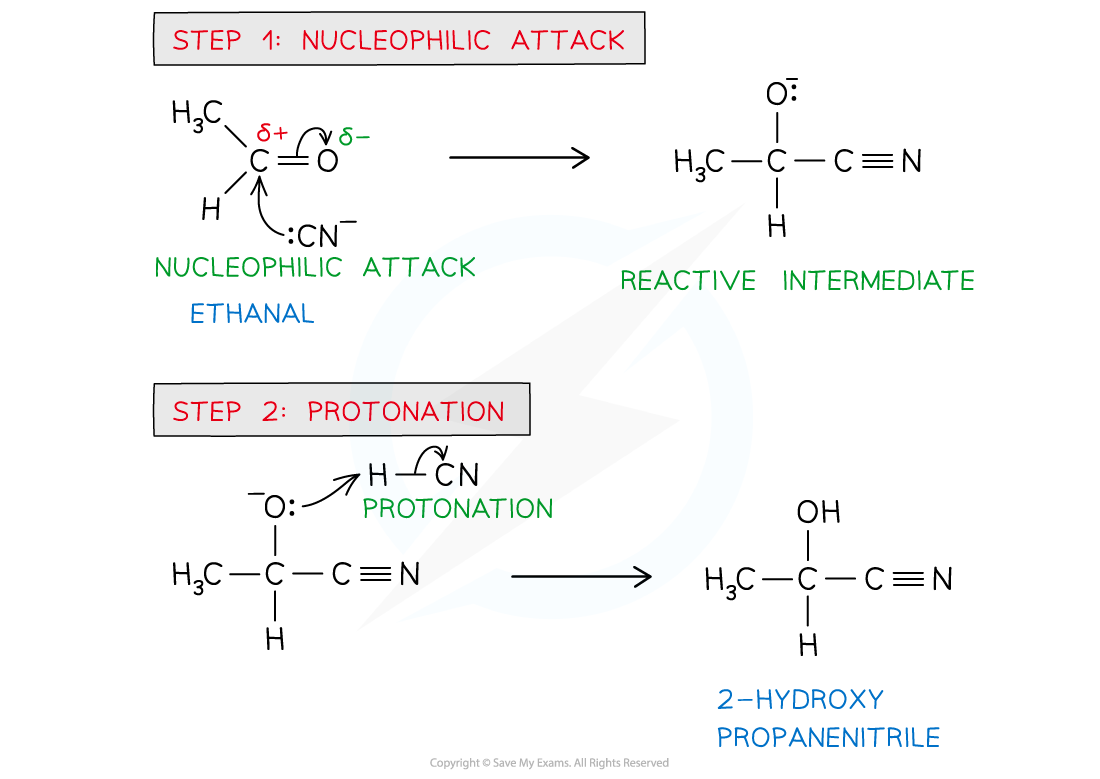
diagram of mechanism for the reaction of carbonyl compounds with NaCN/H+
steps of testing for the carbonyl group in aldehydes and ketones
1) add 5cm3 of a solution of 2,4-DNP in excess into test tube
2) pipette 3 drops of unknown compound and leave to stand
3) if no crystals form add a few drops of H2SO4
4) yellow/orange precipitate forms = C=O present
steps of testing for aldehyde
1) add 3cm3 AgNO3 (aq) into clean test tube
2) add NaOH (aq) into the AgNO3 until a brown precipitate of AgO is formed
3) add dilute ammonia solution until the brown precipitate just dissolves to form a clear colourless solution = Tollens’ reagent
4) pour 2cm3 of unknown solution into clean test tube
3) add 2cm3 Tollens’ reagent
3) leave to stand in beaker of warm water 50°C for 15 minutes. silver mirror = aldehyde present
why does a silver mirror form in the presence of an aldehyde
Tollens’ reagent contains Ag+ ions which act as an oxidising agent in the presence of ammonia
Ag+ ions are reduced to silver as the aldehyde is oxidised to a carboxylic acid
steps to melting point analysis of carbonyl compounds
1) filter impure yellow/orange solid to separate the solid precipitate from the solution
2) recrystalise the solid to produce a pure sample of crystals
3) measure and record the melting point of the purified 2,4-DNP
4) compare the mp to a database to identify the original compound
solubility of carboxylic acids
C=O AND O-H bonds are polar
form hydrogen bonds with water
carboxylic acids with up to 4 C atoms are soluble in water
as number oc C atoms increases, solubility decreases as the non-polar C chain has a greater effect on overall polarity
strength of carboxylic acids
weak acids
partially dissociates in water
HCOOH (aq) = H+(aq) + HCOO- (aq)
functional group of carboxylate
define a hydrogen bond
a type of permanent dipole-dipole interaction found between molecules containing an electronegative atom with a lone pair of electrons and a hydrogen atom attached to an electronegative atom
explain why ice is less dense than water
1) when water freezes hydrogen bonds hold water molecules apart in an open lattice structure 2) the water molecules in ice are further apart than in water
benefits of ice being less dense than water in lakes and ponds
the ice acts as an insulating layer and prevents the water from freezing solid
explain why water has a relatively high melting and boiling point
1) all molecules have London forces between them 2) hydrogen bonds are extra forces over and above the London forces 3) a larger quantity of energy is required to break hydrogen bonds in water
define hydrocarbon
a compound containing carbon and hydrogen only
define homologous series
a family of compounds with similar chemical properties whose successive members differ by the addition of a -CH2 group
define functional group
part of the organic molecule that is largely responsible for the molecule’s chemical properties
alcohol
hydroxy/ -ol
alkene
-ene
aldehyde -CHO
-al
ketone -C(CO)C-
-one
carboxylic acid -COOH
-oic acid
ester -COOC-
-oate
acyl chloride -COCl
-oyl chloride
amine -NH2
amino-/ -amine
nitrite -CN
-nitrite
define aliphatic
carbon atoms are joined to each other in unbranched or branched chains or non-aromatic rings
define alicyclic
carbon atoms are joined to each other in cyclic structures with or without branches
define aromatic
some or all of the carbon atoms are found in a benzene ring
define alkane
containing single carbon-to-carbon bonds
define alkene
containing at least one double carbon-to-carbon bond
define alkyne
containing at least one triple carbon-to-carbon bond
alkyl
-yl
alkane → haloalkane
halogen/ UV
radical substitution
alkene = alkane
H2
Ni catalyst
423K
addition
alkene = haloalkane
hydrogen halide
RTP
addition
alkene = alcohol
H2O (g)/ H3PO4
addition
alcohol = alkene
conc H2SO4
elimination
haloalkane = alcohol
NaOH (aq)
reflux
nucleophilic substitution
alcohol = haloalkane
sodium halide and H2SO4
reflux
nucleophilic substitution
primary alcohol = carboxylic acid
K2Cr2O7/ H2SO4
reflux
oxidation
primary alcohol = aldehyde
K2Cr2O7/ H2SO4
distil
oxidation
secondary alcohol = ketone
K2Cr2O7/ H2SO4
reflux
Oxidation state of Group 1 metals
Always +1