Rate equations yr13
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
Rate of reaction units
Mol dm-3 s-1
Define order of reaction
The power to which the concentration of x affects rate
What does order of reaction tell you?
The power to which the concentration of x effects rate
What does the rate equation show and what is it?
Mathematical link between the concentration of each reactant and the reaction rate
Rate = K [A]x [B]y
What is 1st order of reaction
Rate is directly proportional to the concentration of x
E.g. If concentration is doubled (X 2)→ rate is doubled (X 21 )
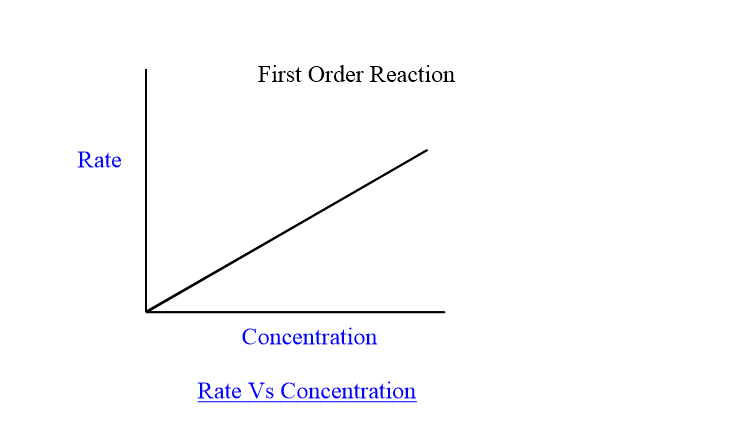
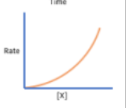
What is 2nd order of reaction?
Rate is proportional to the concentration of X squared
E.g If concentration is doubled ( X 2) → Rate is increased by a factor of 4 ( X 22 )
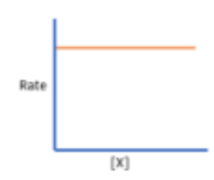
What is 0 order?
Rate does not change despite change in concentration
E.g. if concentration was doubled (X 2) → rate increases by 20 (Which is x 1 so no change)
Anything raised to the power of 0 …
Is not in the rate equation
If a rate/ concentration graph is unclear, how can you determine the order?
Use the equation
Log rate = m log[A] + log k
You work out the log of rate and the log of concentration of the reactant and log of k
This gives you a straight line and gradient m gives you the order
Define overall order
Sum of all the powers to which the reactants are raised to
Why is knowing the overall order helpful?
Helps determine the rate determining step for a reaction
Helps determine the mechanism for a reaction
Gives an understanding of how the rate of reaction would respond to changes in concentration
What is the role of catalysts and involvement in rate equation?
Increase the rate of reaction but are not used up in the reaction
Appear in the rate equation NOT in the chemical equation
What happens when a reactant is in excess?
If the concentration of a reactant is much larger than another, its concentration will only change by a very small proportion
Thus the concentration of excess reactant is constant
Meaning the concentration of the chemical (in excess) is unlikely to affect the rate if reaction
Excess reactant treated as a zero order
How can you make a concentration time graph to determine the order of a reactant?
Make the reaction mixture and start timer
Remove a sample at regular time intervals, to determine leftover reactant concentration
Quench the reaction by neutralising it or placing it in ice
Analyse the concentration of leftover reactant via titration
What does the gradient of a concentration time graph tell you?
Rate of reaction
Explain a concentration time graph depicting 0 order
Depicts a constant decline of the concentration of the reactant
Gradient is unchanged, even though reactant concentration decreases
Therefore rate is constant
Shows 0 order as reactant concentration decreases at a constant rate

Explain concentration time graph for first order
Sloping curve with a constant half life→ time taken for concentration of reactant to half
Order can be found out by using tangents
Gradient = rate
Rate is directly proportional to concentration
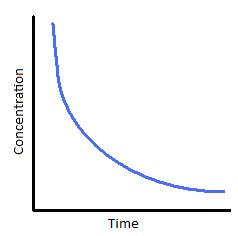
Explain concentration time graph for second order
Curve declines steeply at first then levels out→ increasing half life ( time increases for concentration to halve)
Gradient = Rate
What does the rate constant(K) demonstrate?
Links concentrations to the rate of reaction
Larger the value of K…
The faster the rate of reaction
What does the value of K vary with ?
Temperature
Larger the temperature, the larger the value of K (Rate constant)
Units of rate constant (K)
Depends
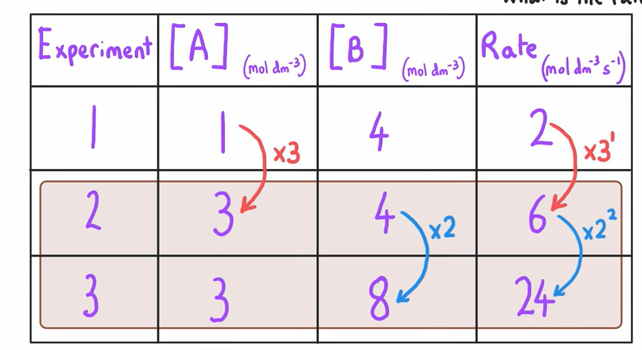
When your trying to deduce order of a reactant from a table of experiments what do you do ?
Find pairs of experiments where only 1 of the reactants has had a change in concentration.
What is a mechanism and how does it link to the rate determining step?
A series of steps through which a reaction progresses often forming intermediate compounds
Each step can have a different rate of reaction
Slowest step is called the RDS
And controls the overall rate of reaction
(So if you want to speed up a reaction focus on RDS)
Chemicals in the RDS…
Will be in the rate equation
What should you know about intermediate products and the rate equation?
Intermediate products do not appear in the rate equation
And instead the reactants that produced the intermediate are included in the rate equation
(Check notes for additional guidance)
The rate equation demonstrates the link between concentration of reactants and reaction rate so any other factor affecting rate must be affecting…..
The value of K
What is the Arrhenius equation and units ?
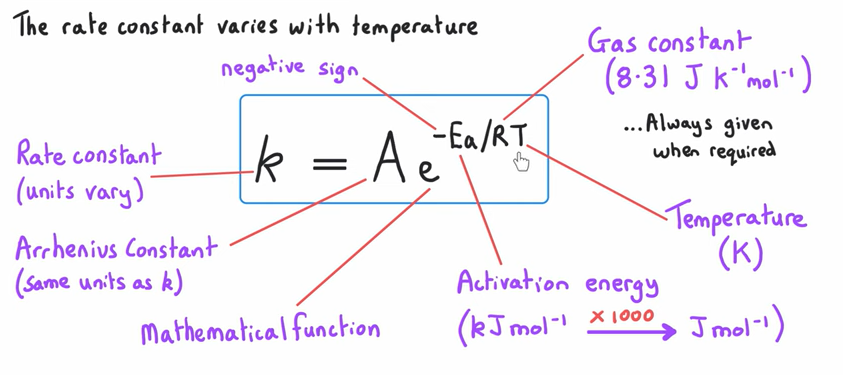
What does the Arrhenius equation depict?
Shows that the rate constant varies with temperature
How do you rearrange the Arrhenius equation to workout Ea?
Ea = (In A- InK) x RT
How else can you rearrange the Arrhenius equation, where it mimics a straight line equation?
May have to use this equation to plot a graph→ To find out Ea
Ink on y axis and 1/T on x
In K has no units and 1/T → k-1

What practical measures the rate of reaction by an initial rate method
Iodine clock
What happens in iodine clock practical
Hyrdorgen peroxide reacts with iodide to form iodine
Thiosulfate ion immediately reacts with iodine
When iodine has reacted with all the limited amount of thiousfate ions present- excess iodine remains in the solution
Reaction with the starch then forms a dark blue solution