3.3 & 3.4- Elements of the p and d blocks
1/128
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
129 Terms
What is the inert pair effect?
Electrons in the p subshell removed first, then s subshell electrons removed before d subshell electrons
What does the inert pair effect do as you go down the group?
Becomes more significant
Why does boron form compounds with the +3 oxidation state alone, but thallium compounds are more stable with the +1 oxidation state?
+1 occurs due to the inert pair of s-electrons
Inert pair effect becomes more significant as you go down the group
What can p block elements do?
Expand their octet into the d subshell, to occupy more electrons in their outer shell
Do all p block elements require a full outer shell?
No eg. BF3
What can amphoteric substances do?
React with both acids and bases as they can both donate and gain electrons
Need more on amphoteric
Why are group 3 elements called such?
Because they have 3 electrons in their outer shell
What does this mean?
They are only able to form 3 covalent bonds, giving 6 electrons in the outer shell of the Group 3 atom
What does this cause?
Electron deficiency
What does this mean for aluminium and boron?
They are able to form co-ordinate bonds with a lone pair of electrons from another atom to form a stable octet.
Therefore, what are they?
Electron acceptors and can form dimers
What can the boron in BCl3 be described as?
Electron deficient
What is meant by the term electron deficient?
Outer shell of electrons is not full
When BCl3 is mixed with ammonia a new species is formed. Identify the new species and explain how it forms.
NH3.BCl3- co-ordinate bond forms between lone pair on N and electron deficient BCl3
Bond angles:
120
107
104.5
180
90
109.5
Trigonal planar
Pyramidal
Non-linear
Linear
Octahedral
Tetrahedral
What is a co-ordinate bond represented as?
An arrow from donating to receiving
What does isoelectronic mean?
The same electronic structure
Why is boron nitride isoelectronic with carbon?
Because both have a total of 12 electrons on the two atoms
Compare hexagonal boron nitride to graphite
1) Atoms in adjacent layers of h-BN are aligned. In graphite, thy are not
2) Pi electrons are more localised in h-BN, as each N atom in h-BN has a lone pair of electrons. Makes h-BN an insulator. Relatively evenly delocalised in graphite
3) Electronegativity difference bewteen B and N which gives polar B-N bonds. No electronegativity difference between the C atoms in graphite.
Why is graphite a good conductor of electricity but h-BN isn't?
Delocalised electrons can move through the layers in graphite, carrying a charge. In h-BN however, the pi electrons are more localised as each N atom has a lone pair
What is h-BN used in?
Electronics for semi-conductors, ceramics and microwave windows
What is the structure of cubic boron nitride?
Same as diamond- giant tetrahedral
Same properties as diamond- strong, hard, high melting point
What does volatility do as you go down group 7?
Decreases as larger surface area of contact and more electrons as larger atoms
Why does reactivity decrease going down group seven?
Larger atoms so greater distance from nucleus to outer electron, so weaker electrostatic forces of attraction between nucleus and outer electron, greater electron shielding, so harder to gain an electron
Are halogens oxidising or reducing agents?
Oxidising
What happens to their oxidising power as you go down the group?
Their oxidising power decreases as harder to gain a new electron
So what happens to their standard electrode potential as you go down the group?
Their standard electrode potential gets weaker
Which out of Cl and I will have the stronger oxidising power?
Cl as easier for it to gain electrons as further up group
Can chlorine displace bromide ions?
Yes, because chlorine has a more positive standard electrode potential than bromine so chlorine can oxidise bromide ions to bromine molecules
Which ions are oxidised by sulfuric acid?
Chloride, bromide and iodide
What happens if you react H2SO4 with NaCl?
Steamy fumes of HCl are produced but cannot be oxidised by sulfuric acid
H2SO4 has a less positive standard electrode potential than Cl2
What happens if you react H2SO4 with NaBr?
Steamy fumes of HBr
Can be oxidised by H2SO4 so redox reaction takes place and bubbles of colourless gas and orange fumes of bromine are produced
What happens if you react H2SO4 with NaI?
Steamy fumes of HI
Can be oxidised
Redox
Purple I2 fumes- can sublime into a black/brown solid
As iodide is an even stronger reducing agent, yellow solid of sulfur can be produced through further reduction
Disproportionation reactions of chlorine:
What is disproportionation?
When the same species is both oxidised and reduced
Chlorine reaction with cold dilute sodium hydroxide?
Cl2 + 2NaOH -> NaCl + NaOCl + H2O
With warm, concentrated?
3Cl2 + 6NaOH -> 5NaCl + NaClO3 + 3H2O
What are the formulas for:
Chlorate (I) ion
Chlorate (III) ion
Chlorate (V) ion
ClO-
ClO2- (as oxidation number of chlorine atom is +3)
ClO3-
What is the most stable oxide of Carbon and why?
CO2
Inert pair effect becomes more significant as you go down the group and carbon is at top of the group
What is the only stable compound to contain carbon in the +2 oxidation state?
CO
What does this mean it can act as?
A reducing agent as it can easily become oxidised from +2 to +4
What is it therefore used for?
Extracting metals from their oxides
eg
Fe2O3 + 3CO -> 2Fe + 3CO2
What can this method only be used for?
The oxides of less reactive metals
Why?
The oxides of more reactive metals are too stable and so will not react
What is the most stable oxide of lead?
PbO
What are all lead (IV) compounds and why?
Oxidising agents as they are easily reduced from +4 to +2 oxidation state
eg
PbO2 + 4HCl -> PbCl2 + Cl2 + 2H2O
What are metal oxides?
Basic
Non-metal oxides?
Acidic
Eg?
CO2 (acidic oxide)
How does CO2 act as an acidic oxide?
It is soluble in water to give carbonic acid
What will CO2 do?
React with alkalis to form a salt
eg
CO2 + NaOH -> NaHCO3
What can some metals form and what does this mean?
Amphoteric oxides- shows both acidic and basic properties
Eg?
PbO (amphoteric oxide)
Give some examples of when PbO acts as an amphoteric oxide?
PbO + 2HNO3 -> Pb(NO3)2 + H2O
PbO + 2NaOH + H2O -> Na2[Pb(OH)4]
Why does CCl4 not react with water but SiCl4 does?
Carbon does not have any d-orbitals in its valence shell, so it is unable to expand its octet to occupy more electrons.
Silicon possesses d-orbitals that allow the water molecules to form co-ordinate bonds with.
What is the equation for this reaction?
SiCl4 + 2H2O -> SiO2 + 4HCl
What type of compounds are Pb(II) compounds?
Ionic
What else are they?
Insoluble in water
What are the two common compounds that dissolve readily in water?
Lead nitrate Pb(NO3)2
Lead ethanoate Pb(CH3COO)2
What happens when you add aqueous sodium hydroxide to a Pb2+ compound?
An initial white precipitate of Pb(OH)2 is formed
Write an equation for this
Pb2+(aq) + OH-(aq) -> Pb(OH)2 (s)
What happens when you react excess aqueous sodium hydroxide with Pb2+?
The white precipitate redissolves in excess OH- to form the tetrahydroxoplumbate (II) ion
Equation?
Pb(OH)2 + 2OH- -> [Pb(OH)4]2- (aq)
HCl with Pb2+ compounds?
A dense white precipitate of lead (II) chloride, PbCl2, is formed
Write an equation
Pb2+ + 2Cl- -> PbCl2
Potassium iodide with Pb2+ (really important to remember)
Bright yellow precipitate of lead (II) iodide is formed
Equation
Pb2+ + 2I- -> PbI2
What is the chlorate (I) ion?
An oxidising agent
Write an equation to show this
ClO- + 2H+ + 2e- -> Cl- + H2O
What else is an oxidising agent?
Elemental chlorine
Equation please
Cl2 + 2e- -> 2Cl-
What is the oxidising power of chlorine and chlorate?
The basis of their use in bleaches
Why?
Because bleaching is an oxidation reaction
What is the oxidised form of the coloured material?
Colourless
What does the oxidising ability of ClO- give it the ability to do?
Kill bacteria as the microbe cells are oxidised.
What is this the basis of?
The chlorination of water supplies to disinfect them
What is a transition metal?
An element that possesses a partially filled d-subshell as an atom or in its stable ions
Why is Zinc not considered a transition metal?
Because it does not possess a partially filled d-subshell as an atom or in its stable Zn2+ ion form
What are the characteristics of transition metals?
-Variable oxidation states
-Complex ion formation
-Formation of coloured ions
-Catalytic activity
Why can transition metals have different oxidation states?
Because the energies of the 4s and 3d orbitals are very similar, so the energy required to remove any of these electrons is similar
What does a transition metal complex consist of?
A central metal ion surrounded by a number of molecules or ions called ligands.
How are the ligands attached to the central ion?
By coordinate bonds
Which atom supplies the two electrons?
The ligand
How can the coordinate bond form?
The transition metal ions have many orbitals available for bonding, so an orbital from the ligand containing a lone pair of electrons can overlap with an empty orbital on the transition metal ion to form a coordinate bond
How are ligands classified?
By the number of coordinate bonds that they can form in complexes
What is a monodentate ligand?
A ligand that is able to donate one pair of electrons to a central metal ion
What is a bidentate ligand?
Molecule that donates two electron lone pairs to a metal ion via two SEPARATE coordinate bonds .
Typically, what are complex ions formed from?
Octahedral- 6 ligands arranged around central metal ion
Tetrahedral- 4 ligands arranged around central metal ion
When drawing the structure of complexes, what must they be represented in?
3D
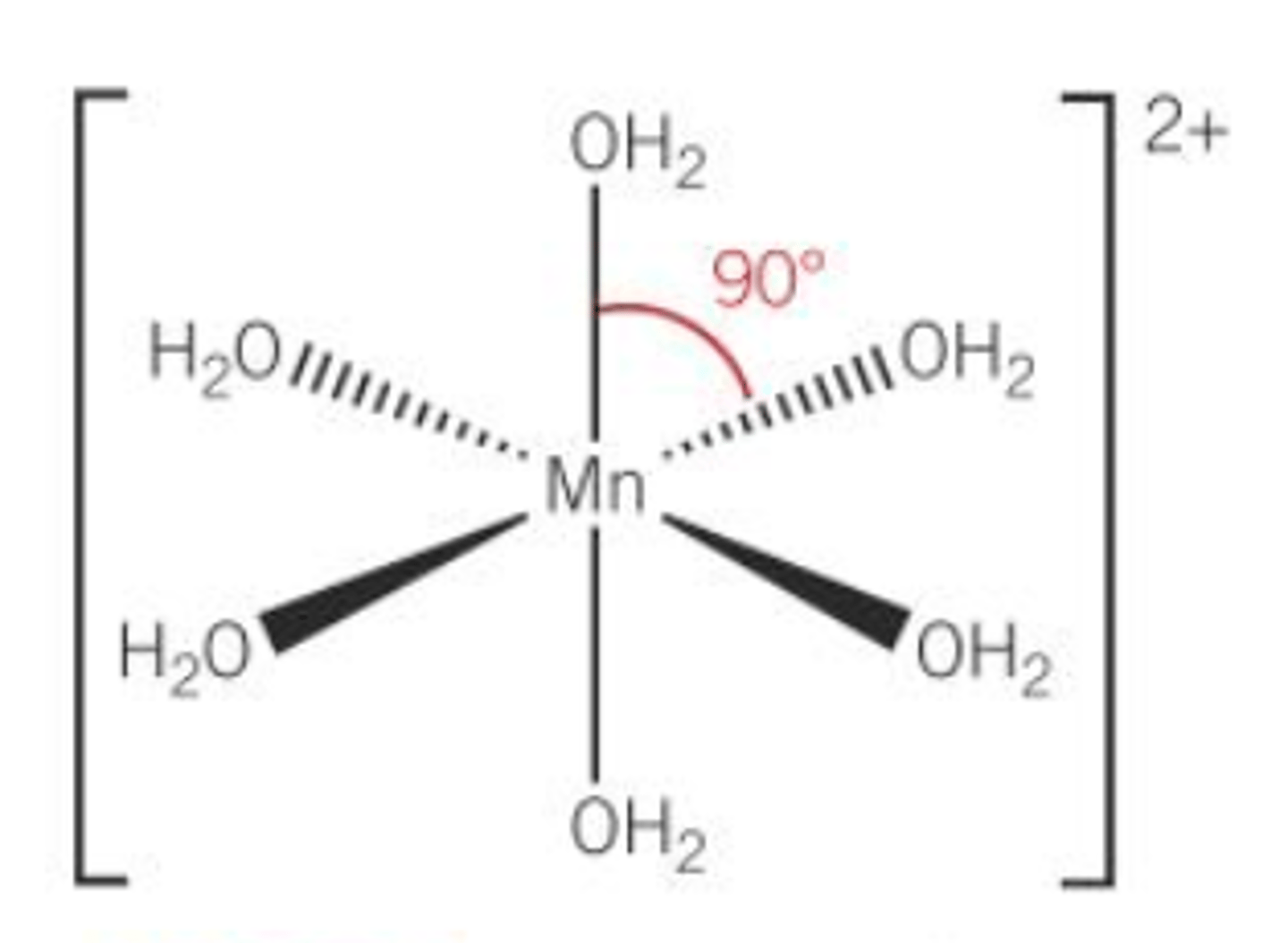
Define a ligand exchange reaction
When one ligand in a complex ion is replaced by a different one
If ammonia solution is added to a solution containing [Cu(H2O)6]2+, ammonia molecules do what?
Replace 4 of the water molecules to form a new complex, [Cu(NH3)4(H2O)2]2+, leaving 4 water molecules
If more ammonia is added, what will happen to the equilibrium?
It will shift to the right, forming more [Cu(NH3)4(H2O)2]2+ which gives a royal blue colour
When are transition metals only coloured?
When they form complexes
Without ligands, what are all d orbitals in a transition metal?
degenerate
define degenerate
orbitals with the same energy
What happens when ligands approach the metal ion?
They cause the energy of three of the d orbitals to become different to the other two
What does this do to the d orbitals?
Splits them into two sets: three of lower energy and two of higher energy in an octahedral complex, three of higher energy and two of lower in tetrahedral
What is this process called?
d-orbital splitting
When can an electron in a 3d orbital move from a lower to higher energy set?
When it can gain sufficient energy