Microorganisms, Infection, and Immunity 2 - MII2, University of Edinburgh
1/575
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
576 Terms
What is the big three
Malaria, HIV/AIDS/, Tuberculosis
What is intra-host adaptation
Immune system localises the the microorganism to one particular site in the body. Anywhere else triggers a response
What is inter-host adaptation
microorganisms that cause species barriers and can be leathal to different species
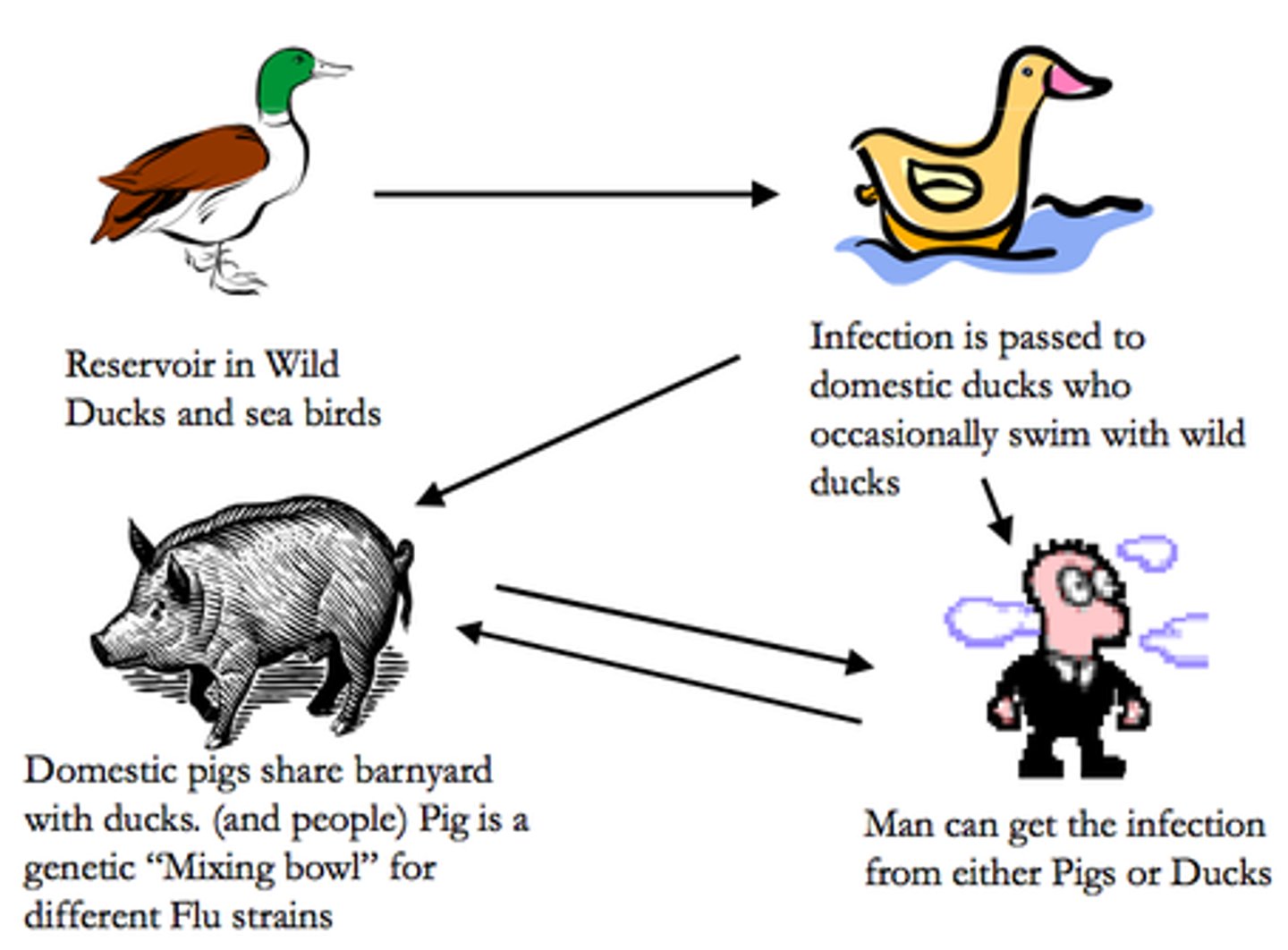
What is zoonosis?
humans to animals
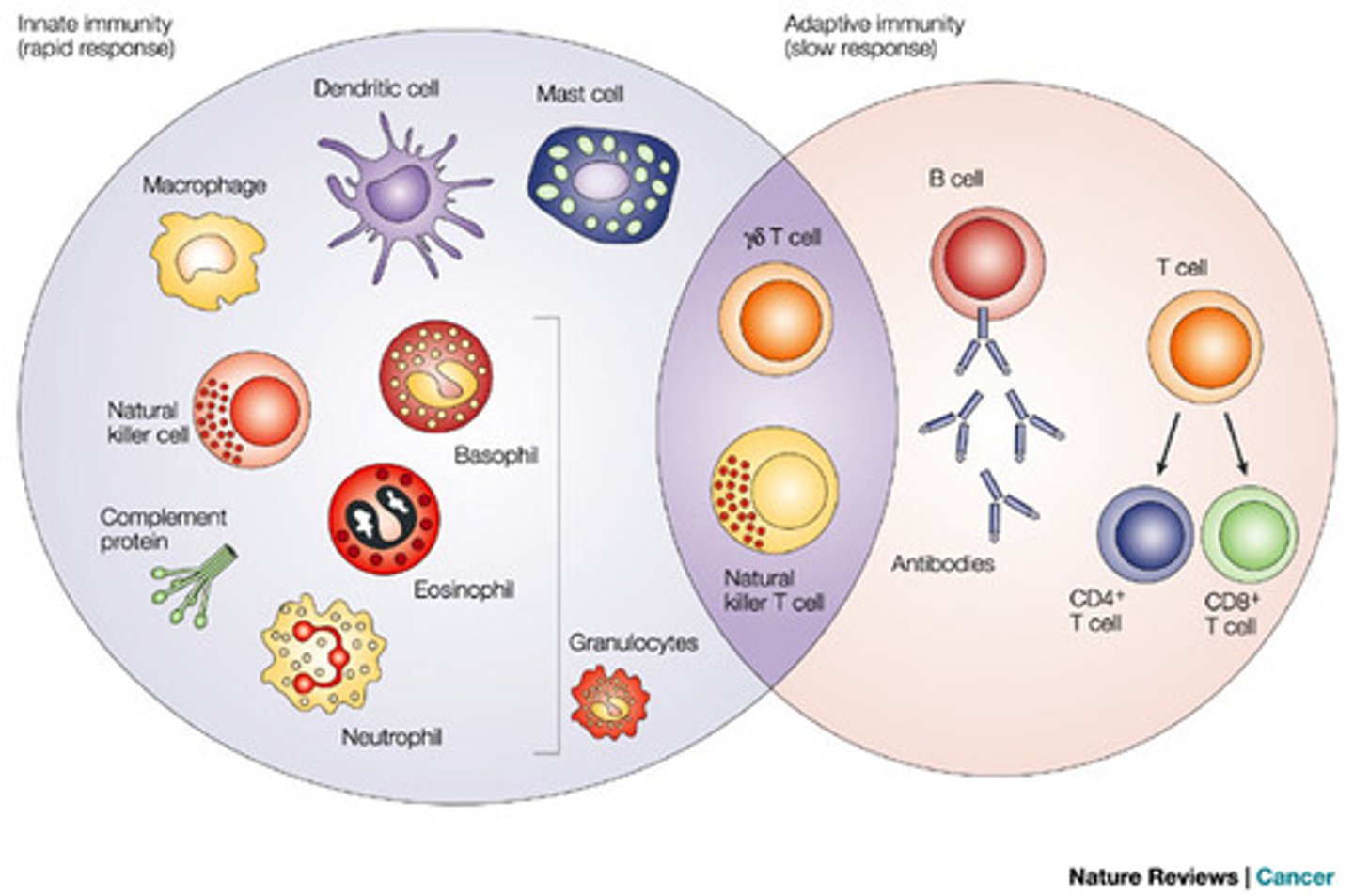
What is innate immunity?
Nonspecific protection against foreign substances indiscriminantly.
All cells but lymphocytes.
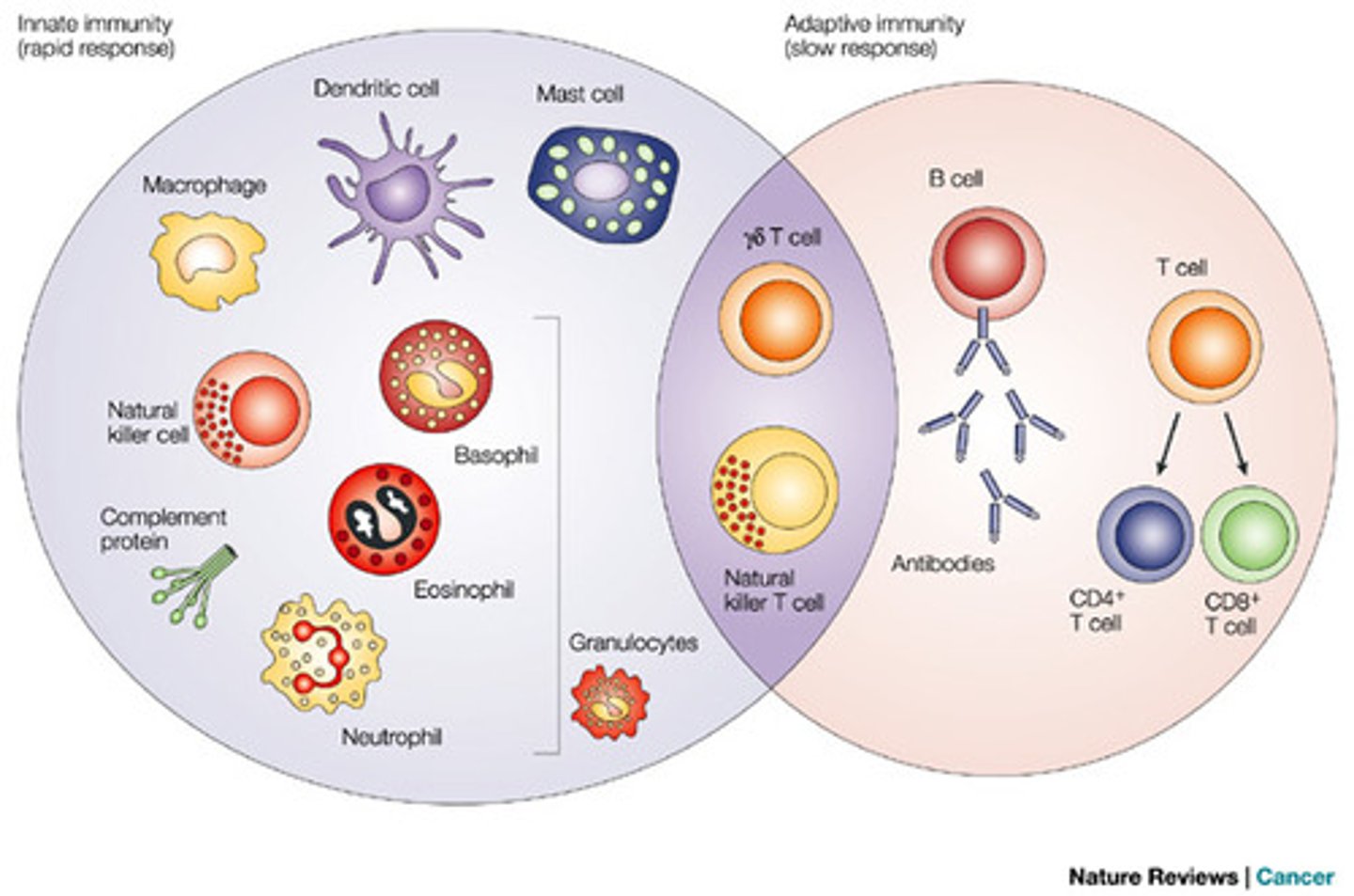
What is adaptive immunity?
the body's ability to recognize and defend itself against distinct invaders and their products
What is CFR
case fatality rate
What are live attenuated vaccines?
They are weakened but still active organisms
What are dead/subunit vaccines?
vaccines using a part of a microorganism. Not as harsh as live attenuated vaccines
What are viral vectors?
where a virus is carried by an organism, and it can go from that organism to another host
Do all microorganisms cause diseases ?
no
Do microorganisms evolve and adapt to hosts?
yes
When pathogens switch hosts. the outcome is _______ (unpredictable/predictable)
Unpredictable
CFRs can influence perception of risk versus hazard (T/F)
T
- CFRs are rarely uniform across all strata of the population (T/F)
T
- CFRs depend on methods used to record death and diagnosis. Therefore they are estimates (T/F)
T
Can the roles of the immune system be not beneficial to us?
Yeah. As seen in graft rejection, transplants, metabolic diseases
What are the 4 means of barrier defence mechanisms?
Skin
Gut
Lungs
Eyes/nose/oral cavities
What are cathelicidins?
a group of antimicrobial peptides that are part of the innate immune system in many vertebrates.
Where are cathelicidins found?
Skin, Gut, Lungs
Does innate immunity have broad specificity?
Yes
Does innate immunity recognise danger signals?
yes
Is innate immunity ready to go?
Yes
Does innate immunity give time for the adaptive immune response to develop?
yes
If there is no innate immunity, what is seen?
No response to infection
Does innate immunity recruit immune cells to infection sites
yes
What are the 4 innate killing mechanisms?
Phagocytosis
Secretion of cytotoxic granules
Killing of infected cells
Complement proteins
Is Adaptive immunity highly specific?
yes
Does adaptive immunity take time to develop>
yes
Does adaptive immunity remember pathogens?
yes
What type of cells are seen in adaptive immunity?
T and B cells
What is the function of Helper T cells?
promotion of B-cell and T-cell activity
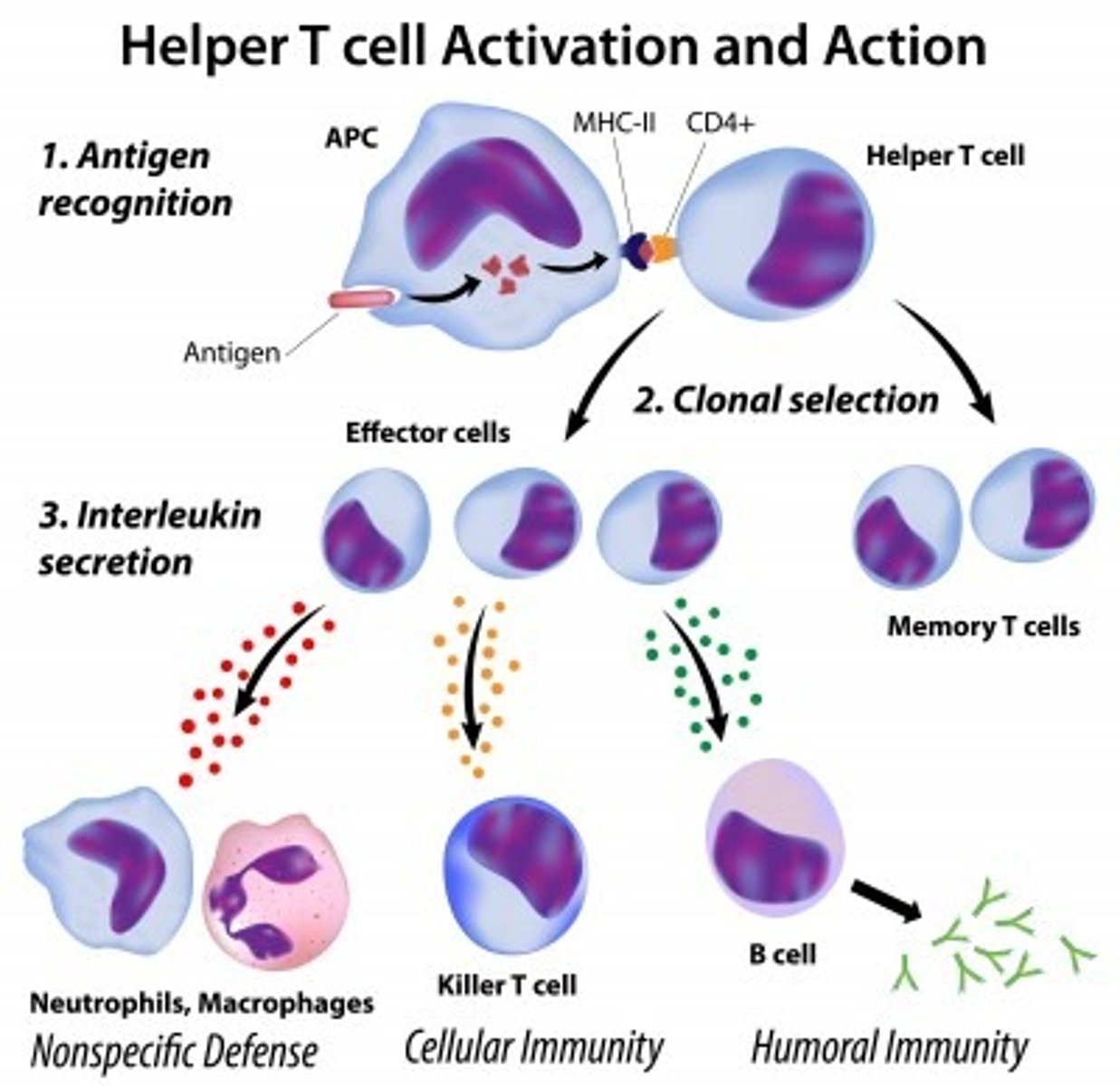
What is the function of Regulatory T cells?
Regulates response levels against a pathogen. Signals to stop overreacting
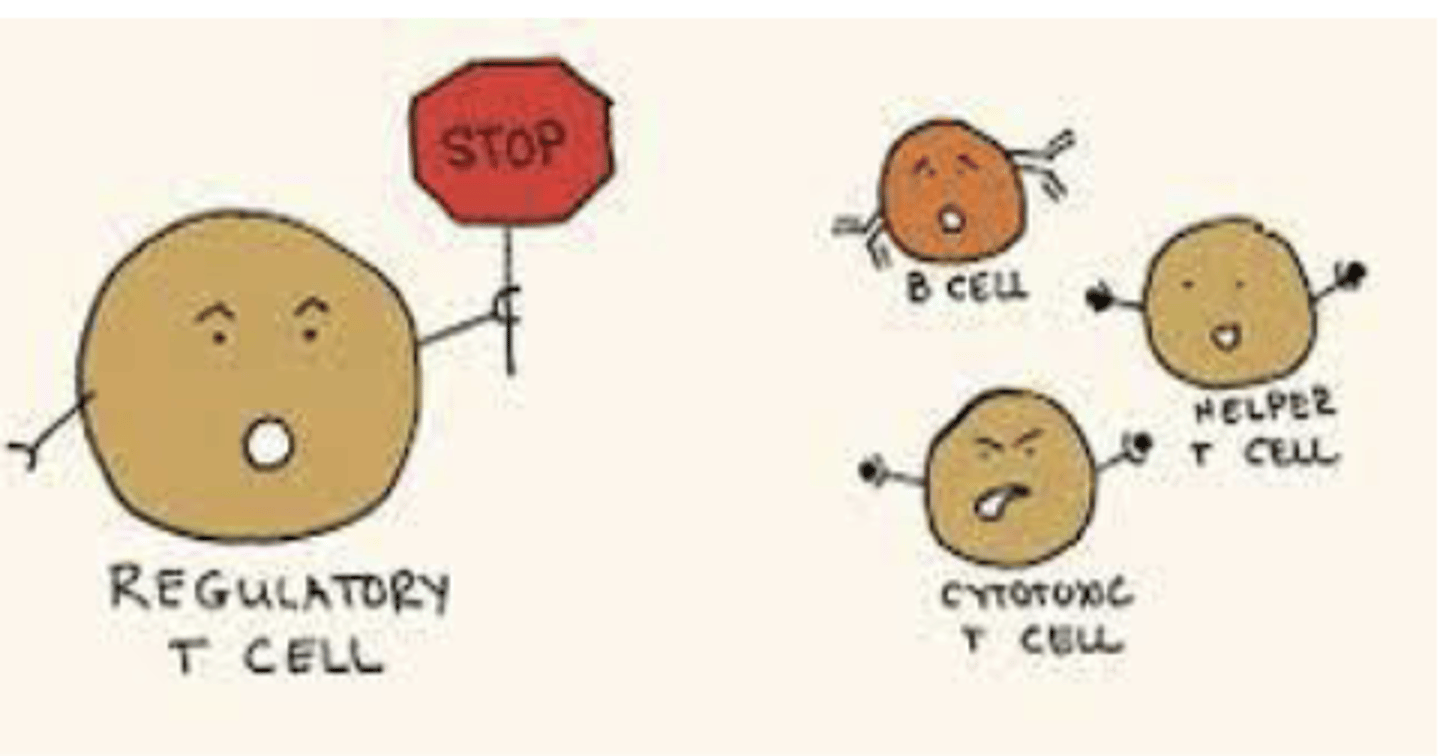
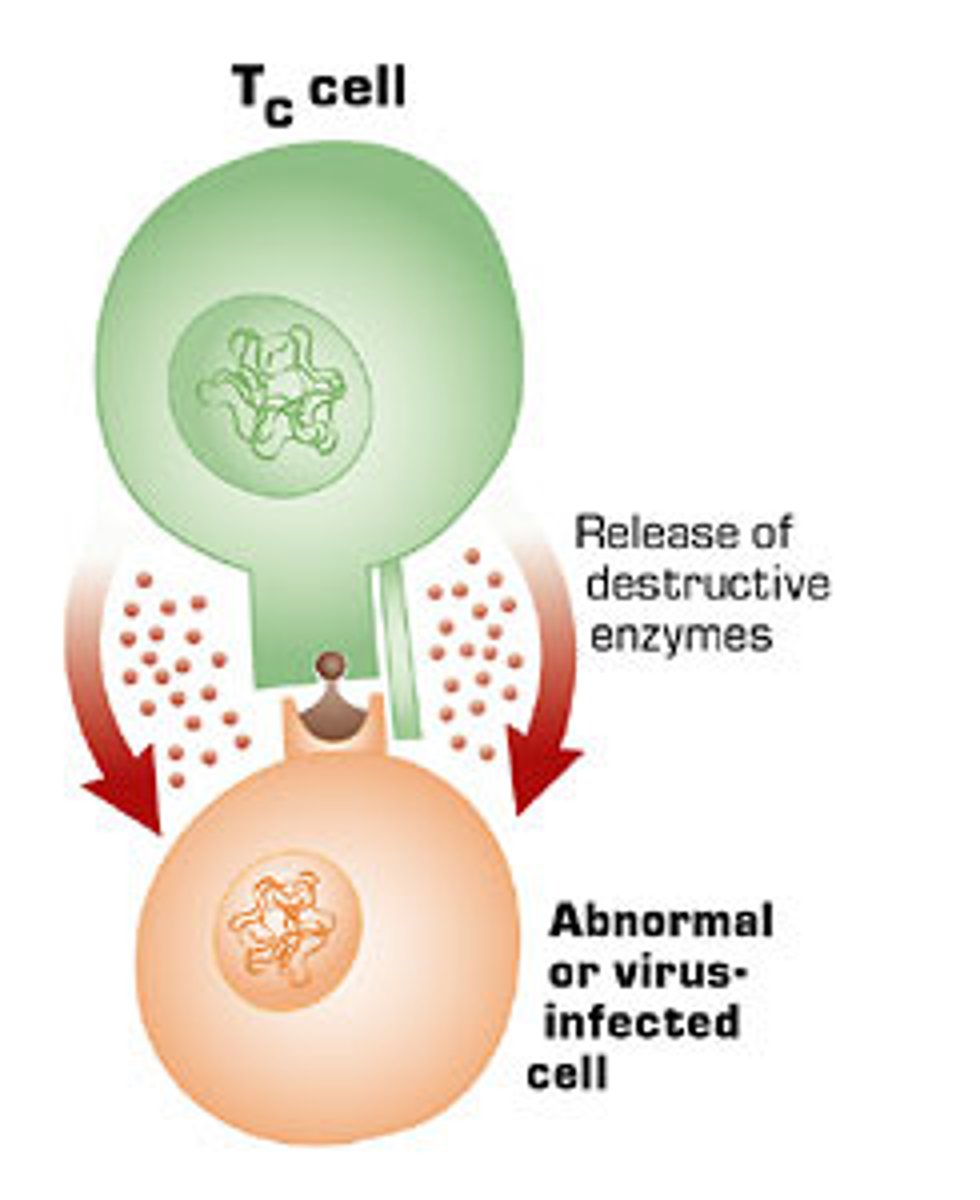
What is the function of cytotoxic T cells?
KILL KILL KILL
If there is no adaptive immunity, what happens?
Infection persists but there is some defence seen from innate immunity
What are the 3 communicators covered in the lectures between innate and adaptive immunity?
Cytokines
Cell to cell communication
Lymph nodes/spleed
Does lymph nodes and spleen play an important role in adaptive immunity responses?
yes. They are the chilling spot for the adaptive immunity cells
What are cytokines?
chemical messengers produced in response to a stimulus
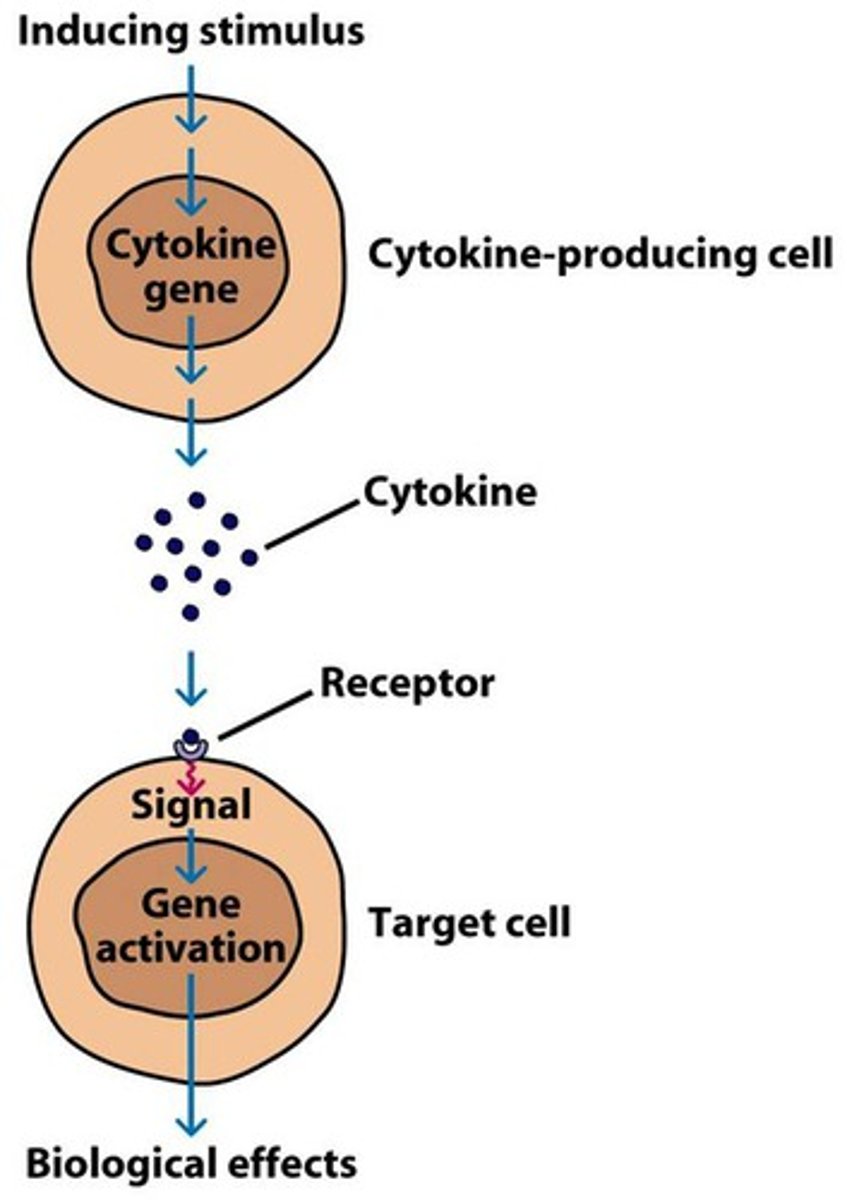
Can cytokines act locally and systematically?
yes
Innate cells recognise generic danger signals via what receptor
pattern recognition receptor
Does somatic recombination help recognise diverse antigens?
yes
Explain the working of the alternate pathway in the complement system
C3 protein (the precursor) forms C3b, that binds to amino and hydroxyl groups on the surface of the bacterium
In the lectin pathway ______ binds to mannose in the surface of the bacterium
MBL (mannose binding lectin)
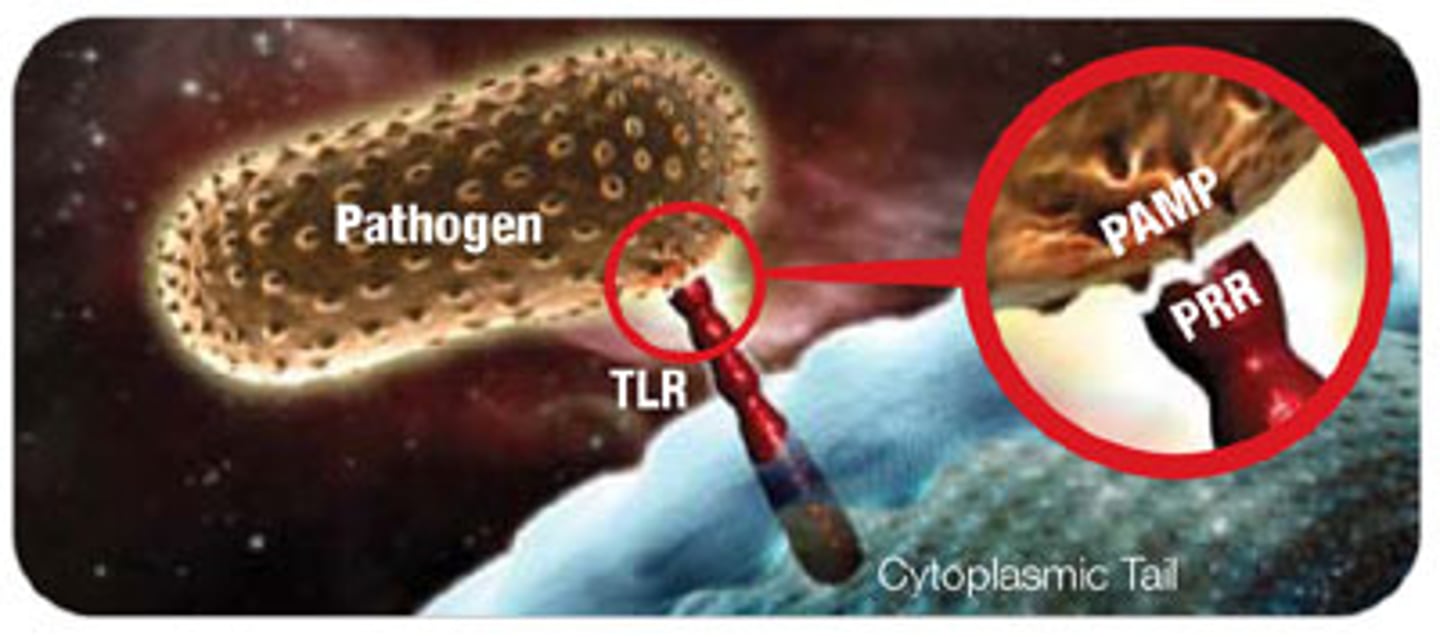
What are PAMPs?
pathogen associated molecular patterns. Conserved microbial molecules that are shared in all microbes
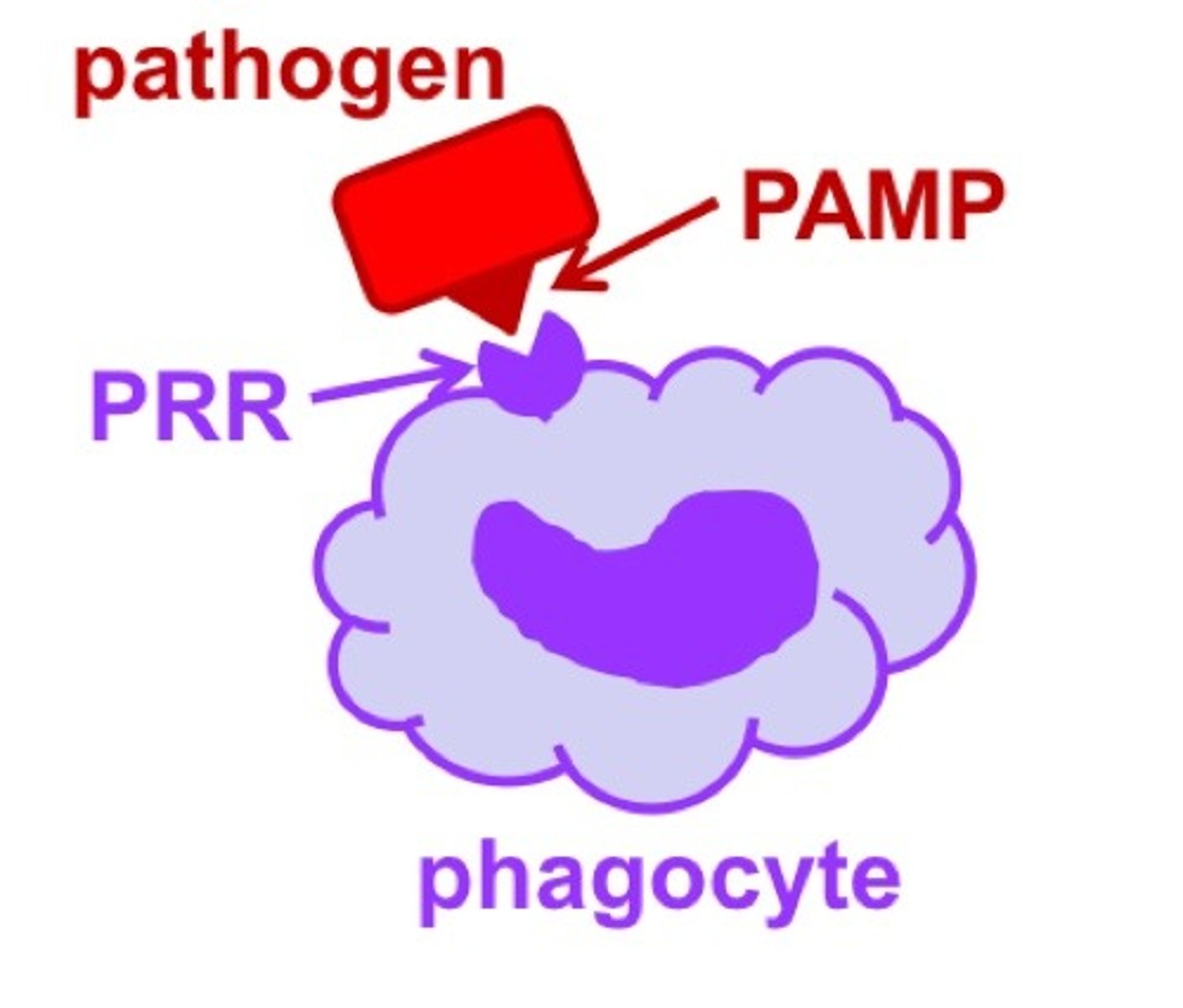
What are PRRs?
pattern recognition receptors. Receptors that recognise PAMPs and DAMPs
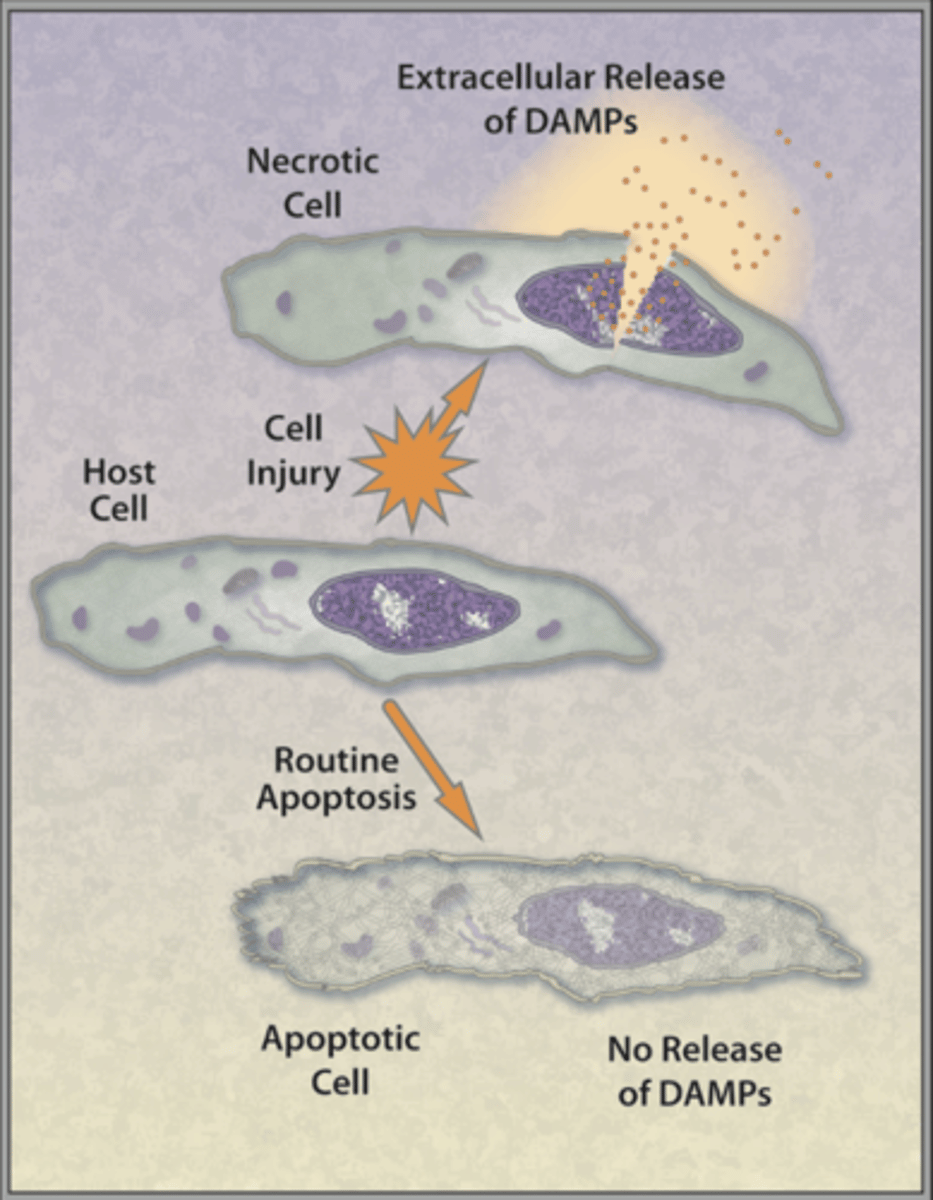
What are DAMPs?
damage associated molecular patterns. Released from dying cells, indicating tissue damage
What are the key immune cells in Innate immunity?
Macrophages
Dendritic cells
Neutrophils
NK cells
function of macrophages?
detect infections and initiate responses
function of dendritic cells?
Present antigens to T cells

What is the function of neutrophils?
phagocytosis
What is the function of NK cells
apoptosis
What is the function TNF-alpha of cytokines?
signals inflammation, attracts macrophages, recruits more B cells and other T cells
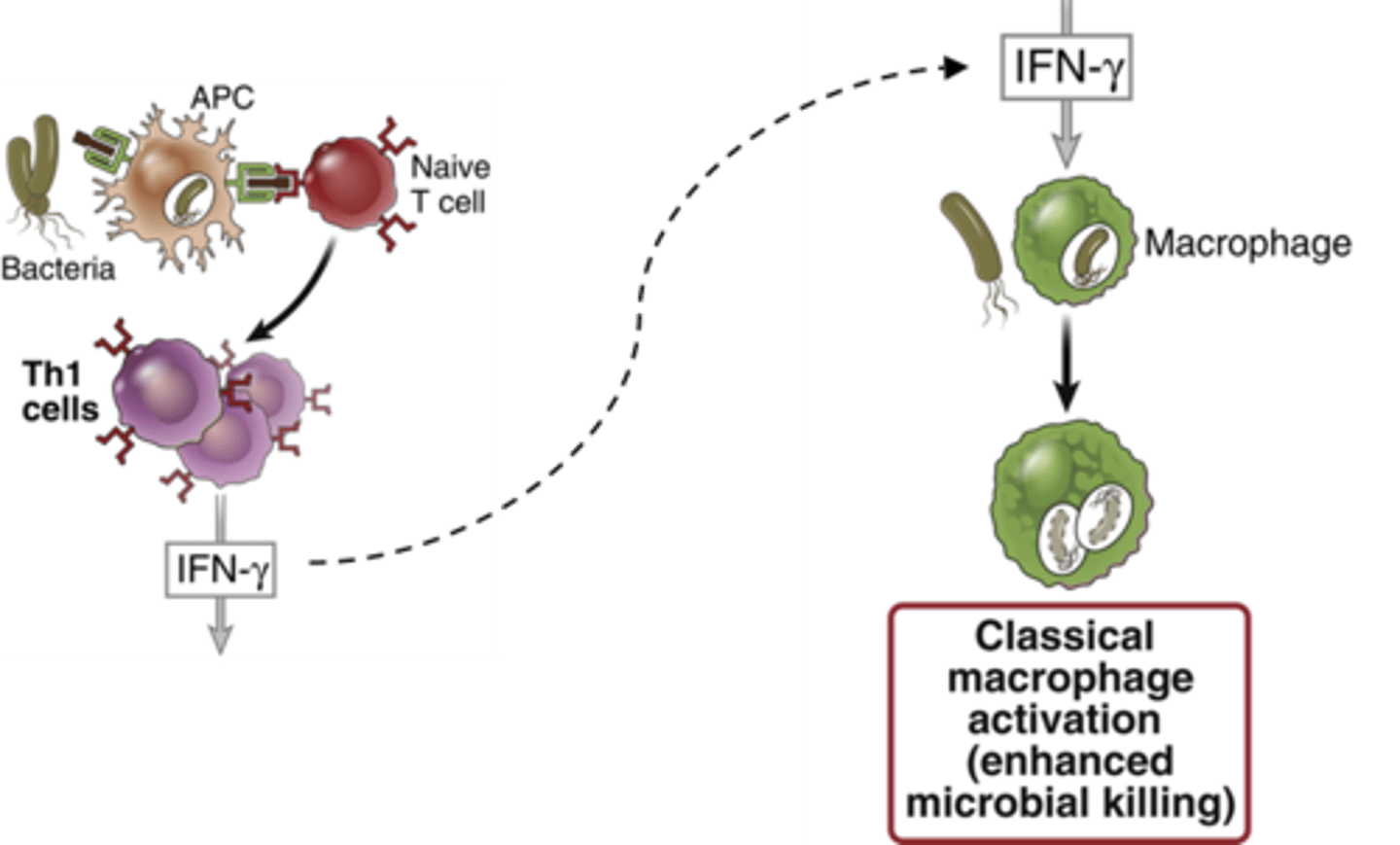
What is the function of interferon-gamma
activates macrophages by producing NK cells
NK cells produce what?
cytokines
During the resting stages what do innate immune cells do?
sample the environment through phagocytosis, normal tissue maintenance
What do annoyed innate immune cells do?
respond to DAMPs and prepare for WARRRRRRRRRRRRRRRR
What do angry innate immune cells do?
WAGE WAR ON THE ENEMIES (foreign particle) !!!!!!!!!!!!!!!!
What is sepsis?
uncontrolled immune response to a systemic infection
Excessive TNF-alpha causes? 3
organ damage, septic shock, bp drops
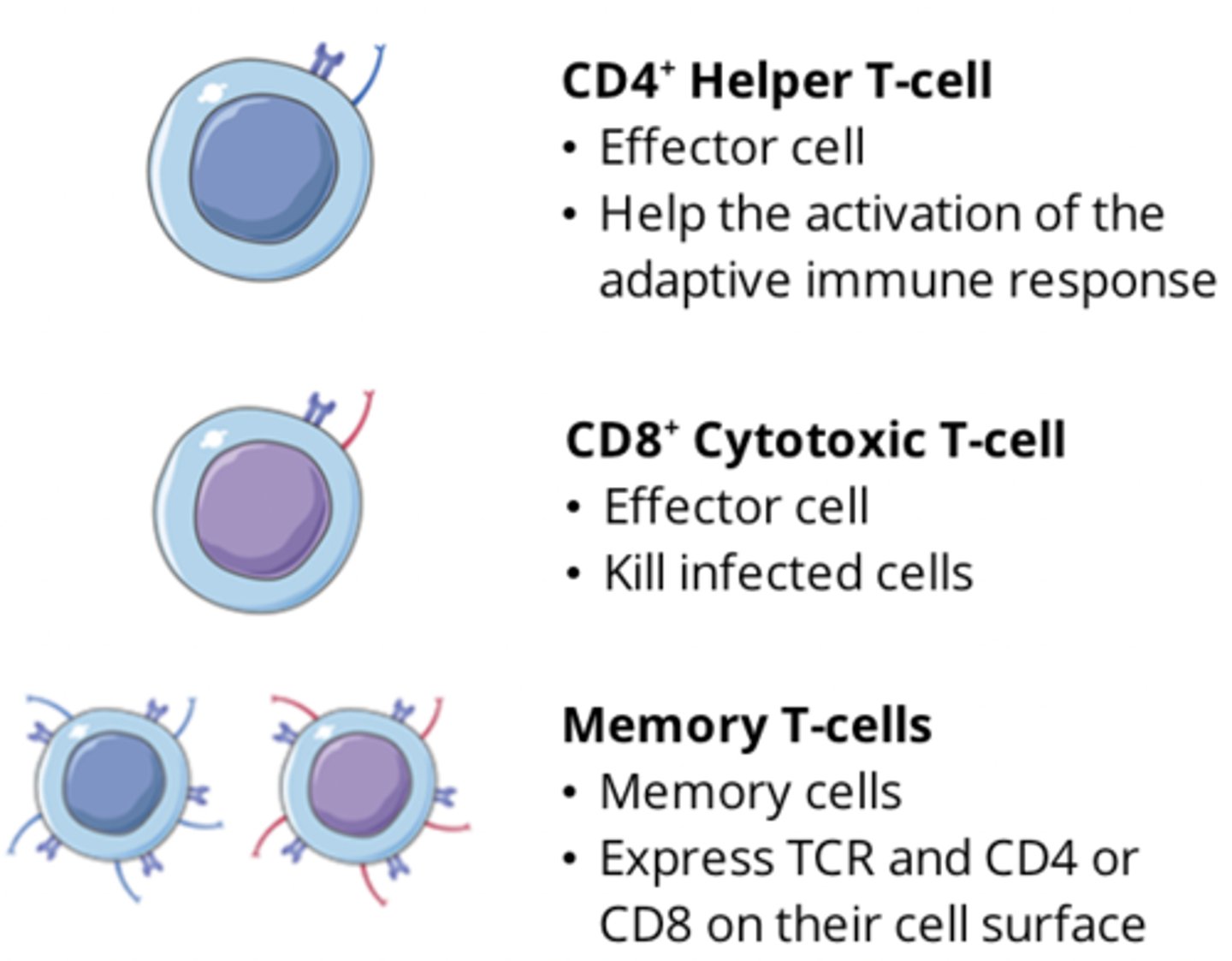
What are the types of adaptive immune cells in the body?
B and T cells
What are the 3 kinds of T cells?
Cytotoxic T cells
Helper T cells
Regulatory T cells
What is an antigen?
molecule recognised by the adaptive immunity
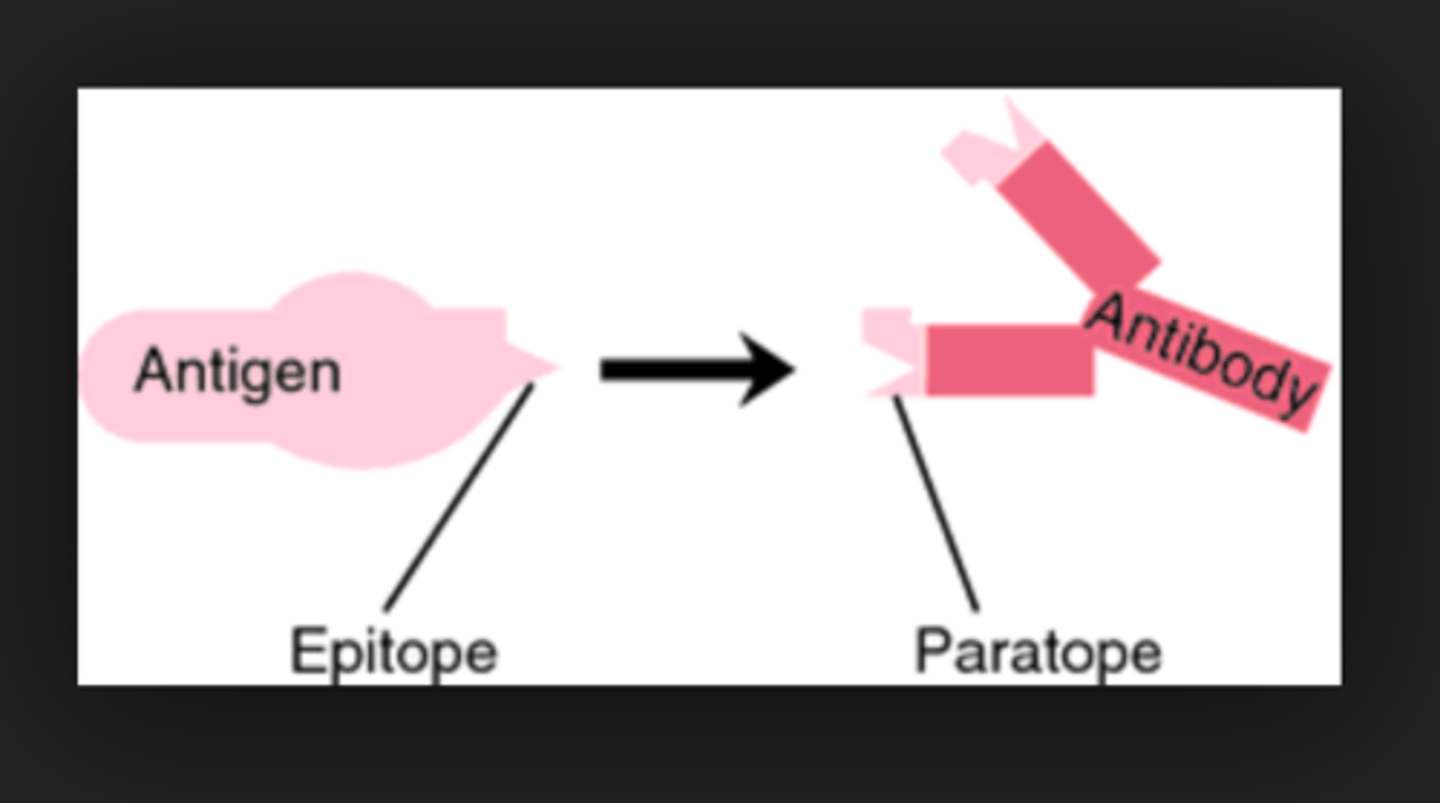
What is an epitope?
A small site of recognition on a large molecule that an antibody recognizes.
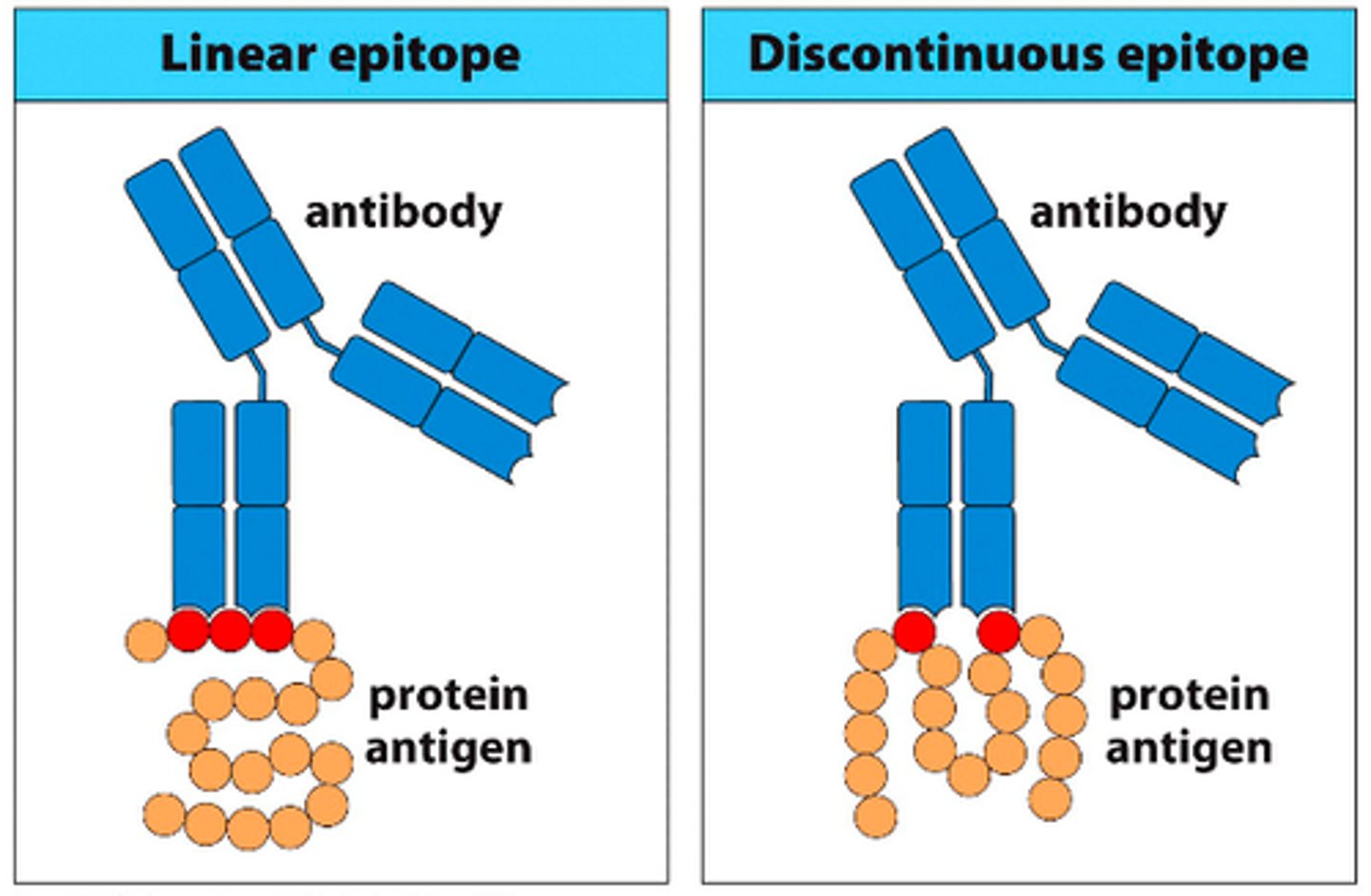
What is a paratope?
part of the antibody that binds to the antigen
What kind of things do B cells recognise?
Pretty much anything organic/ some inorganic too
What kind of things do T cells recognise
Processed information from the APCs
What are APCs?
antigen presenting cells
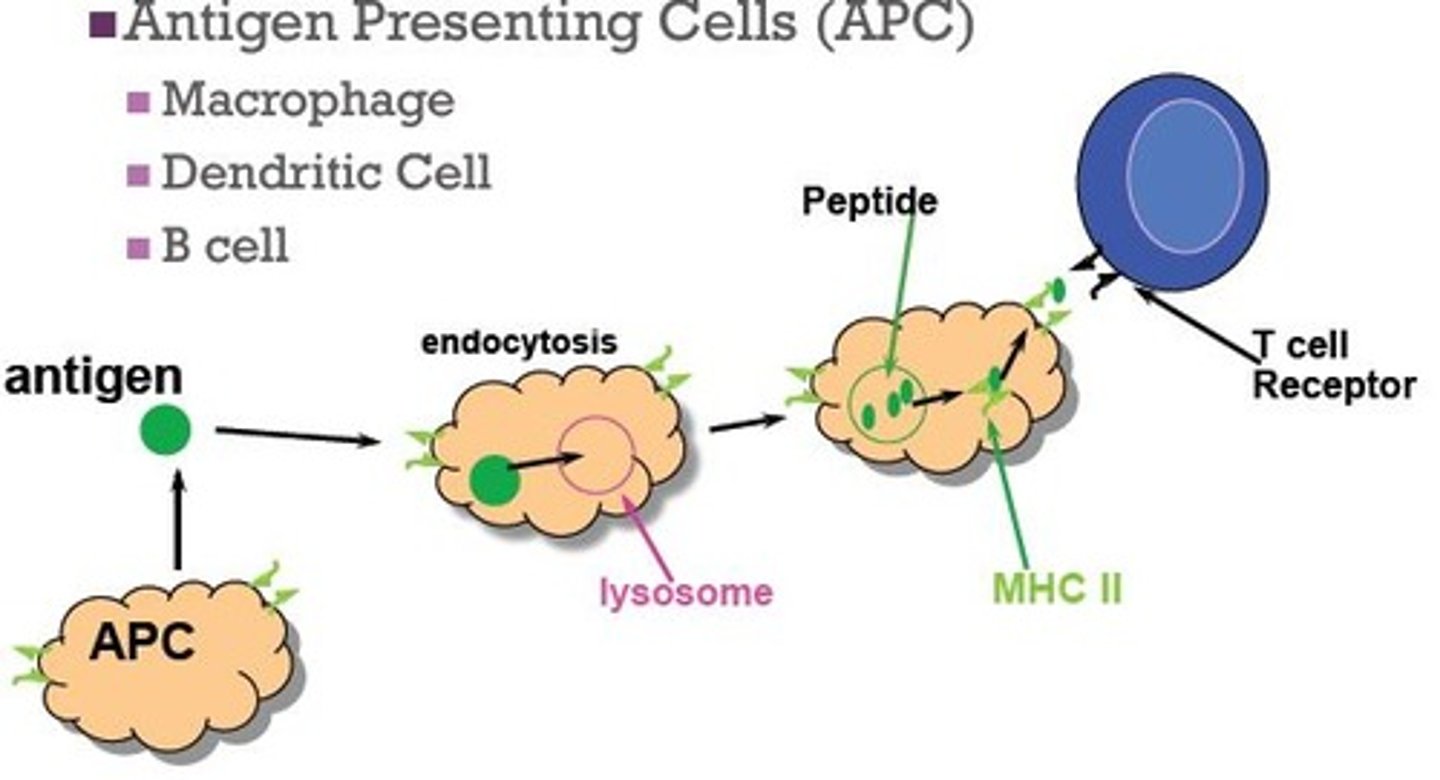
What cells are APCs?
macrophages, dendritic cells, B cells
APCs present processed antigens via ______ molecules
MHC (Major histocompatibility complex)
How many antigen binding sites do B cells have?
2
How many antigen binding sites do T cells have?
1
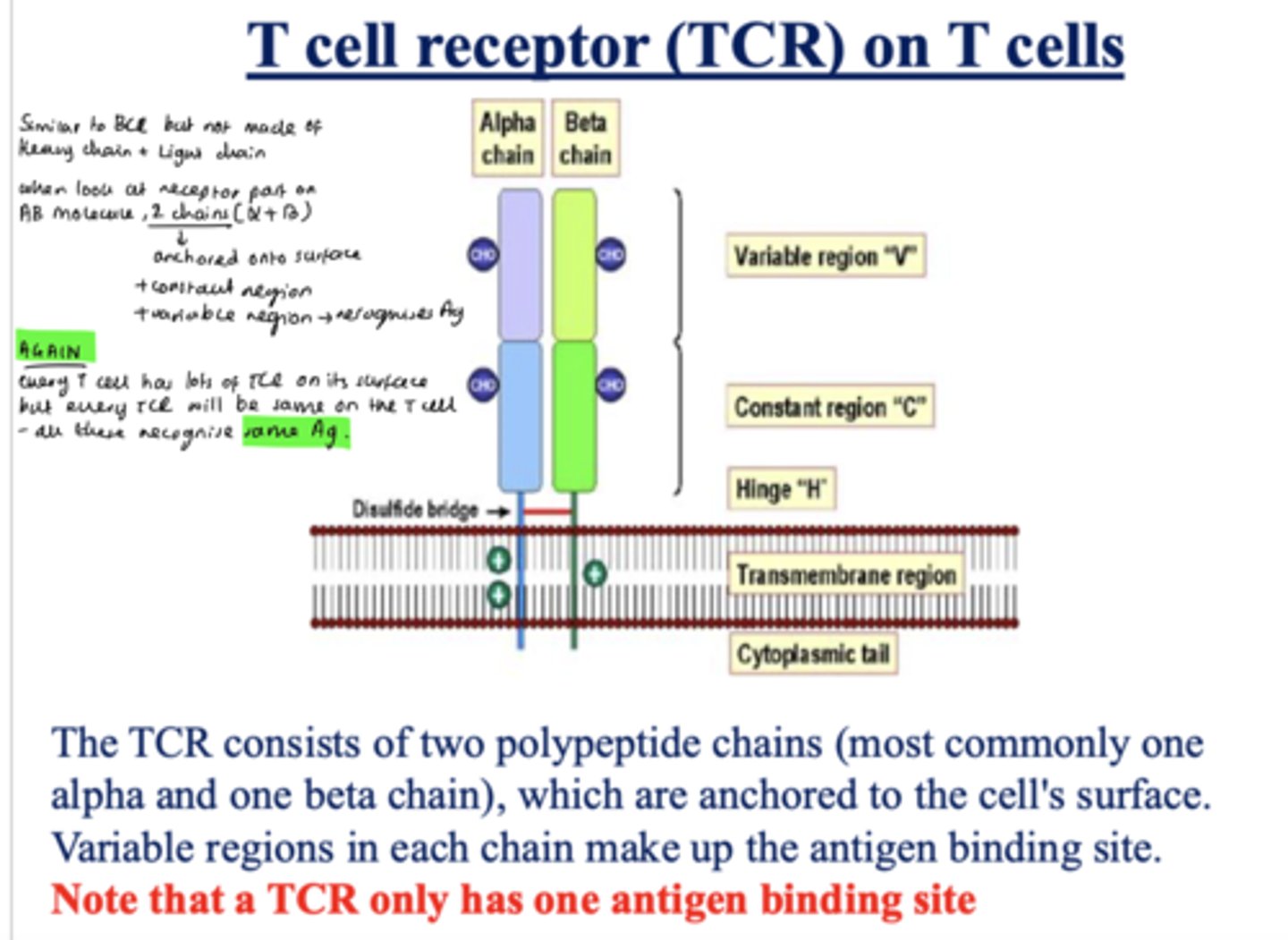
Antigen binds to antibody via _________ (non-covalent/covalent) interactiions?
non-covalent
What are the segments that can undergo somatic recombination in the light chain?
VJ
What are the segments that can undergo somatic recombination in the heavy chain?
VDJ
What are the 3 ways in which junctional diversity helps in generative receptor diversity?
Random nucleotide insertions
Random hairpin structures
VDJ recombination
What is allelic exclusion?
when only one of the parental alleles that code for the TCR or B-cell receptor is functional, ensureing only one receptor specificity per B or T cell by shutting down rearrangement once a successful receptor is produced.
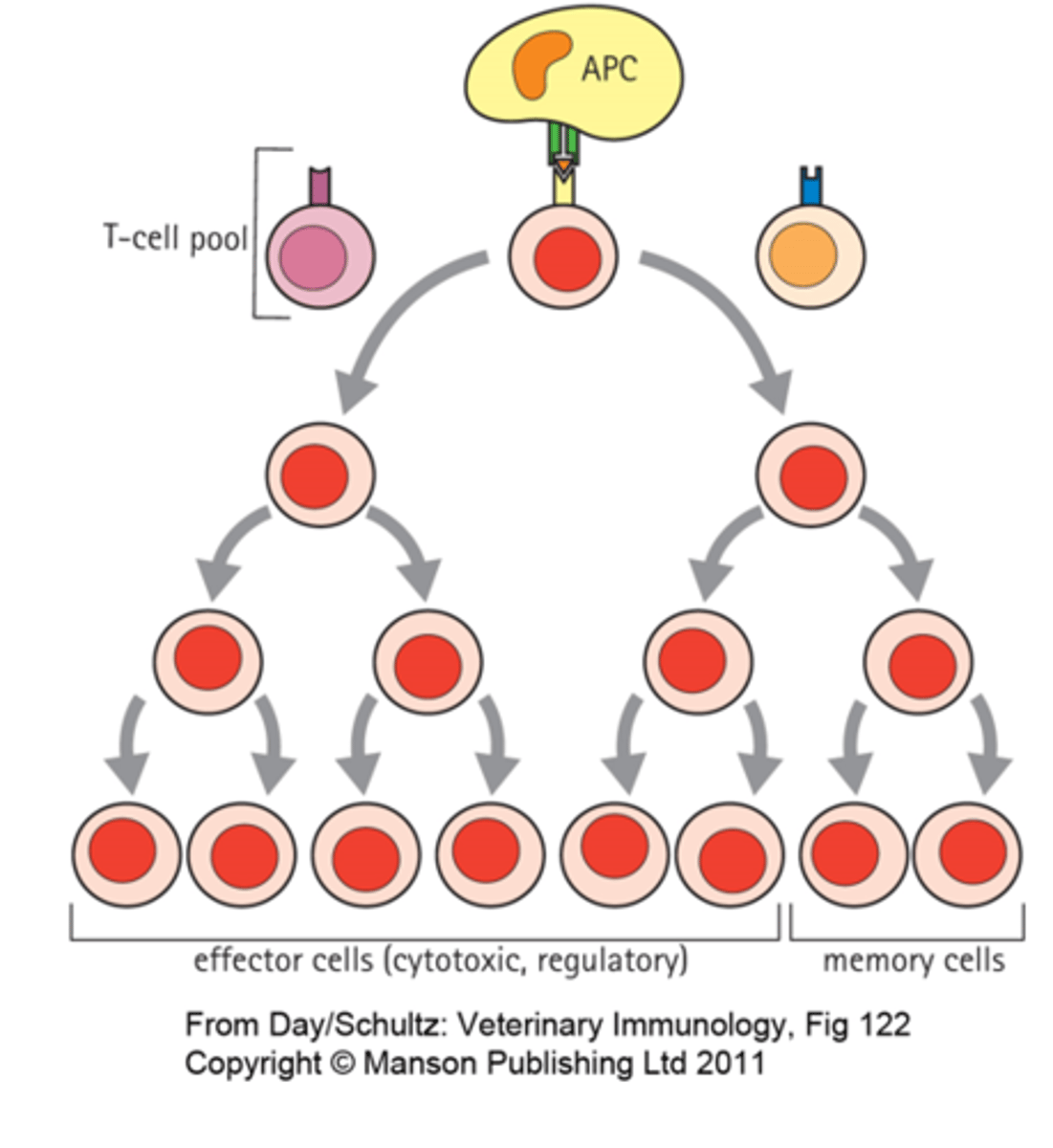
What is clonal expansion?
an increase in the number of cells by mitotic cell division
What is antigenic drift?
gradual accumulation of mutations
What is antigenic shift?
Major antigen changes due to gene exchange (e.g., flu pandemics).
What are the 4 ways pathogens evade immune memory?
Antigenic drift
antigenic shift
high mutation rate
antigen switching
Where are T cells produced?
thymus
Where are B cells produced?
bone marrow
What are the two classes of MHC?
Class I and Class II
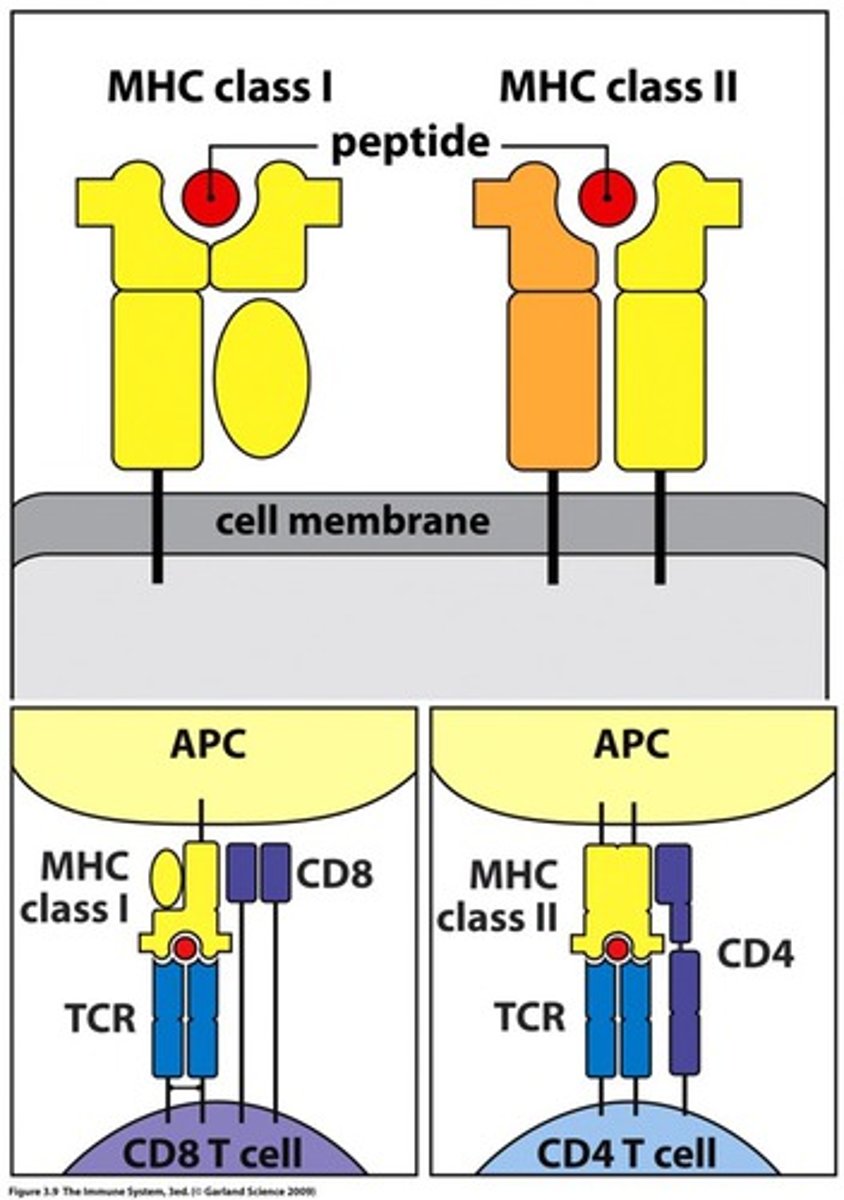
What antigens do MHC I recognise?
intracellular antigens
What antigens do MHC II recognise?
intercellular
What cells are MHC 1 found in?
all nucleated
What cells are MHC2 found in?
APCs
CD8 is present in which MHC class?
1
CD4 is present in which MHC class?
2
Which MHC detects longer amino acids?
2
Cytotoxic T cells detect which MHC class?
1
Helper T cells detect which MHC class?
2
Does the MHC contain a processed part of the antigen when binding to the TCR?
yeah
do high variability in MHC alleles ensures a broad range of peptide presentation.
yeah
- T cells attack grafts with unmatched MHC molecules, leading to _______ (I should know this without fail)
Graft rejection (happens a lot)
Explain the life phases of dendritic cells (HARD but lowkey easy)
Sampler Phase:
- Takes up molecules via phagocytosis.
- Expresses PRRs to detect DAMPs/PAMPs.
Traveller Phase:
- Samples the infection site, then stops sampling to prevent picking up self-antigens.
- Migrates to lymph nodes (LNs) with captured antigens.
Presenter Phase:
- Mature antigen-presenting cell in LNs.
- Upregulates MHC Class II and costimulatory molecules B7 (CD80/CD86).
How do T cells know if its food or a deadly life threatening disease that can cause havoc on the physical body?
MHC-peptide-TCR complex verifies antigen specificity
PAMPs/DAMPs cause up regulation of B7 that binds to the CD28 receptor in T cells that actually conveys if it is dangerous
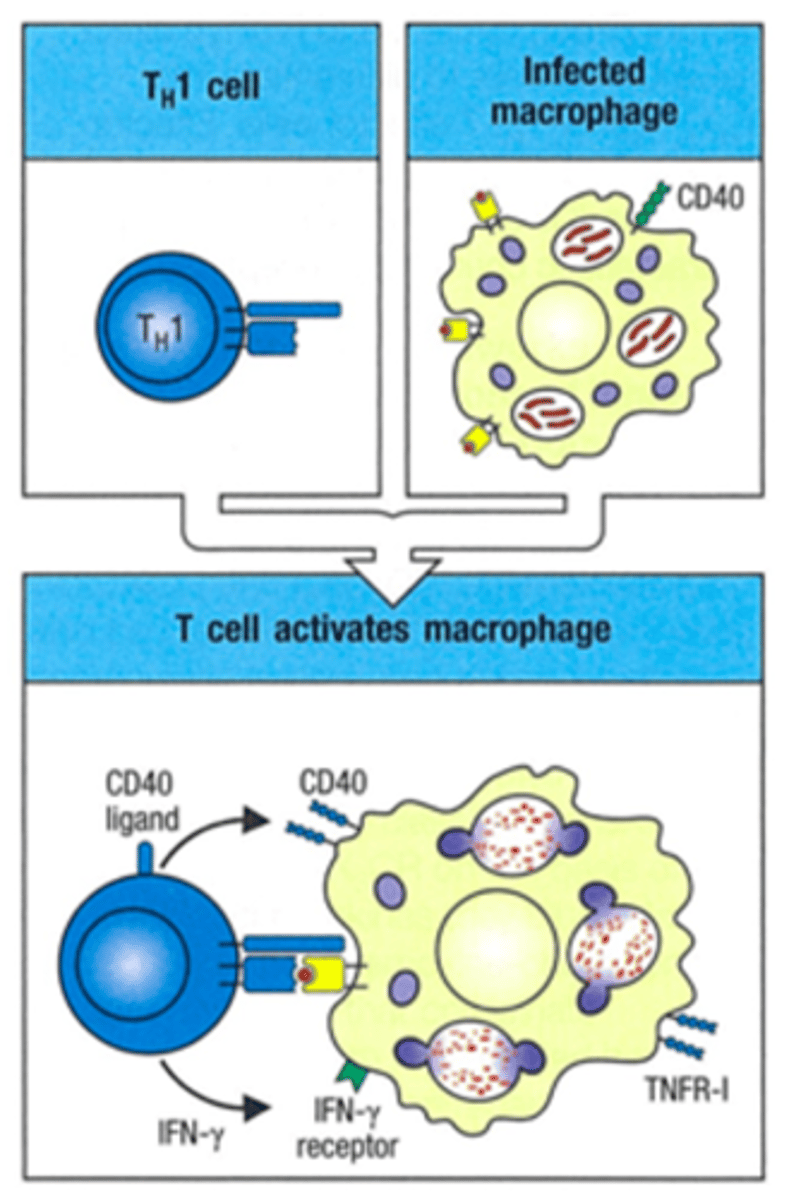
What is the role of macrophages as an APC?
present antigen to T cell,
Requires T cell's help to activate more macrophages though (IFN-gamma)
What is the role of B cells as APCs?
Require T cell "permission" to activate and differentiate
it has a T-dependent and T-independent antibody responses.
What is the function of Cytokine IL-10?
it is an inhibitory cytokine