Heterogenous Catalysis
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Outline the basic steps to heterogenous catalysis
Diffusion of the reactants through to fluid to the catalyst.
Diffusion of the reactant into the catalyst pore system
Adsorption of the reactant onto the catalyst surface.
Conversion of the reactant on the catalyst surface.
Desorption of the product from the catalyst surface.
Diffusion of the product through the catalyst pore system.
Diffusion of the product from the catalyst to the fluid.
What are the two ways catalytic poisoning can occur?
Reactant adsorbs to catalyst too strongly - reactant posioning.
Product poisoning
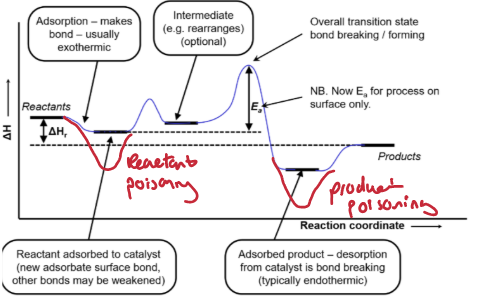
Outline the steps in a water-gas shift reaction
CO + * → CO* (CO adsorption)
H2O + 2* → H* + OH* (dissociative H2O adsorption)
OH* + CO* → HCOO* + * (formate formation)
HCOO* + → CO2* + H* (formate decomposition)
CO2* → CO2 + * (CO2 desorption)
2H* → H2 + 2* (H2 desorption)
What is water-gas shift reactions beneficial for?
WGS is used to adjust the syngas to the desired ratio – very important for efficient use of carbon feedstock/energy used in syngas production.
Syn-gas formed used in MeOH synthesis, FT synthesis, methanation.
Draw a reaction bed profile, explaining what it shows.
Start of bed: low/no product concentration. Bulk of product formation. High initial rate as forwards reaction only
Middle: Forward reaction dominants but reverse reaction also occurring.
End of bed: thermodynamic equilibrium achieved, observed rate of products forming appears to slow down. Forwards and backward reaction are equal.
Outline temperature programmed desorption experiments - what can this technique be used to study?
Catalyst is exposed to an adsorbate at low T, which binds to available surface sites. Temperature is linearly increased → desorption. Amount desorbing monitored by mass spec → TDP spectrum of desorption rate vs T.
Area under peak is proportional to the number of desorbed molecules.
Desorption temperature tells you about interaction/binding strength (chemisorbed vs physiosorbed)
Desorption shape can tell you about the mechanism.
Outline XPS experiments - what can this technique be used to study?
Core electrons excited and ejected. KE of electrons detected → binding energy. Can tell us which elements and their oxidation states.
Surface specific however cannot be done in situ.
Outline X-ray absorption experiments - what can this technique be used to study?
Excite electrons → edge jump in the X-ray absorption. Further energy → electrons ejected.
Element specific but bulk analysis
NEXAFS (Near-edge X-ray absorption fine structure): atomic chemical environment - can give MO information, oxidation state.
EXAFS: near neighbour environment - can give information about coordination number and bond lengths.
What is X-ray diffraction used for in catalysis?
X-rays produce a diffraction pattern that can be used to determine the crystal structure, and crystallite particle sizes.
Bulk-analysis, and requires long range order (i.e. cannot be used for very small or amorphous crystals).
How do TEM, SEM and SPM microscopic techniques all differ (i.e. when should you use each).
Transmission Electron Microscopy: scattering by electron dense atoms gives contrast (darker areas of image = heavier atoms, easy to pick out the metal particles of catalyst). Spot-type analysis of nanostructure.
Scanning Electron Microscopy: surface electron scattering. Wider field of view compared to TEM. Electron dense atoms gives a signal. Good at looking at morphology and support structure.
Scanning Probe Microscopy: gives information about the electronic structure at surface. Finer resolution.
Explain what information is given by IR and DRIFTS - why is this useful compared to other techniques?
Can be employed under reactor conditions/in situ.
DRIFTS measure IR absorption by molecules adsorbed to the surface, giving information on the vibrational modes of surface species. Can be used to monitor changes e.g. with temperature changes. Shows:
whether adsorption is molecular or dissociative.
how strongly bound a species is.
What is BET analysis used for in heterogenous catalysis
Determines the specific external and internal surface area of a catalyst by measuring amount of gas (N2) adsorbed onto the surface.
Gives information on the pore system and pore diameters.
Can indicate sintering or coking.
How does gas chemisorption work and what information does it provide?
Treats catalyst with H2 or CO for adsorption. Remove physisorbed molecules e.g. by increasing temperature to get only chemisorbed. Can plot pressure vs V absorbed → chemisorbed appears linear, since we can only get a chemisorbed monolayer.
Outline the mechanism for ammonia synthesis - state the RDS. Name the catalyst.
H2(g) + 2* → 2H* (dissociative adsorption).
N2(g) + 2* → N2* + * → 2N* (RDS for N2 dissociation)
N* + H* → NH* + *
NH* + H* → NH2* + *
NH2* + H* → NH3* + *
NH3* → NH3(g) + *
Catalyst = Fe2O3 with K2O , Al2O3 and CaO
Explain using d electron count why Ru and Fe are optimal for ammonia synthesis.
Electron density in d-orbitals -> pi backbonding into N2, weakening N2 bond for dissociation.
H, start to populate antibonding orbital once d-orbitals are more than half fill -> N2 won’t want to stay absorbed to surface.
Fe and Ru give optimal d electron count for adsorption and dissociation - balanced. Ru = higher activity but more expensive.
Explain how Al2CO3 and CaO act as structural promoters, and how K2O acts as an electronic promoter.
Al2O3 and CaO reduces sintering (sticking together) of Fe2O3. Helps maintain high surface area of iron therefore high activity.
K2O = K+ provider which uses dipole-dipole interactions to favour back donation, aiding N2 dissociation and NH3 removal.
What is the advantage of using heat exchangers in the reaction bed?
Ammonia syntehsis = exothermic → increases temperature as produced, shifting equilibrium against NH3.
Intermittent cooling not in presence of catalyst.
Catalysis → Cool → Catalysis → Cool etc.... Allowing for higher overall conversion.
Give the mechanism for methanol synthesis after WGS has occurred.
CO2* + H* → HCOO*
HCOO* + H* → H2COO* + *
H2COO* + H* → H3CO* + O* RDS, breaking C-O bond.
H3CO* + H* → H3COH* + *
CH3OH* → CH3OH + *
What is the role of zinc in the Cu/ZnO/Al2O3 catalyst for methanol synthesis
ZnO acts as a support for the Cu. Stabilises Cu surface area and can act as a chemical promoter, withdrawing electron density from Cu
How is the surface area of Cu metal measured (chemisorption only)
Uses N2O, which reacts with Cu to form N2 and CuO. Binds to Cu only. Can use low T to look at reactive sites only.
What is the role of CO (other than for WGS formation of syngas) in methanol synthesis
CO helps with (some) reduction of ZnO, leading to better Cu wetting (flattening over surface) increasing surface area for reaction.
Outline a basic description for Fischer Tropsch synthesis
intitiation - either CO dissociates to surface CHx*, then chain growth OR CO initially hydrogenated to surface hydroxymethane type intermediates, then chain growth.
Chain growth. Process analogous to polymerisation.
Termination → alkene, alkane and alcohol products.
How does Fe and Co catalysts control the length of the chain formed.
Chain growth probability = alpha.
Typical Fe catalyst, alpha = 0.65-0.70 I.e. making lighter (shorter chain) molecules.
Typical Co catalyst, alpha = 0.75-0.85, makes longer chains typically.
Why do longer residence times lead to less alkenes formed?
Surface alkene chain termination can occur via reaction with hydrogen or via beta-hydrogen abstraction.
Alkene product can easily reabsorb which may reinitiate chain growth or hydrogenate therefore longer residence time often leads to less alkenes (more likely to be hydrogenated to alkane)
What is the role of precious metal promoters and alkali promoters in FT synthesis.
Precious metal promoter aids hydrogen dissociation/reducibility.
Alkali promoters increase chain growth probability.
How can WGS sometimes be an advantage to FT synthesis?
Can help to improve amounts of H2 in H2 poor feeds. Occurs more in Fe catalysts. Happens because H2O = product.
Which technique can be used to look at the build up of hydrocarbonaceous surface layer formation in Fe catalysts in FT synthesis?
Use temperature-programmed oxidation - CO2 evolution as temperature increases → bulk iron carbide peaks at higher T.