Pentose Phosphate Pathway (PPP)
1/135
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
136 Terms
what is the pentose phosphate pathway (PPP) also called
the hexose monophospahte shunt
where does the PPP occur
cytosol
what major metabolic pathways is PPP closely related to
- glycolysis
- gluconeogenesis
- fatty acid synthesis
- nucleotide synthesis
why is PPP considered biomedically important
essential for:
- NADPH production
- oxidative stress defense
- biosynthesis (fat, cholesterol, nucleotides)
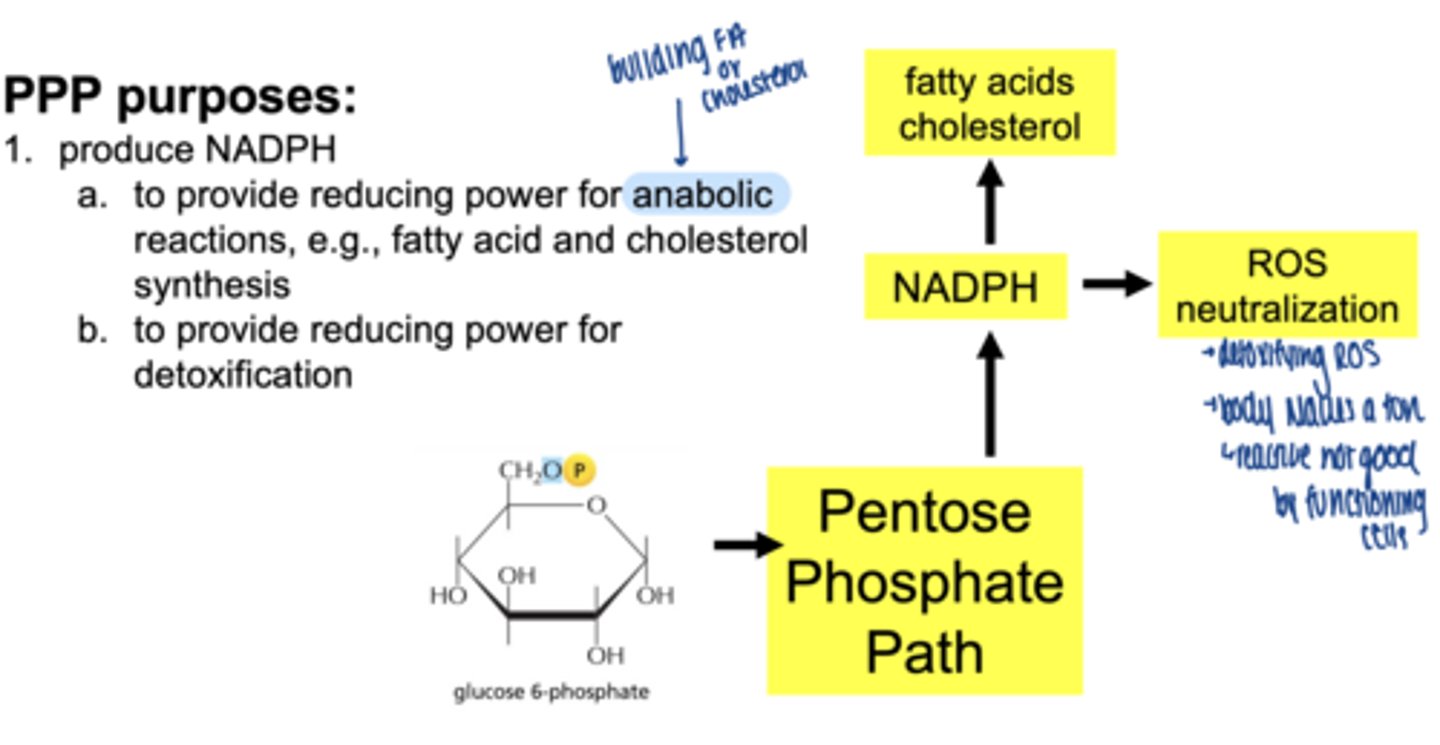
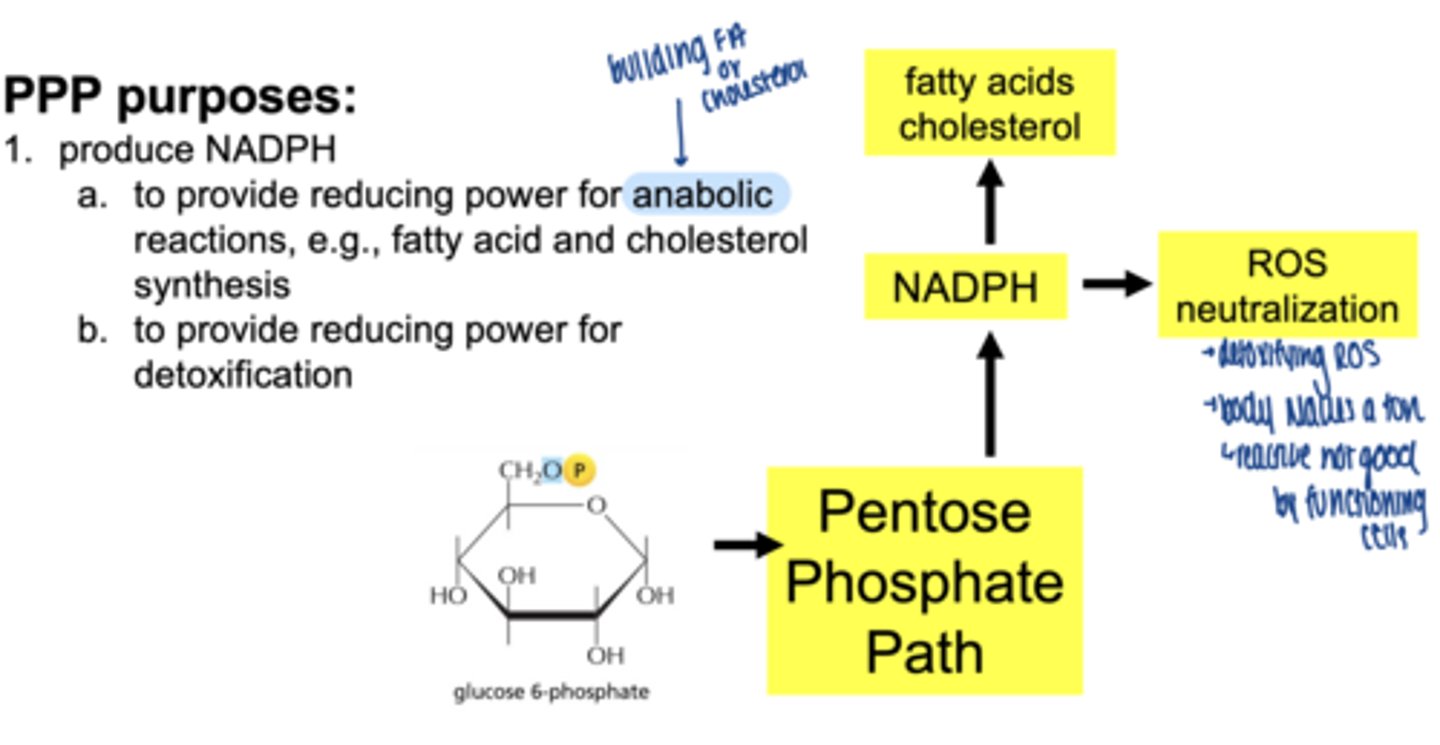
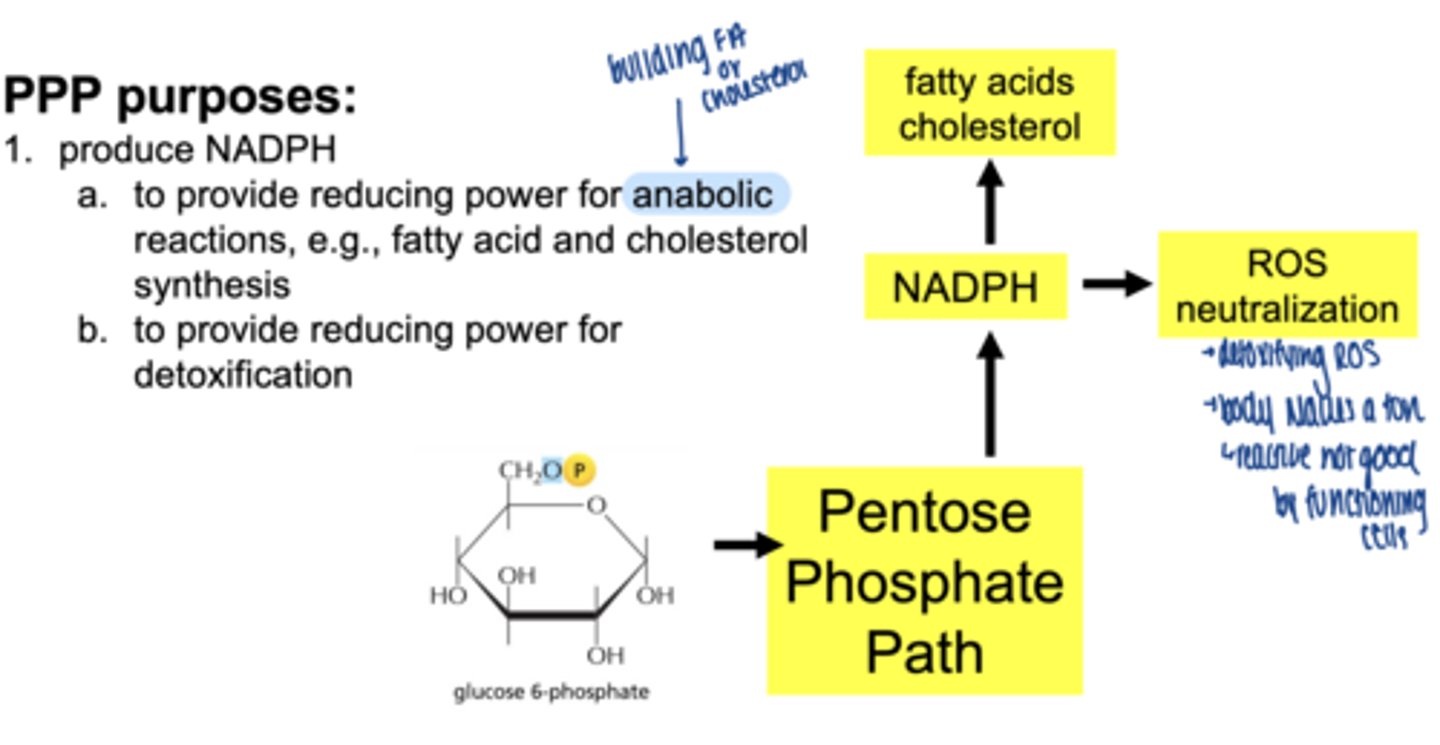
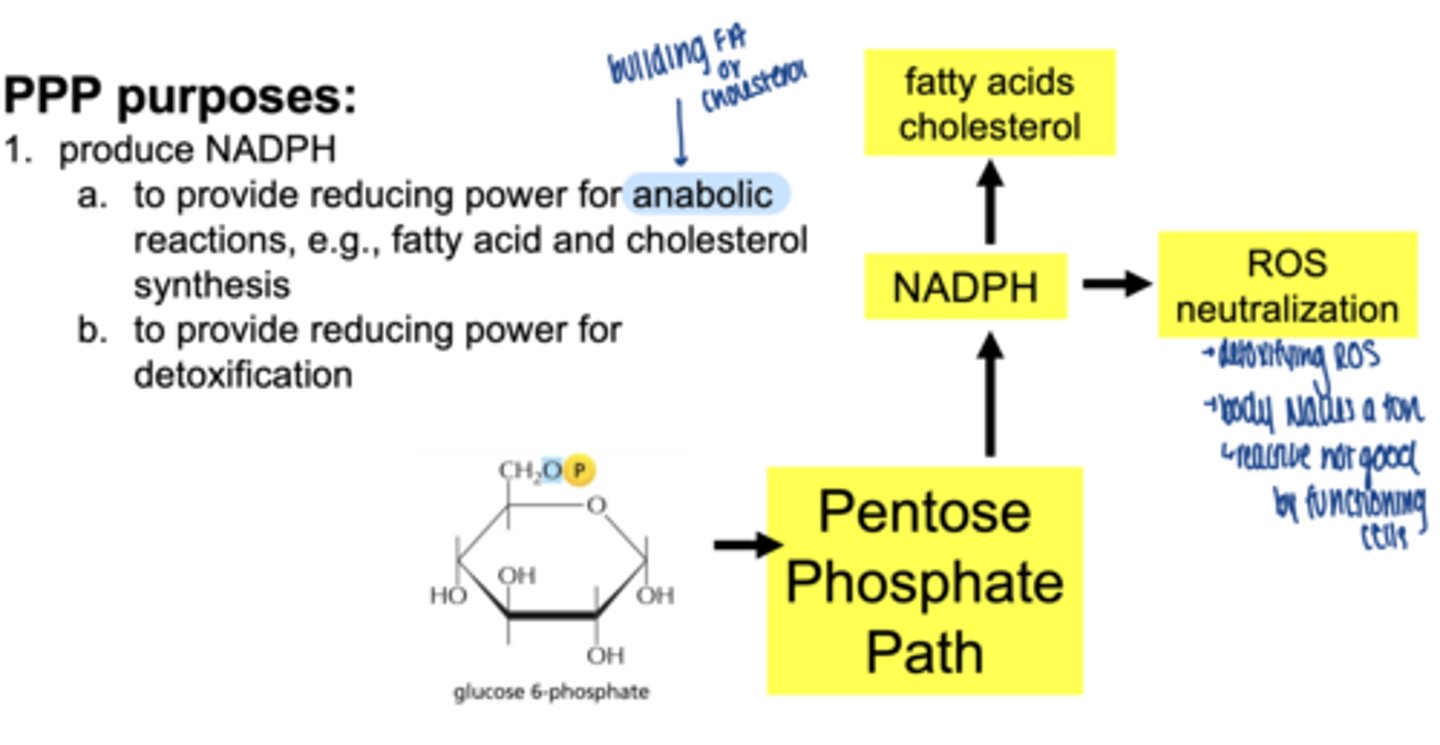
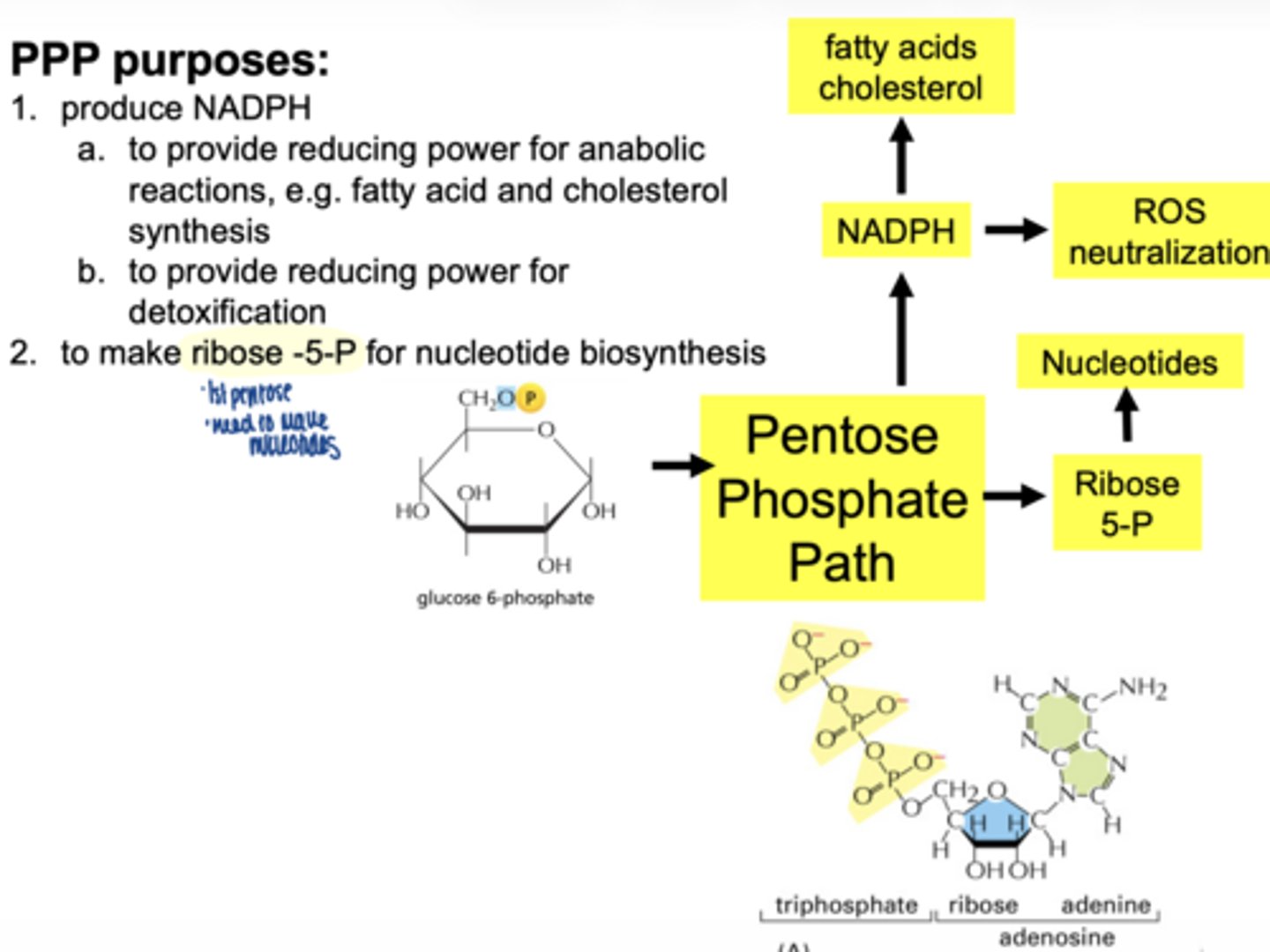
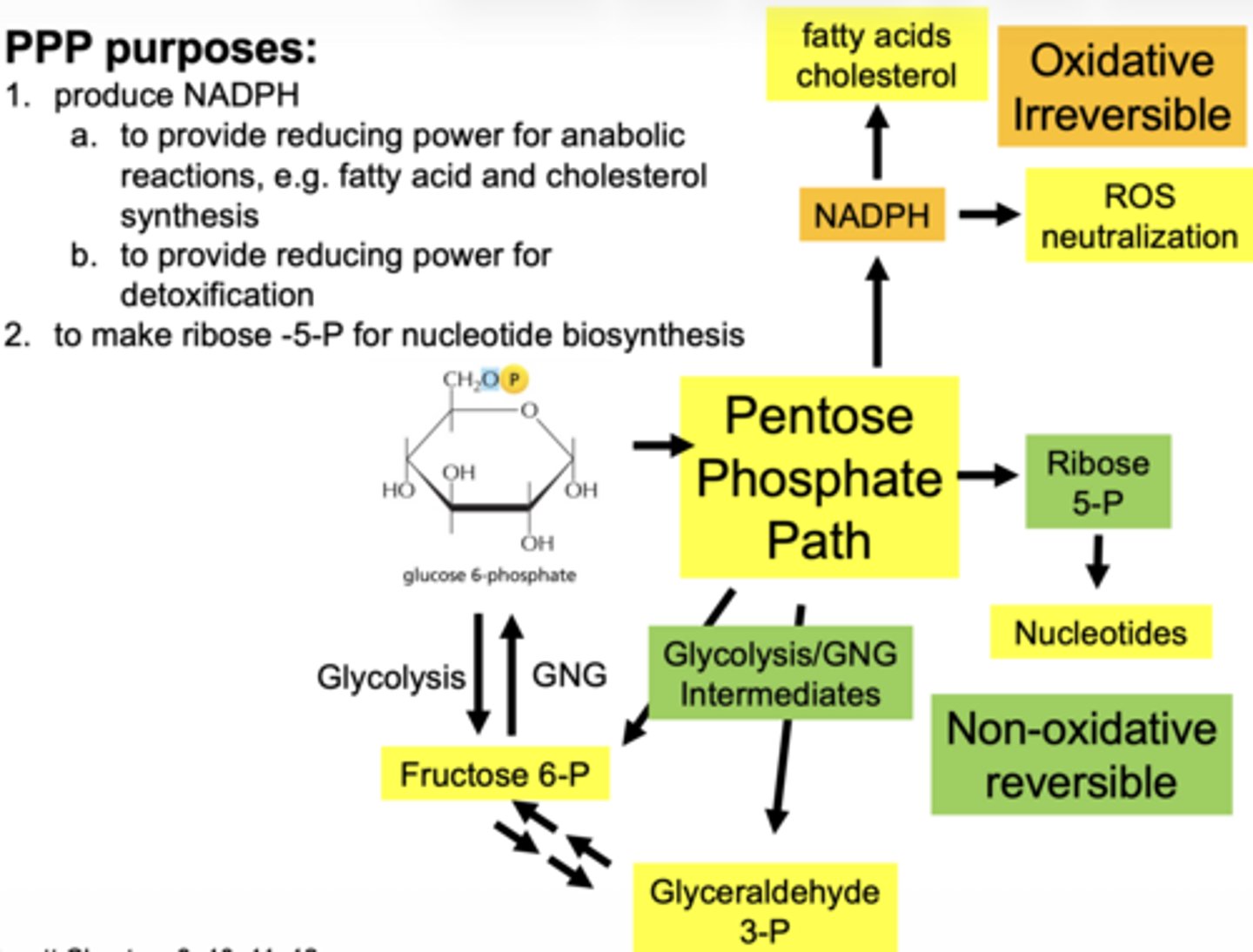
what are 3 main functions of the PPP
1. produce NADPH
2. reduce oxidative stress (ROS) detoxification
3. produce ribose-5-phosphate for nucelotide synthesis
what is NADPH
a reducing molecule similar to NADH but with an extra phosphate
what is main role of NADPH
reducing power for anabolic reactions
what is NADH mainly used for
ATP production (energy generation)
which type of metabolism uses NADPH
anabolic (building pathway)
examples of processes that require NADPH
- fatty acid synthesis
- cholesterol synthesis
- detoxification reactions
what are reactive oxygen species (ROS)
highly reactive oxygen-containing molecules that can damage cells
natural byproducts of oxidative metabolism & oxidative stress from drug or toxin exposure
examples of ROS
Hydrogen peroxide (H₂O₂)
Superoxide (O₂⁻)
Hydroxyl radical (OH•)
Hypochlorite (OCl⁻)
why are ROS dangerous
they damage:
- DNA
- proteins
- cell membranes
what conditions are associated with excess ROS
- cancer
- aging
- inflammation
- reperfusion injury (heart attack damage)
are ROS ever beneficial
yes... used by the immune system to kill pathogenic microbes
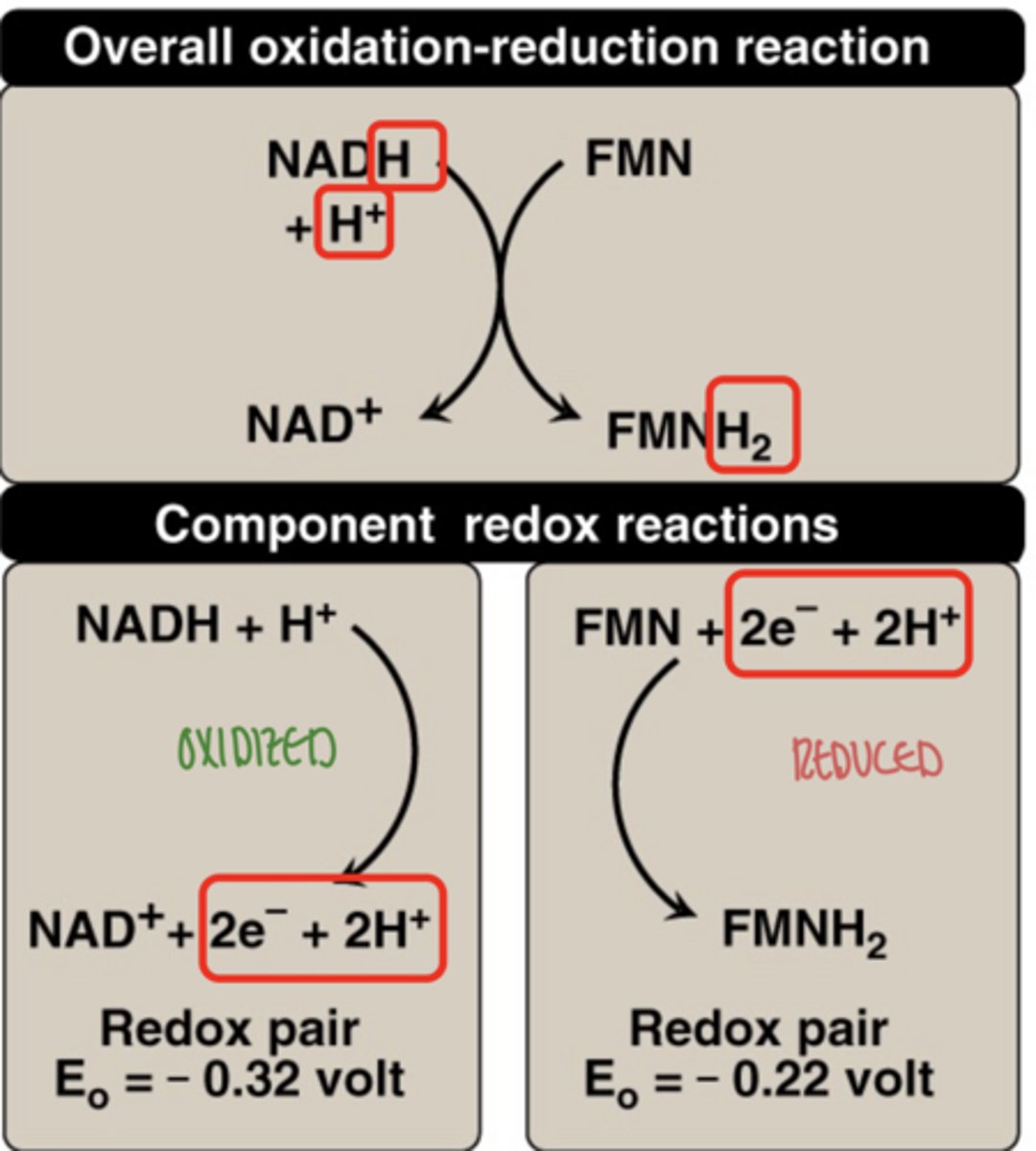
what does oxidation mean
loss of electrons
what does reduction mean
gain of electrons
OIL RIG
why must oxidation and reduction occur together
electrons must be transferred between molecules
(paired reactions)
you need a _____ molecule to neutralize reactive oxygen species
reduced
what is glutathione
a tripeptide antioxidant containing cysteine
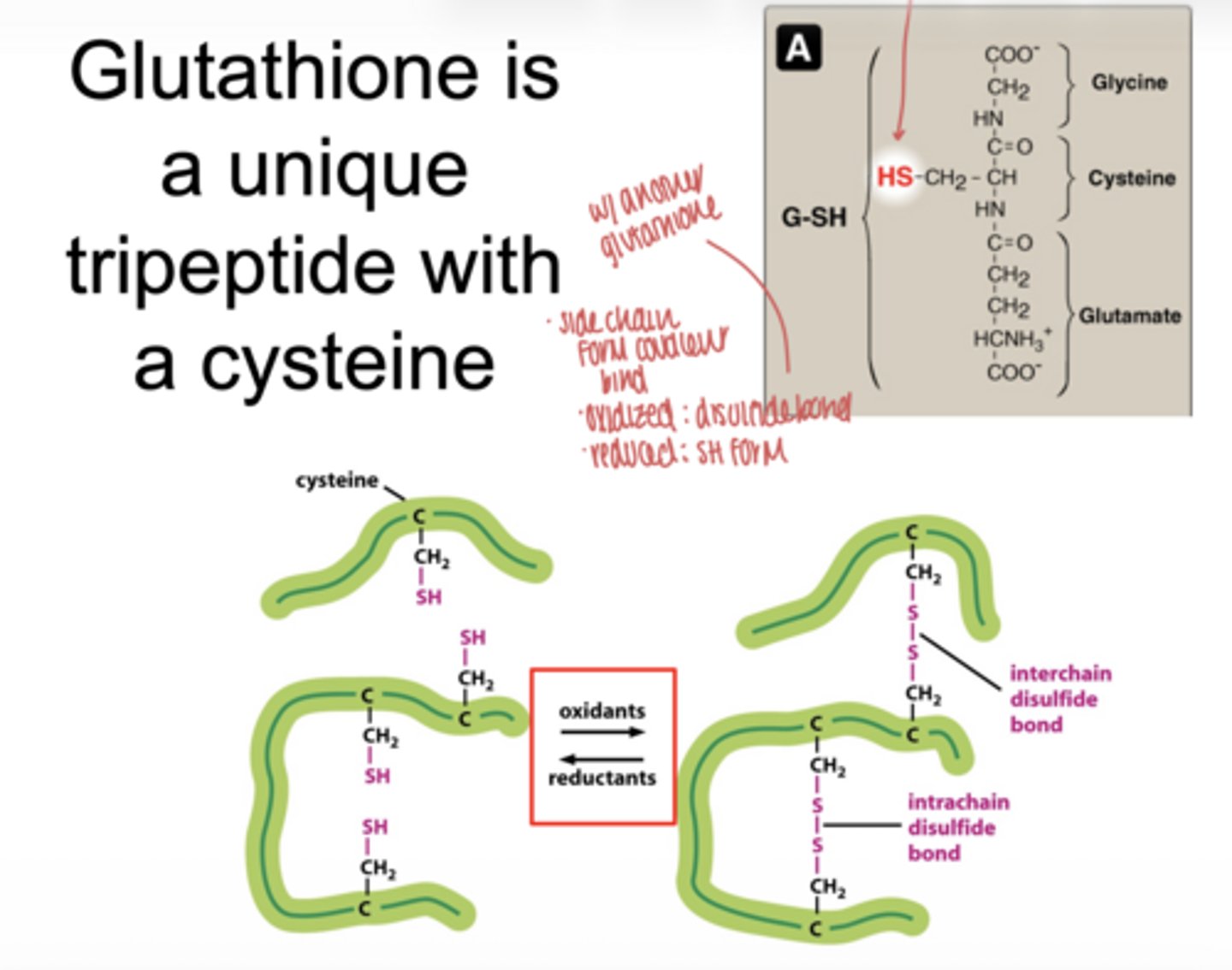
what functional group makes glutathione important
the -SH (thiol) group on cysteine
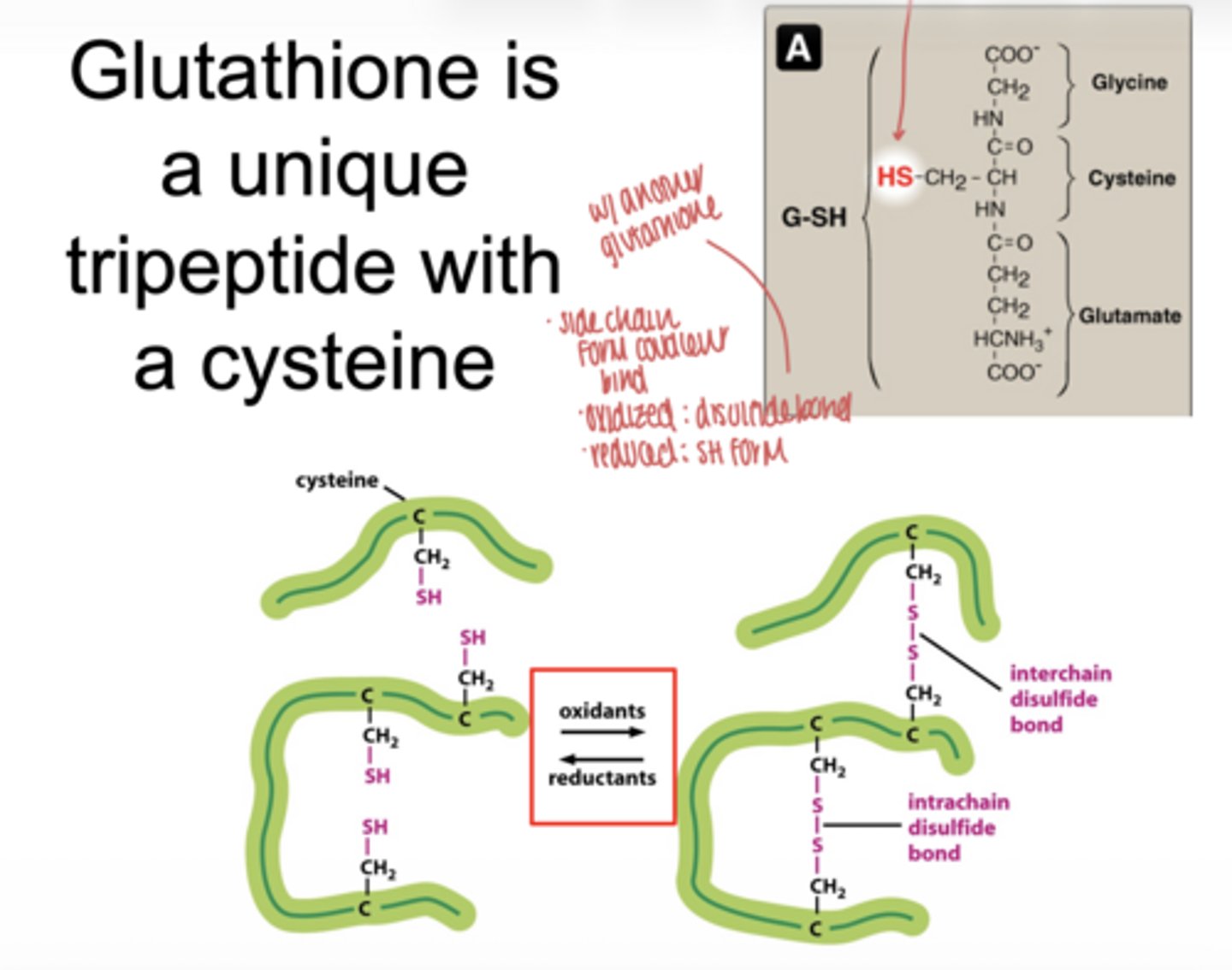
what happens when glutatione is oxidized
forms a disulfide bond (GSSG)
what happens when glutathione is reduced
exists as two separate SH groups (GSH)
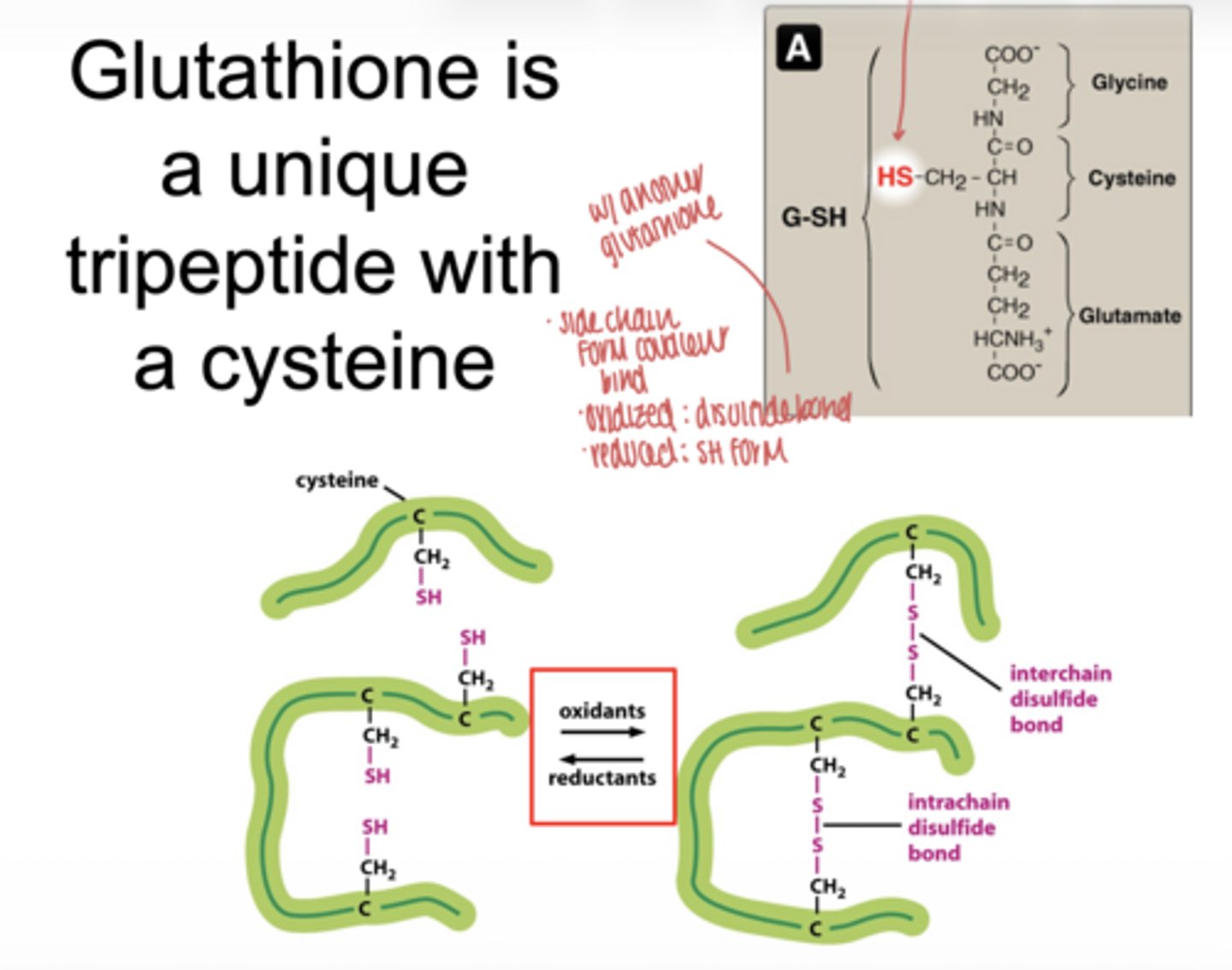
what enzyme reduces oxidized glutathione (GSSG)
glutathione reductase
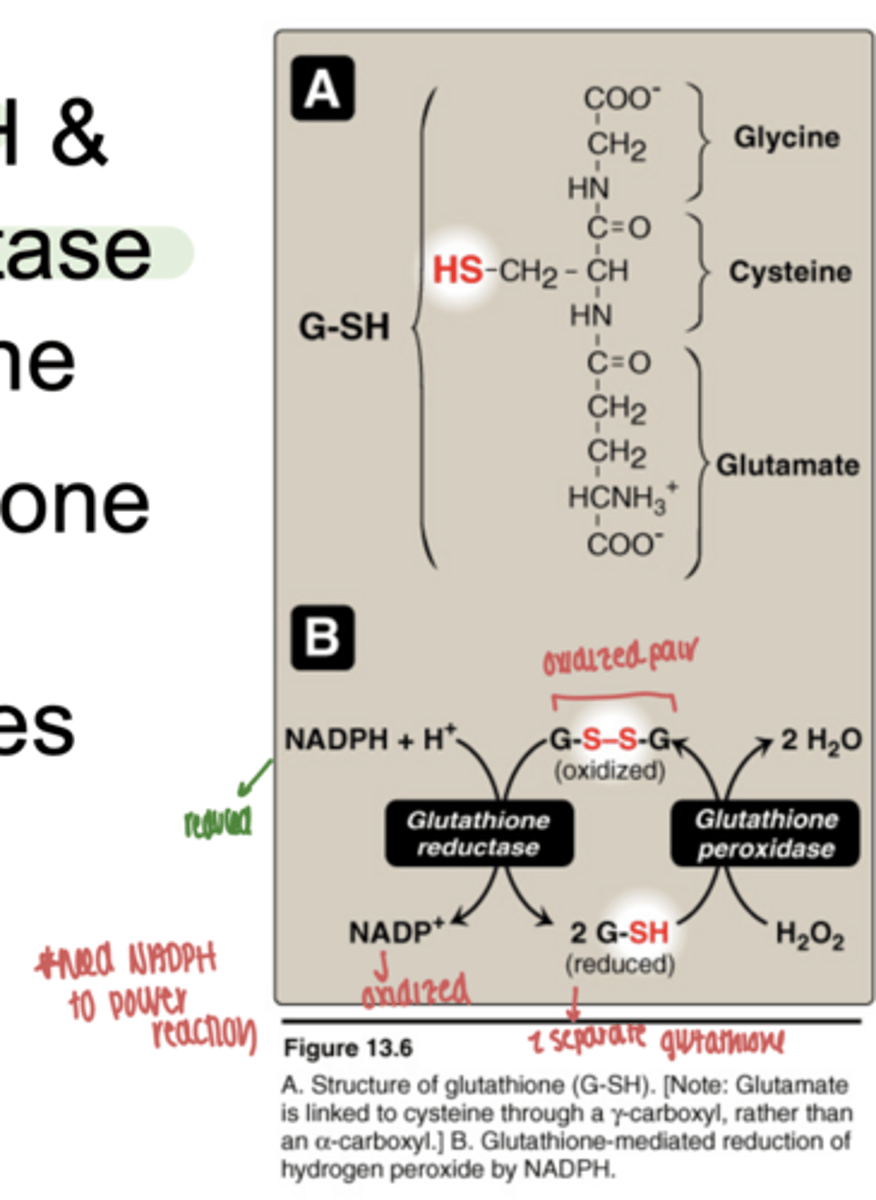
what molecule provides the reducing power for this reaction
NADPH
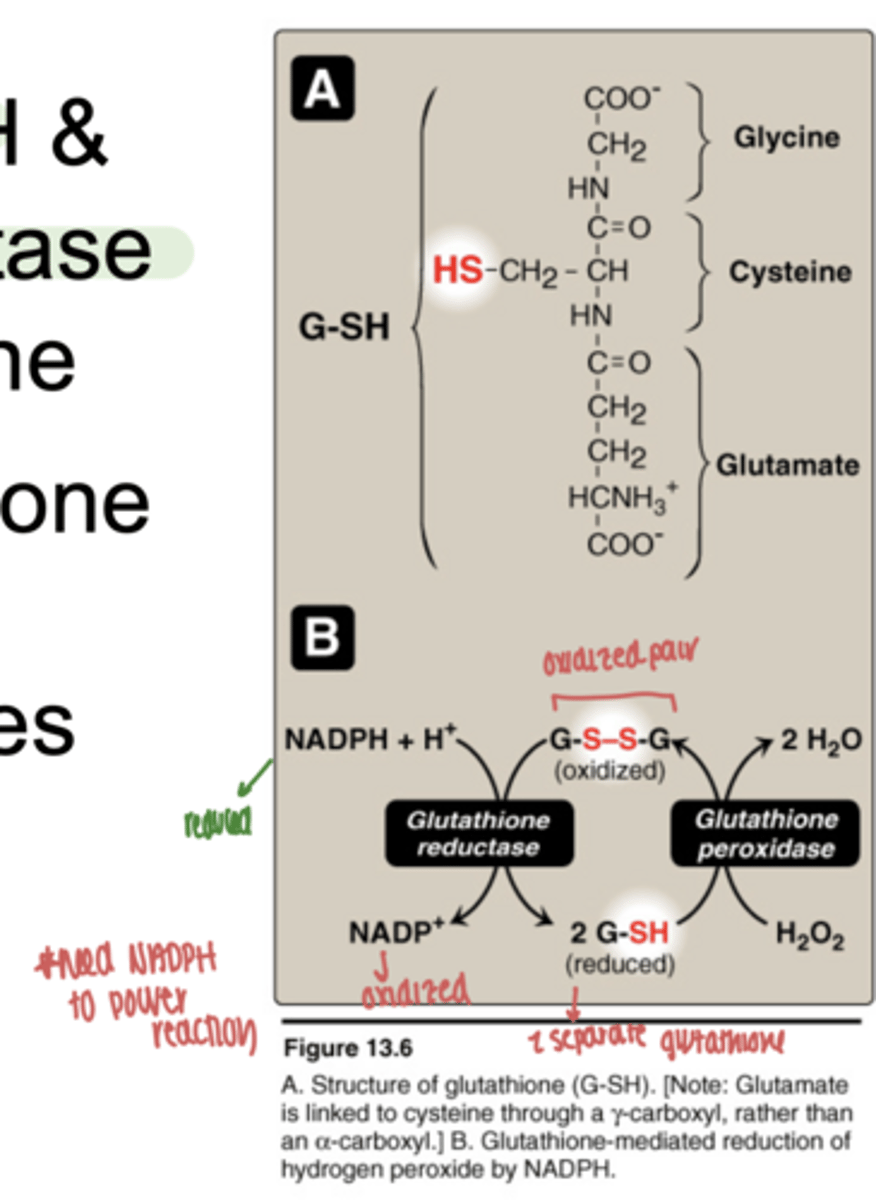
what enzyme uses glutathione to detoxify ROS
glutathione peroxidase
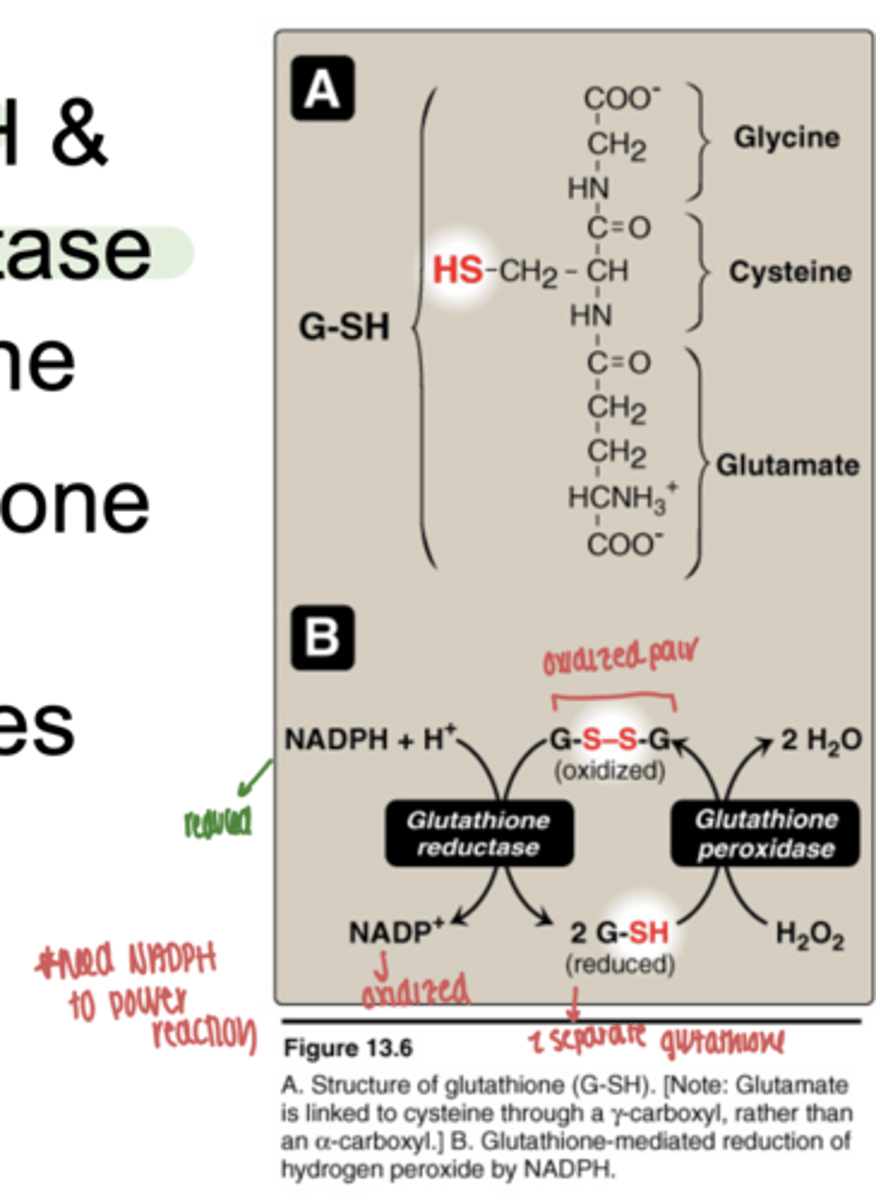
what reaction does glutathione peroxidase perform
converts H₂O₂ → H₂O
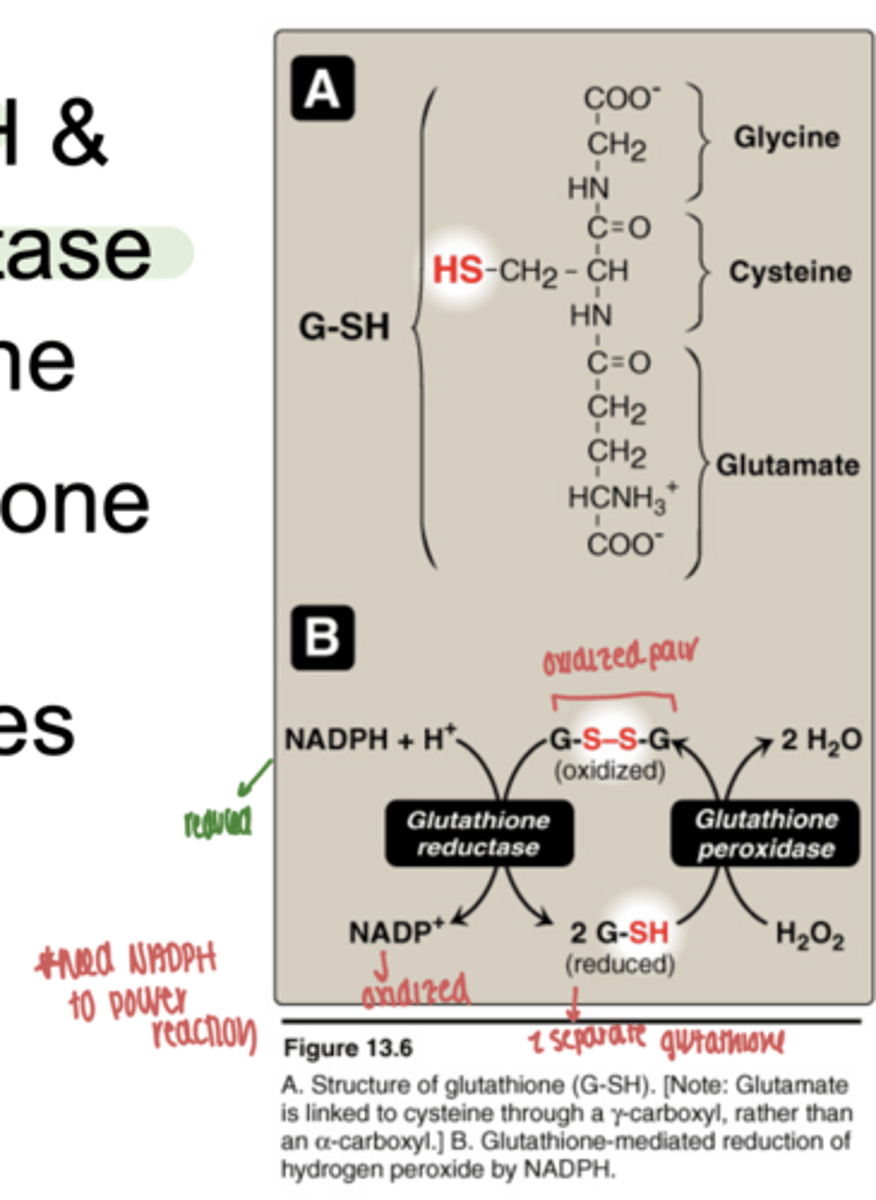
why is NADPH essential here
continously regenerates reduced glutathione
what happens if NADPH is low
- cannot regenerate glutathione
- ROS accumualtes
- leads to cell damage
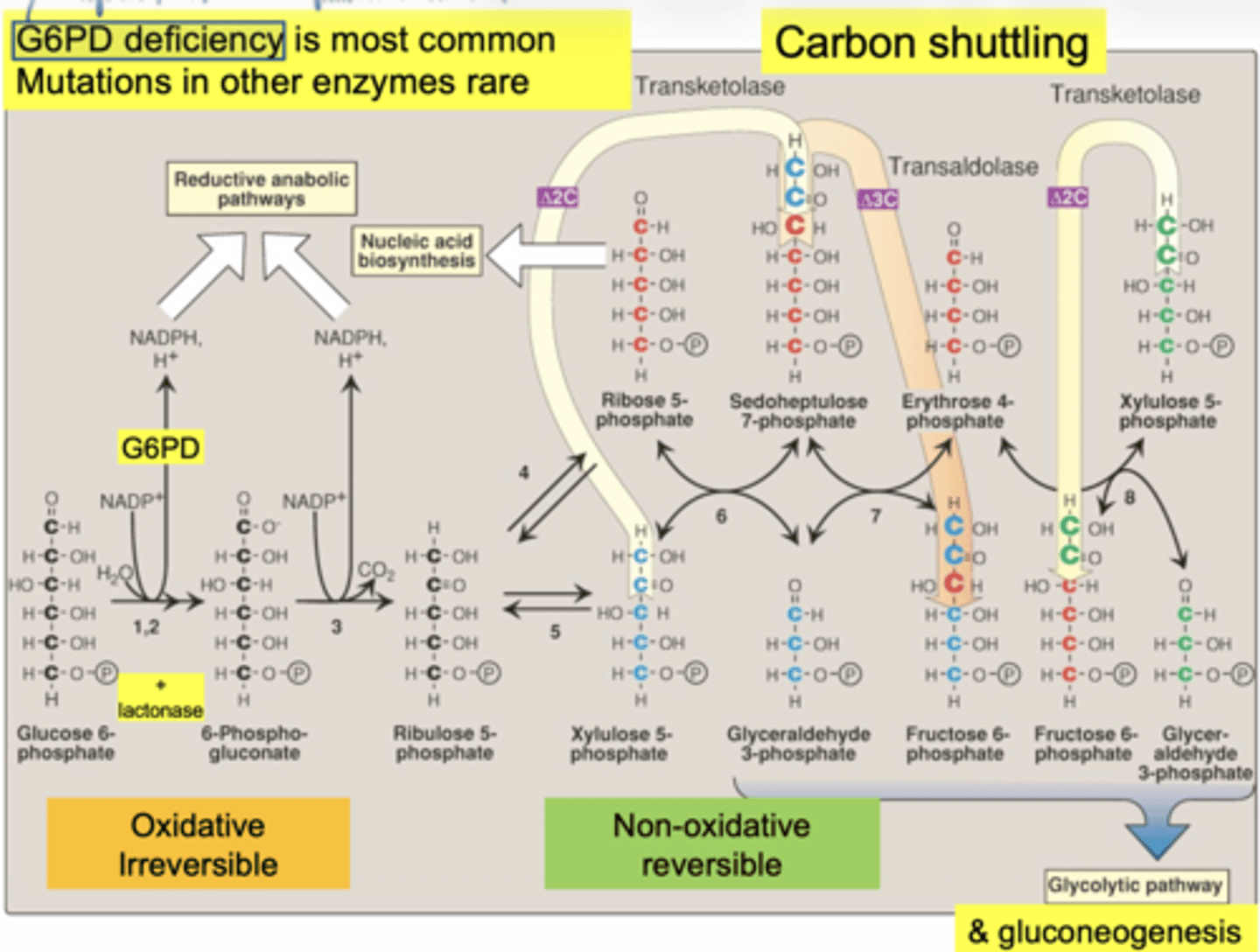
what molecule enters the PPP (oxidative phase start)
glucose-6-phosphate (G6P)
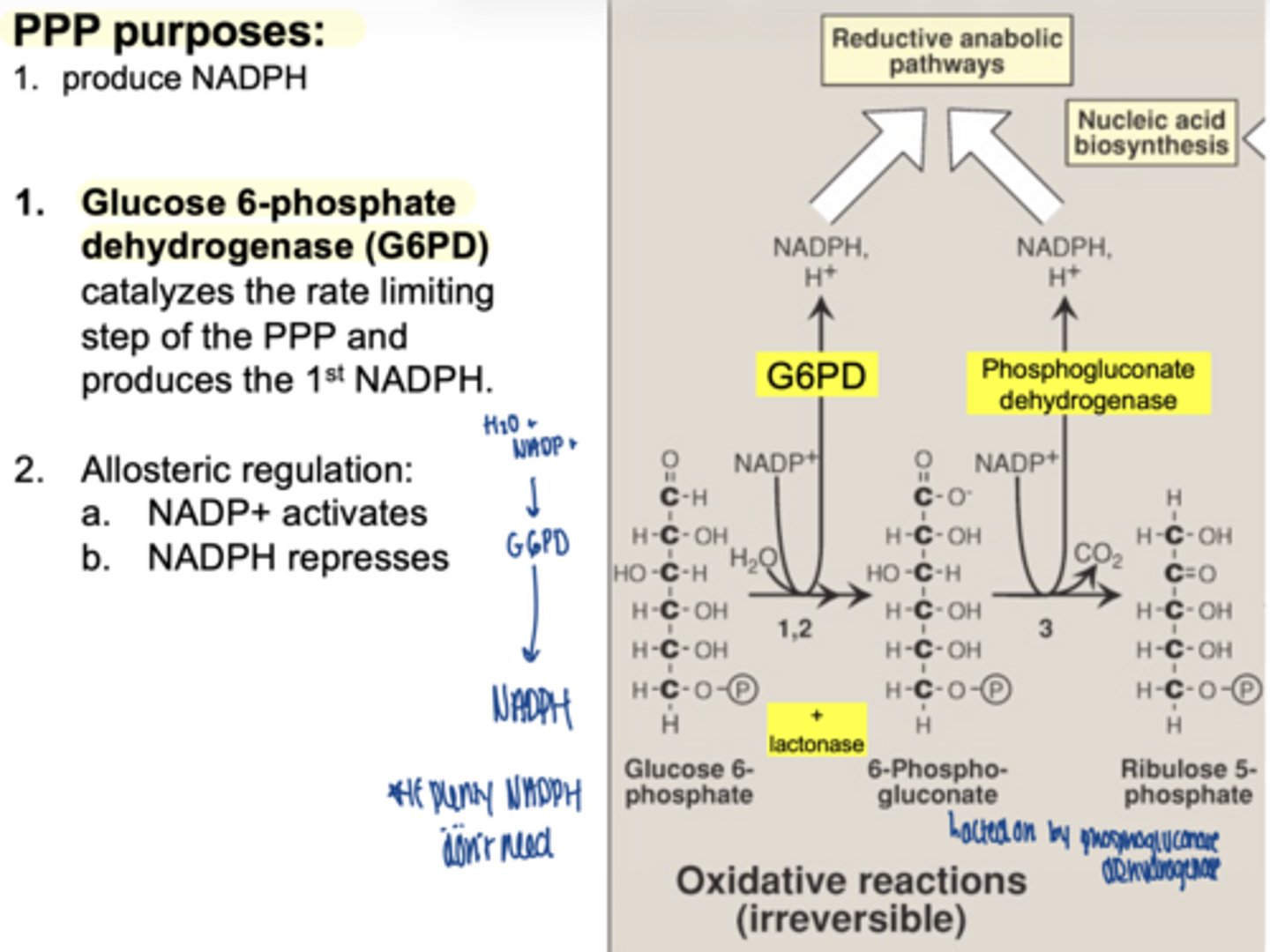
what is the first enzyme of PPP
glucose 6 phosphate dehydrogenase (G6PD)
what does G6PD produce
1 NADPH
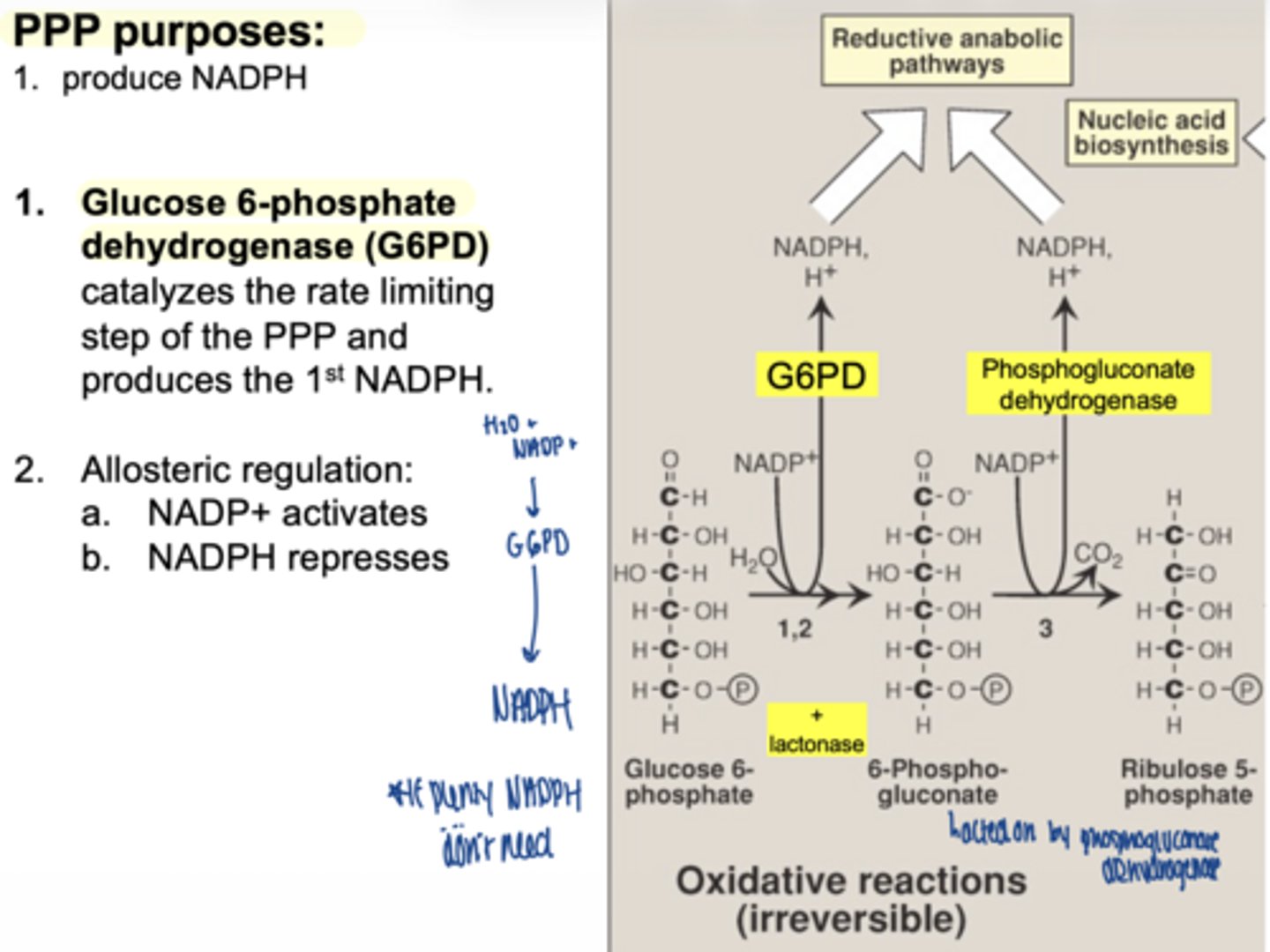
why is G6PD important
it is the rate-limiting enzyme
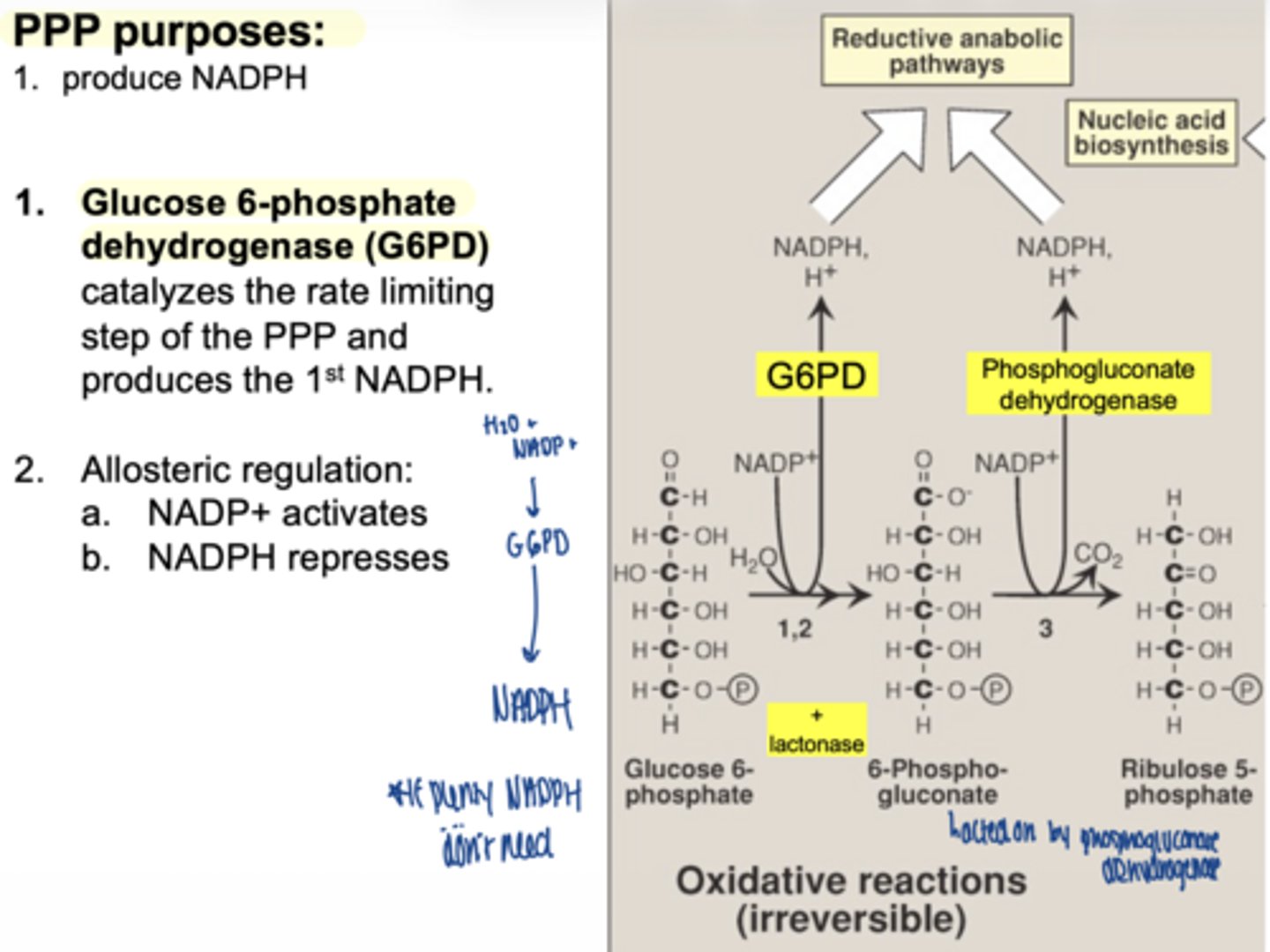
what activates G6PD
NADP+ (allosteric)
what inhibits G6PD (allosteric)
NADPH
hormonal regulation of PPP
insulin activates it
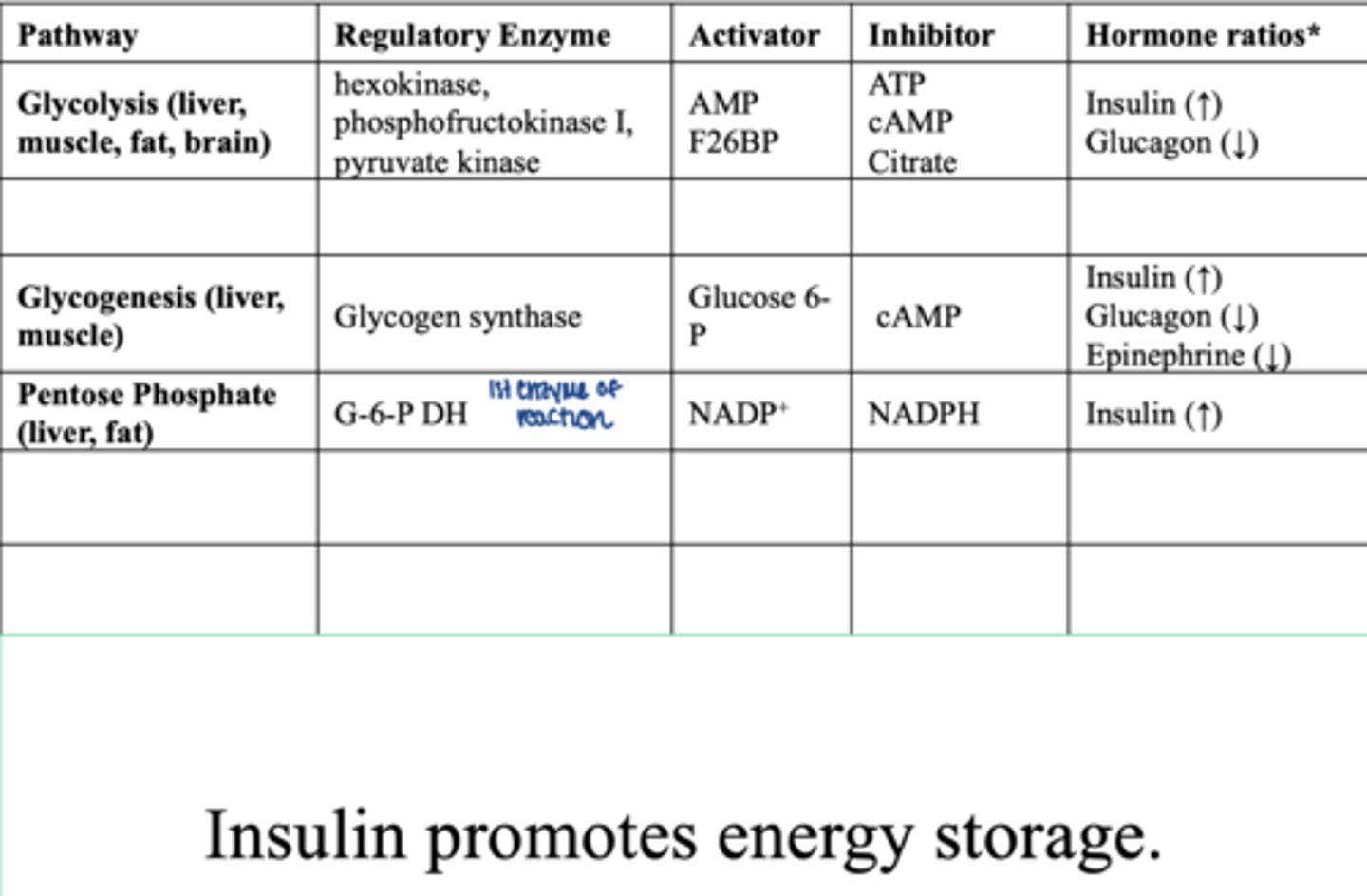
pentose phosphate pathways (liver, fat)
regulatory enzyme:
- G-6-P DH
activator:
- NADP+
inhibitor:
- NADPH
hormones:
insulin (increases)... promotes energy storage
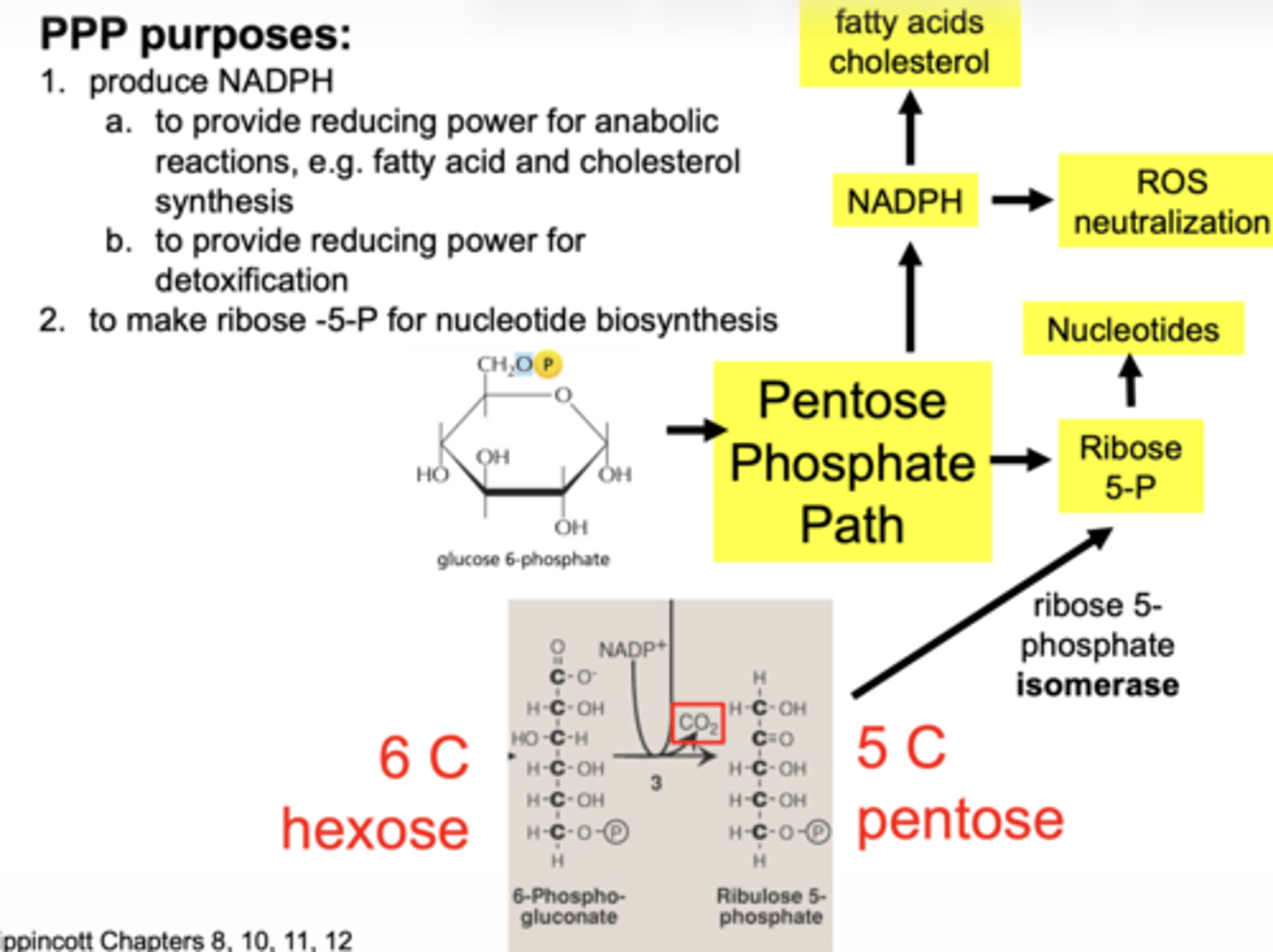
what happens in the second oxidative step
another NADPH is produced
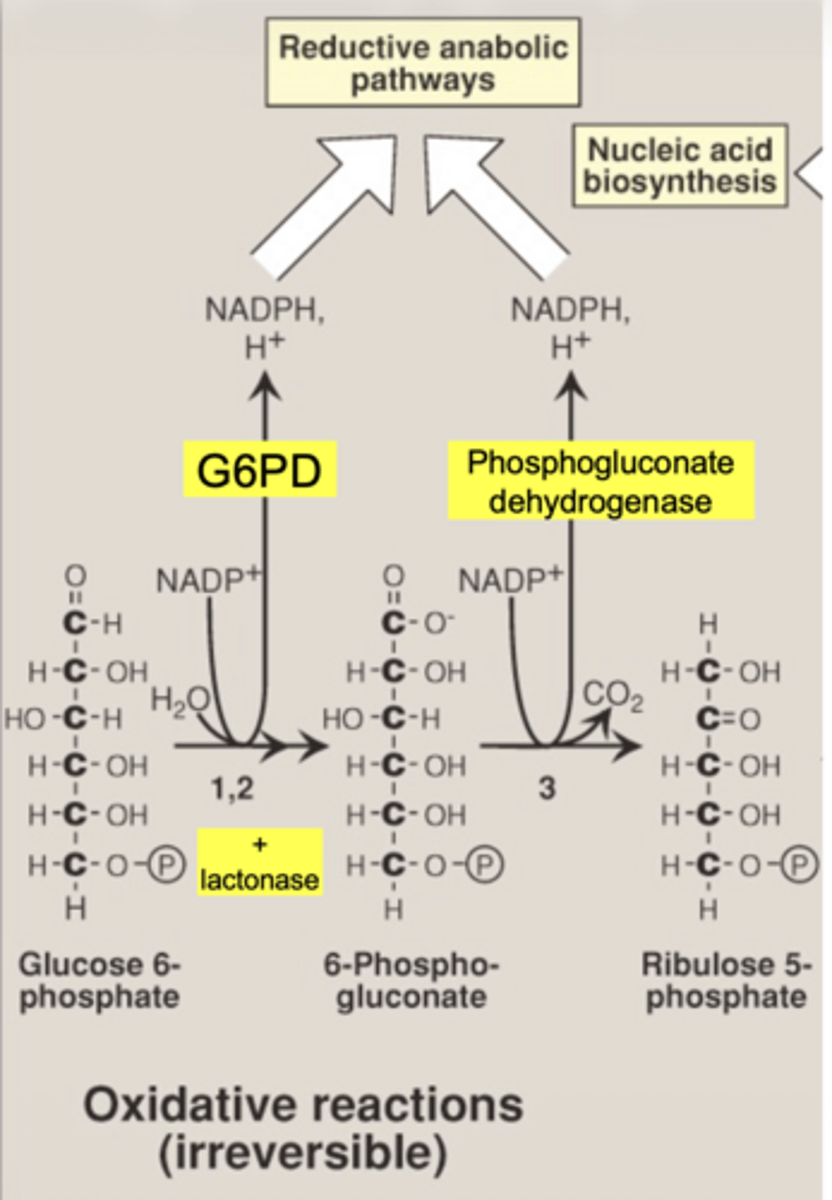
what enzyme produces the second NADPH
6-phosphogluconate dehydrogenase
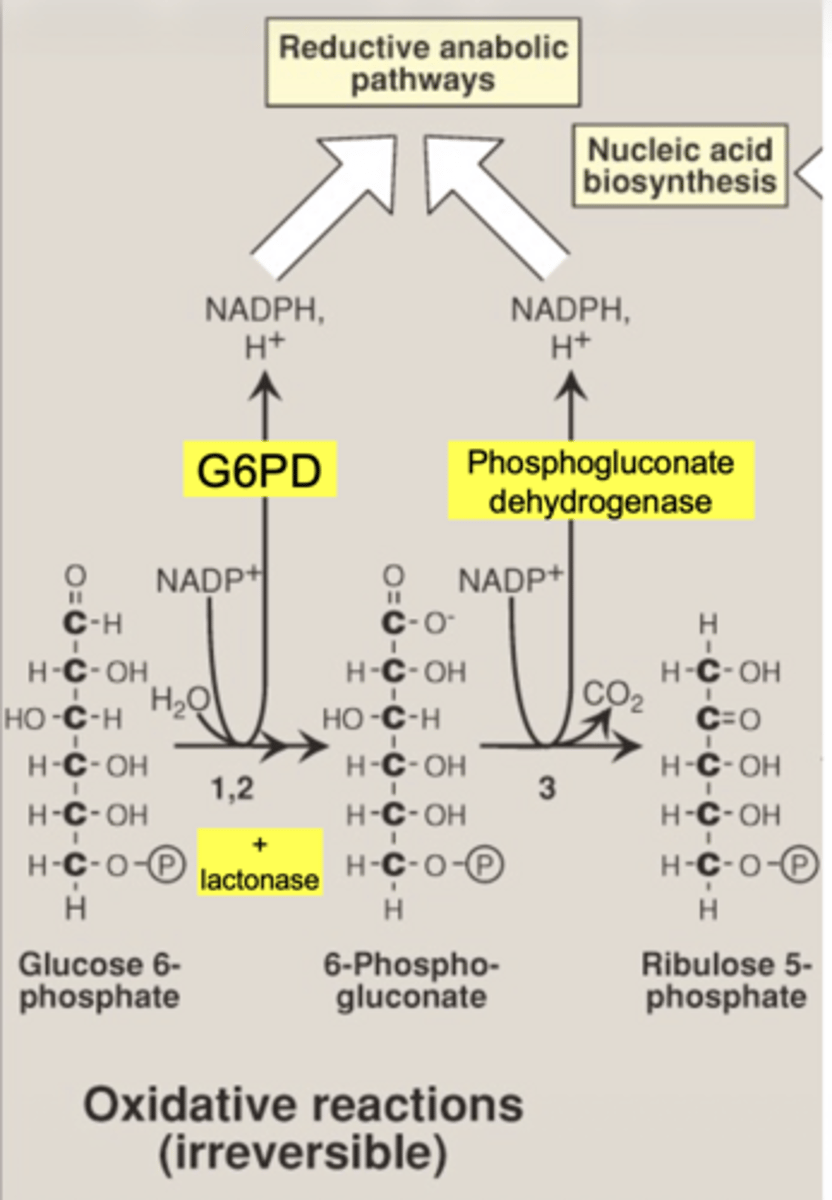
what important structural change occurs here
6-carbon sugar → 5-carbon sugar
(Hexose → Pentose)
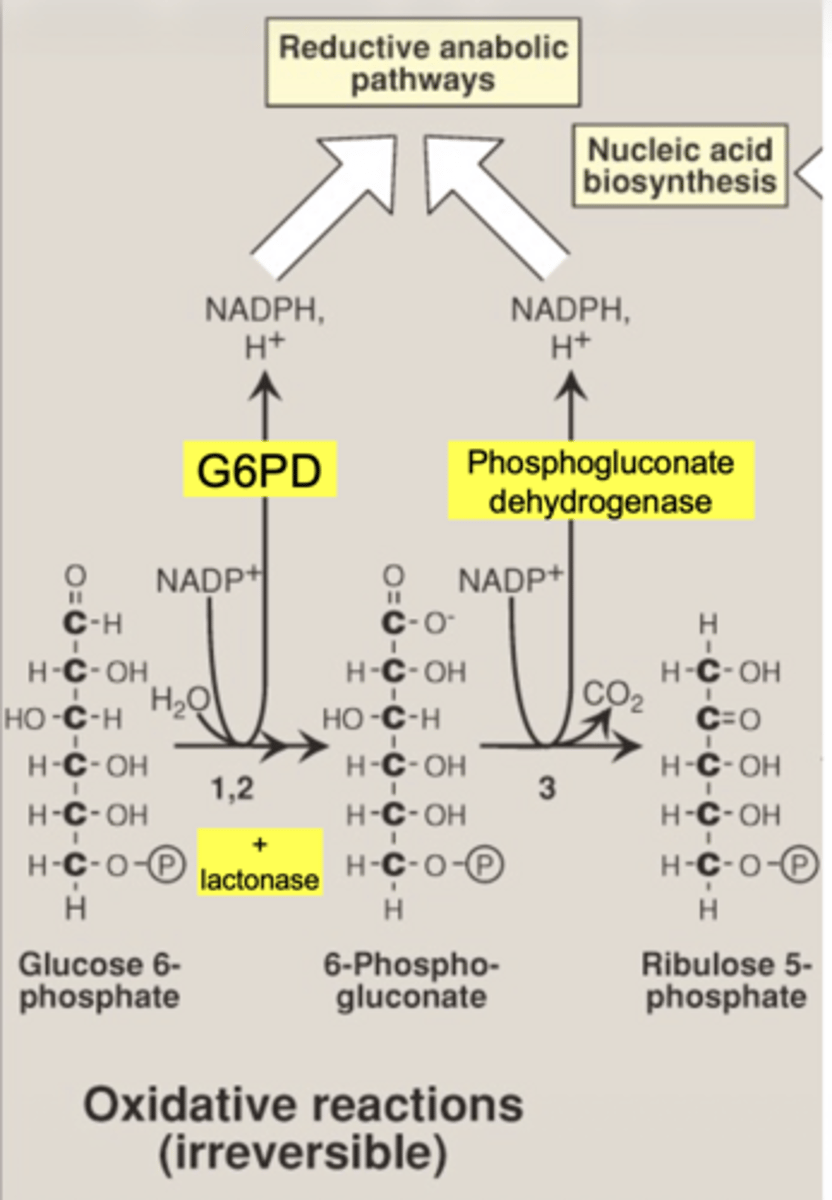
what are the 2 main products of oxidative phase
2 NADPH
ribose-5-phosphate
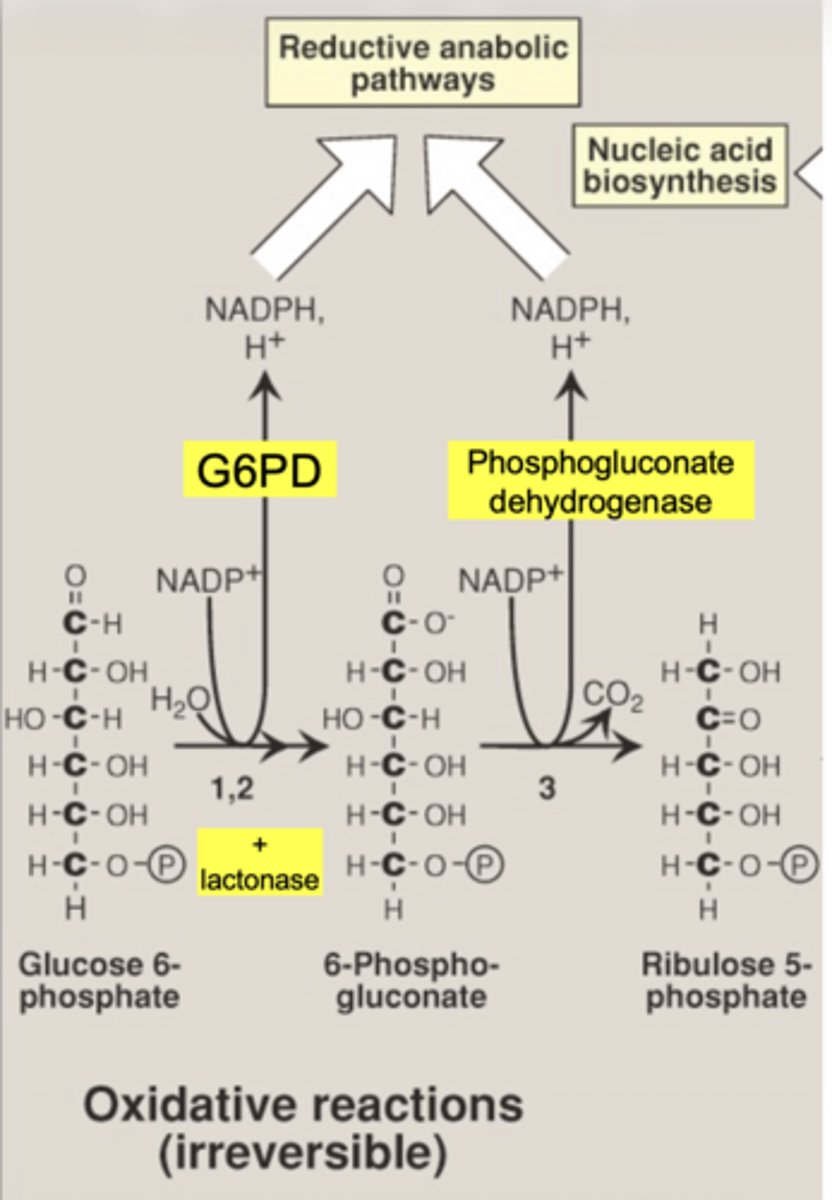
is the oxidative phase reversible
no... irreversible
why is ribose-5-phosphate important
it is used to make nucleotides (DNA/RNA)
is the non-oxidative phase reversible or irreversible
reversible
what is the main function of this phase
carbon shuttling between sugars
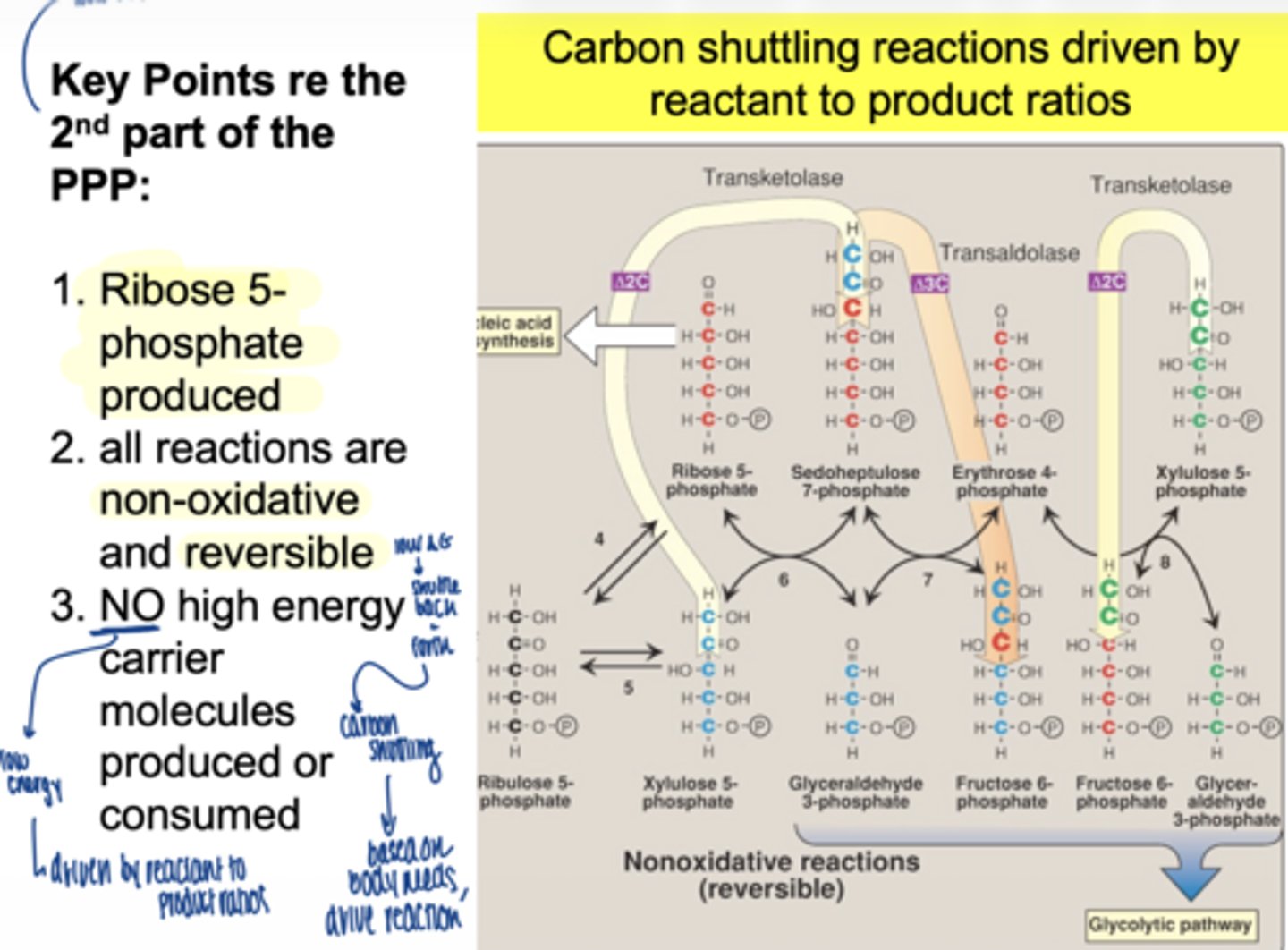
does the second part of PPP (non-oxidative) produce NADPH
no
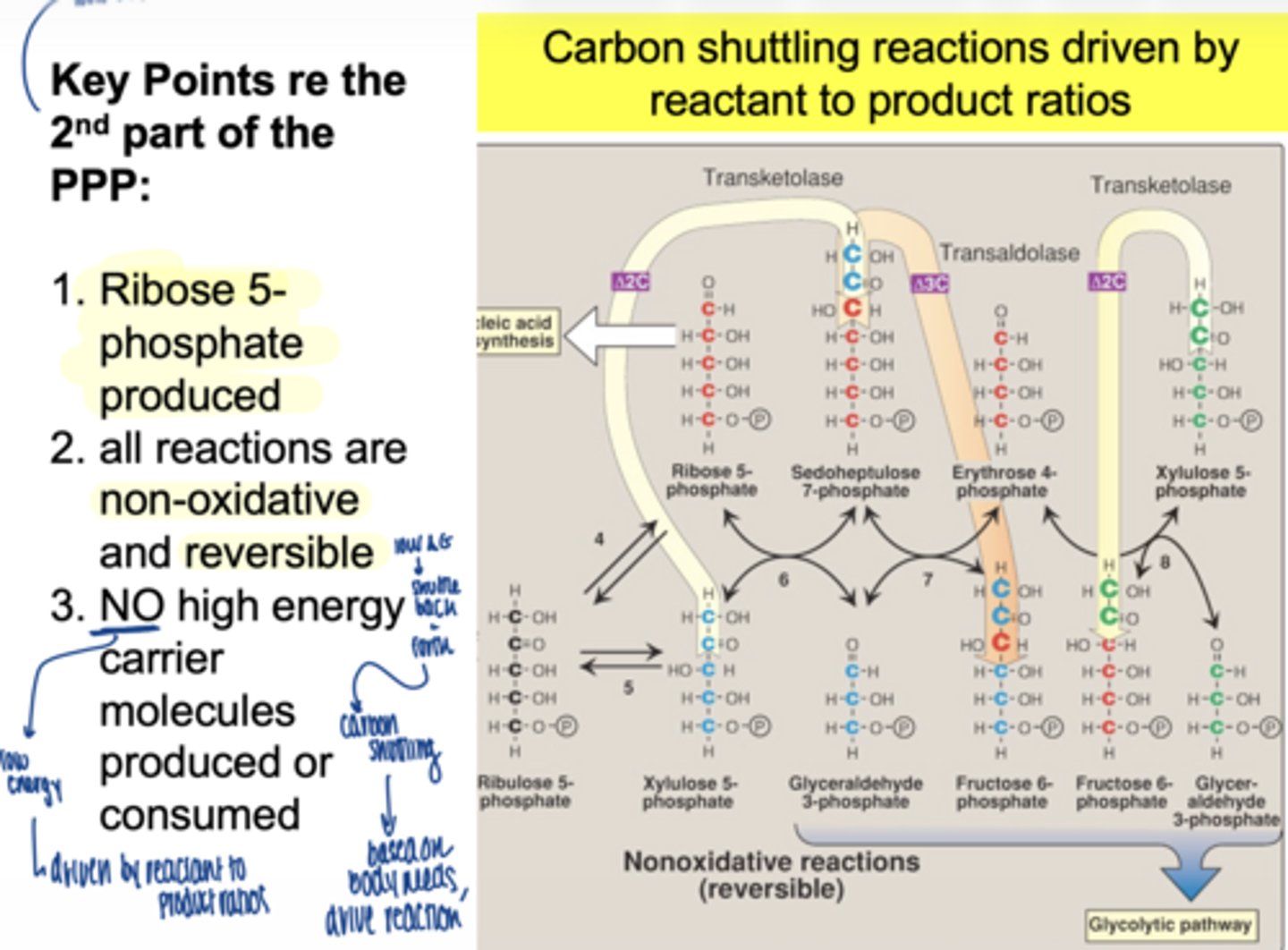
does it use ATP
no
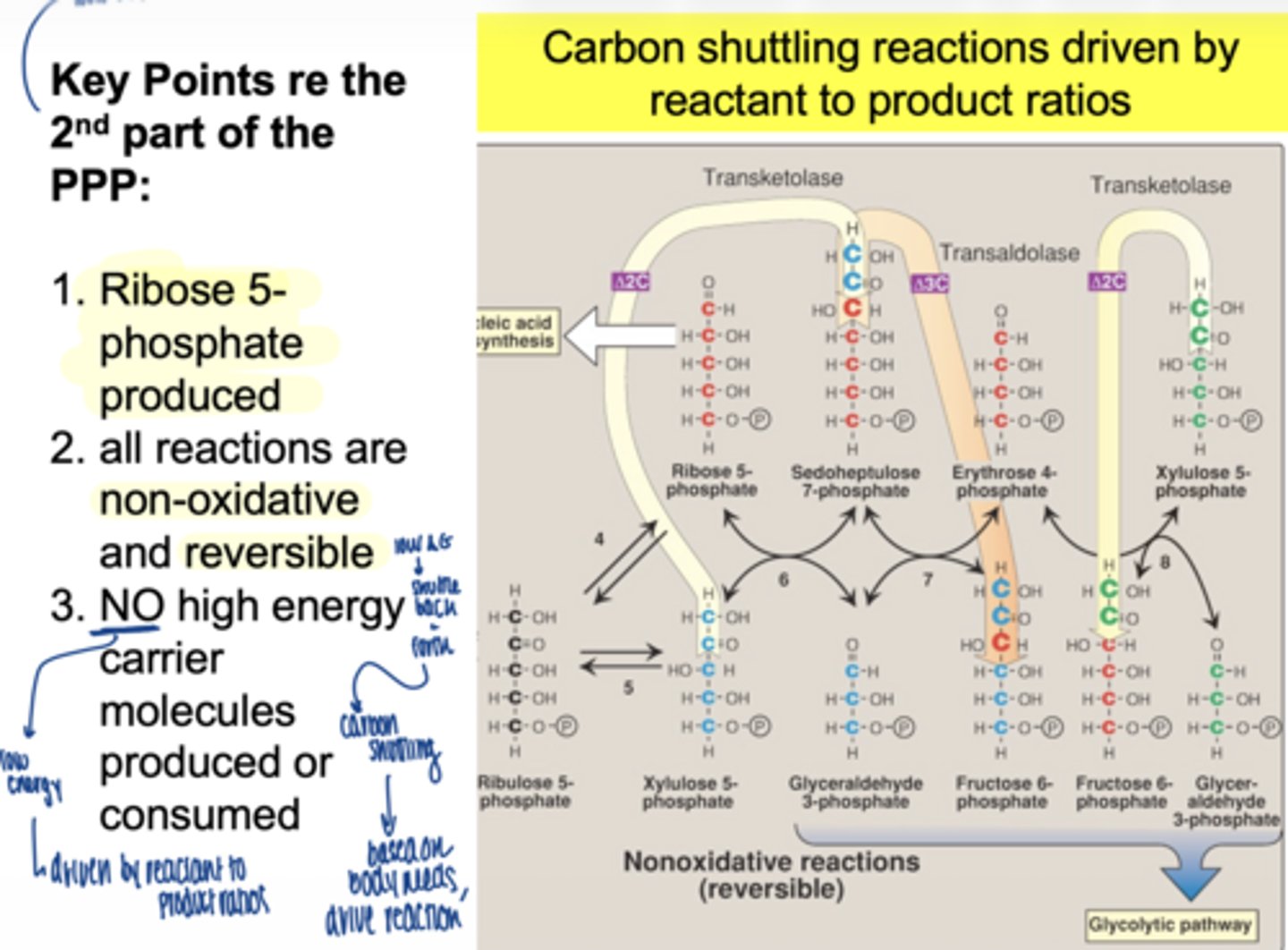
no high ____ ____ molecules produced or consumed in non-oxidative PPP
energy carrier
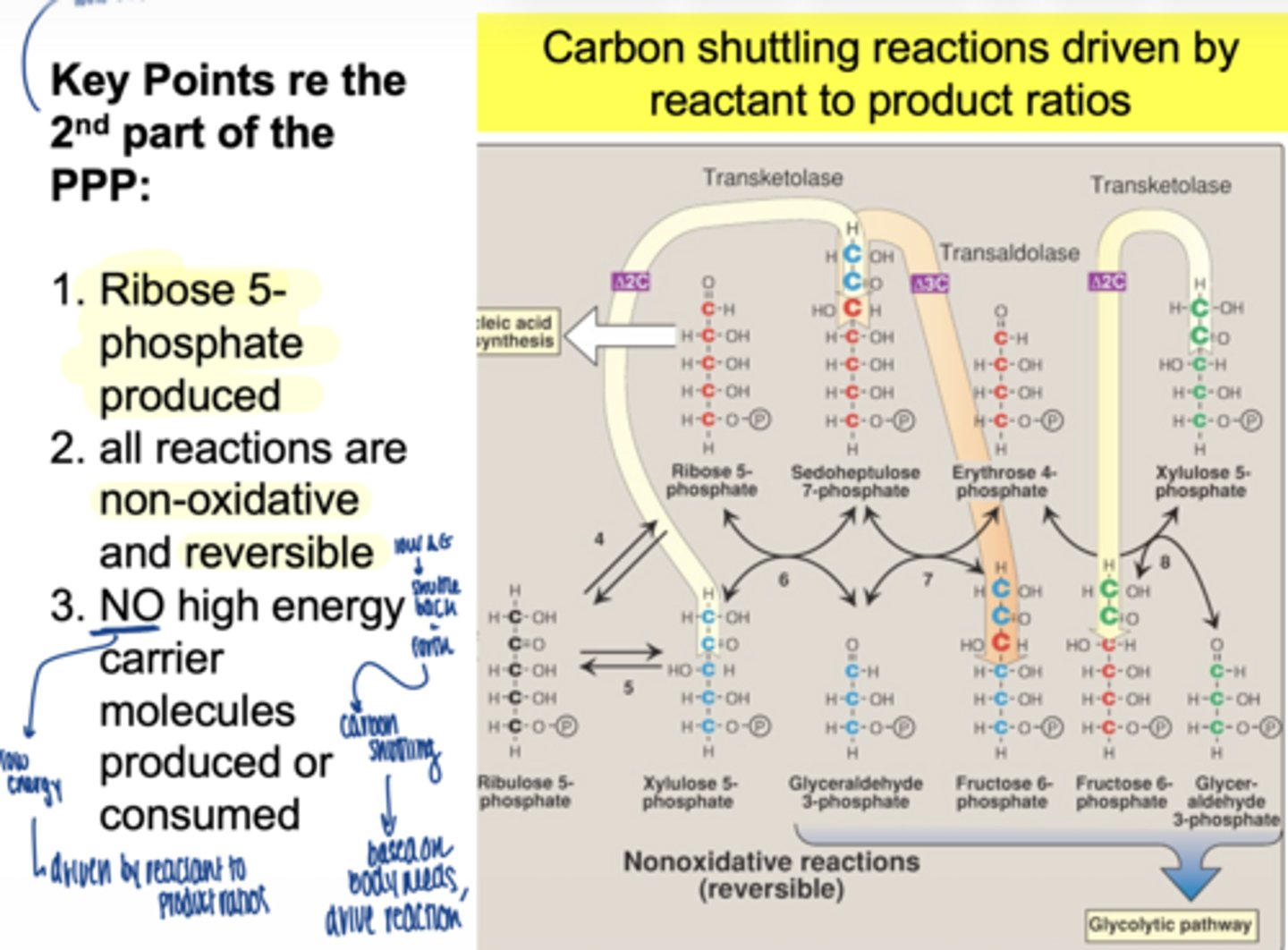
what drives the non-oxidative phase
mass action (reactant/product ratios)
what glycolysis intermediates are produced (non-oxidative)
fructose-6-phosphate
glyceraldehyde-3-phosphate
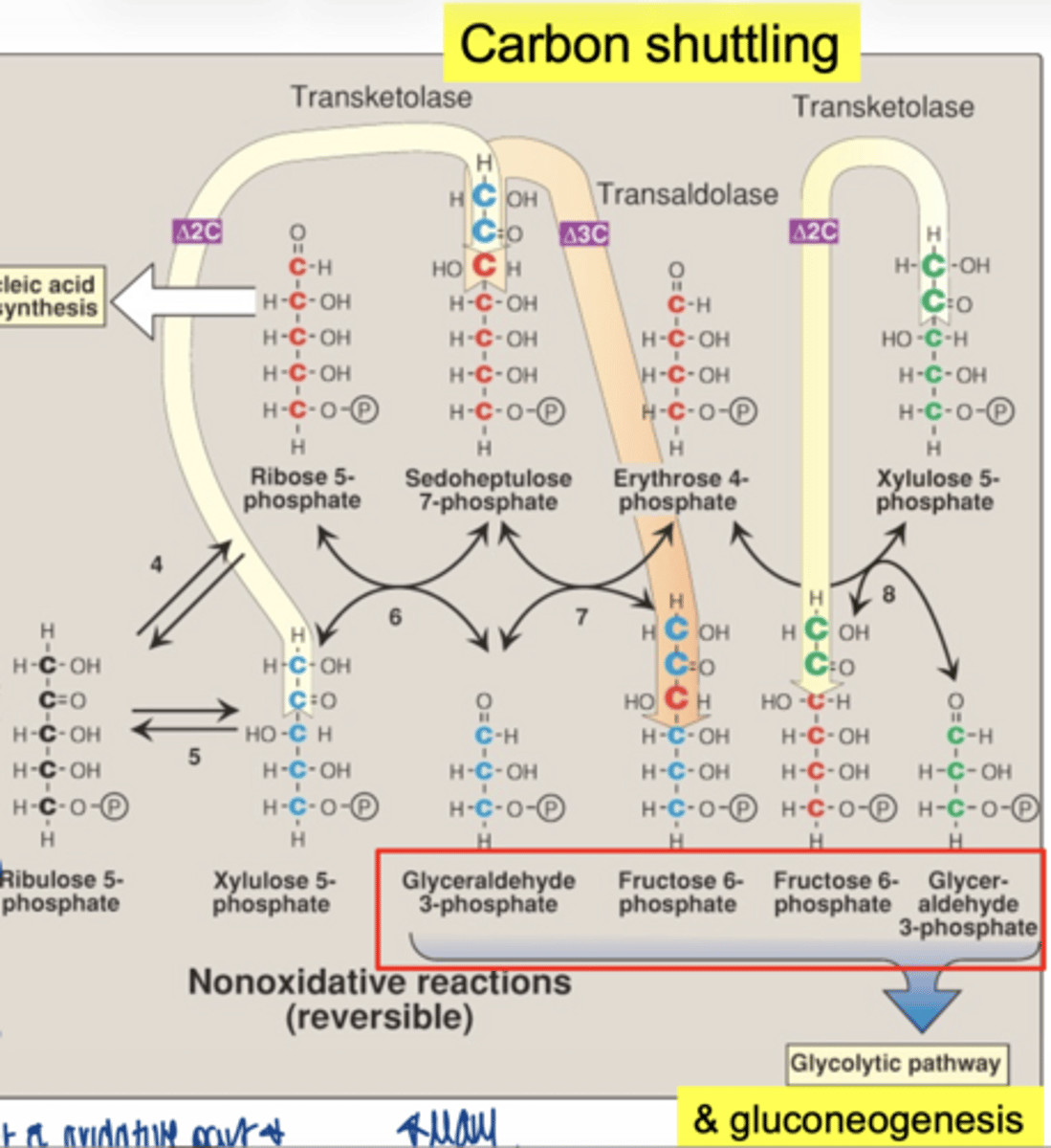
why are the intermediates important
PPP connects to:
- glycolysis
- gluconeogenesis
**feed into this pathways as needed**
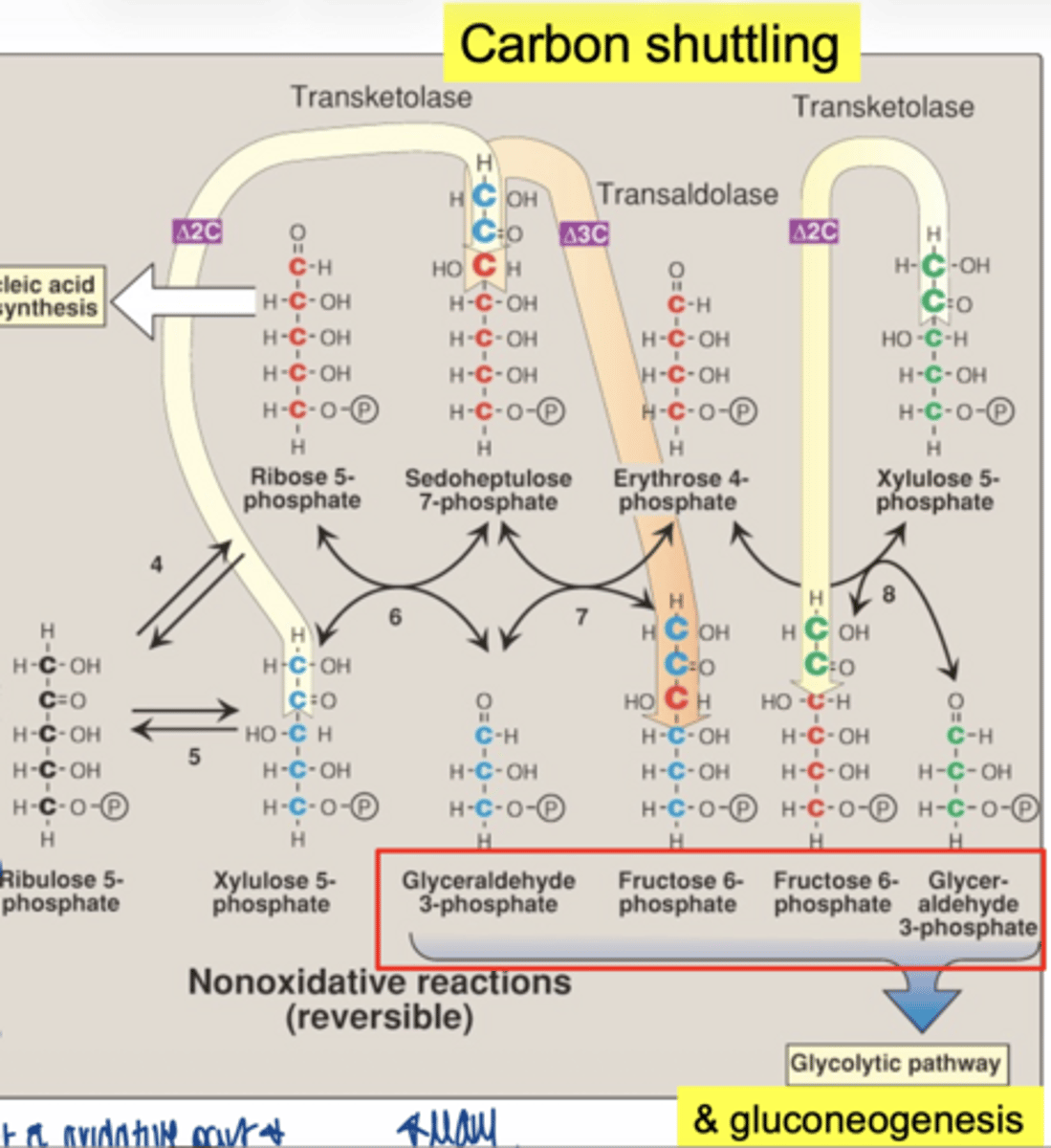
key difference between oxidative vs non-oxidative PPP
oxidative:
- NADPH produced
- irreversible
- function = NADPH + ribose
non-oxidative:
- NADPH not produced
- reversible
- function = carbon shuffling
what is the most common PPP disorder
G6PD deficiency... mutations in other enzymes are rare
why are red blood cells especially vulnerable
- no mitochondria
- PPP is their ONLY source of NADPH
**rely solely on cytosolic paths: glycolysis & pentose phosphate path**

G6PD protects against oxidative damage from...
1. naturally oxidative compounds (fava beans, henna)
2. oxidative drugs (ex. anti-malarials, antibotics, analgesics)
3. inflammatory injections
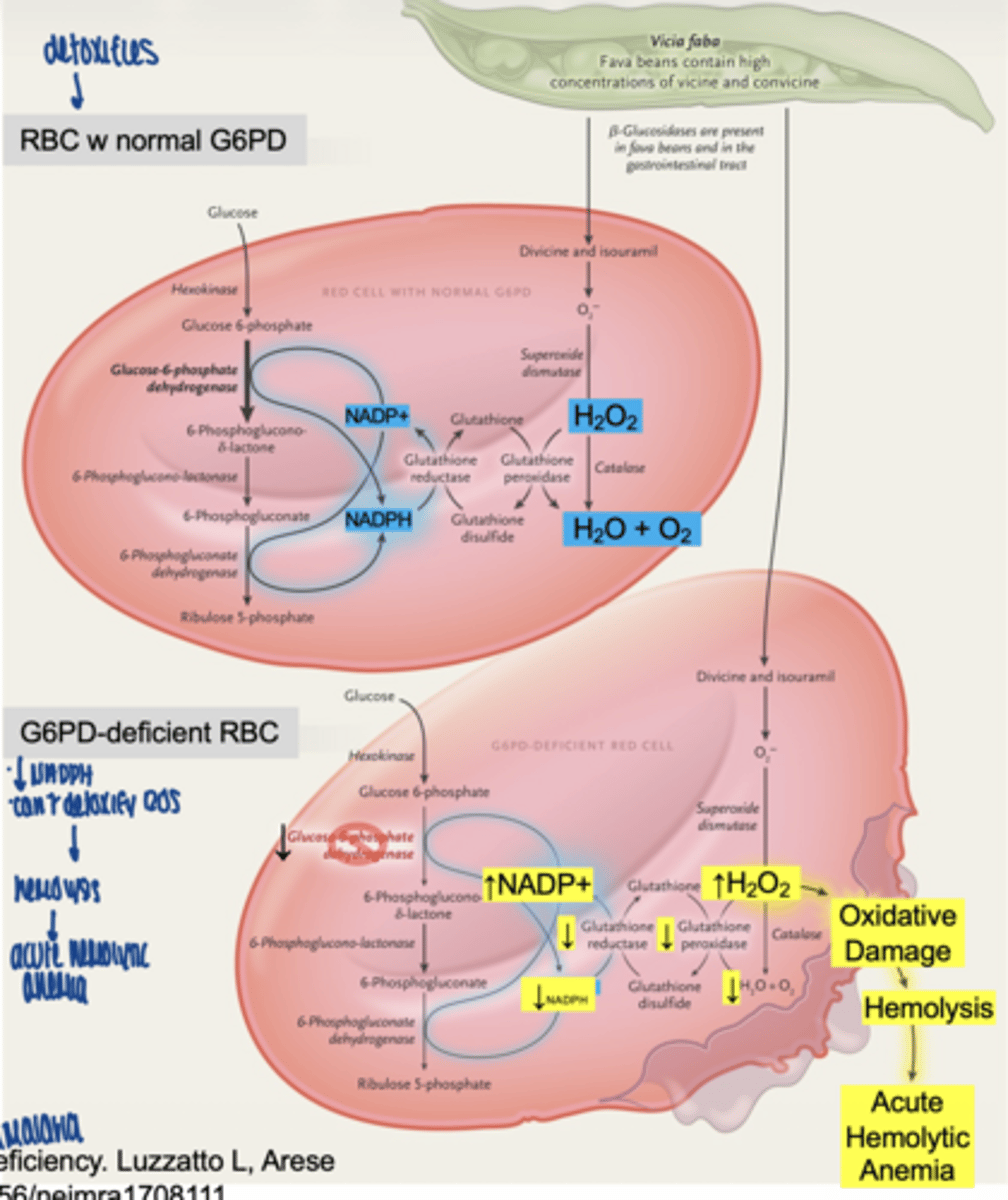
what happens in G6PD deficiency
↓ NADPH
↓ glutathione
↑ ROS
→ hemolysis
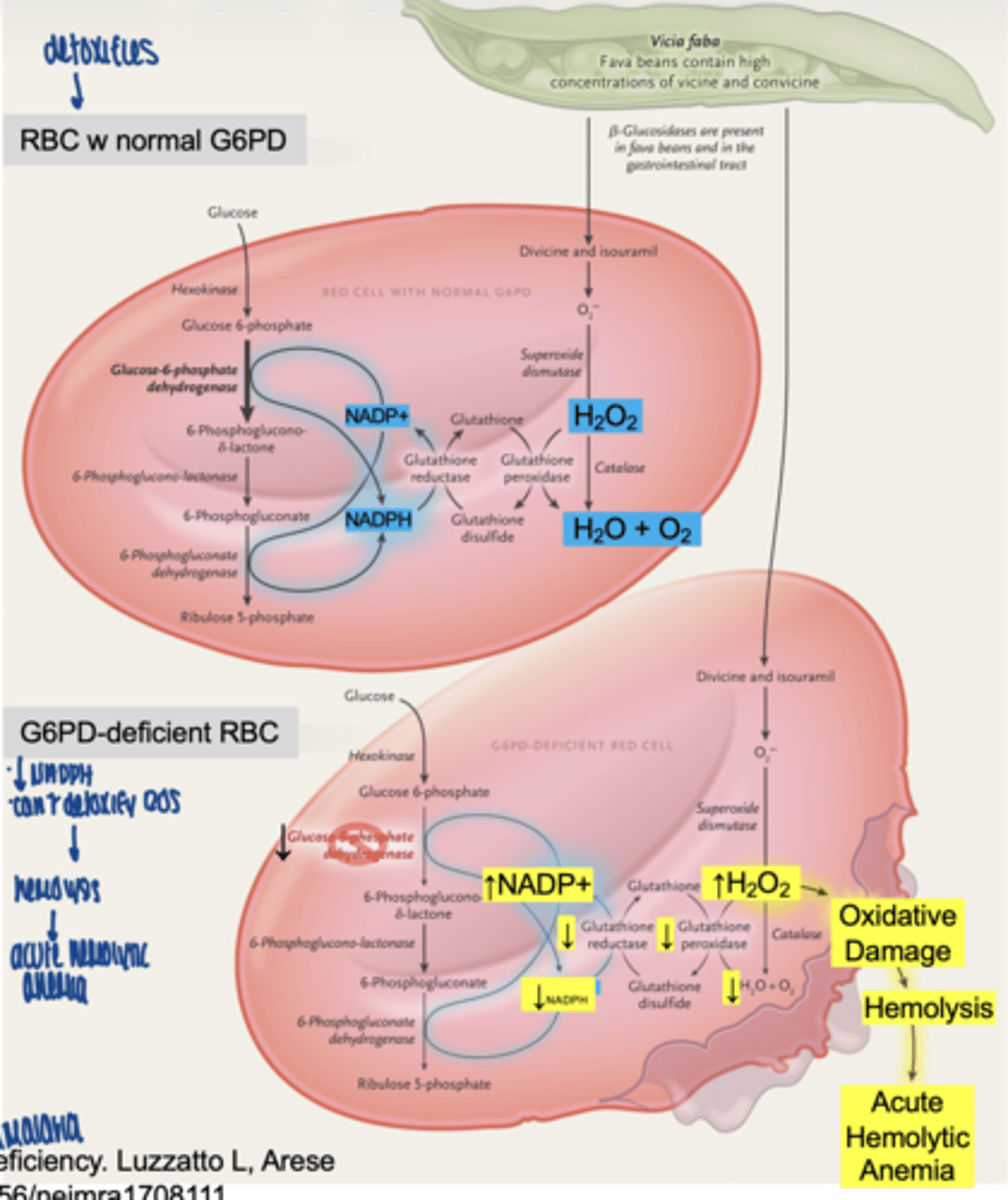
what can trigger hemolysis in G6PD deficiency
- fava beans
- drugs (antimalarials, antibiotics)
- infections
what condition results from G6PD deficiency
acute hemolytic anemia
why is G6PD deficiency maintained in populations
provides protection against malaria
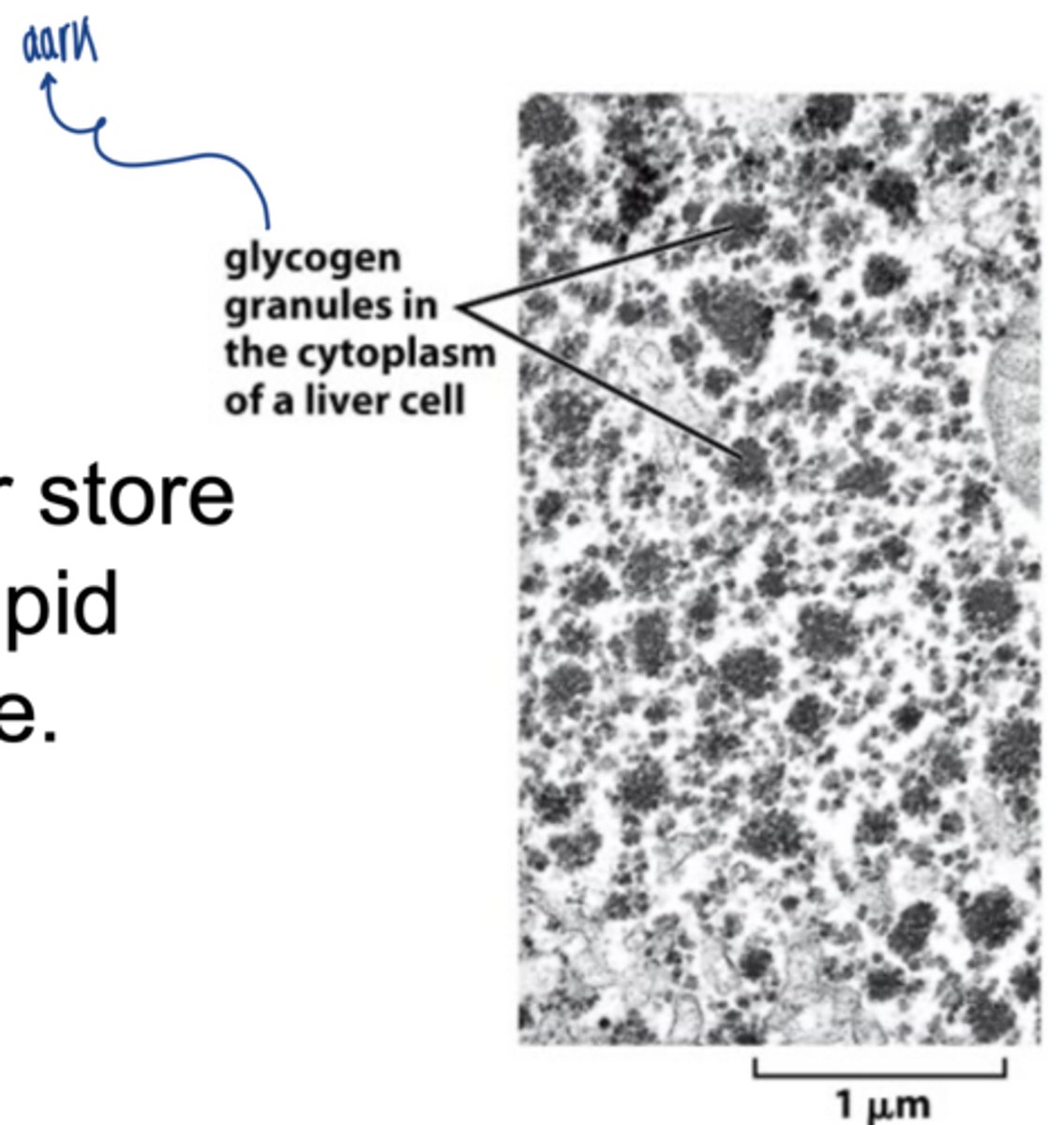
what is glycogen
branched polymer of glucose used for energy storage in animals
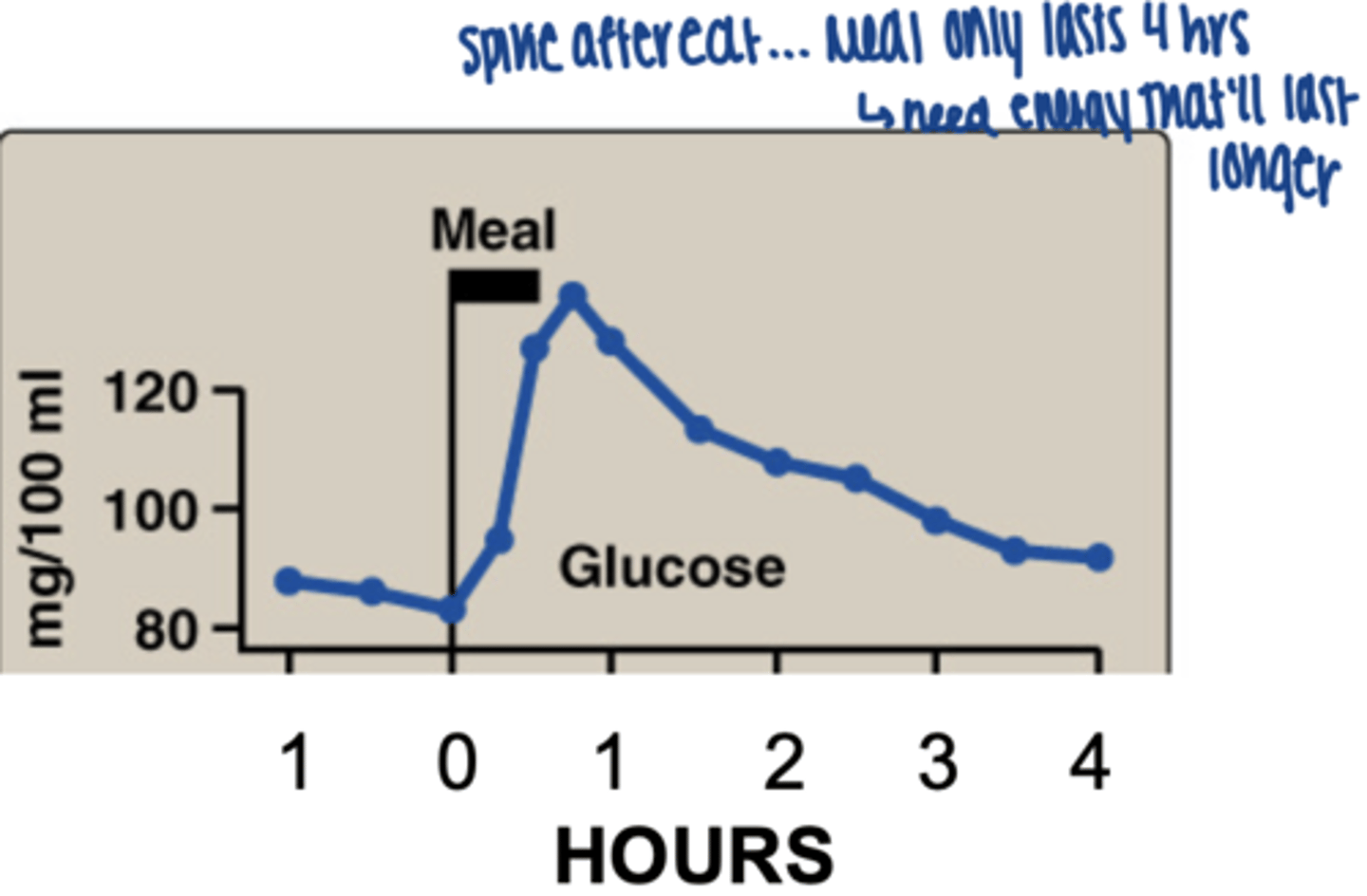
glucose from a meal only lasts hours in bloodstream
Longer term fuels needed:
• Glycogen (Carbohydrates = 4 kcal/g)
• Fat = 9 kcal/g)
• Protein = 4 kcal/g)
(Not good fuel, but alcohol = 7 kcal/g)
where is glycogen stored
primarily in:
liver → maintains blood glucose
skeletal muscle → used for muscle contraction
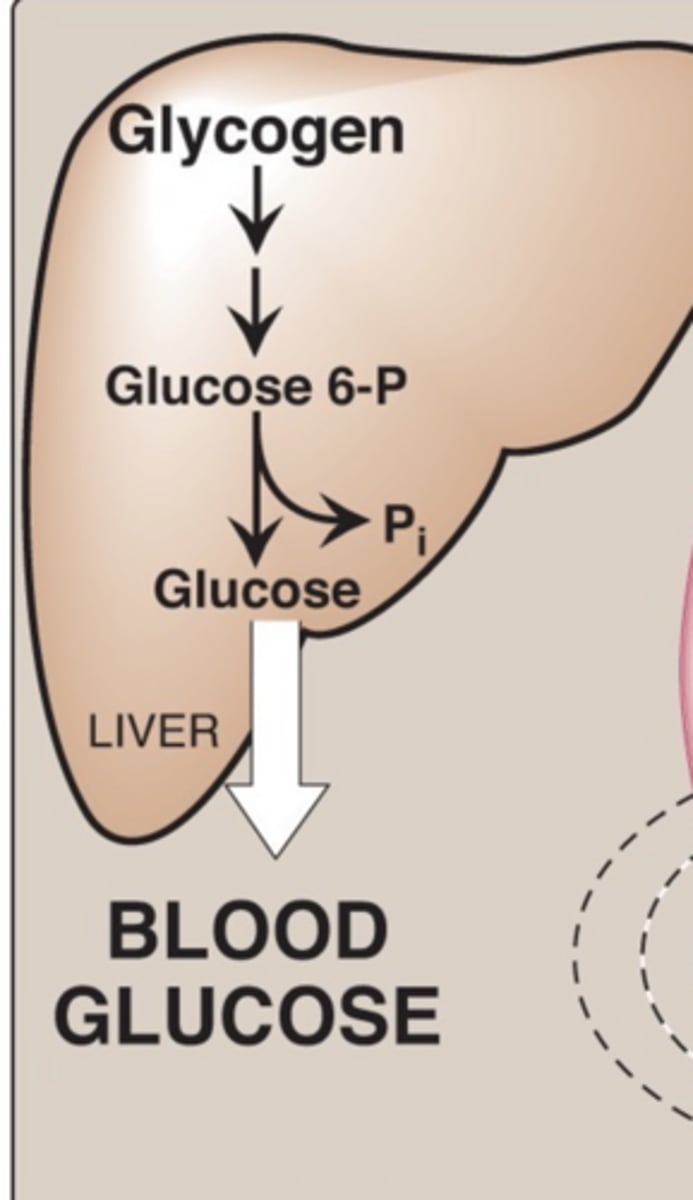
glycogen stored in ____ can maintain blood glucose levels for ~24 hrs; glycogen stores reflect fed or ___ state
liver, fasting
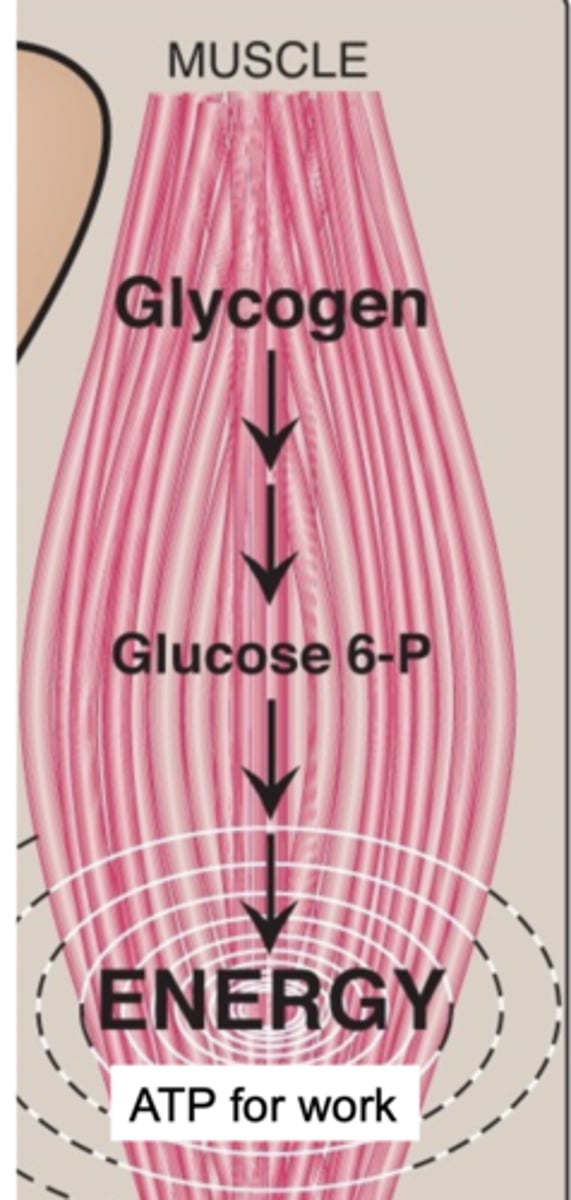
glycogen stored in muscle fuels ____ production for muscle contraction; glycogen stores reflect exercise intensity
ATP
glycogen → glucose 6-P → energy (ATP for work)
how many glucose molecules make up glycogen
55,000
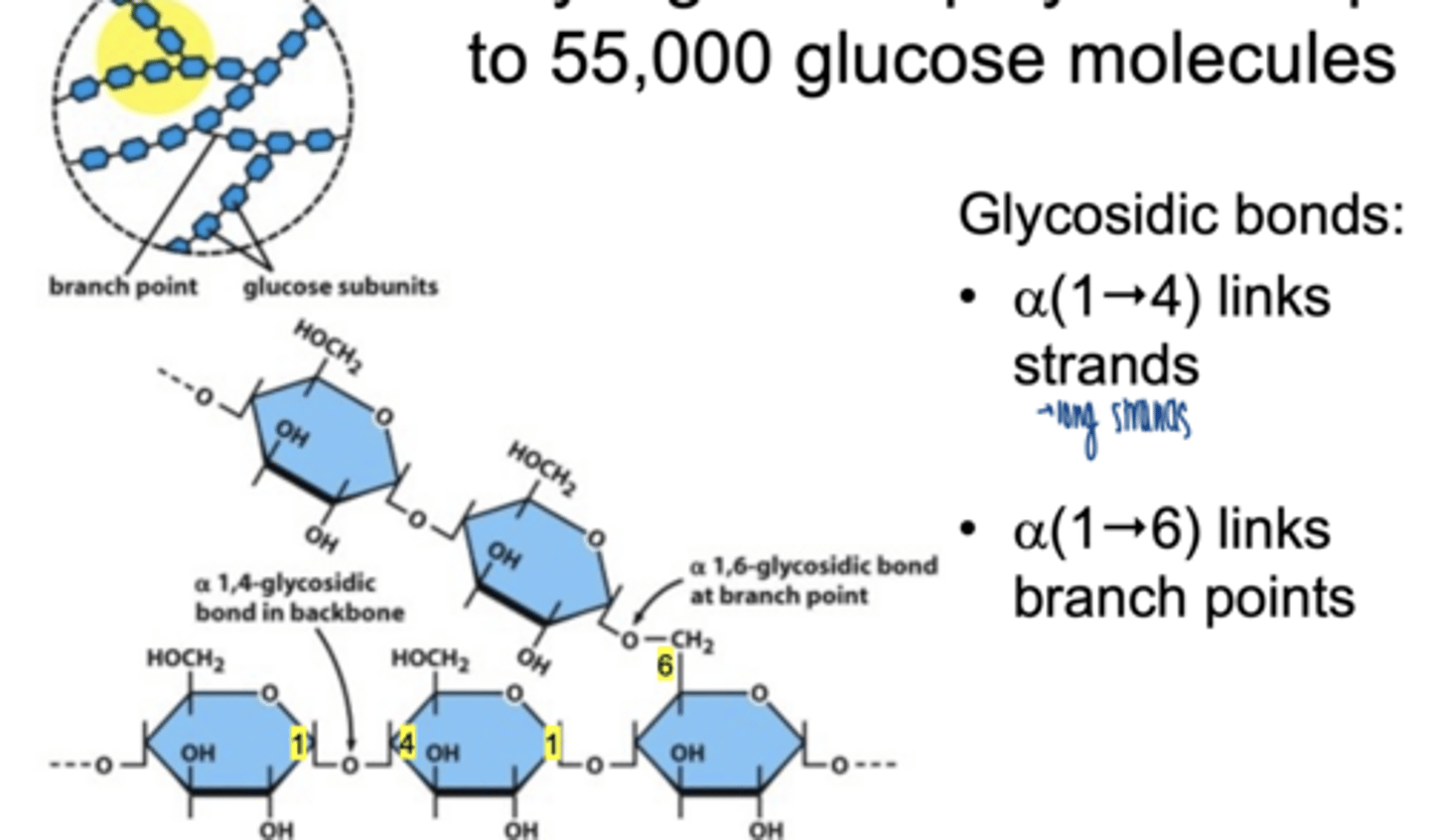
structural features of glycogen
α-1,4 glycosidic bonds → linear chains
α-1,6 glycosidic bonds → branch points (every 8-12 residues)
highly branched → allows rapid synthesis and breakdown
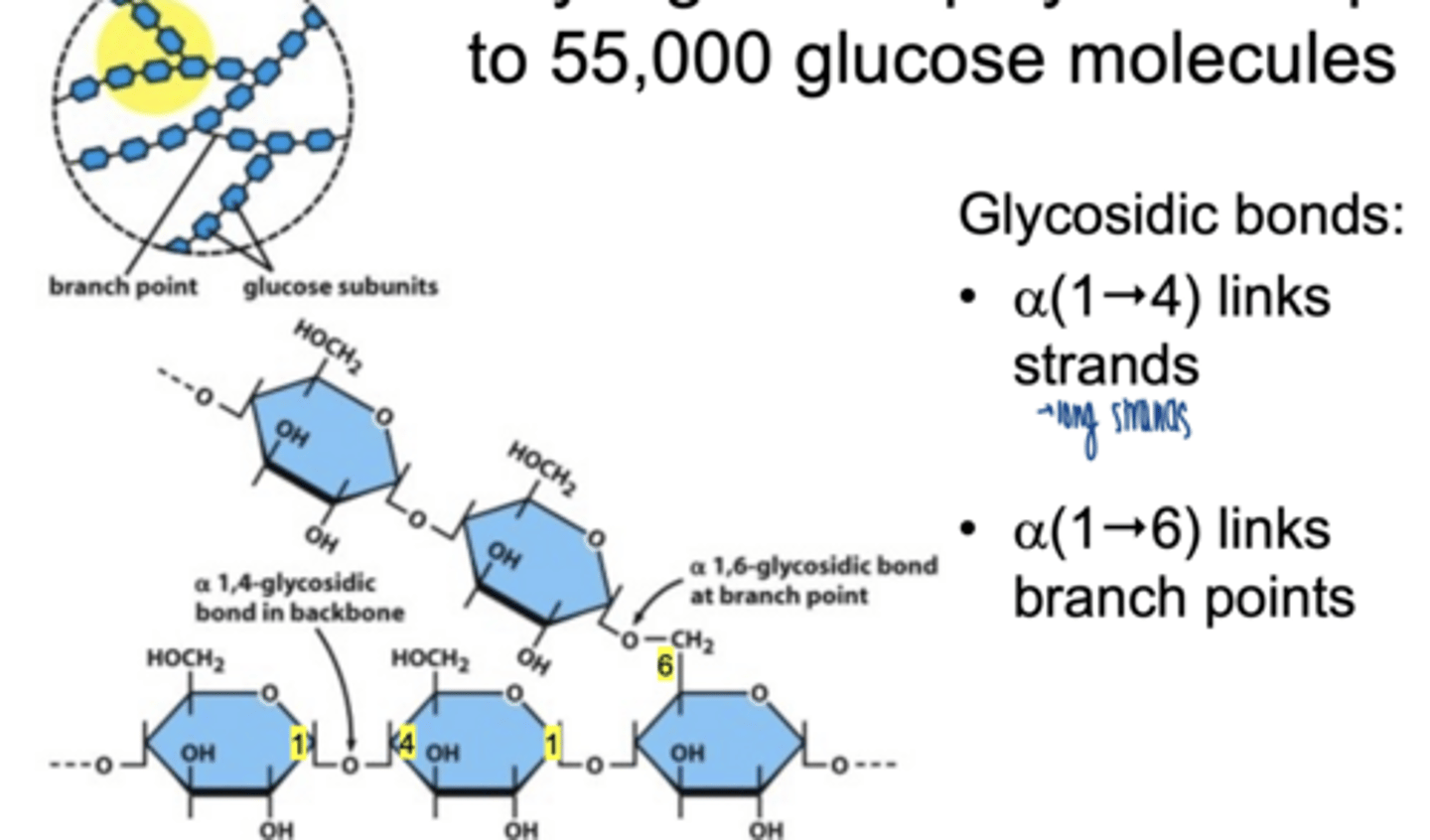
why is glycogen highly branched
increases solubility
provides multiple ends for enzymes → rapid glucose release
what is glycogeneis
process of synthesizing glycogen from glucose
when does glycogenesis occur
in the fed state (high glucose, high insulin)
what is the overall goal of glycogenesis
convert excess glucose → glycogen for storage
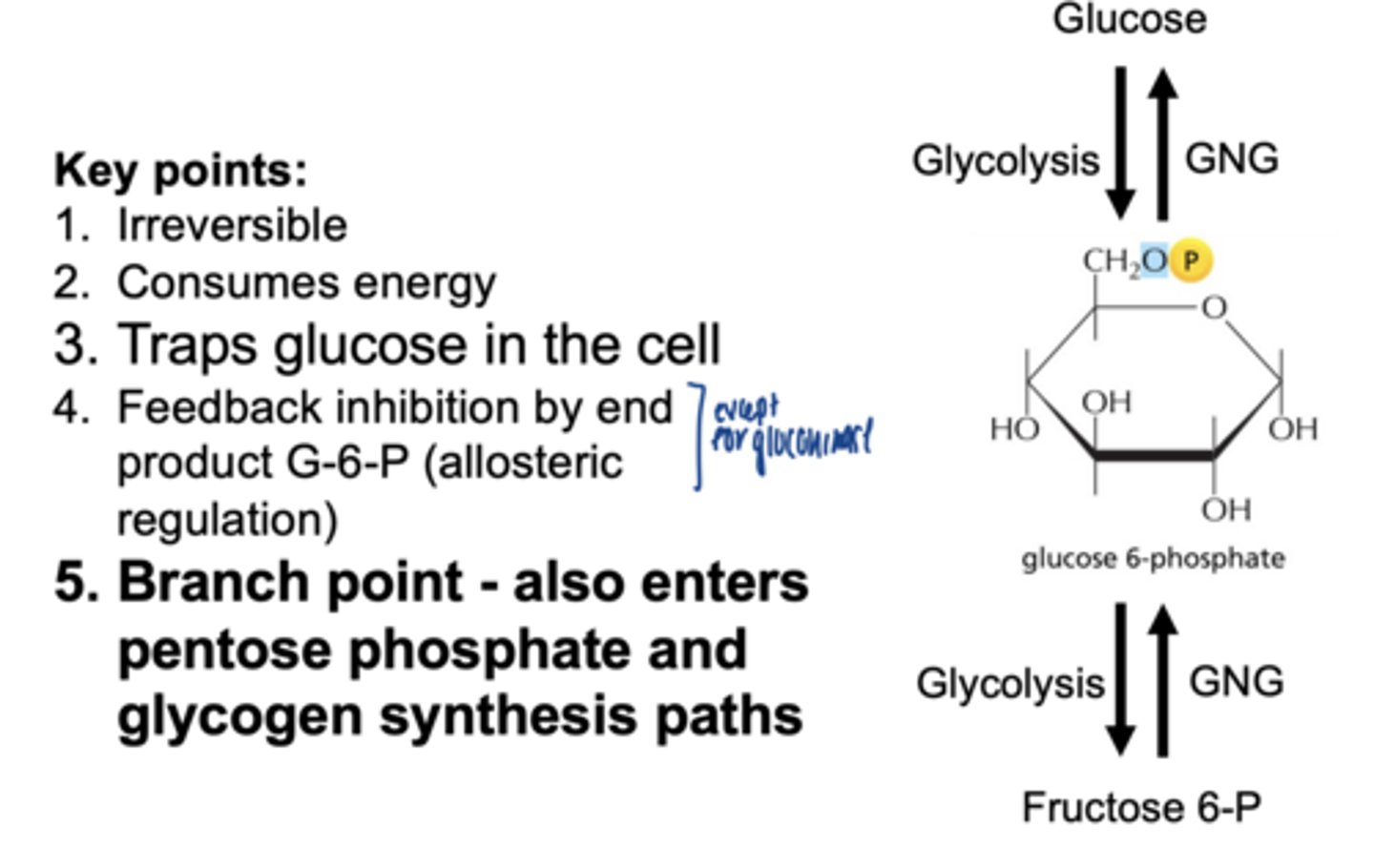
key points of glucose phosphorylation
- irreversible
- consumes energy
- traps glucose in the cell
- feedback inhibition by end product G6P (allosteric)
- branch point-- also enters pentose phosphate & glycogen synthesis pathways
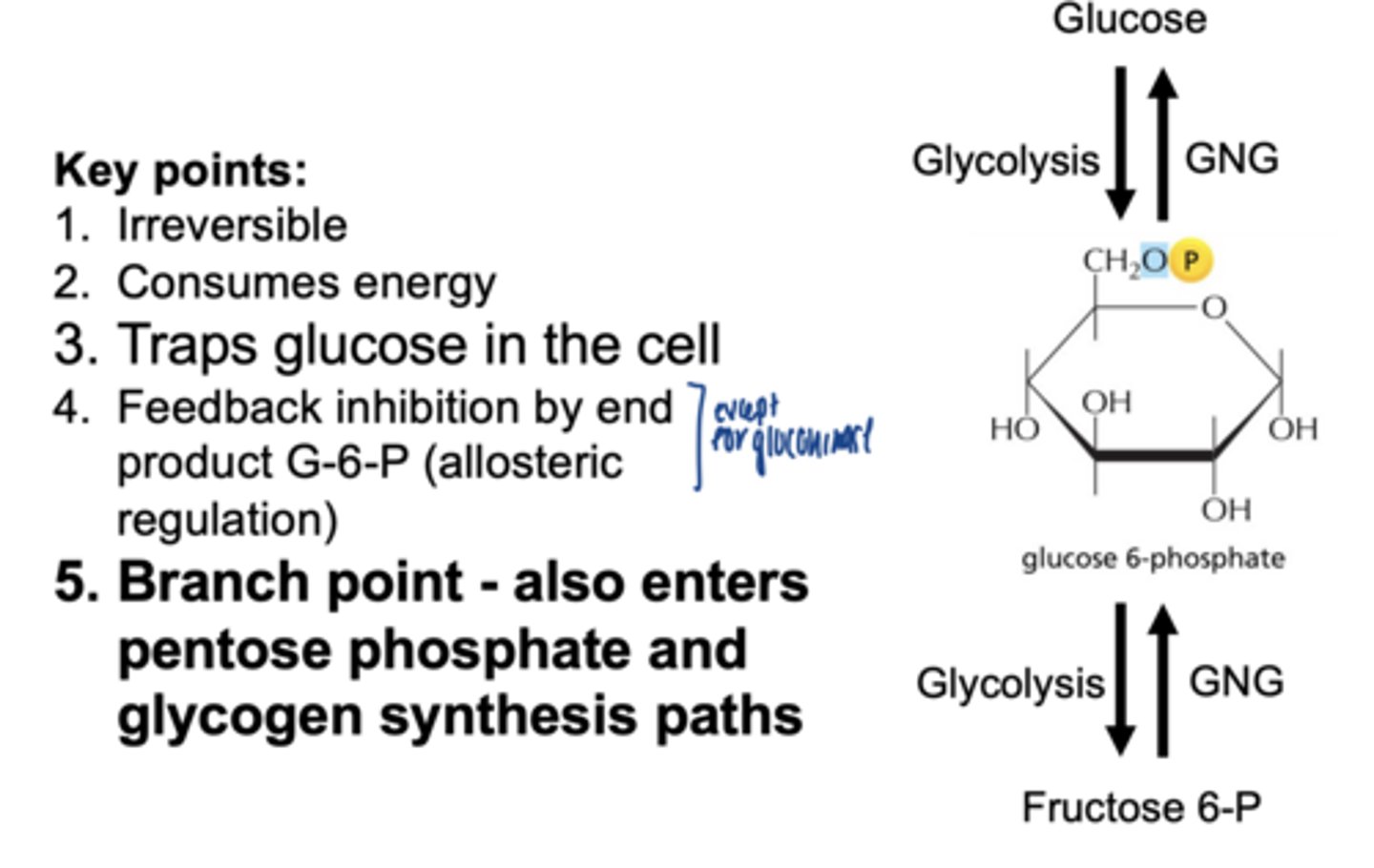
what is the first step of glycogenesis
glucose → glucose-6 phosphate (G6P)
phosphorylation of glucose
what catalyzes this step
hexokinase (most tissues)
glucokinase (liver)
why is phosphorylation important
traps glucose inside the cell
prepares it for metabolism
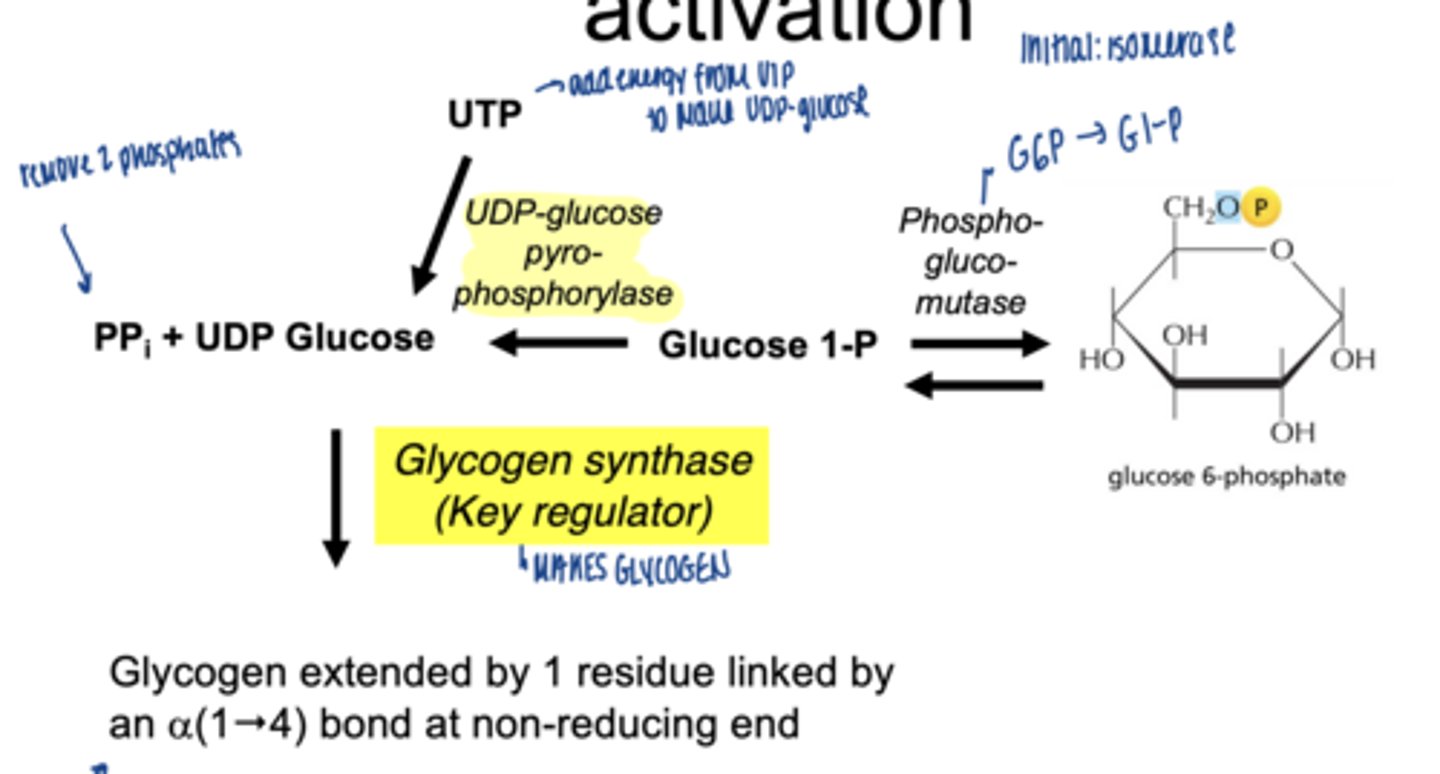
what happens to glucose-6-phospahte next (isomerization)
converted to glucose-1-phosphate (G1P)
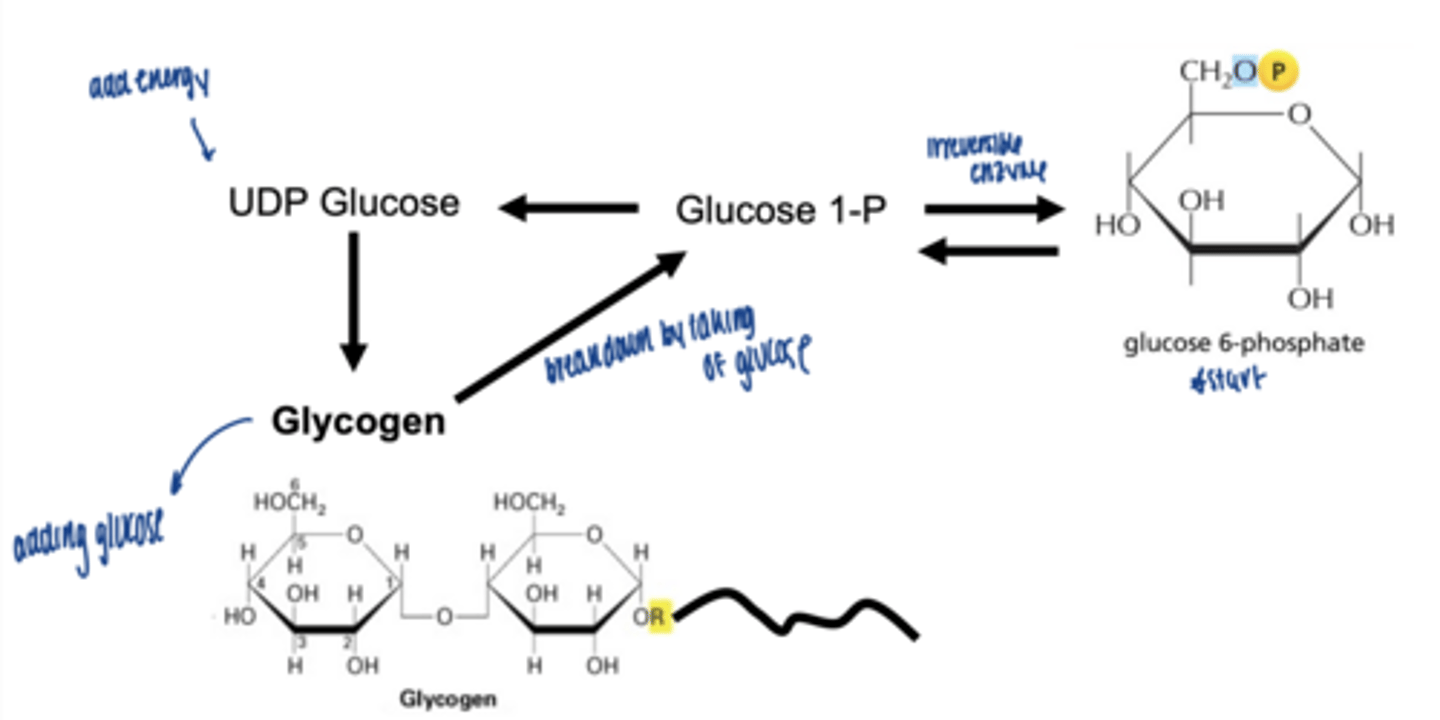
which enzymes catalyzes G6P to G1P
phosphoglucomutase (irreversible)
why is G1P important
direct precursor for glycogen synthesis
what happens to glucose-1-phosphate
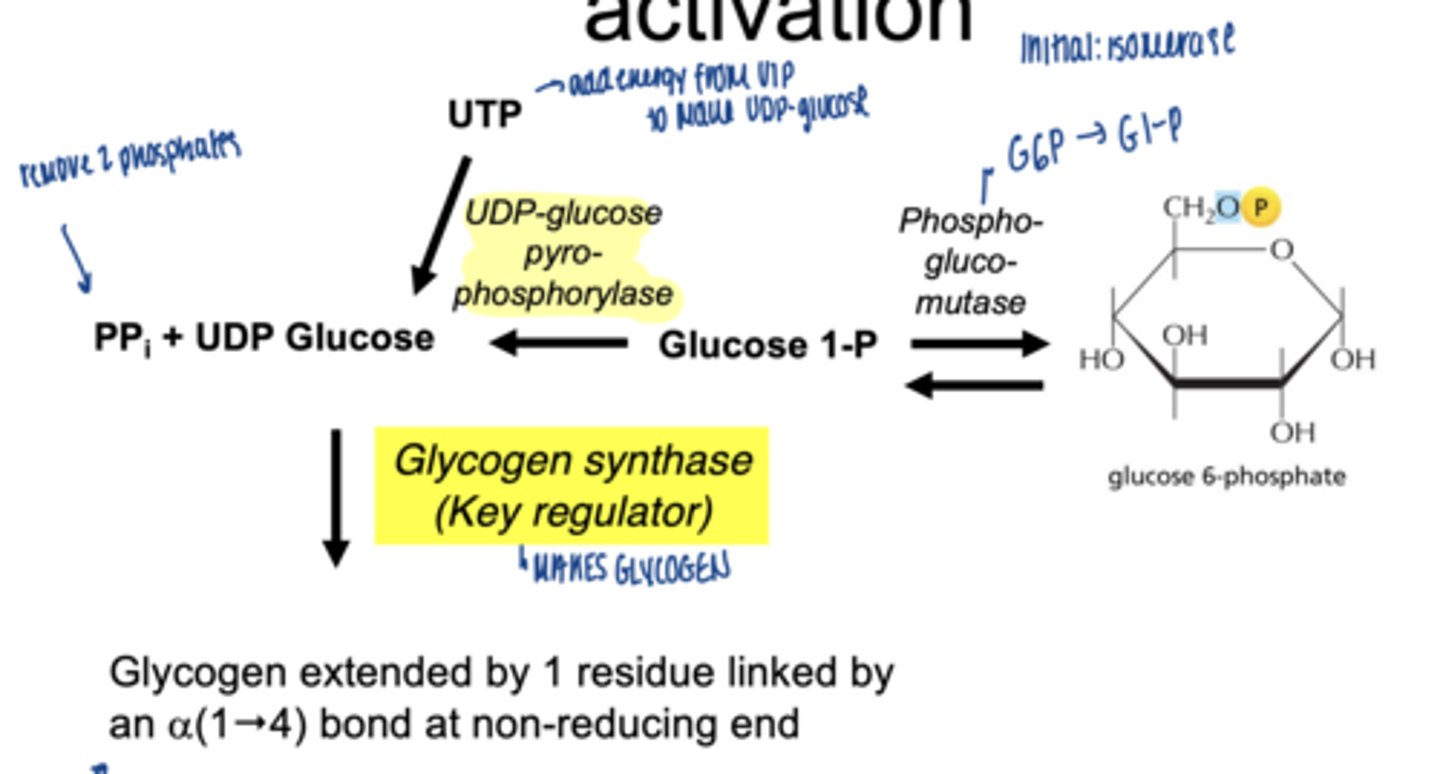
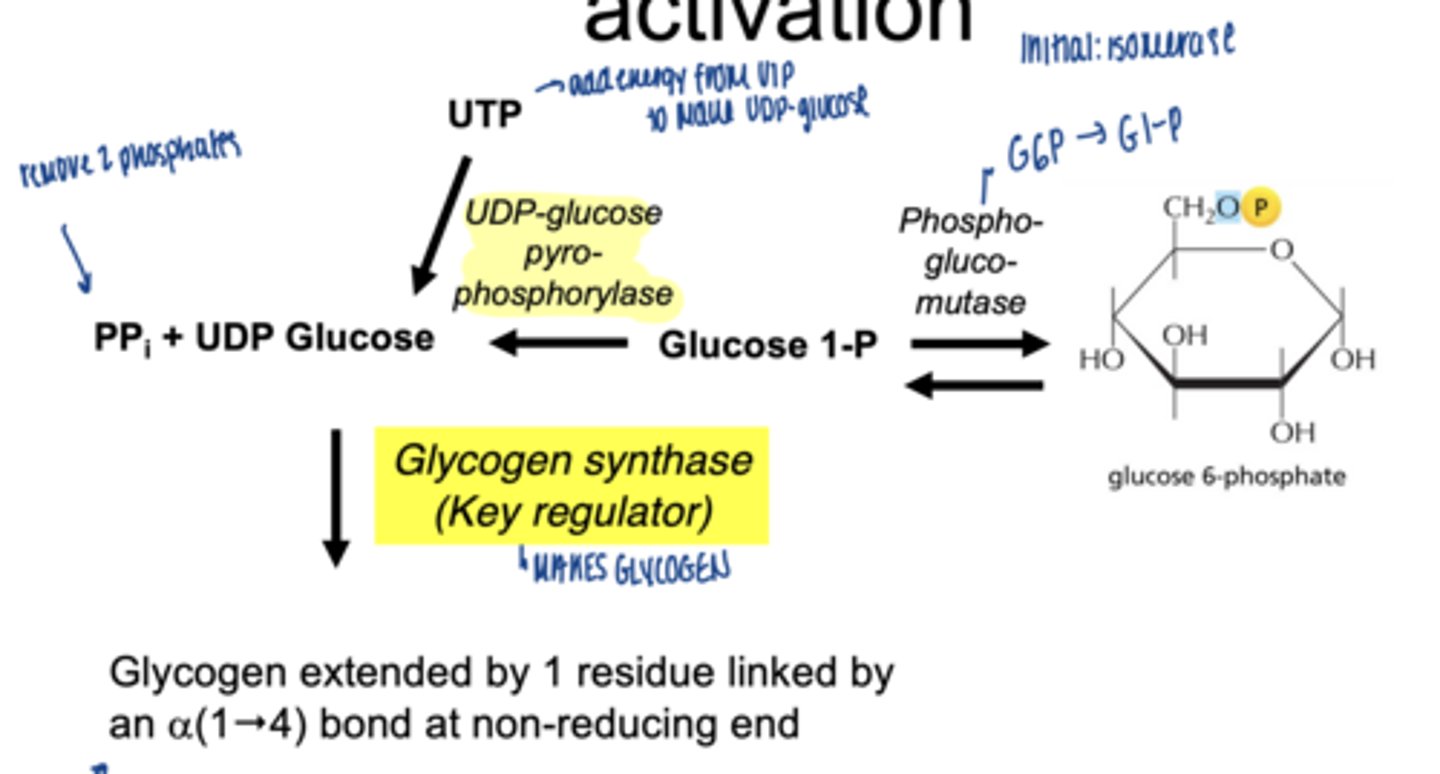
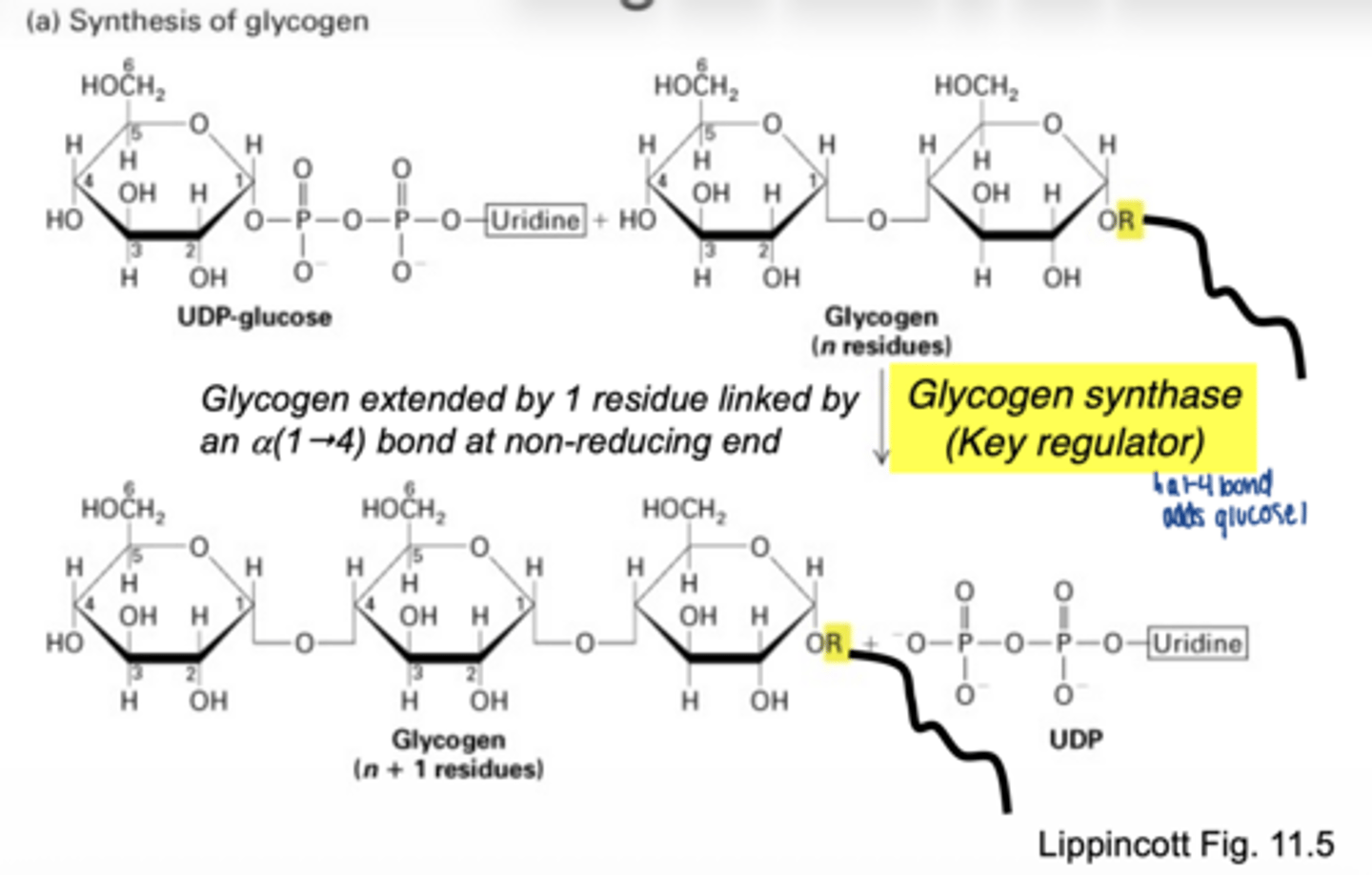
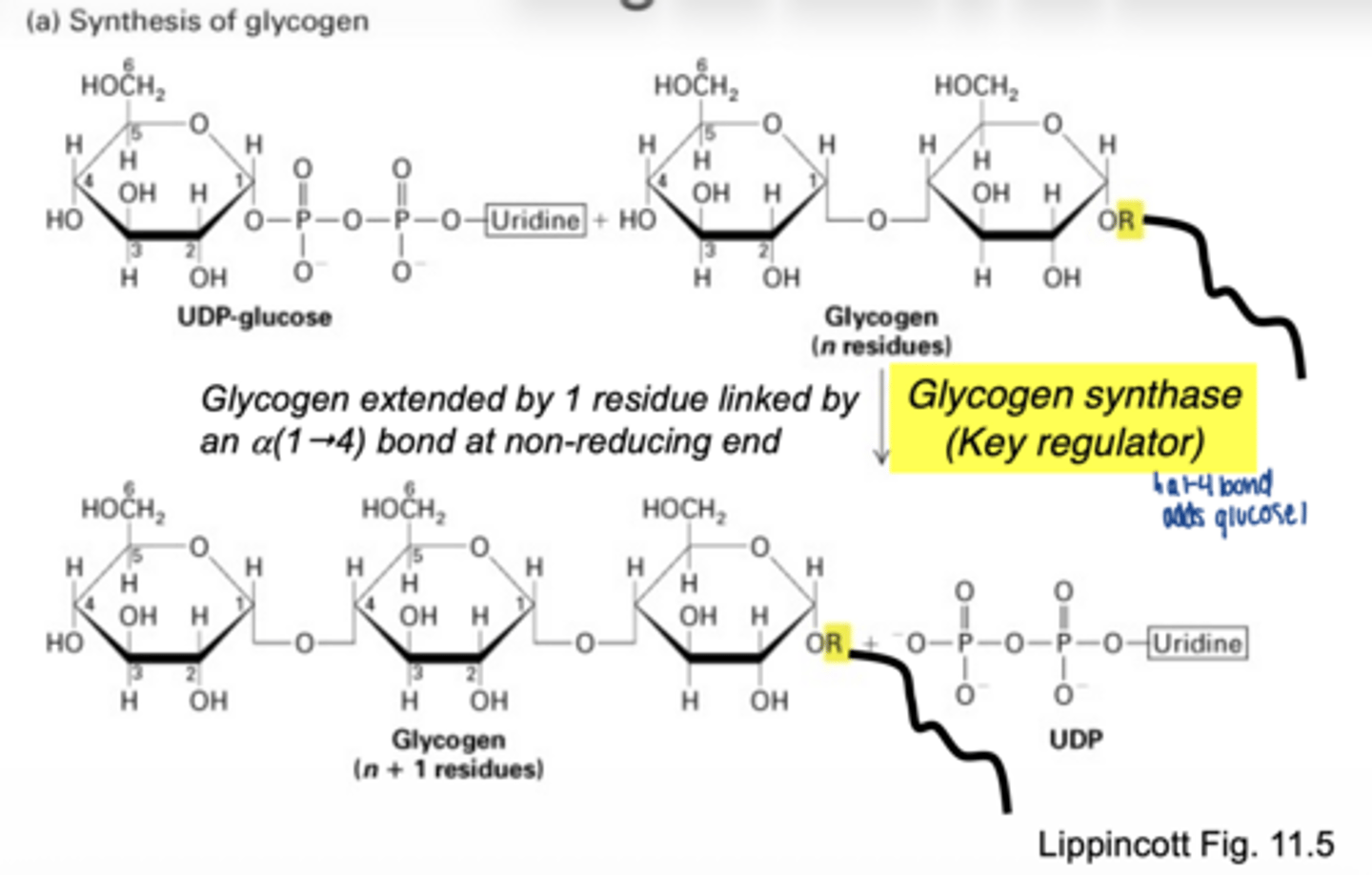
converted to UDP-glucose
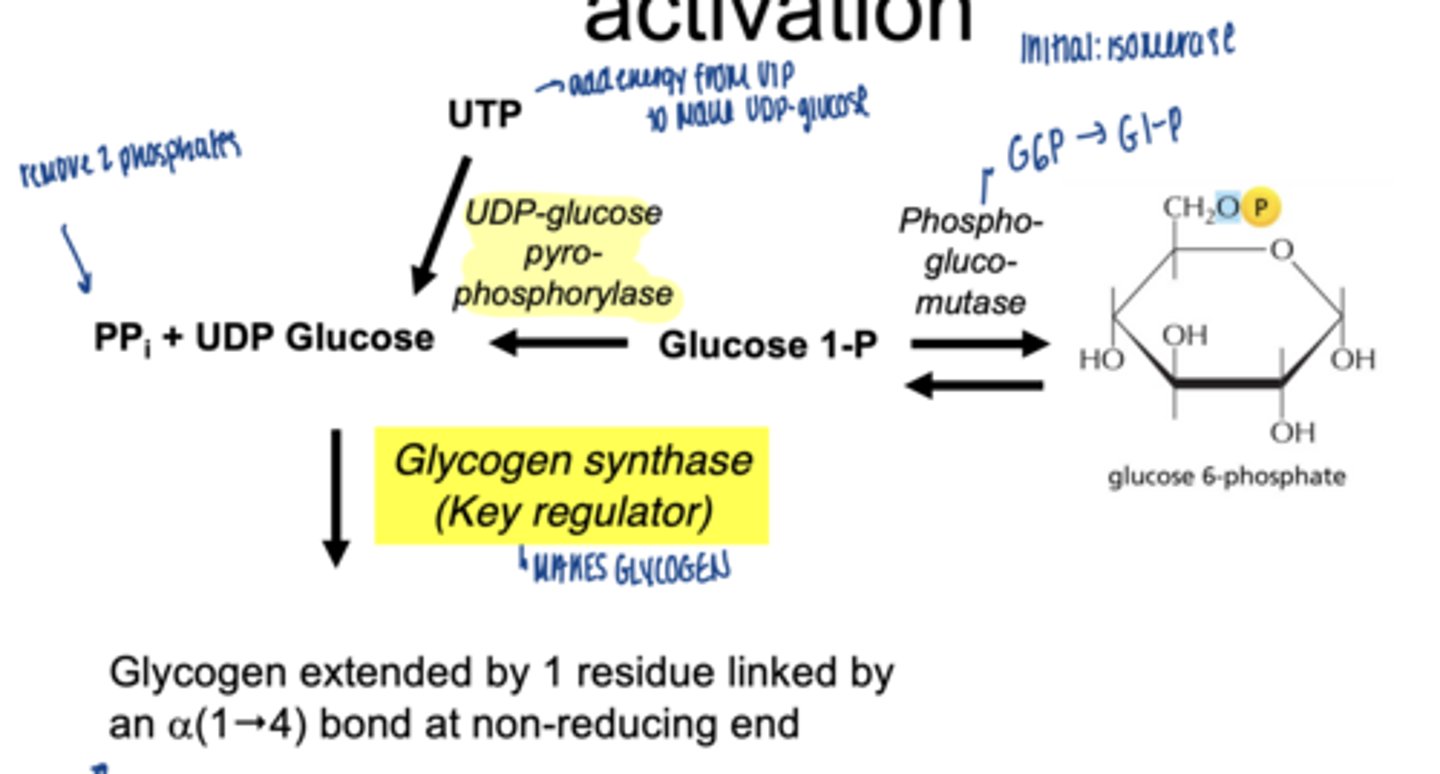
which enzyme is involved
UDP-glucose pyrophosphorylase
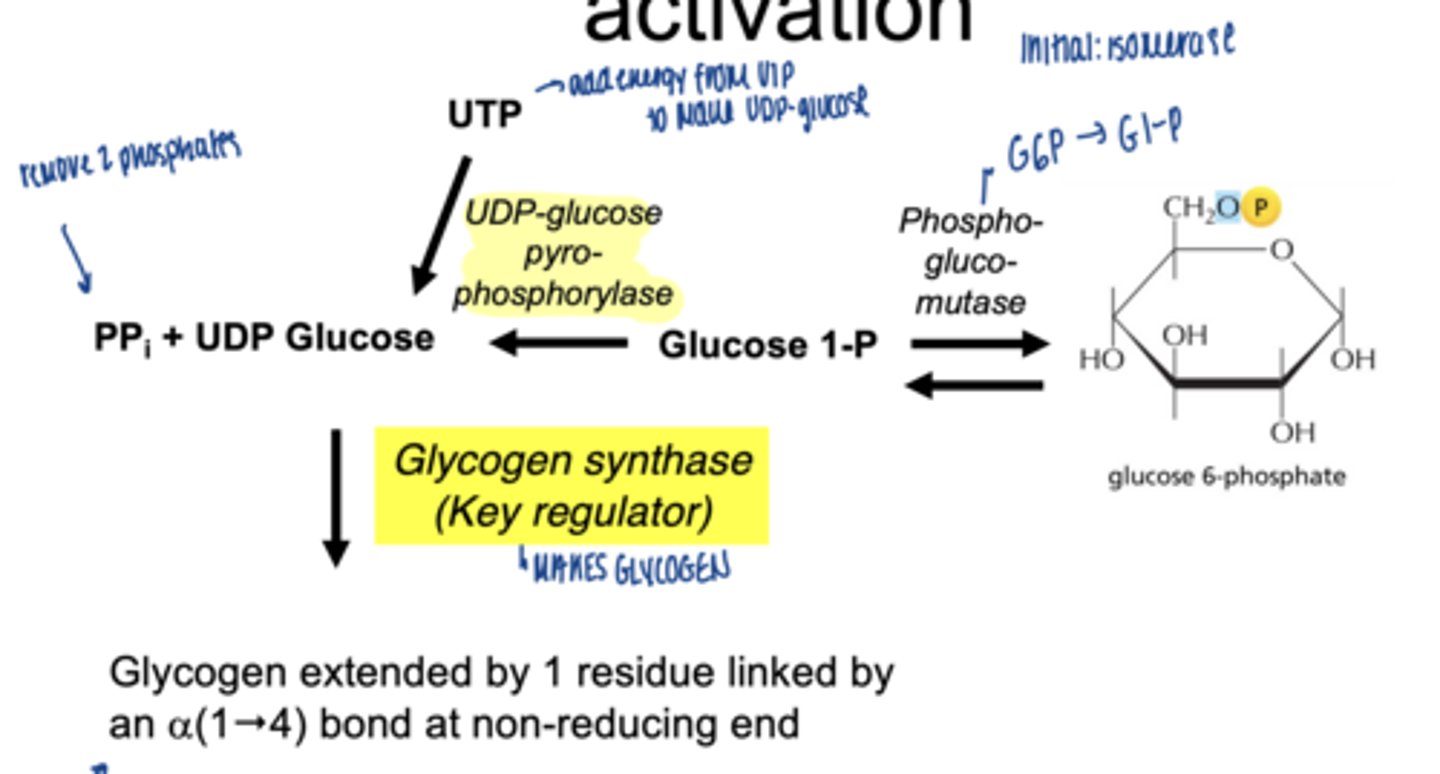
why is UDP-glucose needed
- activated form of glucose
- provides energy for bond formation
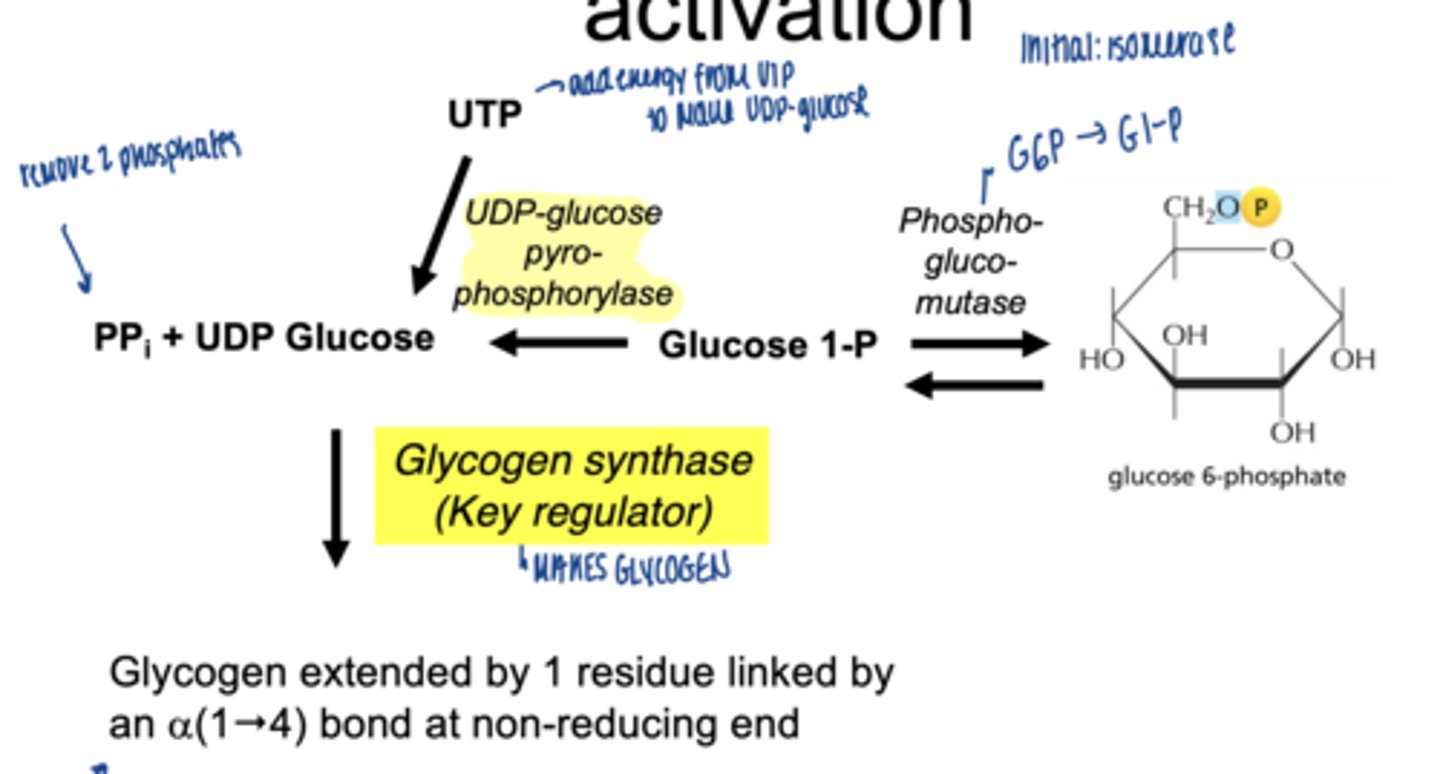
what drives this reaction forward
breakdown of pyrophosphate PPi
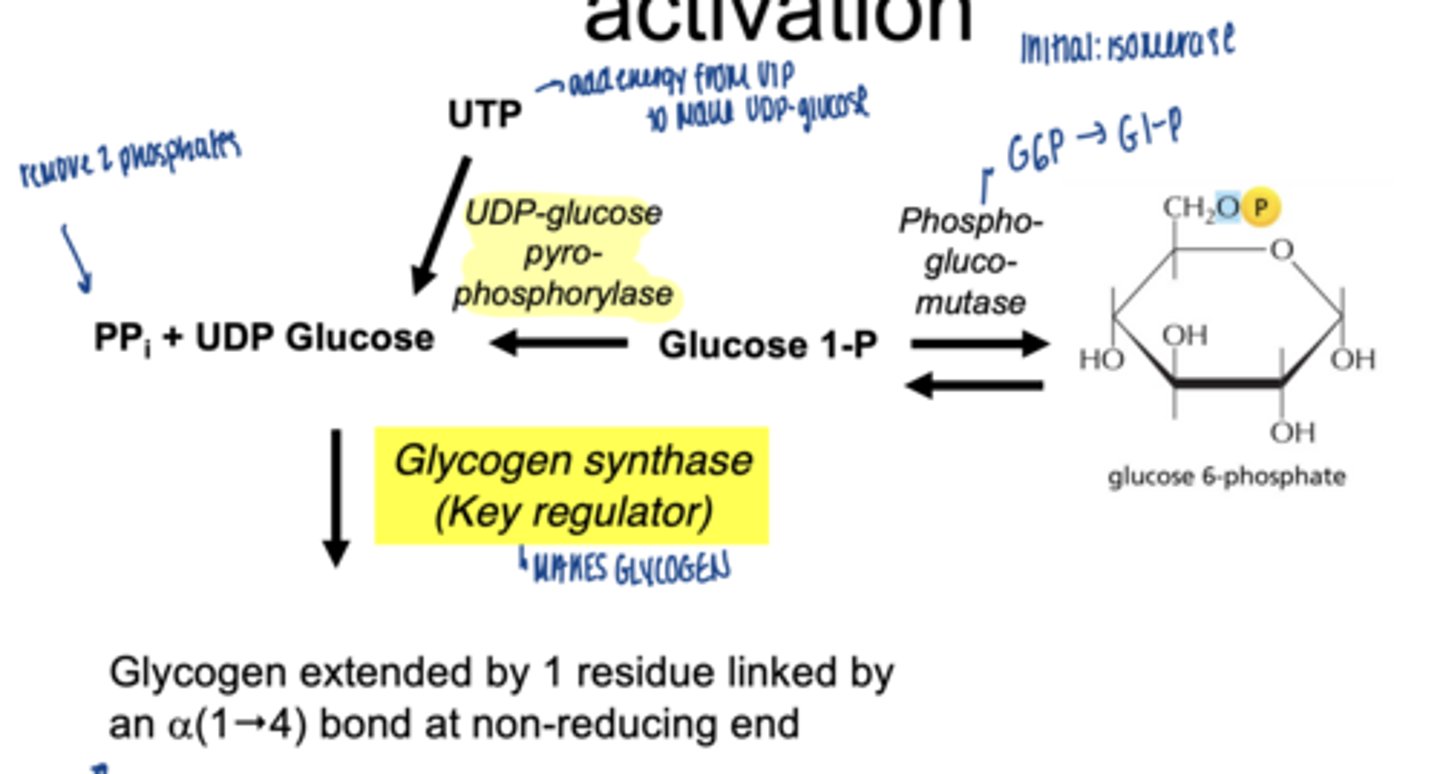
what enzyme builds glycogen chains
glycogen synthase
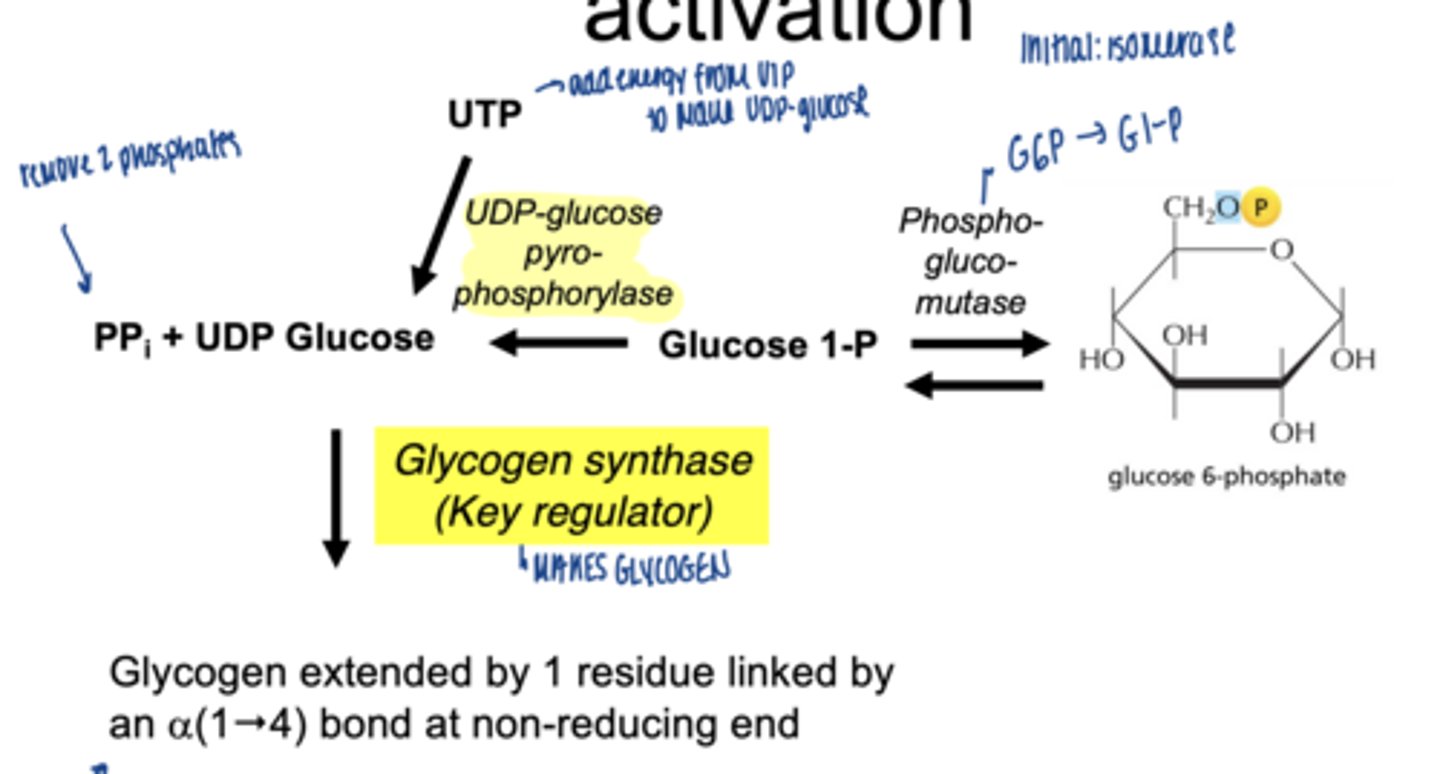
what type of bonds does glycogen synthase form
α-1,4 glycosidic bonds at non-reducing end
can glycogen synthase start a new chain from scratch
no
what is required to start glycogen synthesis
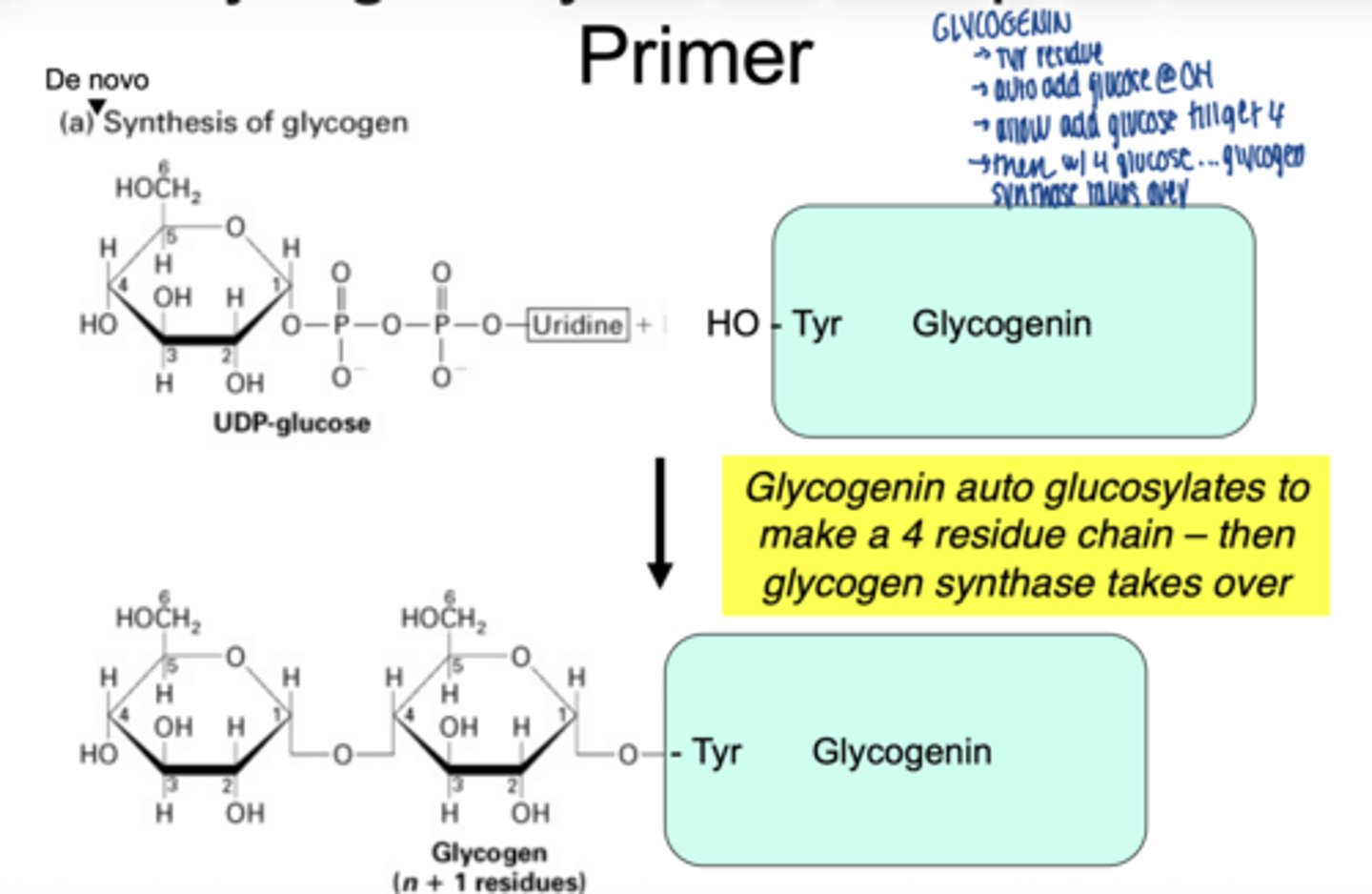
a primer protein called glycogenin
how does glucogenin work
- attached first glucose to a tyrosine residue
- builds a short chain (~4 residue chain
- glycogen synthase then takes over
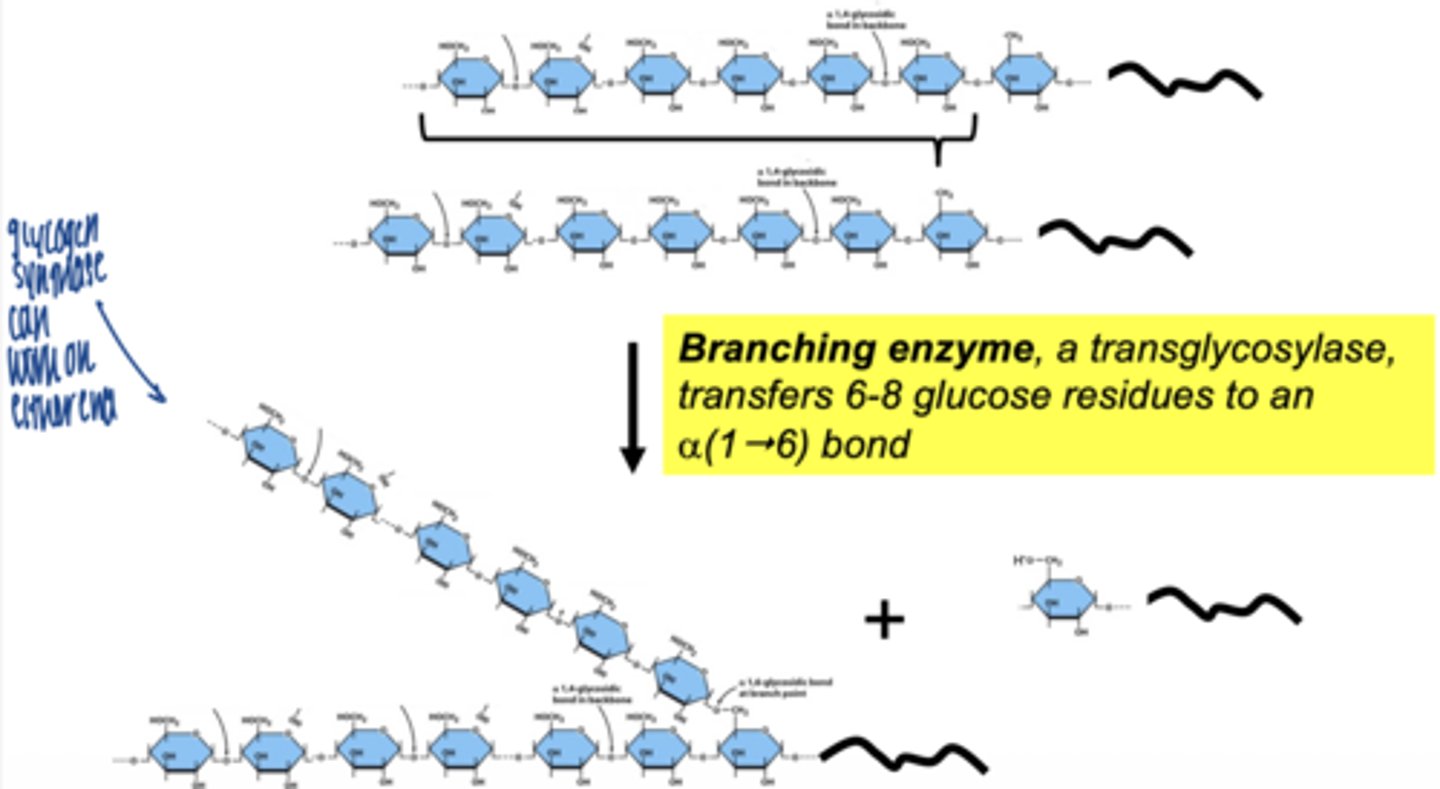
what enzyme creates branches in glycogen
branching enzyme (amylo-1,4 → 1,6 transglycosylase)
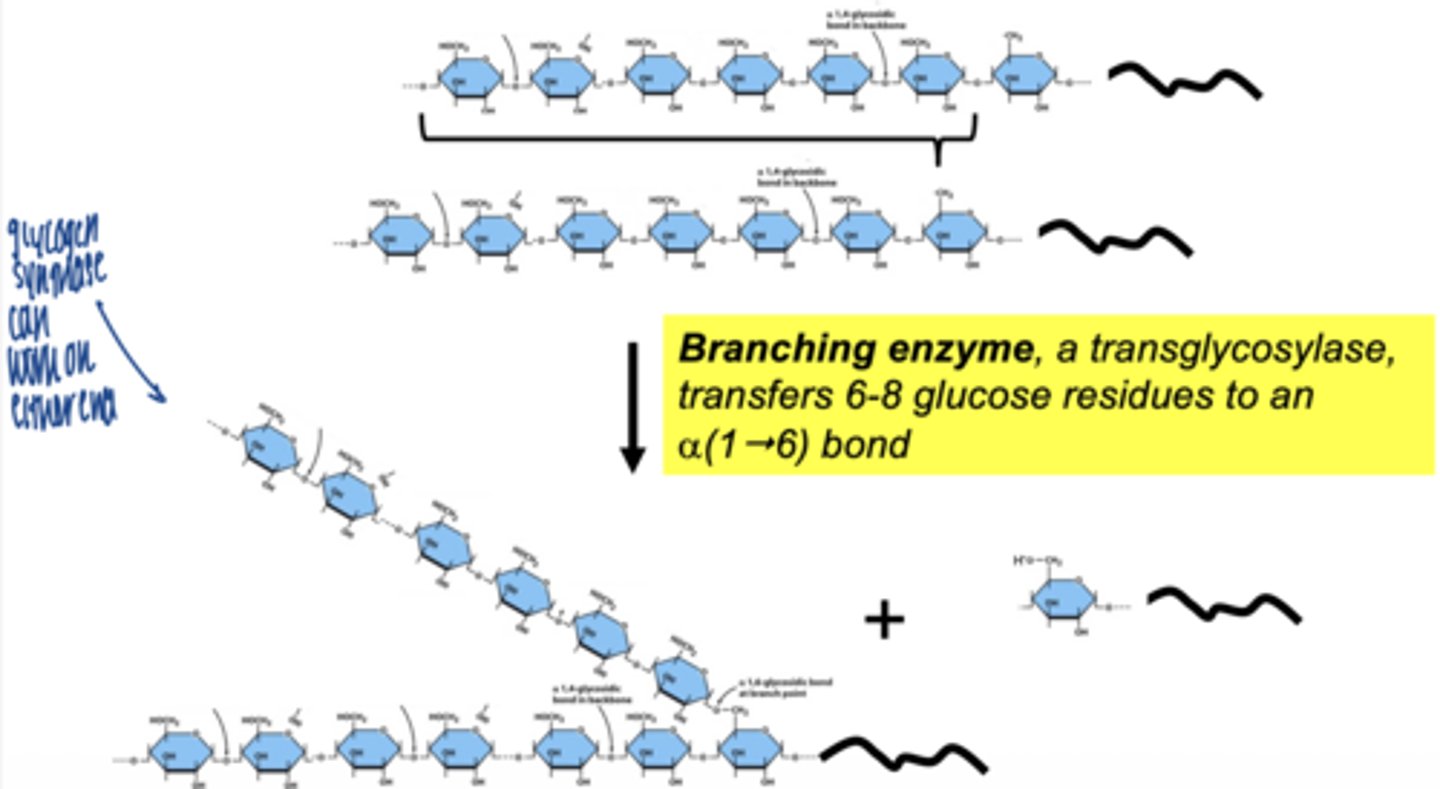
what does branching enzyme do
- transfers a segment of glucose chain
- creates α-1,6 bond
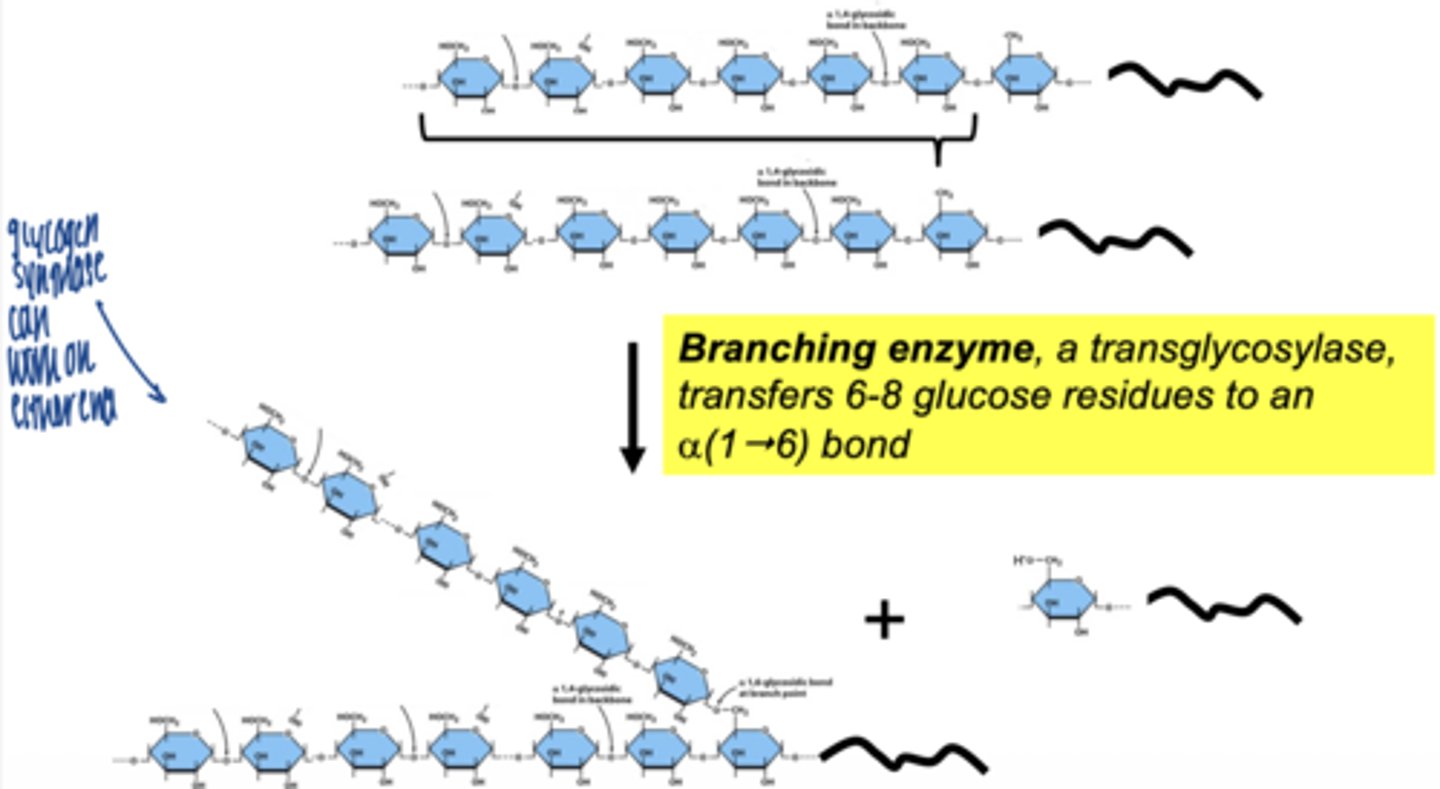
why are branches important
- increase solubility
- allow rapid glucose release
what is glycogenolysis
breakdown of glycogen → glucose
**chain shortening**
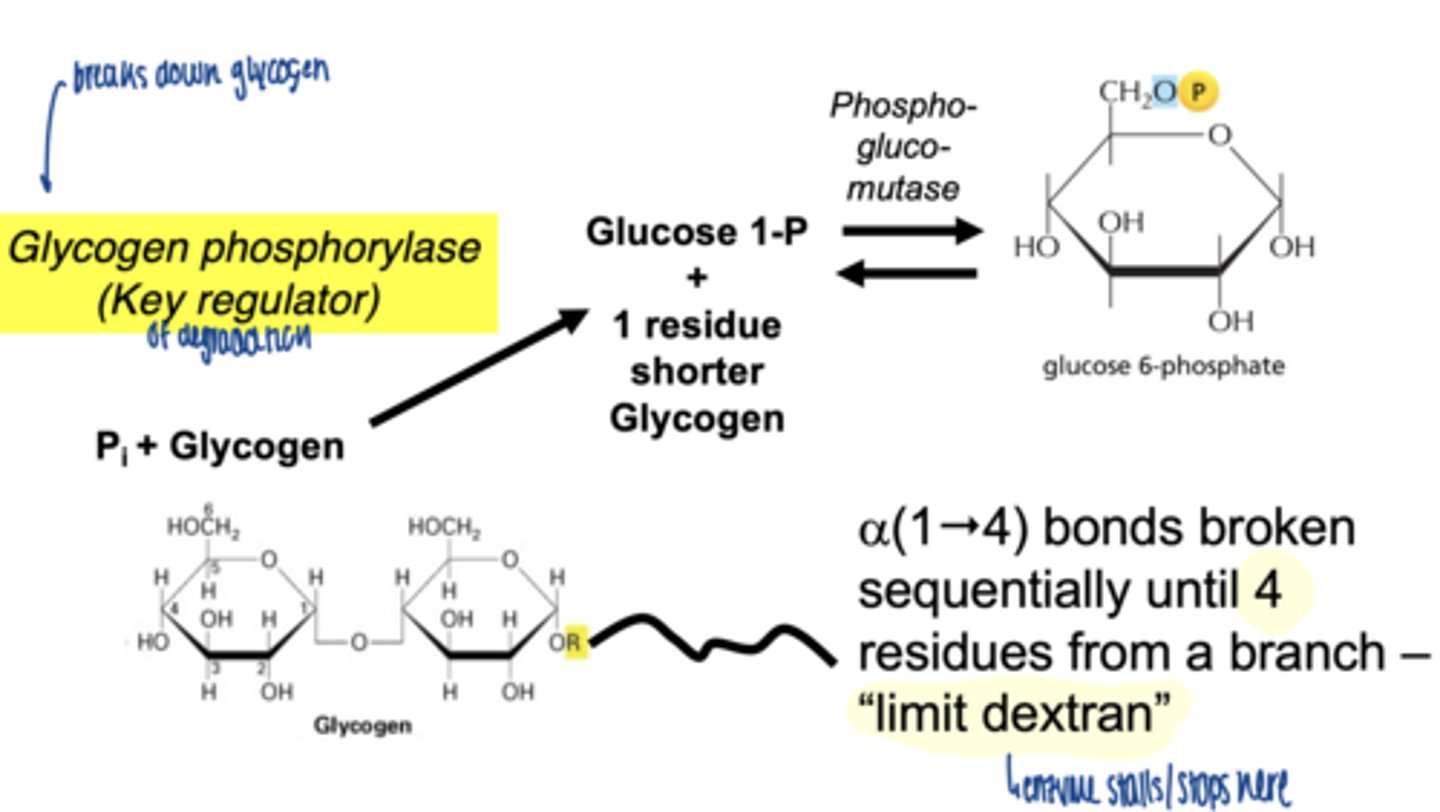
when does glycogenolysis occur
- fasting state
- exercise
what is the key enzyme in glycogen breakdown
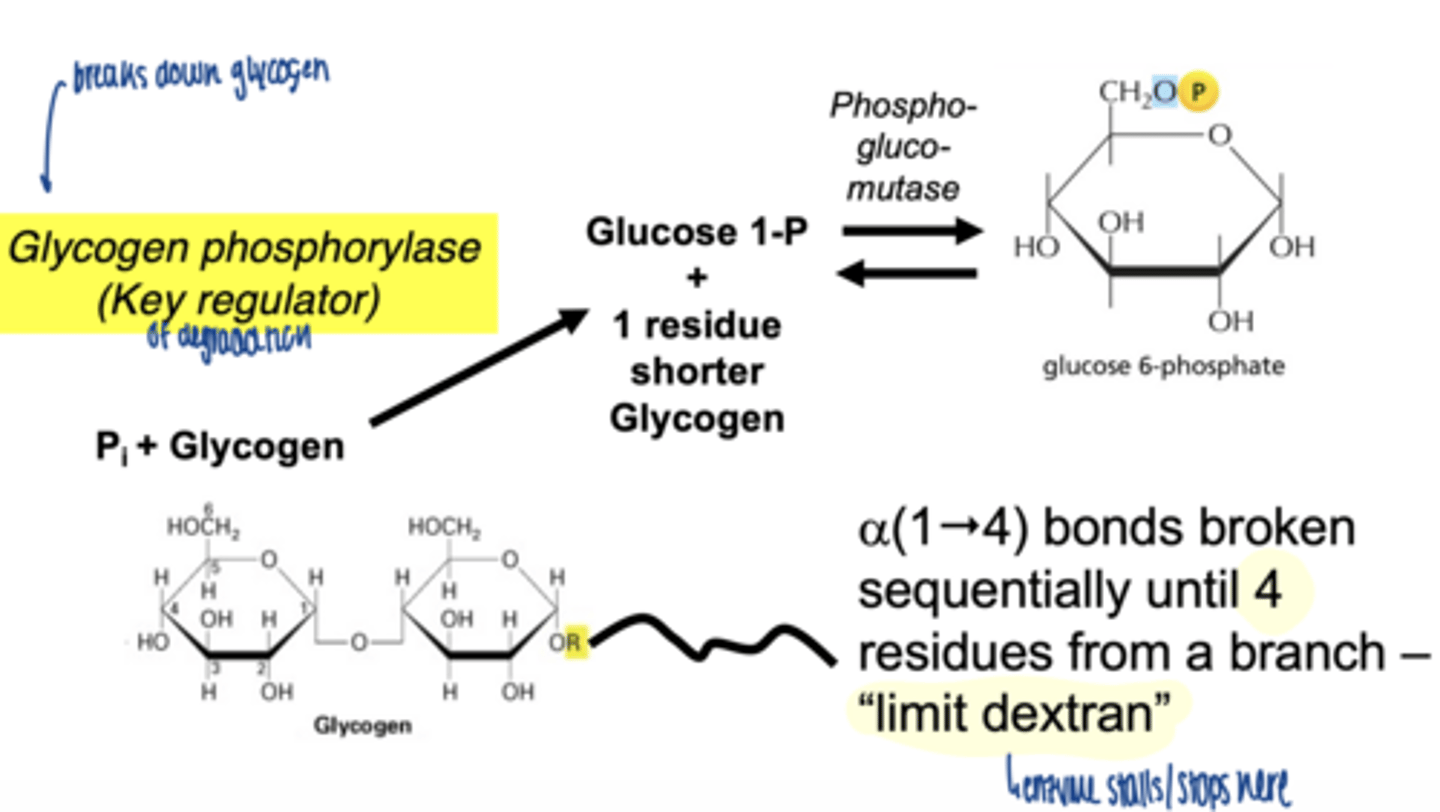
glycogen phosphorylase
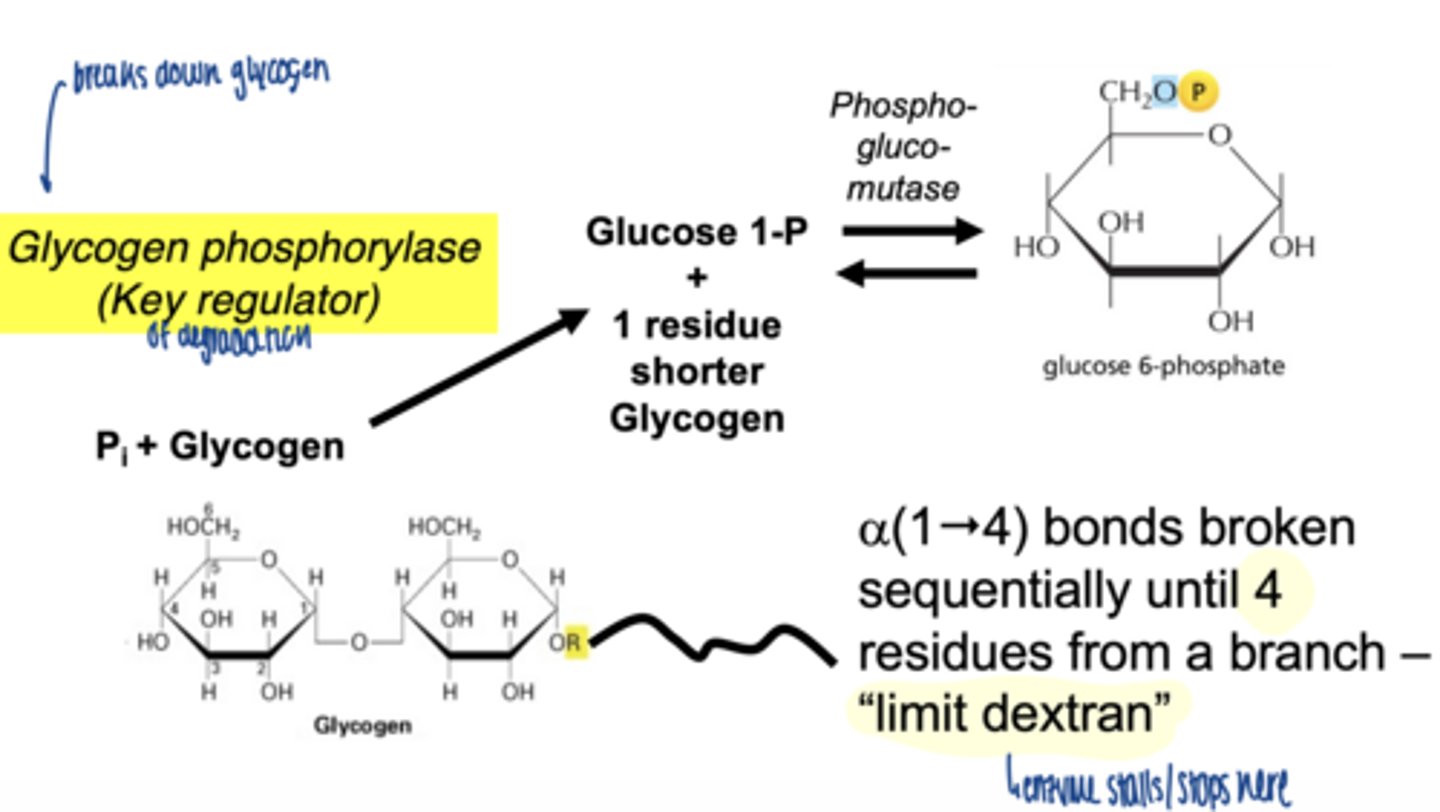
what is role of glycogen phosphorylase
-cleaves α-1,4 bonds
-produces glucose-1-phosphate
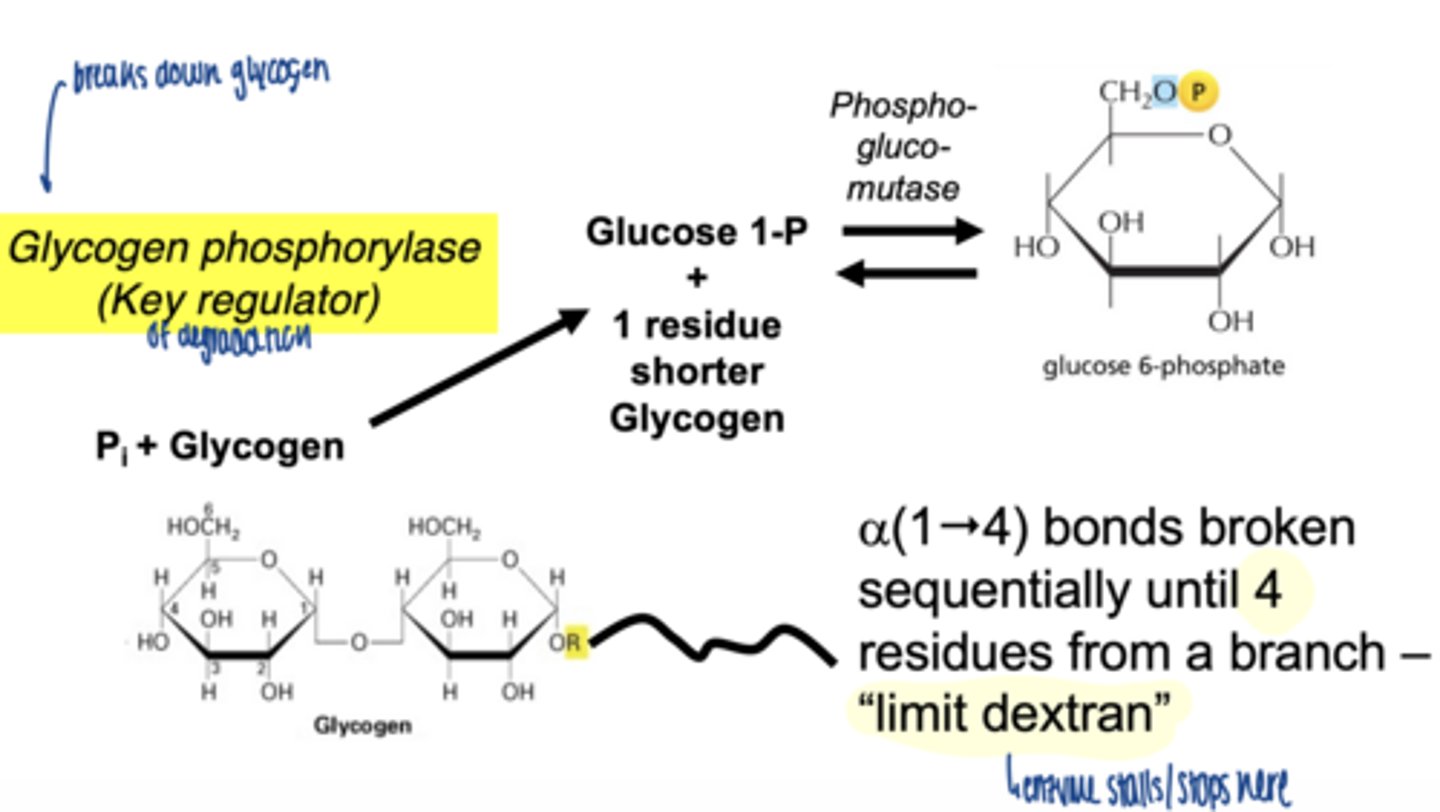
why phosphorylation instead of hydrolysis
- conserves energy
- produces G1P (already phosphorylated)
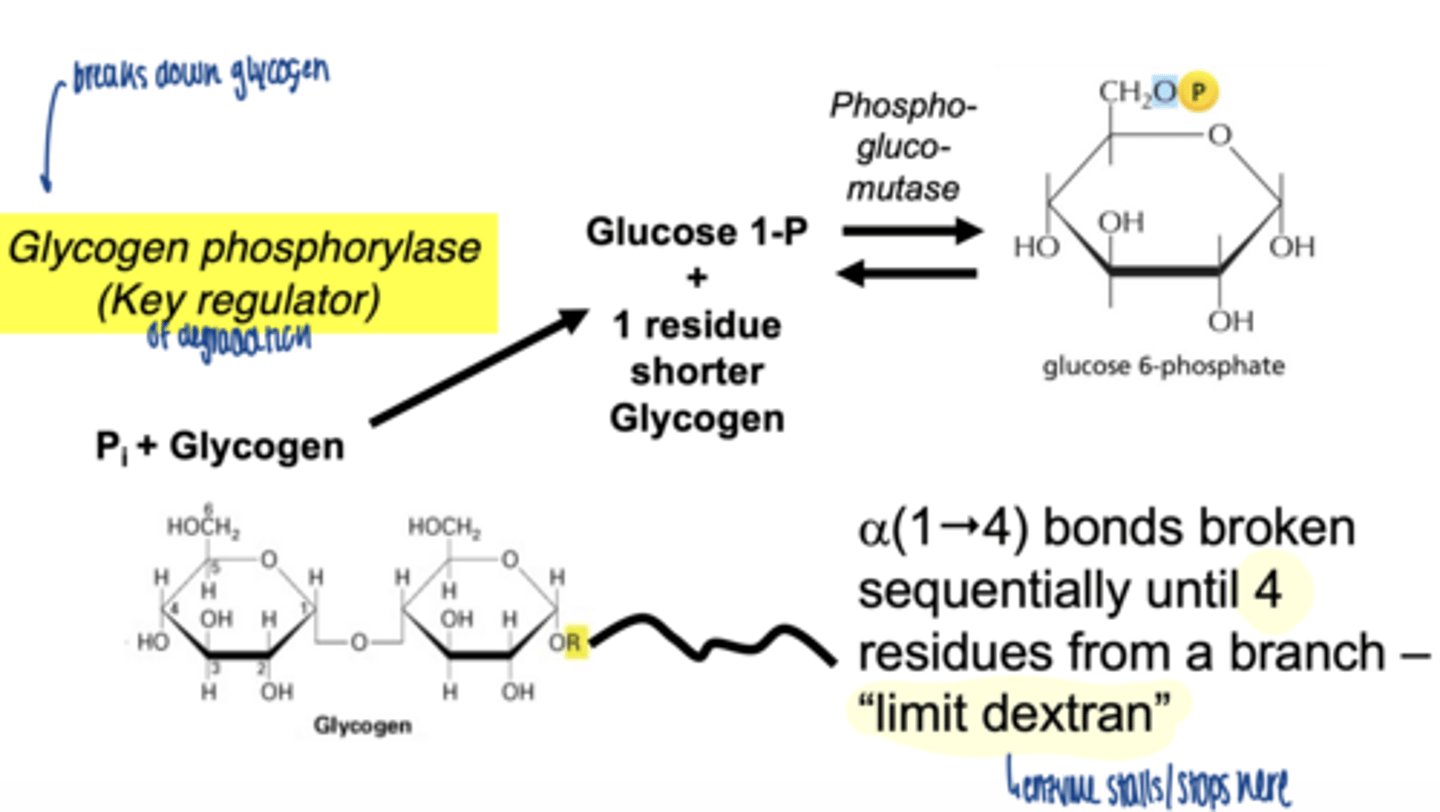
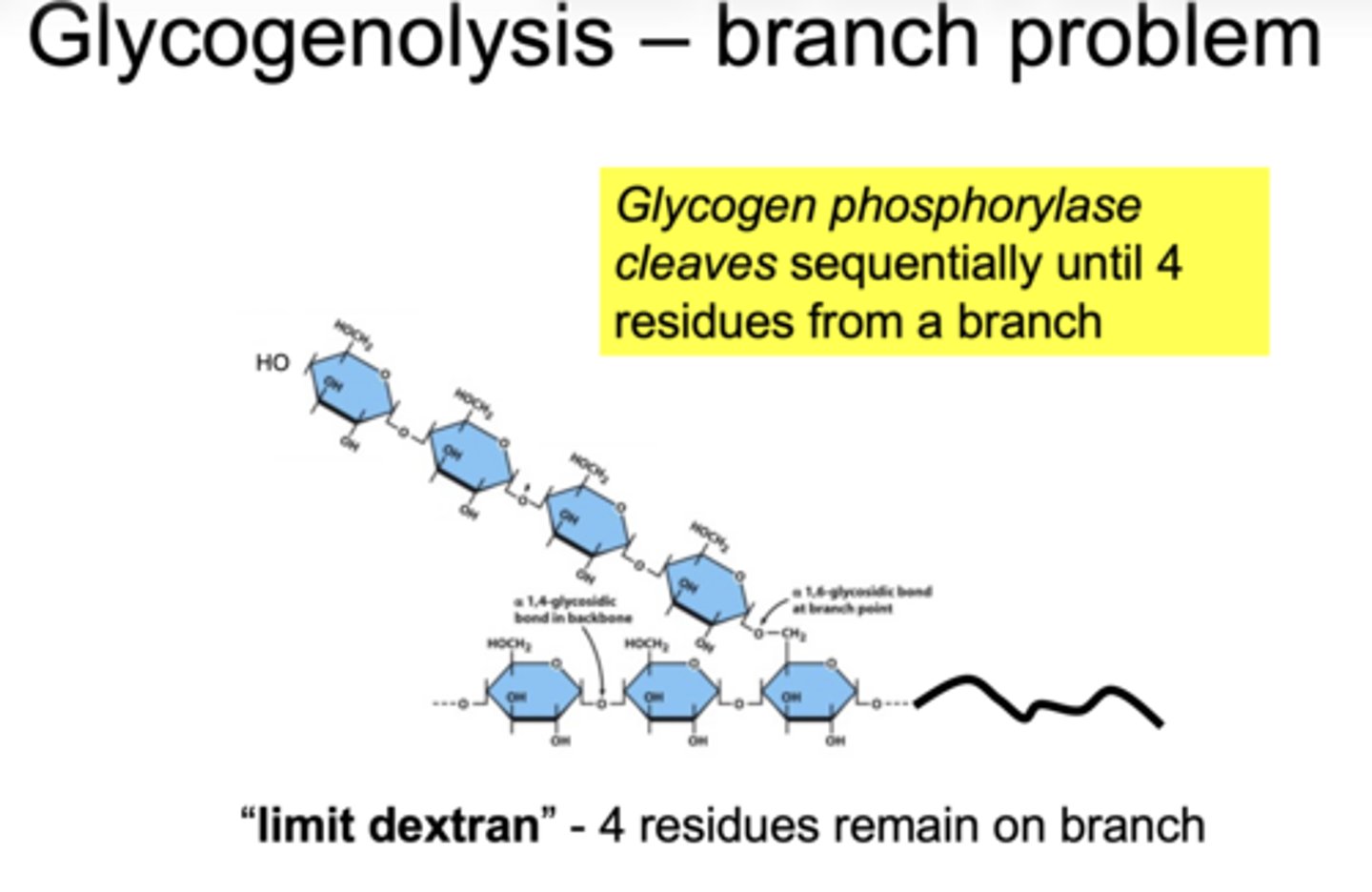
how far can glycogen phosphorylase degrade glycogen
stops 4 residues before a branch point
a(1"4) bonds broken sequentially until 4 residues from a branch - "limit dextran"
glycogen phosphorylase cleaves sequentially until ___ residues from a branch remain
4
what enzyme handles branch points
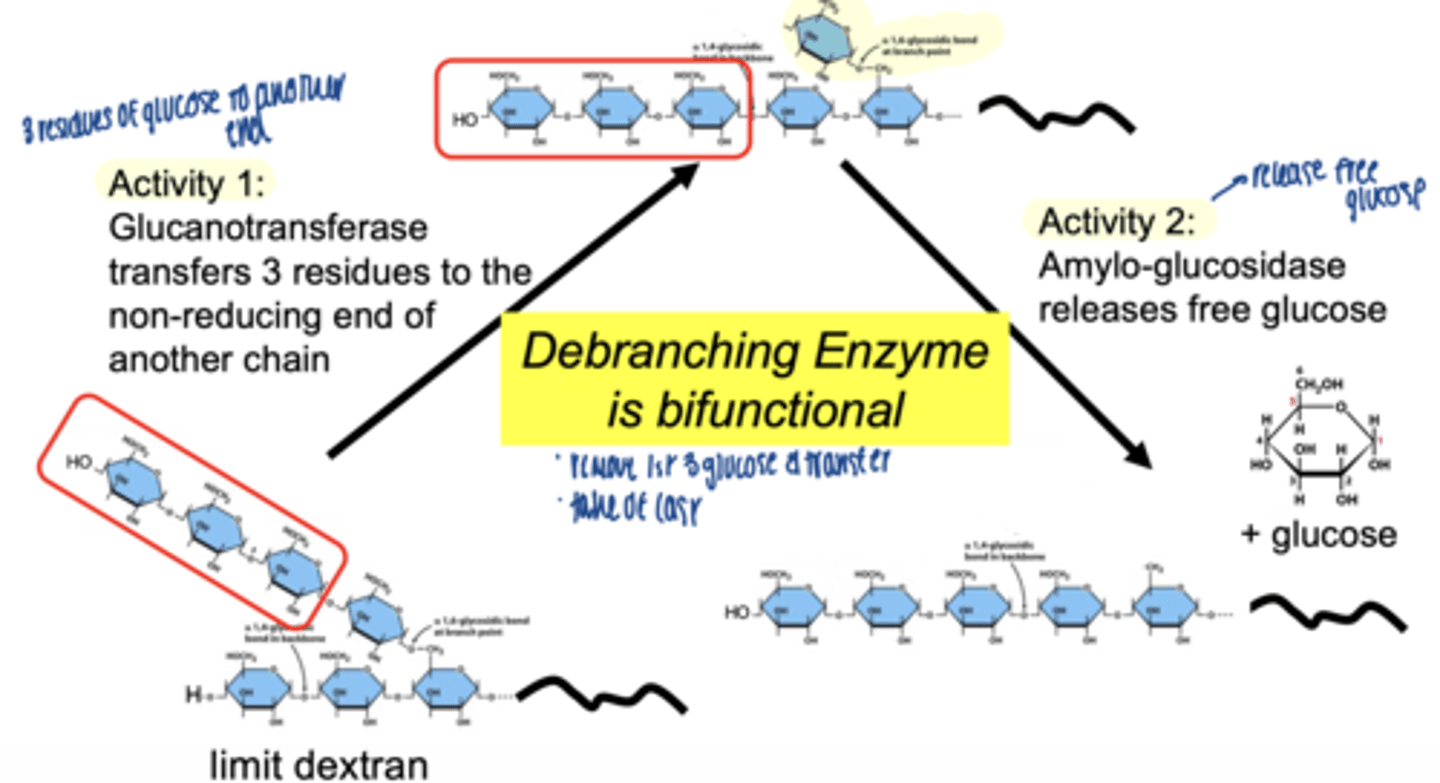
debranching enzyme
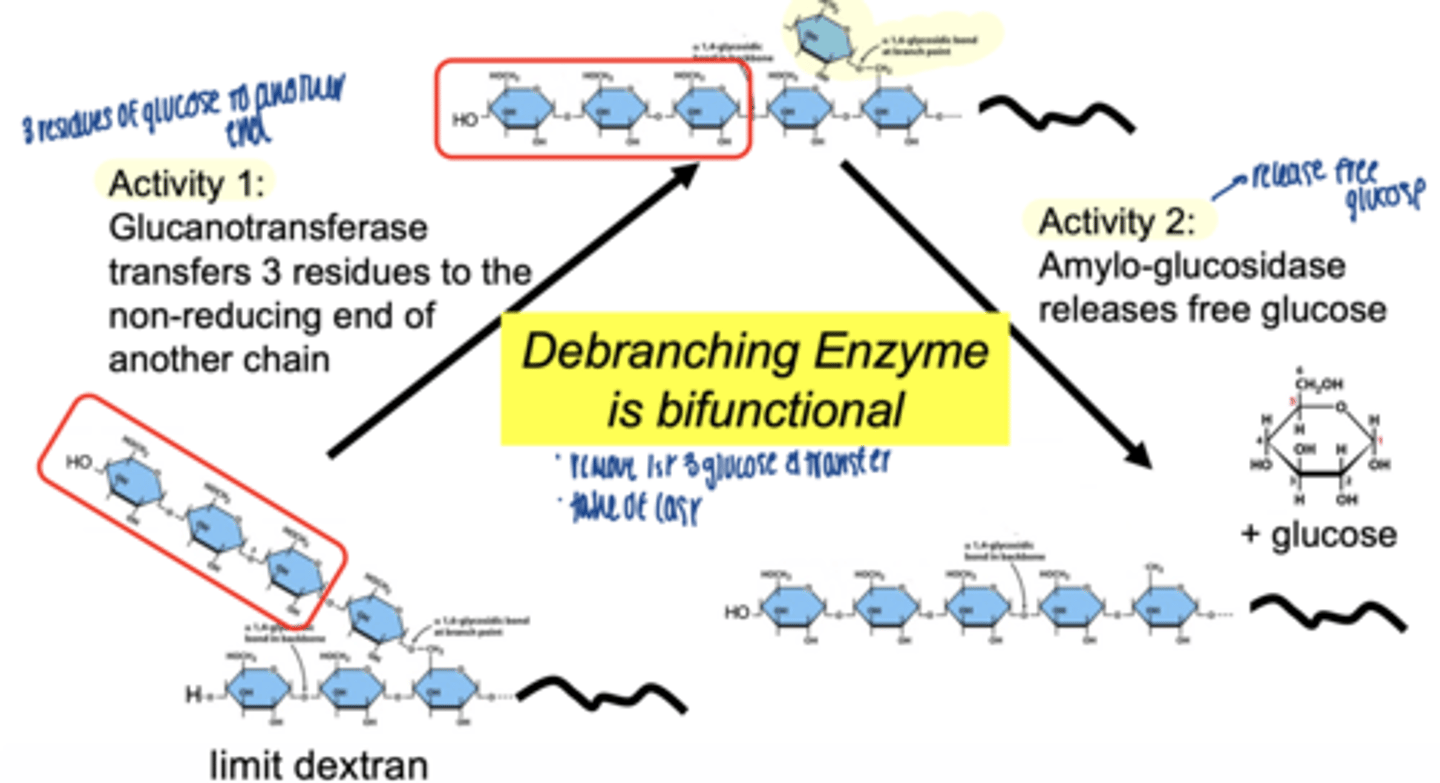
two activities of debranching enzyme
glucotransferase activity → moves 3 glucose units to non-reducing end of another chain
α-1,6 glucosidase (amylo-glucosidase)→ removes branch point glucose & releases free glucose
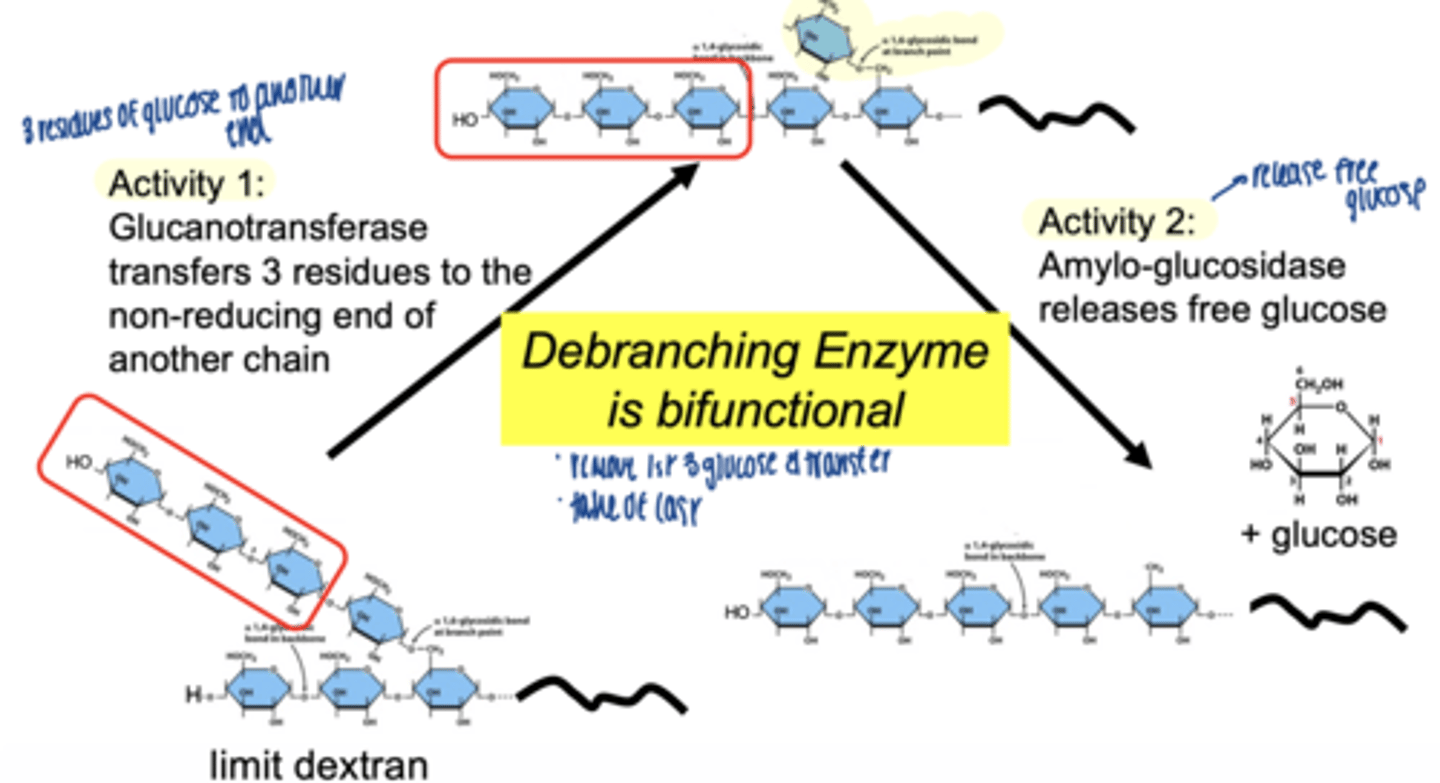
debranching enzyme (bifunctional)
Glycogen phosphorylase breaks a-1,4 bonds, releasing Glucose-1-Phosphate, but stops 4 residues from a branch
the transferase activity of GDE moves 3 glucose residues
a-1,6 glucosidase: hydrolyzes the remaining
a-1,6 glycosidic bond at the branch point.
free glucose molecule is released (not Glucose-1-Phosphate).