6.3 - Analysis (done)
1/29
Earn XP
Description and Tags
Chromatography (gas + TLC), NMR (13C + 1H), doesn't include tests since already explored in other decks)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
What is chromatography used for, and what are the stationary phase and mobile phase?
Chromatography is used to separate the components of a mixture.
The stationary phase does not move.
The mobile phase moves through or over the stationary phase.
What are the stationary phase and mobile phase in TLC chromatography?
stationary phase = solid on the plate
mobile phase = solvent
How are substances separated in thin-layer chromatography?
Substances are separated by their different attractions to the stationary phase and mobile phase.
A substance with a strong attraction to the stationary phase moves more slowly.
A substance with a weaker attraction to the stationary phase moves further up the plate.
What is adsorption in thin-layer chromatography?
Adsorption is when particles stick to the surface of the solid stationary phase.
What is affinity for the stationary phase in TLC?
It means how strongly a substance is attracted to the stationary phase.
Higher affinity means moves less.
Lower affinity means moves further.
What is the Rf value and how is it calculated?
The Rf value compares how far a substance moves relative to the solvent front.
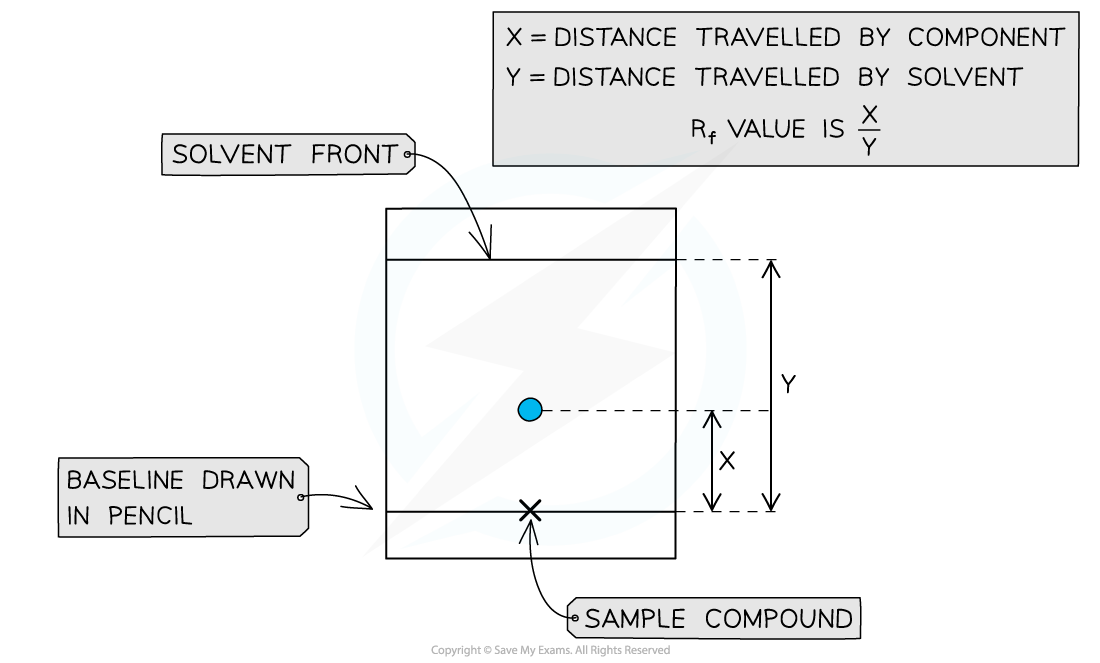
How is a substance identified using a TLC chromatogram?
Calculate its Rf value and compare it with the Rf value of a known substance (in a database) under the same conditions.
What do high and low Rf values show?
Low Rf means strong attraction to the stationary phase, so the substance moves less.
High Rf means weaker attraction to the stationary phase, so the substance moves further.
How can a TLC chromatogram show whether a sample is pure or a mixture?
One spot suggests a pure substance.
More than one spot suggests a mixture.
What are the key points to remember when carrying out TLC?
Draw the baseline in pencil.
Keep the solvent below the baseline.
Mark the solvent front before it evaporates.
Use the chromatogram to calculate Rf values.

Why is the baseline drawn in pencil and why must the solvent be below it?
Pencil is used because it does not dissolve in the solvent.
The solvent must be below the baseline so the sample does not dissolve straight into the solvent.

What is retention time in gas chromatography?
Retention time is the time taken for a substance to pass through the column to the detector.
How are substances identified using gas chromatography?
Substances are identified by comparing their retention times with those of known substances, using a database
What does a gas chromatogram show?
Each peak represents a different substance.
The retention time helps identify the substance.
The peak area shows the amount of the substance present.

How are the proportions of substances in a mixture found from a gas chromatogram?
By comparing the peak areas.
Percentage of substance X = (peak area of X / total peak area) x 100.
How is a calibration curve used in gas chromatography?
Calibration curve (GC):
👉 Run the same substance at known concentrations
👉 Record their peak areas
👉 Draw a graph:
peak area (y) vs concentration (x)
👉 For the unknown:
Find its peak area
Use the graph to read off the concentration

What is one limitation of identifying substances by TLC or gas chromatography?
Different substances can sometimes have the same or very similar Rf values or retention times, so identification may not be certain from one result alone
What are the stationary phase and mobile phase in gas chromatography?
stationary phase = high boiling point liquid in the column
mobile phase = inert carrier gas (eg helium)
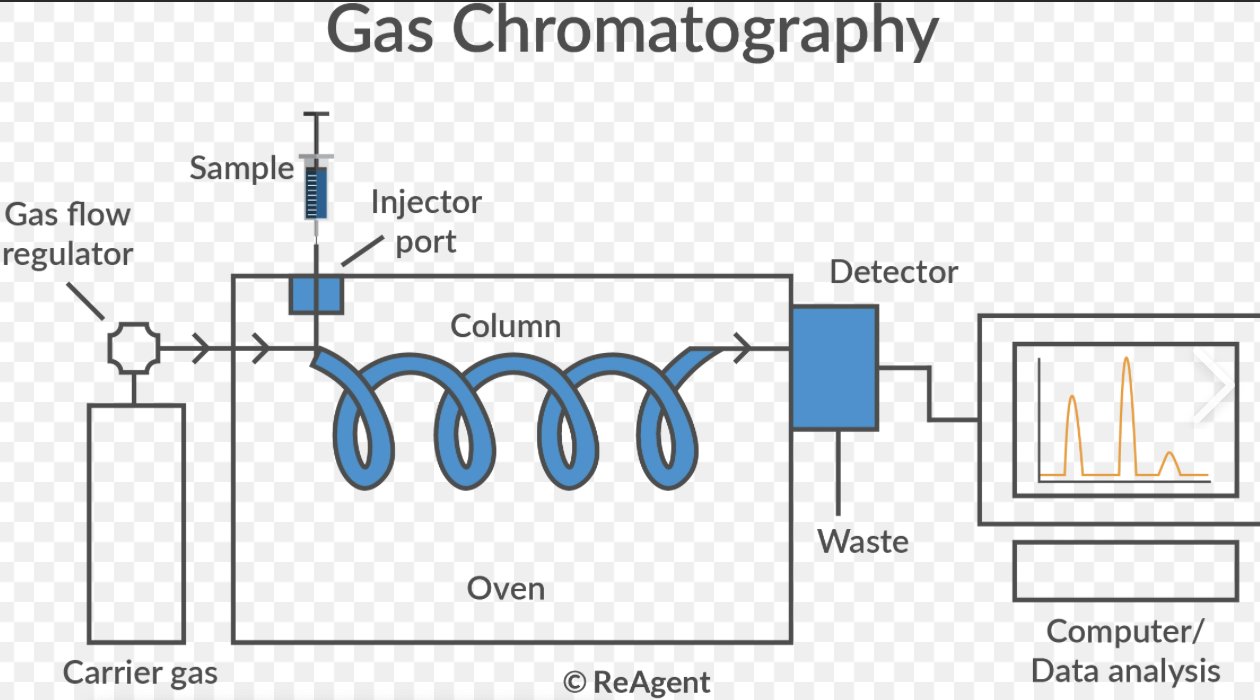
How does a gas chromatograph work?
A small sample of a volatile mixture is injected into the gas chromatograph.
The mobile phase, an inert carrier gas (ie He) carries the sample through a column.
The column contains the stationary phase, a high boiling point liquid.
Different substances move through the column at different speeds depending on their interactions with the stationary phase, so they leave the column at different times.
These are their retention times.
A detector records this as a chromatogram with separate peaks.
How do you determine the number of carbon environments from a carbon-13 NMR spectrum?
The number of peaks represents the different carbon environments.
Each unique carbon environment (with different bonding or surroundings) shows a separate peak.
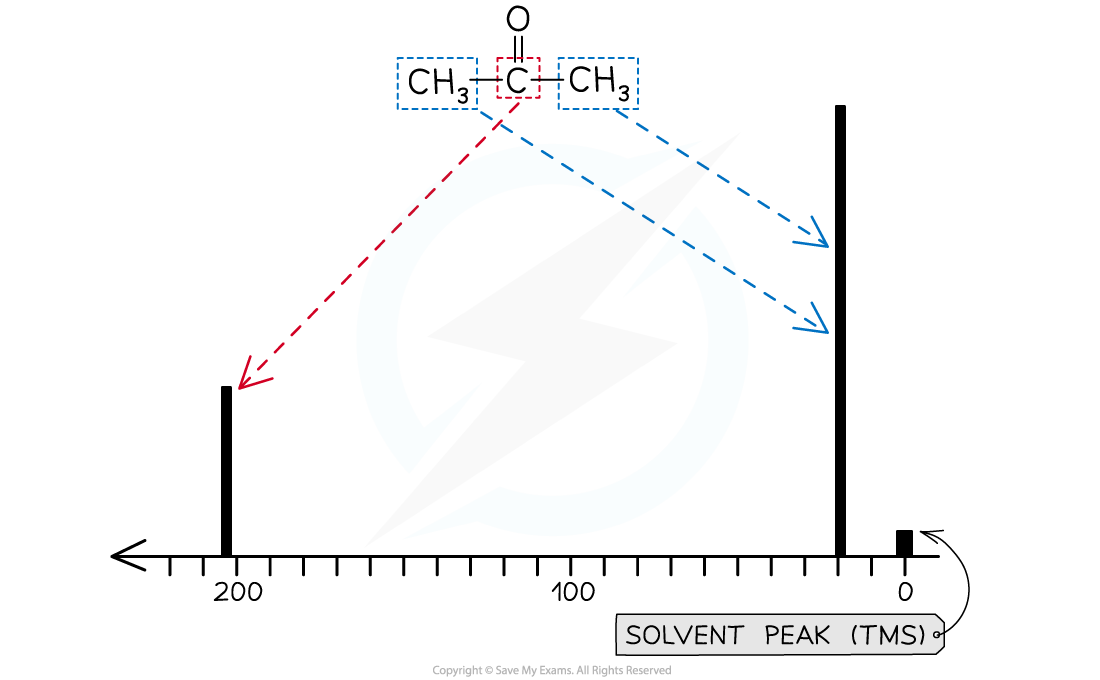
What do the chemical shifts in a carbon-13 NMR spectrum tell you?
The chemical shift indicates the type of carbon environment.
Peaks at different δ values correspond to different types of bonds (e.g., C–C, C–Cl, C–C=O).
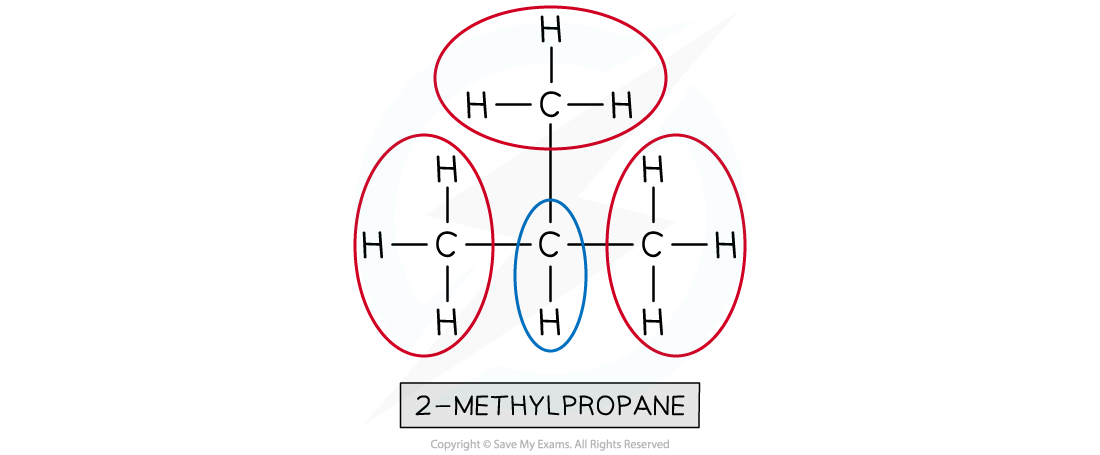
How do you determine the number of proton environments from a proton NMR spectrum?
The number of peaks indicates the different proton environments.
Each unique proton environment gives a distinct peak.
What do chemical shifts in proton NMR spectroscopy represent?
The chemical shift tells you the type of proton environment (e.g., alkyl, alkene, aromatic).
How are integration traces used in proton NMR spectroscopy?
Integration traces show the relative number of protons in each environment.
The area under each peak corresponds to the number of protons contributing to that peak.
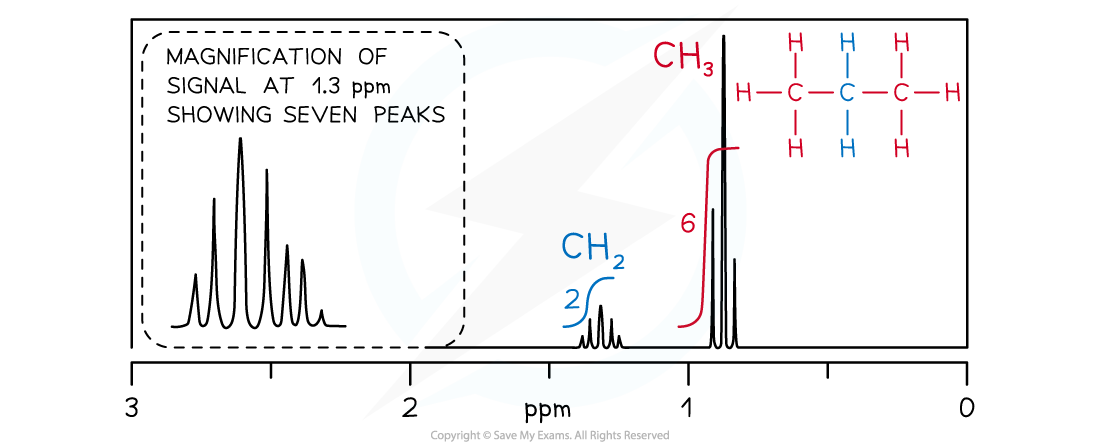
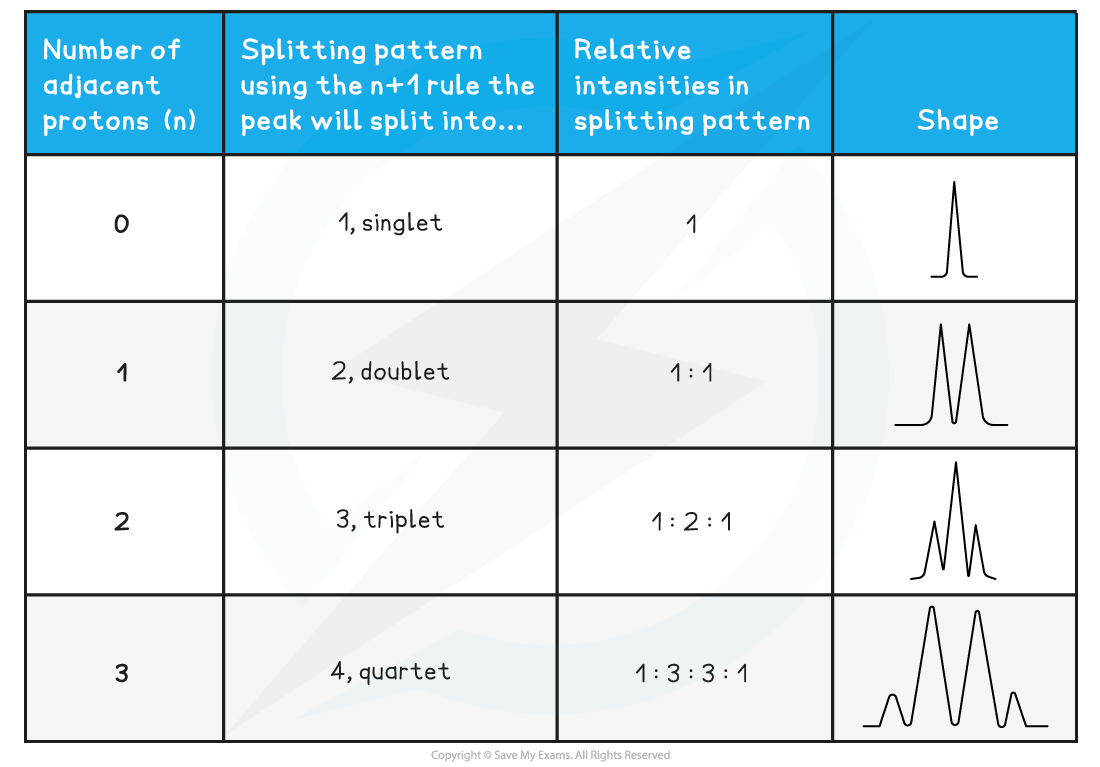
How is spin-spin coupling analysed using the n+1 rule?
For n protons adjacent to the observed proton, the pattern is n+1 peaks.
Singlet: No adjacent protons.
Doublet: One adjacent proton (or CH).
Triplet: Two adjacent protons (or CH₂).
Quartet: Three adjacent protons (or CH₃).
Heptet: Six adjacent protons (or 2 x CH₃)
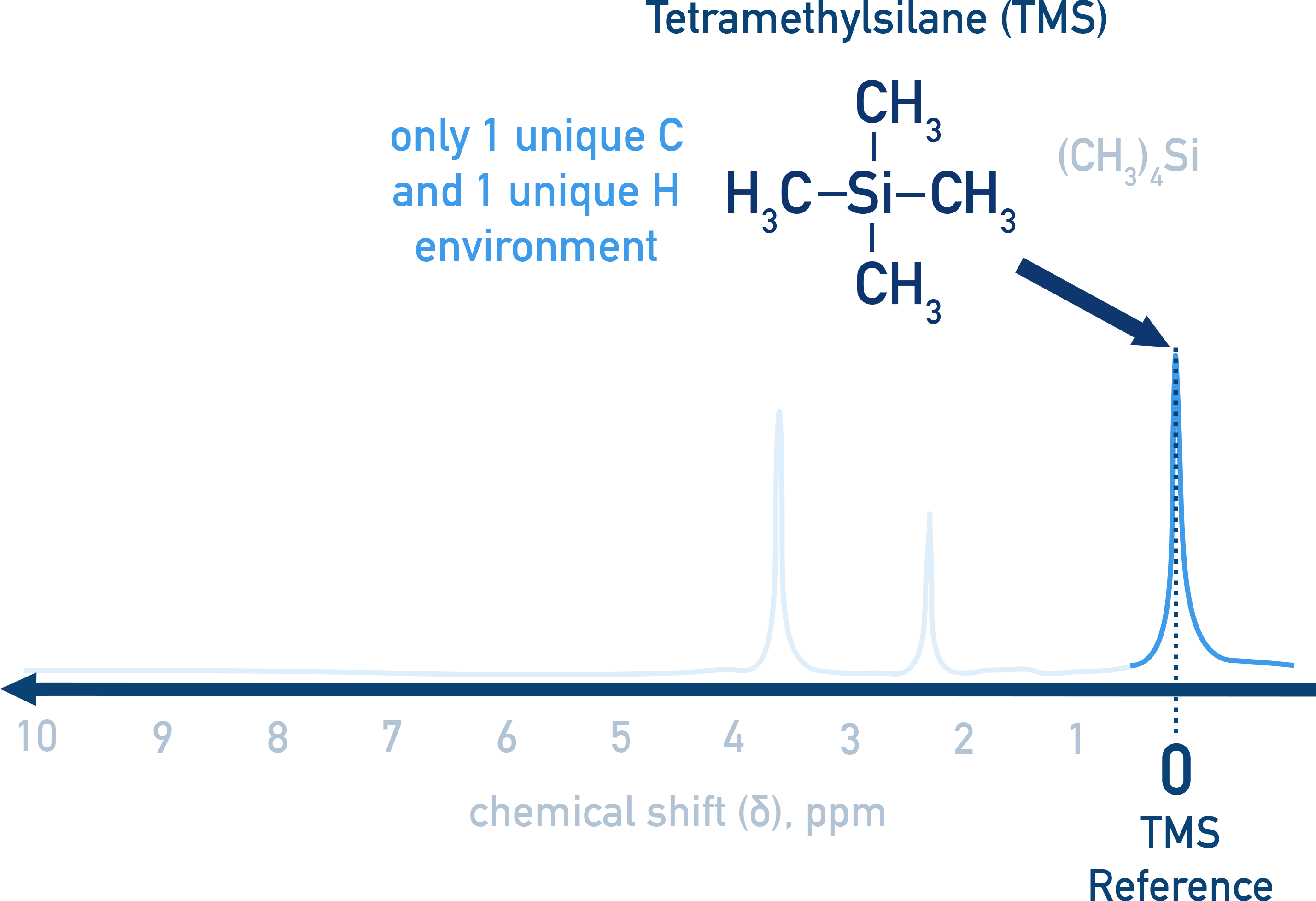
Why is tetramethylsilane (TMS) used as a reference in both NMR spectroscopies ?
TMS has no splitting and a sharp, distinct signal at 0 ppm, making it a universal reference point for chemical shifts.
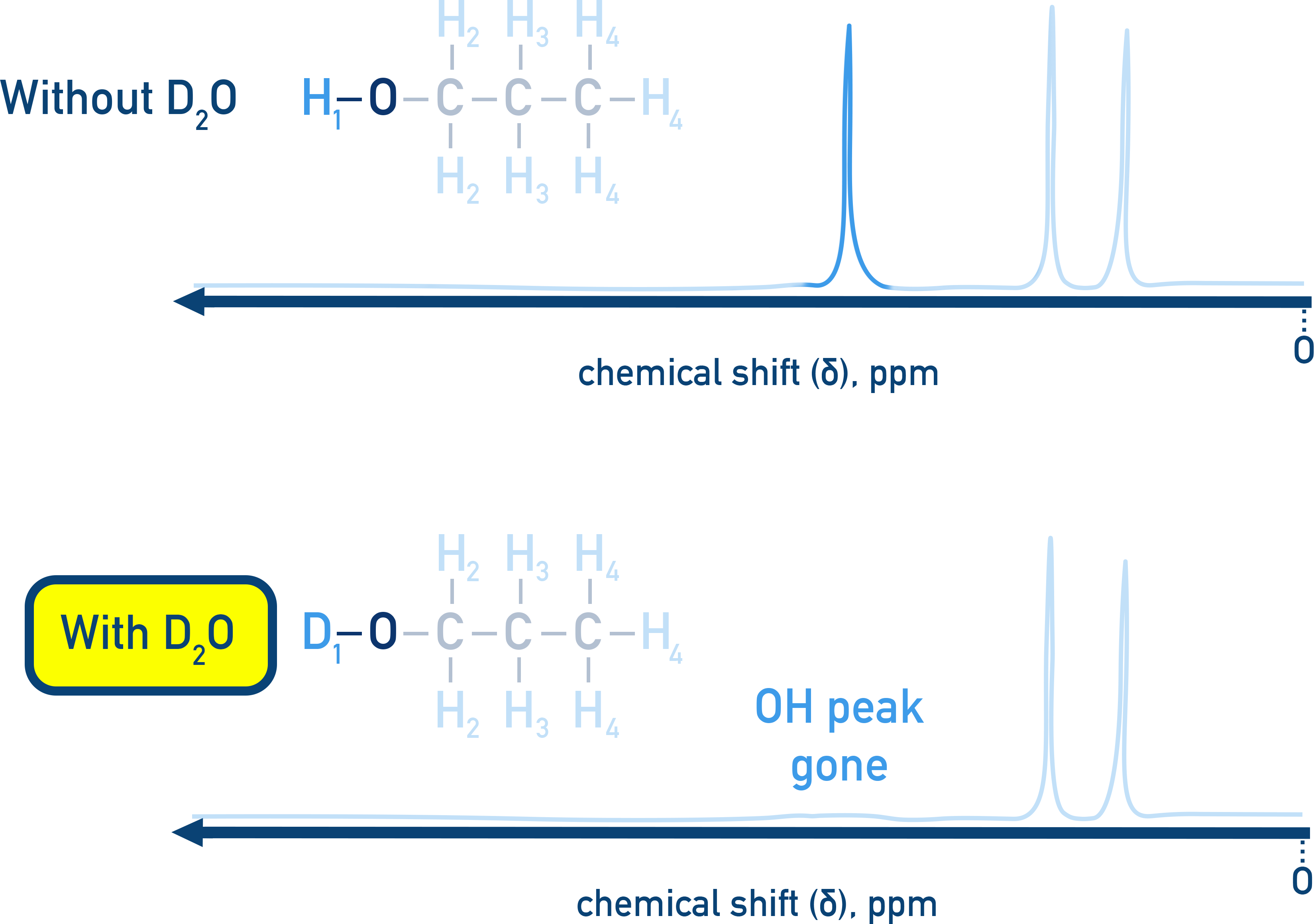
How can O-H and N-H protons be identified in proton NMR?
They show up as broad peaks of variable chemical shift with no splitting pattern, to identify them:
Run a normal H NMR spectrum.
Add D₂O, shake, and run a second spectrum.
O-H and N-H peaks disappear due to exchange with D₂O.
H replaced with D [D not detected by H NMR]
What does each of elemental analysis, mass spectra, IR spectra and NMR spectra tell us about an organic molecule?
Elemental → empirical formula
Mass → molecular mass and fragments of structure
IR → bonds present and functional groups
NMR → environments and structural formula
Why are deuterated solvents used in NMR? (+ give example)
Deuterated solvents, like CDCl₃, do not produce NMR signals because hydrogen is replaced by deuterium avoiding interference with proton NMR signals.
Common splitting patterns
Triplet and quartet → CH3CH2
Heptet and doublet → CH(CH3)2