AP CHEM MASTER SET
1/170
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
171 Terms
Phosphate

Hydroxide
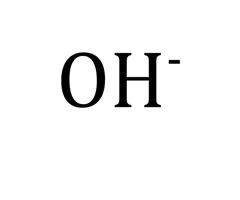
Sulfate
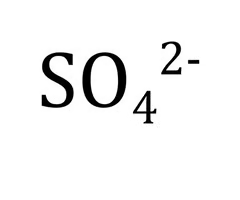
Acetate
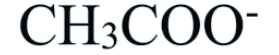
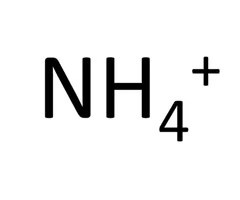
Ammonium
Chlorate
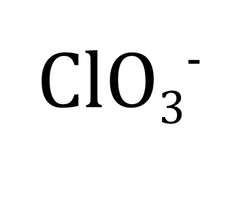
Nitrate
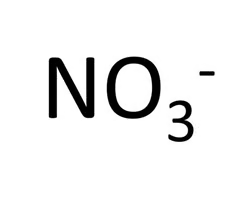
Permanganate
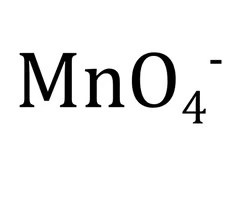
Chromate
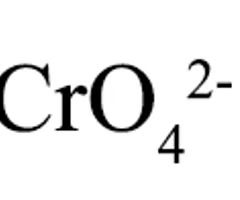
Carbonate
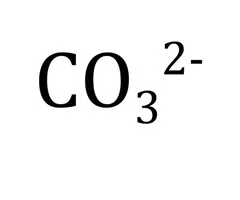
Cyanide

DiChromate
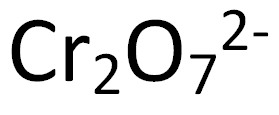
Nitrite
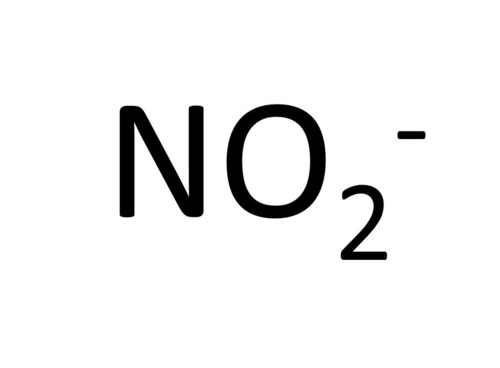
Sulfite
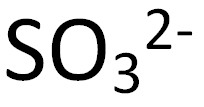
Hydrogen Carbonate
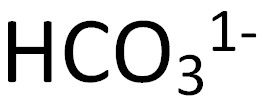
Perchlorate
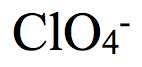
Chlorite
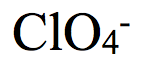
Hypochlorite
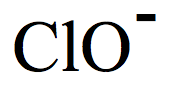
The oxidation state of neutral atoms not bonded to atoms of other elements is…
an oxidation state of 0
The oxidation state of of an ion is…
equal to the charge on that ion
In most compounds the oxidation state of Oxygen is…
-2
The Oxidation state of oxygen in peroxides is…
-1
The oxidation state of Hydrogen that’s bonded to a nonmetal is…
+1
The oxidation state of Hydrogen that’s bonded to a metal is…
-1
In the absence of oxygen the oxidation state of the most electronegative element is…
equal to its most common charge
The combined oxidation states in a neutral atom is equal to… and in an polyatomic ion its equal to…
0 and the charge on that ion
The name and angle of the VSEPR structure w/ a steric no. of 2 and 0 lone pairs
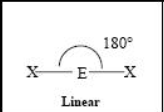
The name and angle of the VSEPR structure w/ a steric no. of 3 and 0 lone pairs
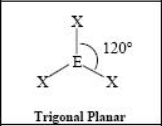
The name and angle of the VSEPR structure w/ a steric no. of 3 and 1 Dlone pairs
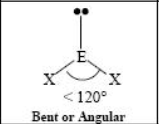
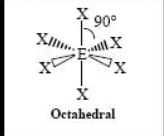
The name and angle of the VSEPR structure w/ a steric no. of 4 and 0 lone pairs
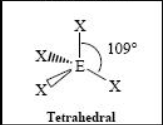
The name and angle of the VSEPR structure w/ a steric no. of 4 and 1 lone pairs
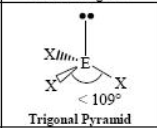
The name and angle of the VSEPR structure w/ a steric no. of 4 and 2 lone pairs
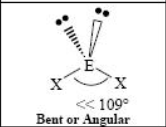
The name and angle of the VSEPR structure w/ a steric no. of 5 and 0 lone pairs
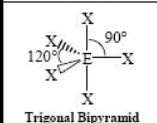
The name and angle of the VSEPR structure w/ a steric no. of 5 and 1 lone pairs
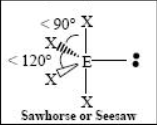
The name and angle of the VSEPR structure w/ a steric no. of 5 and 2 lone pairs
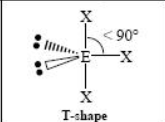
The name and angle of the VSEPR structure w/ a steric no. of 5 and 3 lone pairs
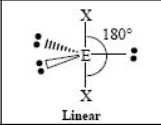
The name and angle of the VSEPR structure w/ a steric no. of 6 and 0 lone pairs
The name and angle of the VSEPR structure w/ a steric no. of 6 and 1 lone pairs
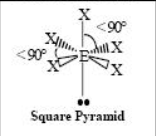
The name and angle of the VSEPR structure w/ a steric no. of 6 and 2 lone pairs
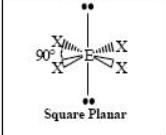
The name and angle of the VSEPR structure w/ a steric no. of 6 and 3 lone pairs
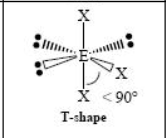
The name and angle of the VSEPR structure w/ a steric no. of 6 and 4 lone pairs
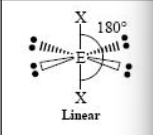
A variant of a chemical element with the same no. of protons and a different amount of neutrons…
Isotopes
The atomic model and theory that treated atoms as solid featureless spheres and it was the first atomic theory based on empirical evidence…
Dalton’s Atomic Theory
Described atoms as uniform spheres of positively charged soup or jelly w/ negatively charged electrons throughout
Thomson’s Plum Pudding Model
The model that was discovered with gold foil experiment
Rutherford’s Nuclear model
Described electrons being localized in levels around the nucleus
Bohr’s Atomic Model
Aufbau principle
Electrons will fill up orbitals with the lowest energy level first
Exceptions to the Aufbau principle
Chromium and Copper
The energy required to remove an electron
Ionization Energy
Ionization energy Periodic Trend
Increases Left to Right
Decreases down a group
The tendency of an atom to attract and form bonds with electrons
Electronegativity
The energy released when an electron is gained by a neutral version of an atom
Electron Affinity
Electronegativity Trends
Increases left to right
Decreases down a group
Atomic Radius Trends
Decreases left to right
Increases down a group
When inner shell electrons repel outer shell electrons and push them away from the positive attraction from the nucleus
Electron shielding
Ionization energy will always be…
positive
Factors that affect Ionization energy
Nuclear charge
Electron sheilding
Zeff
Distance of valance electrons from nucleus
Atomic Theory
All matter is composed of atoms
Elements of the same type are composed of similar atoms
Chemical reactions are merely rearrangement of atoms
Wavelength and Frequency of light are…
inversely proportional
Electrons will fill empty orbitals first before pairing up and they require different spins
Hund’s Rule
Bonds formed by metals and nonmetals and occurs during the transfer of electrons
Ionic bonds
Bonds formed between non-metals and from the sharing of electrons
Covalent bonds
Properties of Ionic Bonds
Low conductivity due to localized electrons for solids
High conductivity in aqueous solutions due to free moving electrons
Non-malleble
High melting points
High boiling points
Properties of Covalent Bonds
Very low conductivity
Non-malleble
Lower melting points
non-ductile
A single bond consists of…
One sigma bond
A double bond consists of…
One sigma and one pi bond
A triple bond consists of…
One sigma and two pi bonds
Caused by differences in polarity, the higher electronegative atom has a negative pole
Dipole-Dipole Force
A specific type of another bond that’s due to H being very positive due to its low electronegativity and being attracted to very electronegative atoms such as (F,O,N)
Hydrogen bonding
Random movement of electrons create temporary dipoles
London dispersion forces
The thing to be dissolved
Solutes
The thing its dissolved in…
Solvent
Assumptions made by Kinetic molecular theory
Gases are in constant random motion
All collisions between are elastic
The volume of molecules is negligible
There is no attraction between molecules
All gasses in a sample have the same avg. KE
Gases deviate from ideal…
high pressures
low temperatures
as pressure increases volume decreases
Boyle’s Law
as temp increases volume increases
Charles Law
Solubility Rules; Compounds with nitrates are…
usually soluble
Solubility Rules; Compounds with any group 1 metal salts are…
usually soluble
Solubility rules; compounds with ammonium are…
usually soluble
solubility rules; compounds with acetates are…
usually soluble
Solubility rules; Compounds with Phosphates are…
usually insoluble
Solubility Rules; compounds with hydroxides are…
usually insoluble
Solubility rules; compounds with carbonates are…
usually insoluble
1 mol of a gas is equal to…
22.4 liters
The measure of the energy thats released or absorbed by the substance when bonds are formed or released during a reaction
Enthalpy (ΔH)
ΔH Formula (change in enthalpy)
ΔH = Hproducts - Hreactants
When products have more energy than the reactants at the end of a reaction
Endothermic reaction
When reactants have more energy than the products at the end of a reaction
Exothermic reaction
A reaction where ΔH is positive
Exothermic
A reaction where ΔH is negative
Endothermic
In an Exothermic reaction the E’a is…
greater than the Ea
In an Endothermic reaction the E’a is…
less than the Ea
Four ways to speed up a reaction
Raise the temperature
Increase the concentration
Decrease particle size
Add a catalyst
The reaction is a 0th order reaction if the graph of …. is a straight line
[A] vs time
The reaction is a 1st order reaction if the graph of … is a straight line
ln[A] vs time
The reaction is a 2nd order reaction if the graph of … is a straight line
1/[A] vs time
elementary step with 2 reactants
bimolecular step
elementary step with 1 reactant
unimolecular step
a elementary step with 3 reactants
termolecular step
an unstable high-energy transition state molecule formed after the reactant state but before the product state
Activated complex