AP Bio Unit One Study Set (created by Wes Besikof)
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
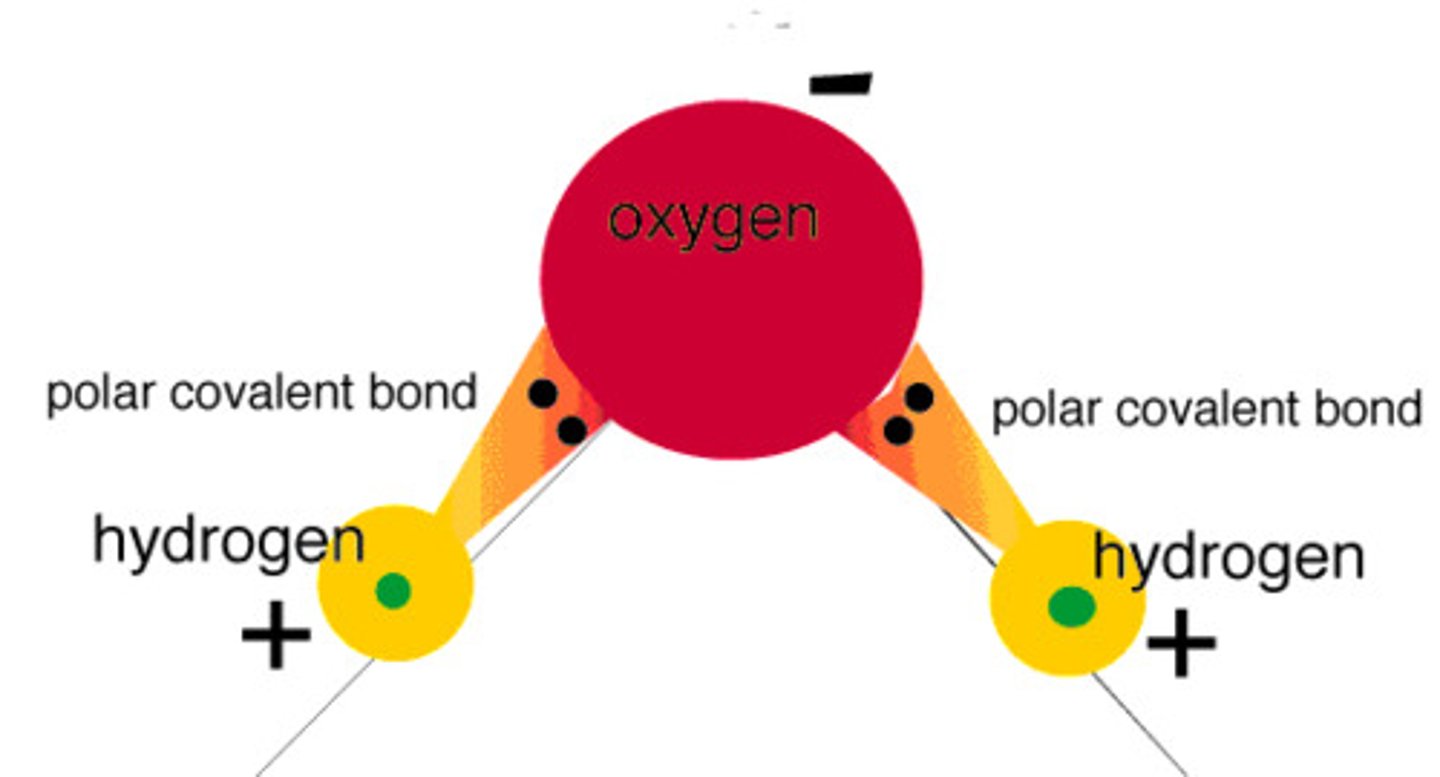
Why is water polar?
Oxygen is very electronegative and Hydrogen is not, so the electron distribution is off centered
Define Cohesion
Attraction between molecules of the same substance

Define Adhesion
attraction between molecules of different substances

Why does water have a high specific heat?
it takes energy to break the hydrogen bonds
Why does ice float?
When water freezes, the hydrogen bonds form crystal lattices that are spacious. (The H-bonds in liquid water are constantly forming and breaking...) This makes ice less dense than water, so it floats
Does water dissolve polar or non polar substances? Why?
Polar (or fully ionic) substances because the negatively charged Oxygen is attracted to the other positively charged atoms, while the positively charged hydrogen is attracted to negatively charged atoms. This separates polar molecules (or ions) from one another.
What is formed when water dissociates?
H3O+ and OH-
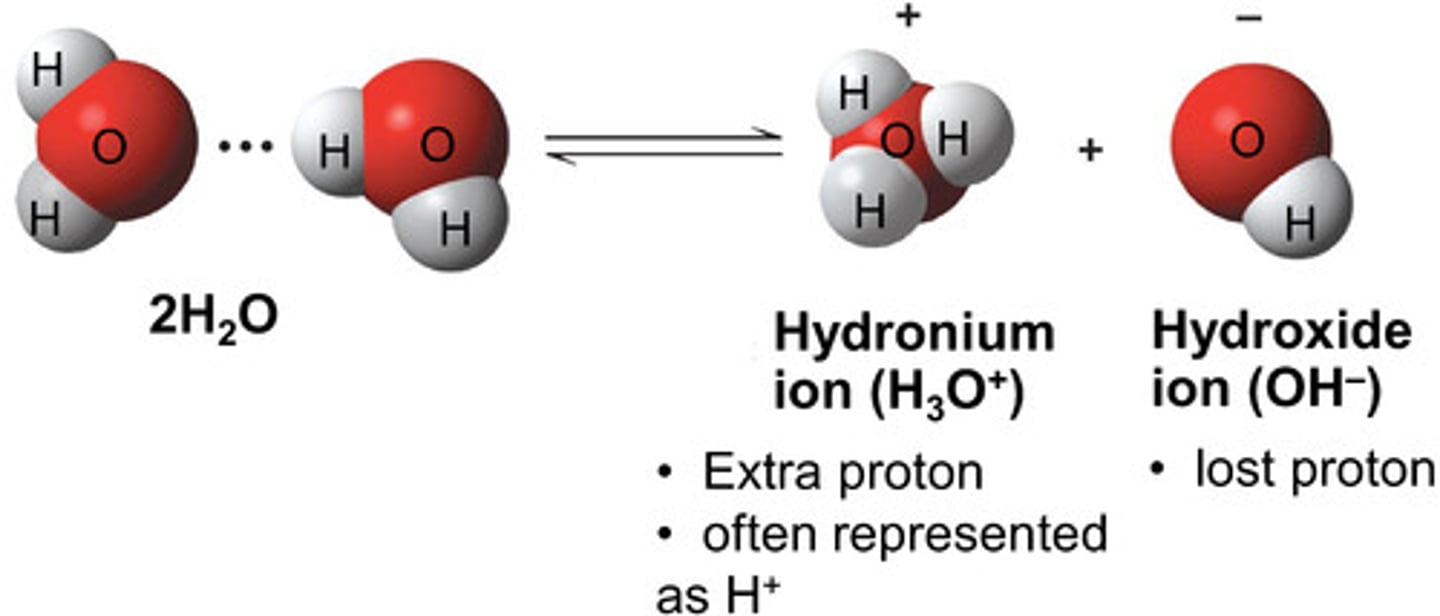
In water, the amount of H3O+ and OH0- are...
equal. This is why water's pH is 7 (neutral)
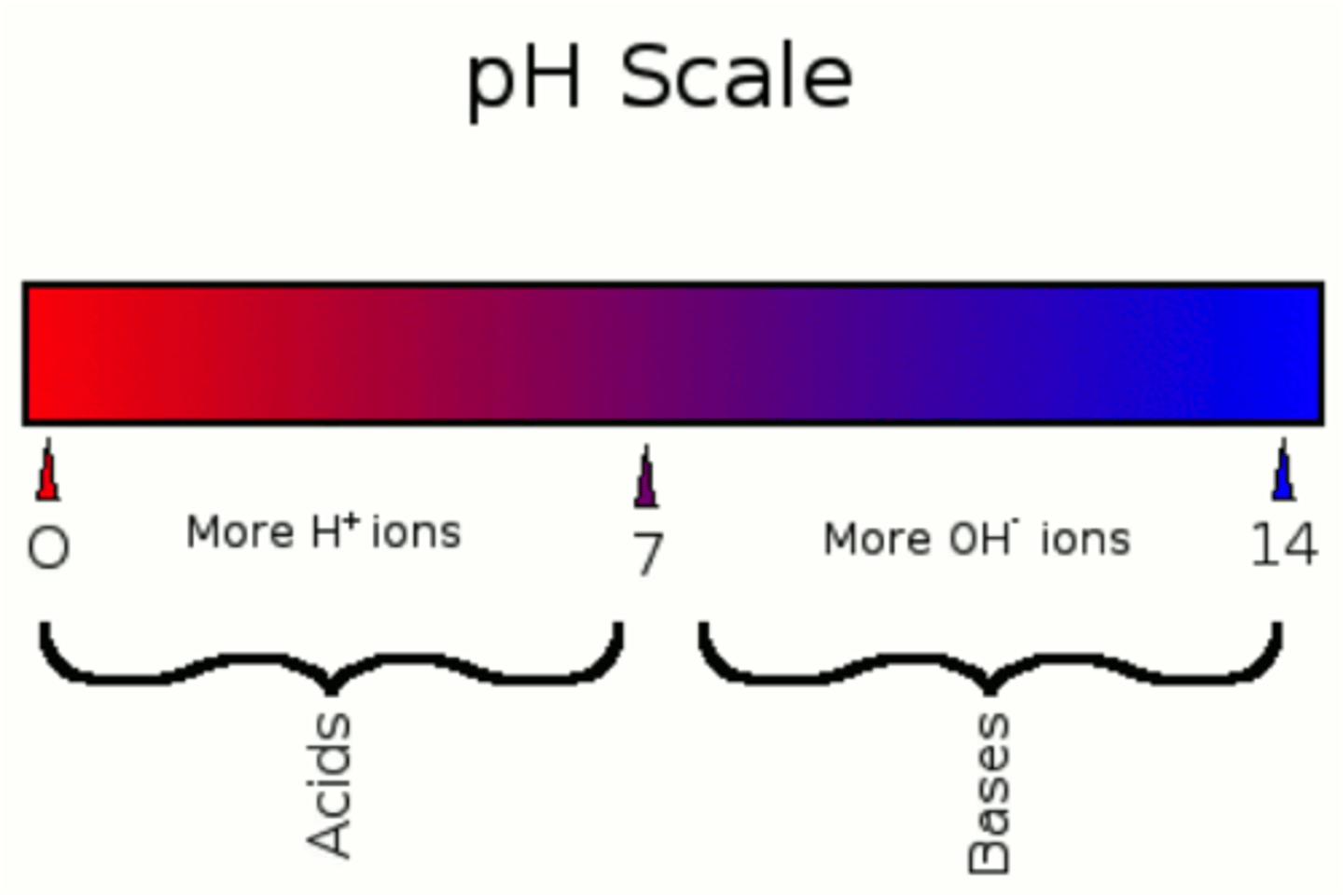
More hydrogen ions means...
lower pH (acidic)

Less hydrogen ions means...
higher pH (basic)

Acids donate or accept protons?
donate protons [H+]
![<p>donate protons [H+]</p>](https://knowt-user-attachments.s3.amazonaws.com/c88f4afe-cb33-4000-8ce5-136cf7ab7992.jpg)
Bases donate or accept protons?
accept protons [H+]
![<p>accept protons [H+]</p>](https://knowt-user-attachments.s3.amazonaws.com/df2cb835-91a4-4402-bb7b-9cd333daa58e.jpg)
Why is carbon so fundamental?
it's tetravalent, which means it makes 4 bonds to become stable. This leads to infinite variety
---OH
Hydroxyl Group
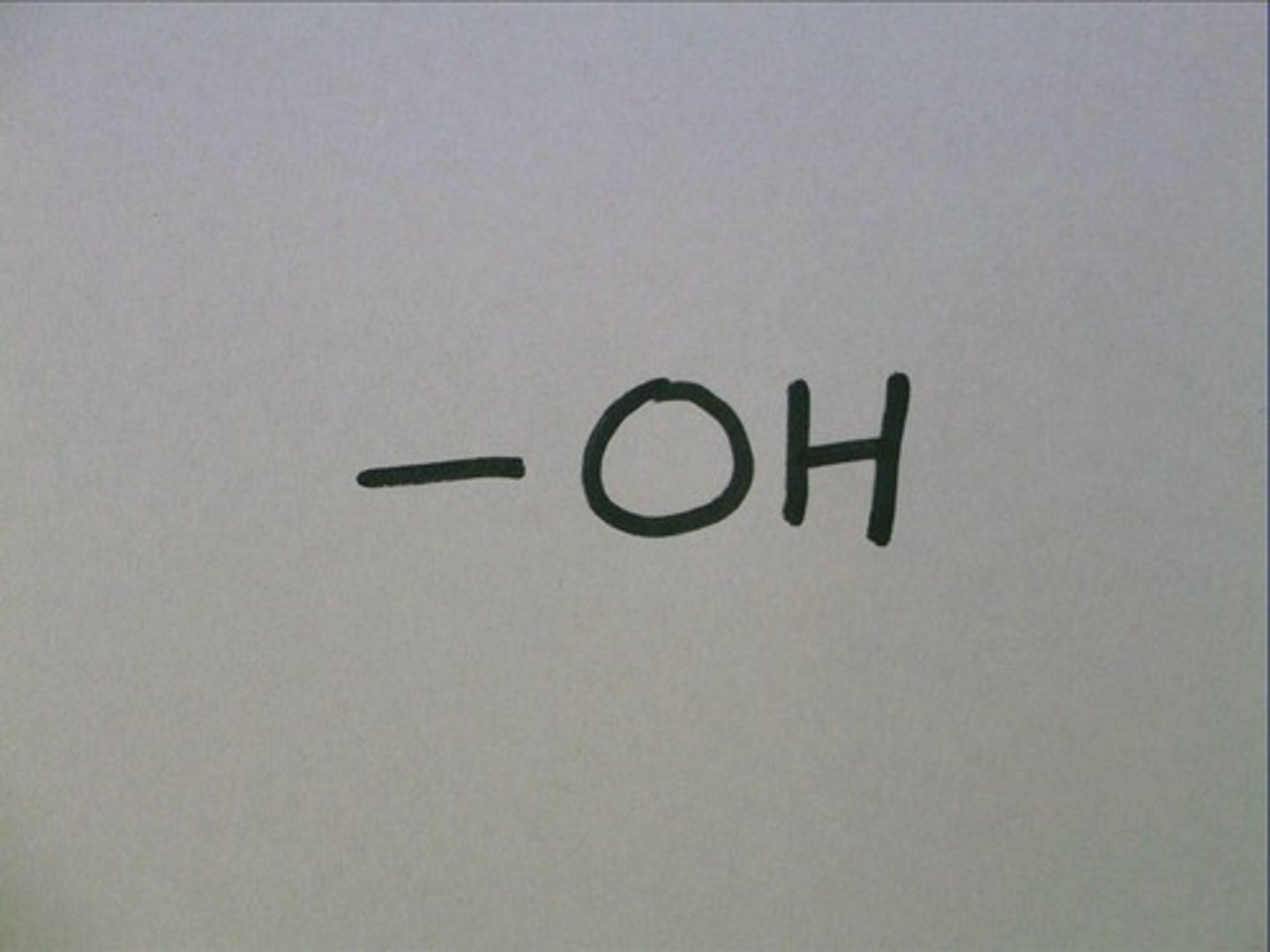
Hydroxyl group properties
polar (hydrophilic), can form hydrogen bonds
--C==O
carbonyl group
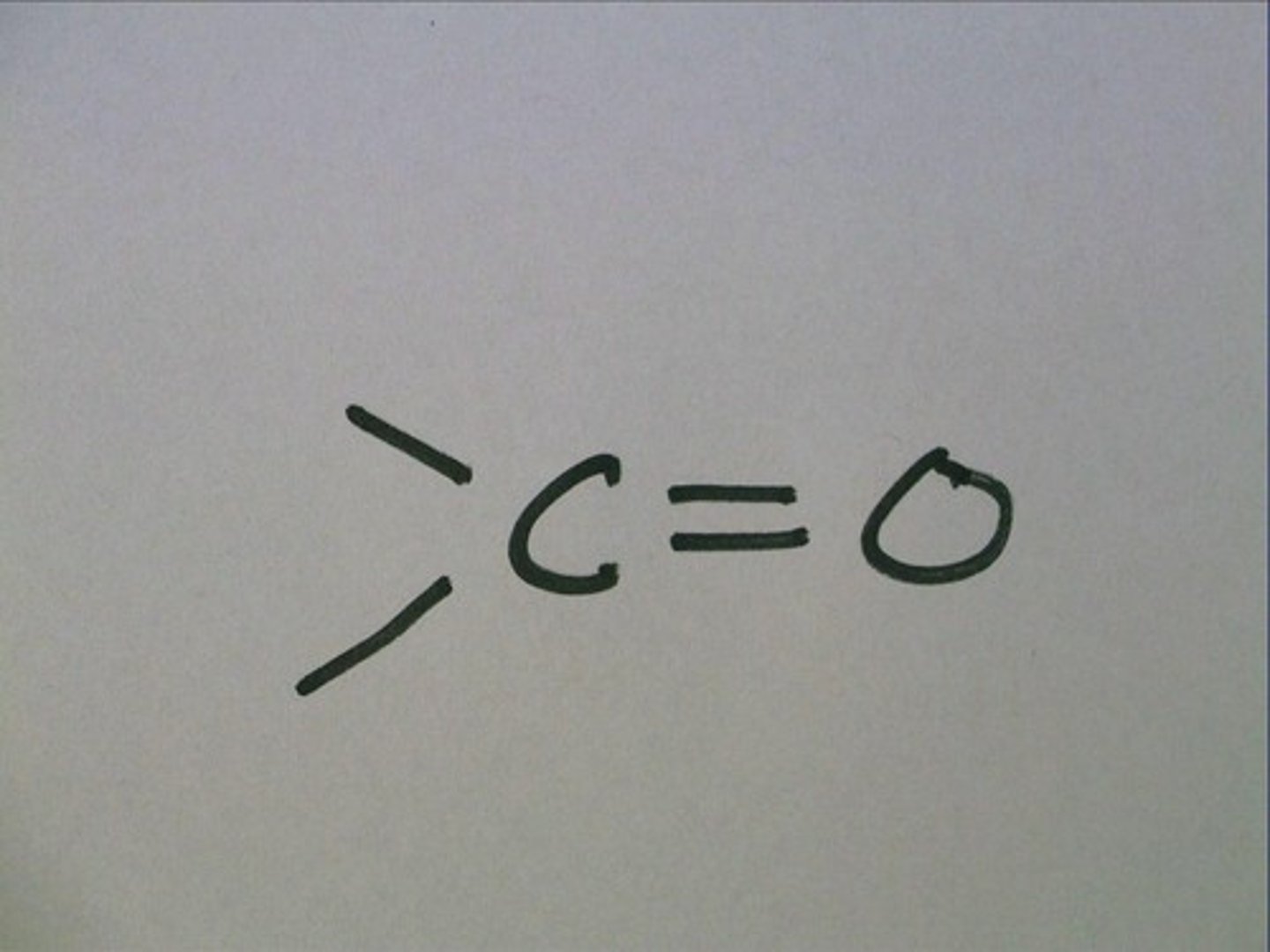
Carbonyl group properties
2 types of carbonyls
Ketone: a carbonyl group within the carbon skeleton
Aldehyde: a carbonyl group on the end of a carbon skeleton
--COOH
carboxyl group
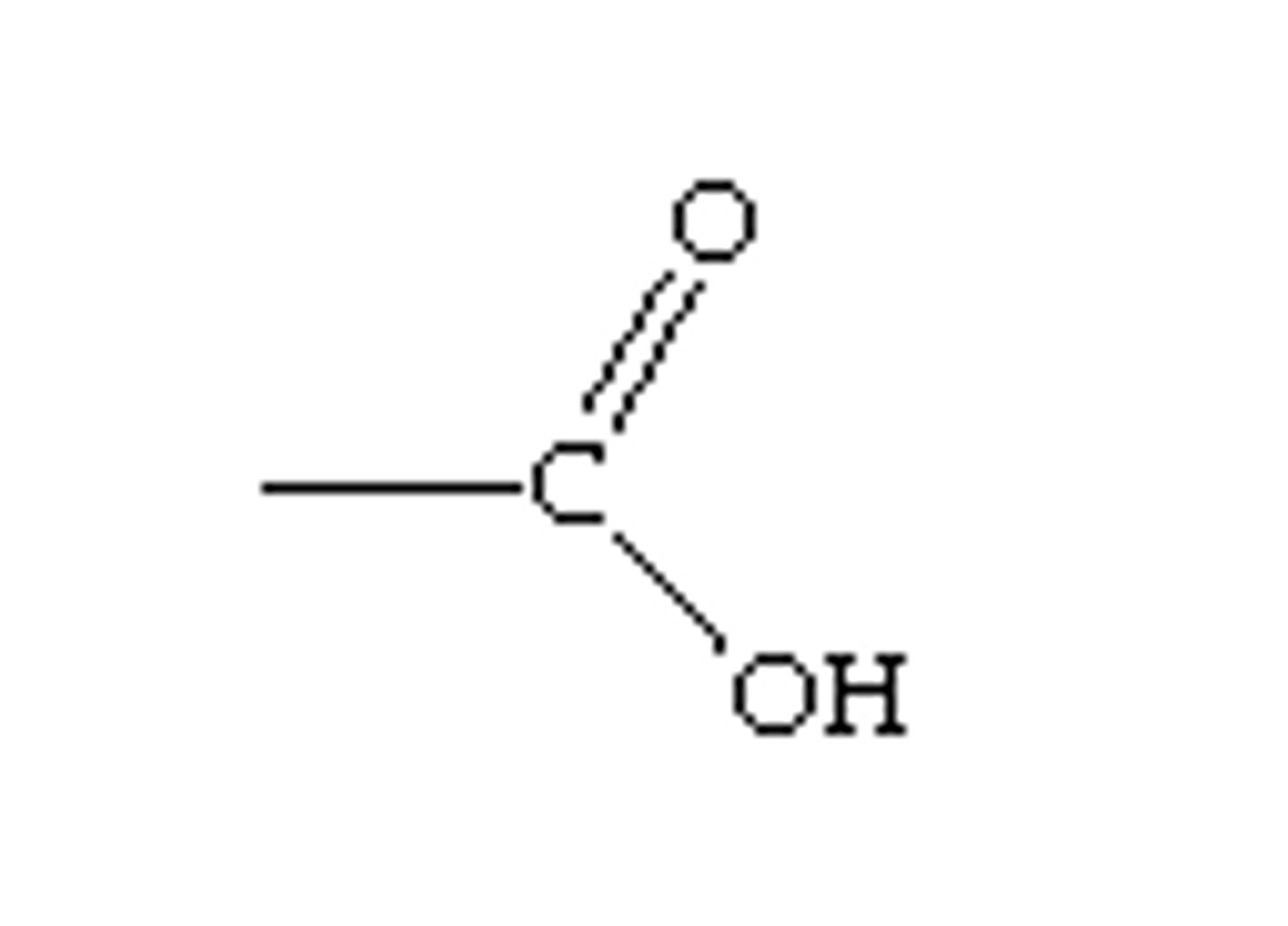
Carboxyl (acid) group properties
acts as an acid (can donate H+) because the covalent bond between oxygen and hydrogen is so polar
--NH2
Amino Group
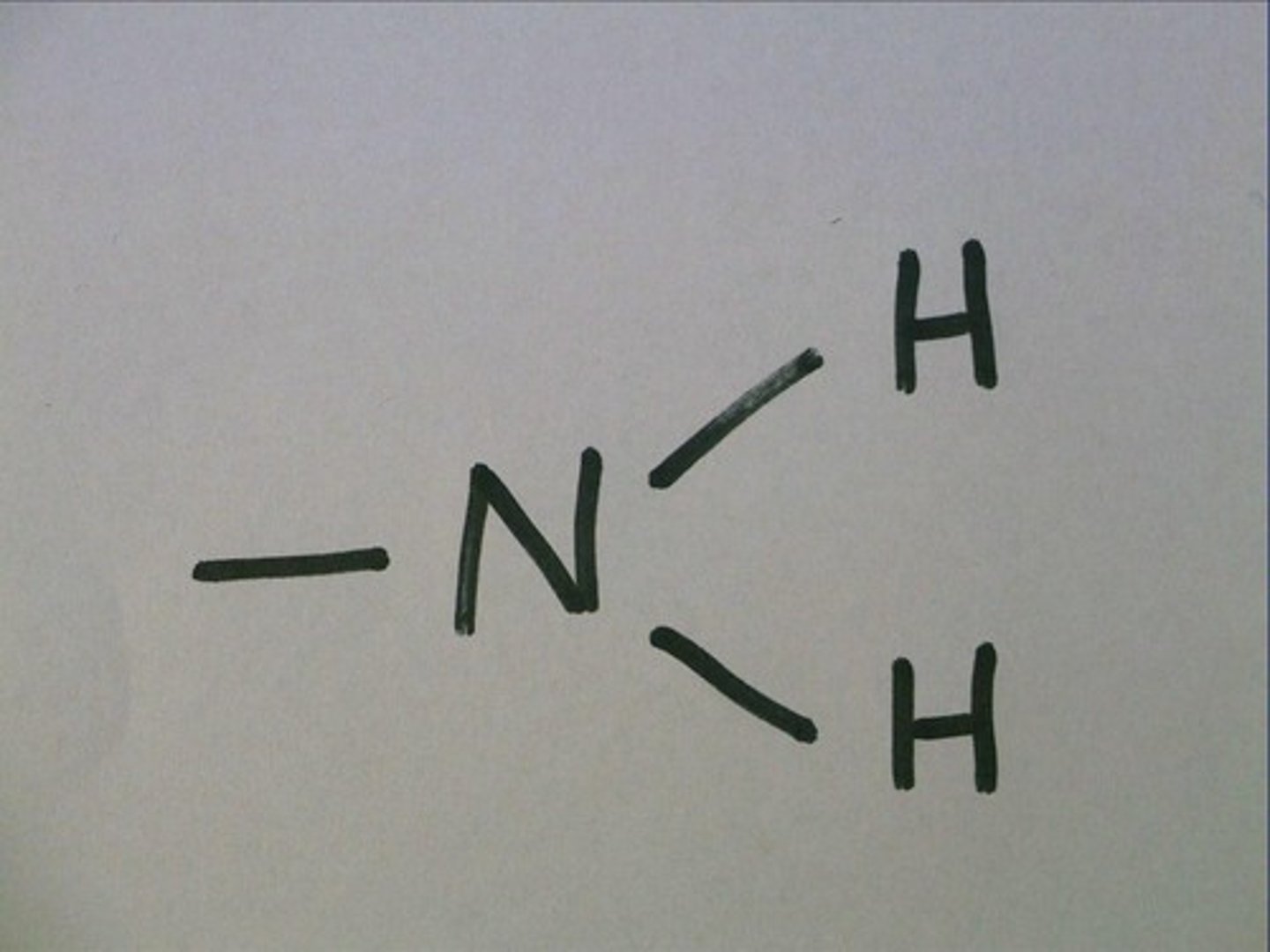
Amino properties
acts as a base, can pick up a proton (H+) from surrounding solution
--SH or --HS
Sulfhydryl Group
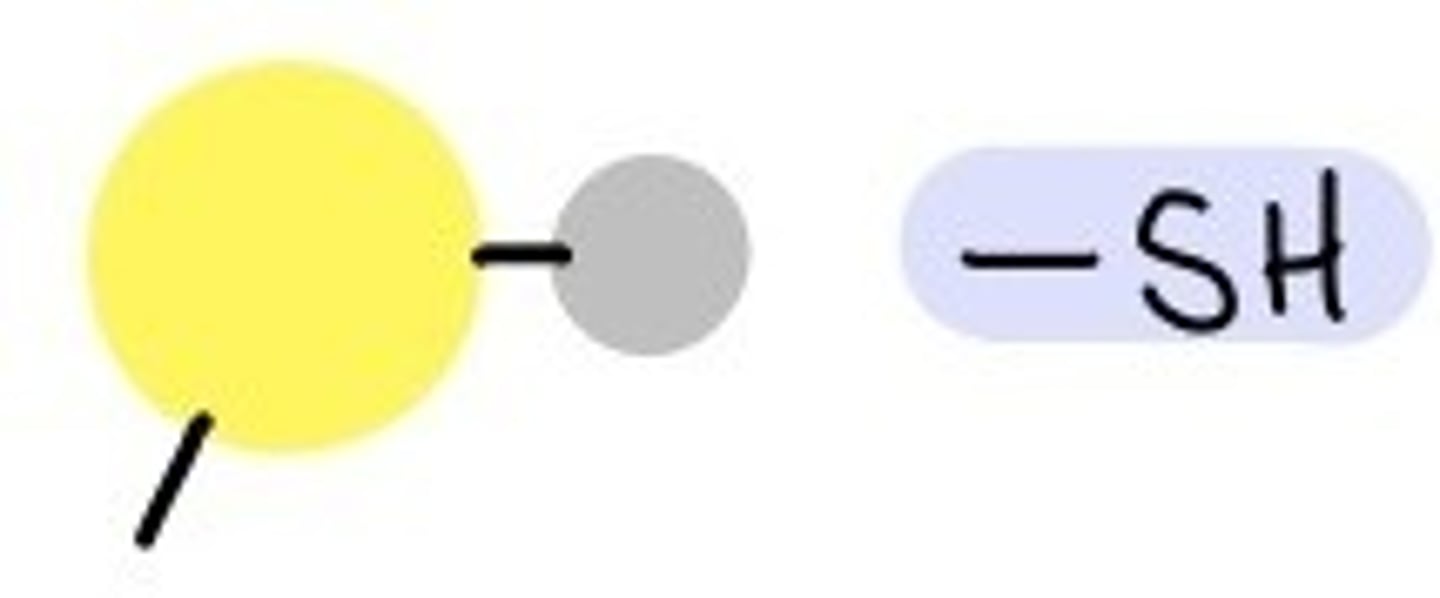
Sulfydral group properties
2 sulfhydral groups form a bridge (cross-linking) to stabilize a protein structure
--CH3
Methyl Group
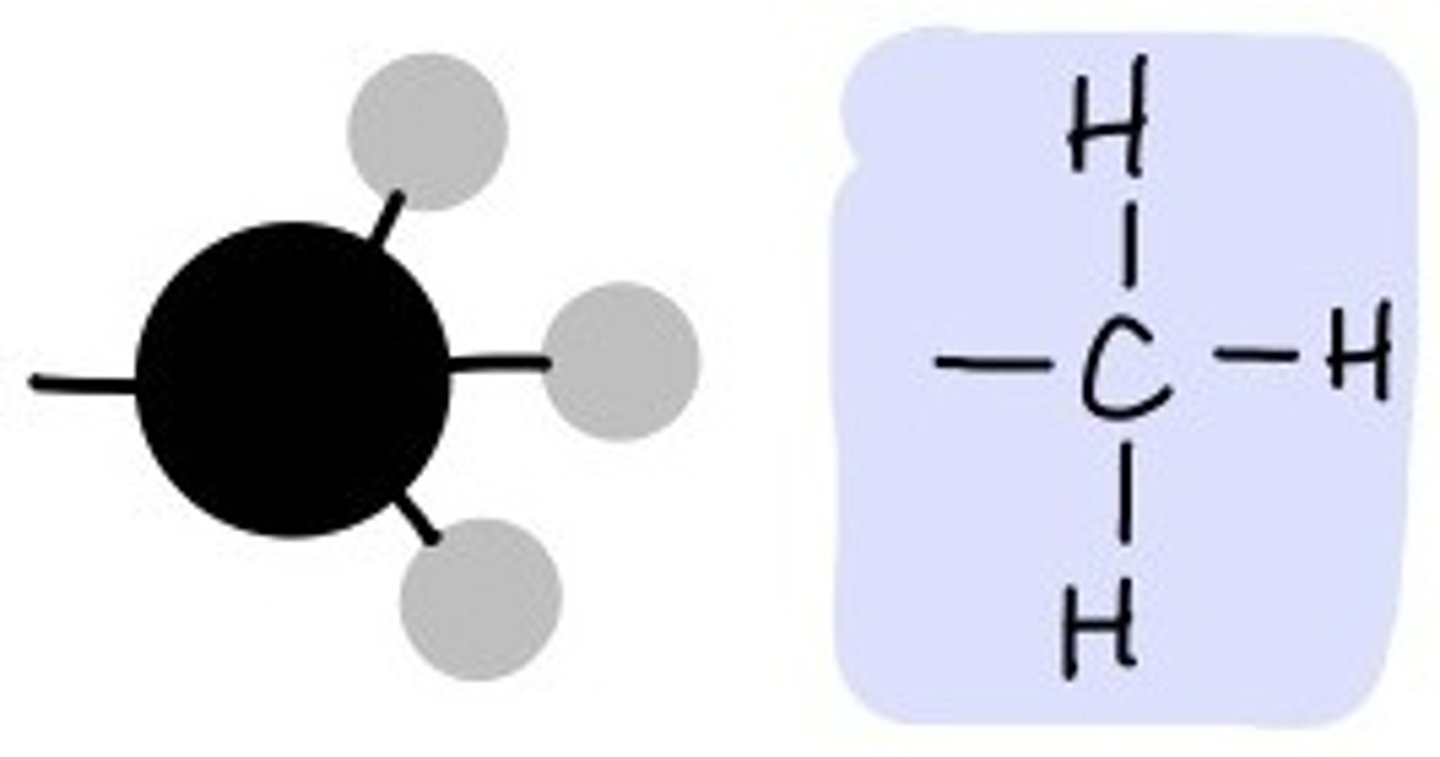
Methyl group properties
non-polar (hydrophobic);
addition of a methyl group to DNA, or to molecules bound to DNA affects the expression of genes (it essentially "shuts off" the expression of DNA)
--PO4
Phosphate Group
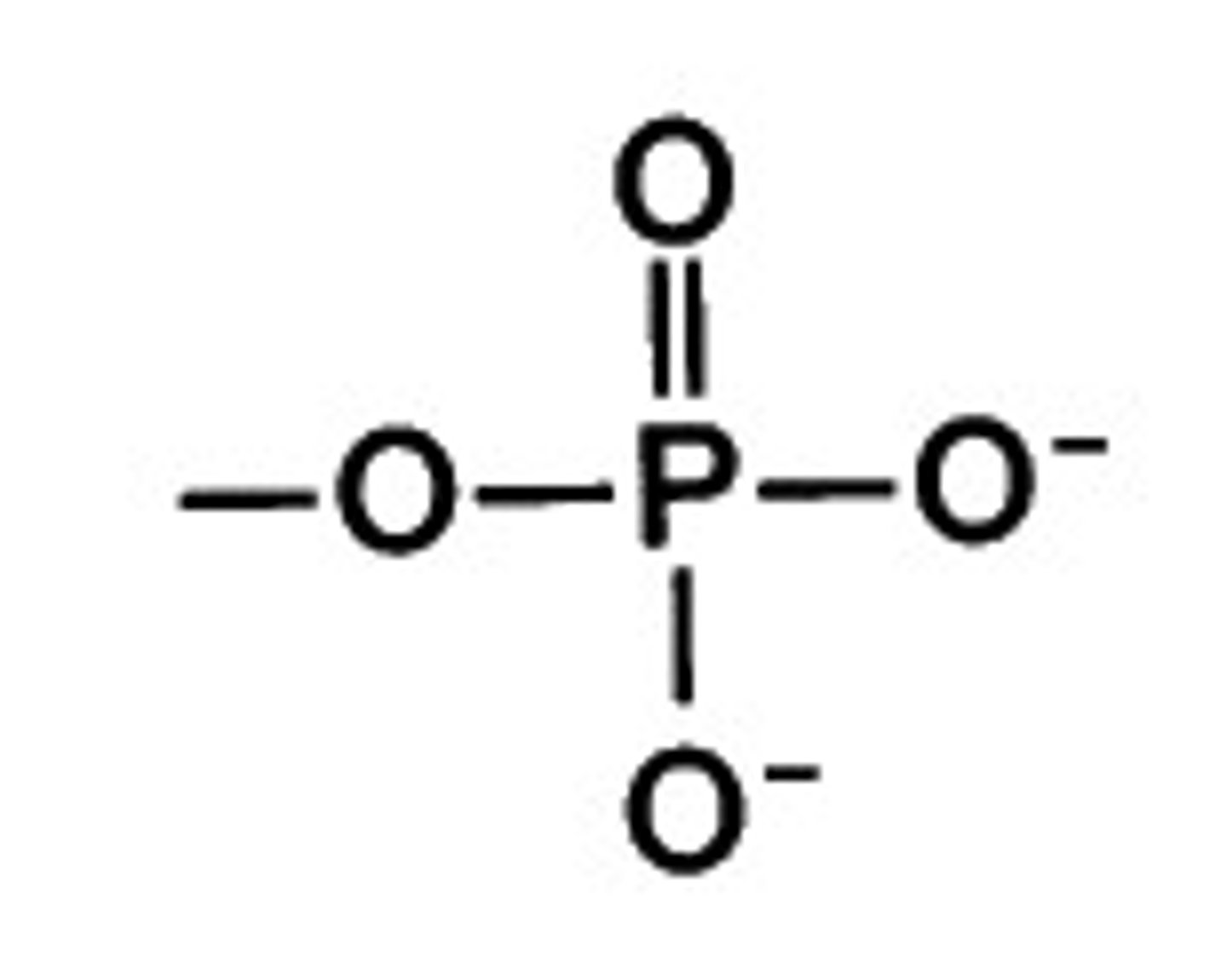
Phosphate group properties
Ionic Group (In biology, it's often referred to as just polar); Found in nucleotides, the monomers of nucleic acids, and in phospholipids, the building blocks of cell membranes.
What is a macromolecule?
a large molecule made of a few, common atoms. They accomplish all life functions and can be very complex
Name the 4 main macromolecules
Carbohydrates, Lipids, Proteins, and Nucleic Acids

except for lipids, macromolecules exist in two forms. What are they?
monomers and polymers
What is dehydration synthesis?
the process of joining two or more monomers to form a polymer by removing water to form a bond (opposite of hydrolysis)
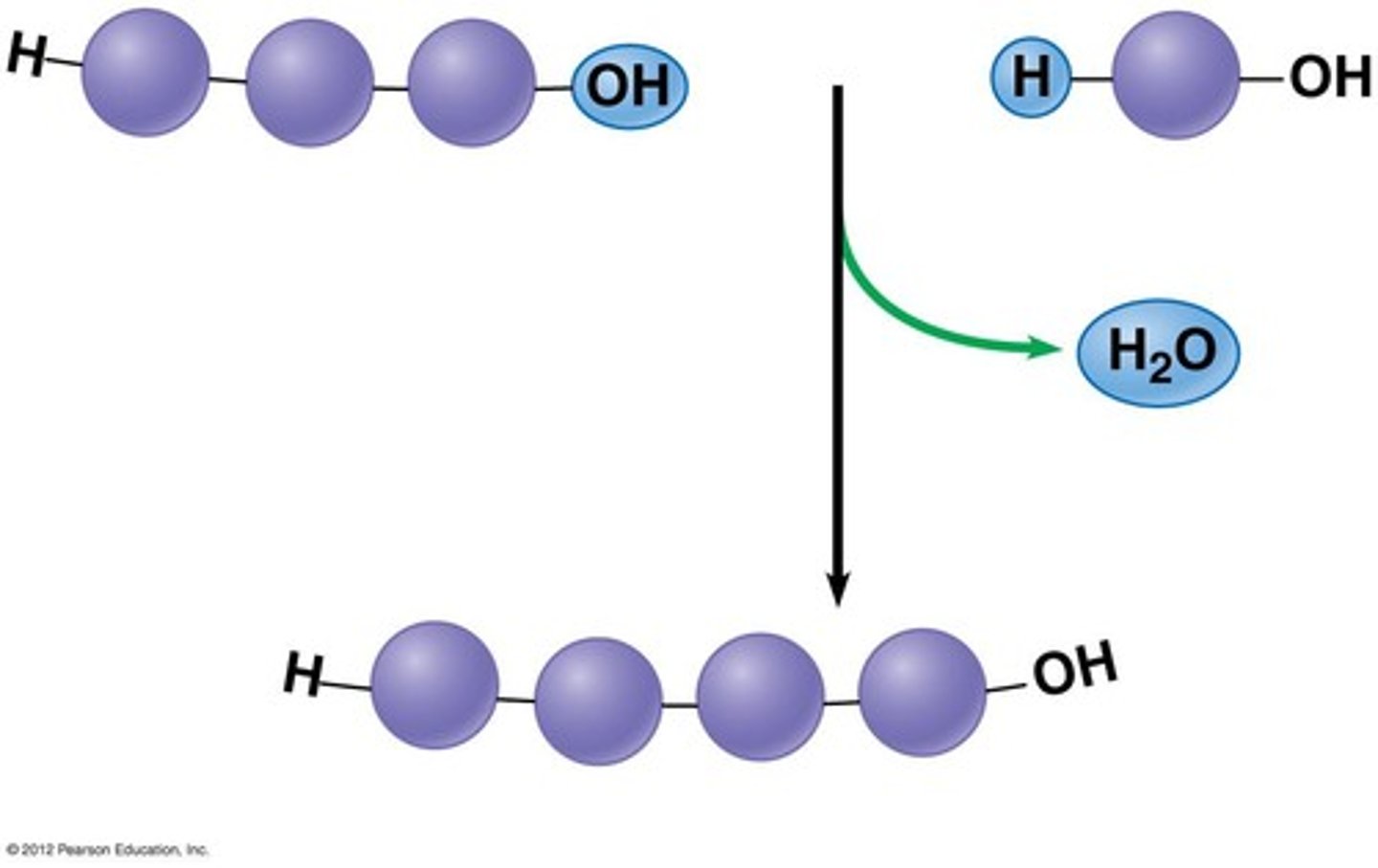
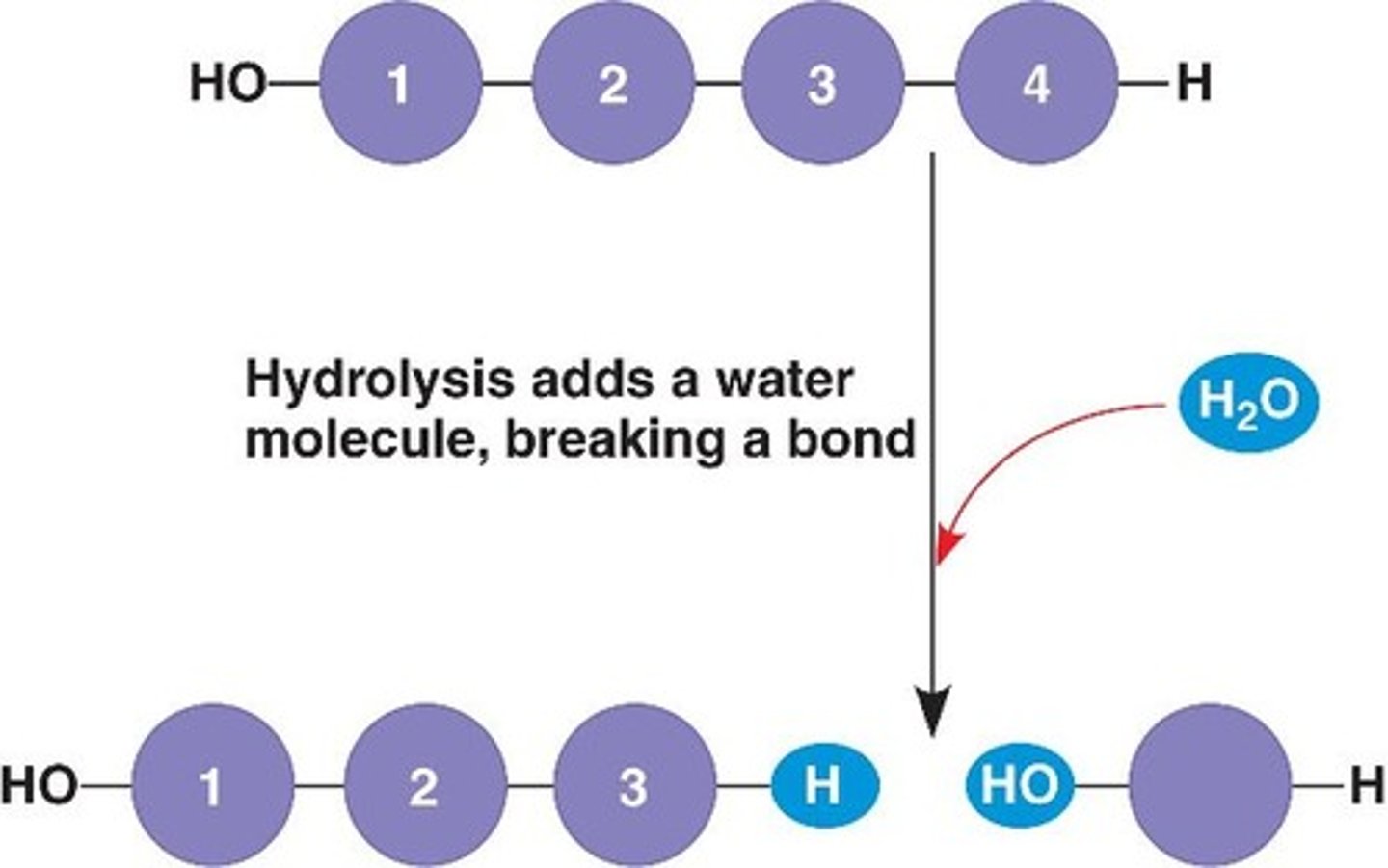
What is hydrolysis?
the process of breaking apart a polymer to form monomers by adding water (opposite of dehydration synthesis); "hydrolyzing" a molecule means adding water to break it up.
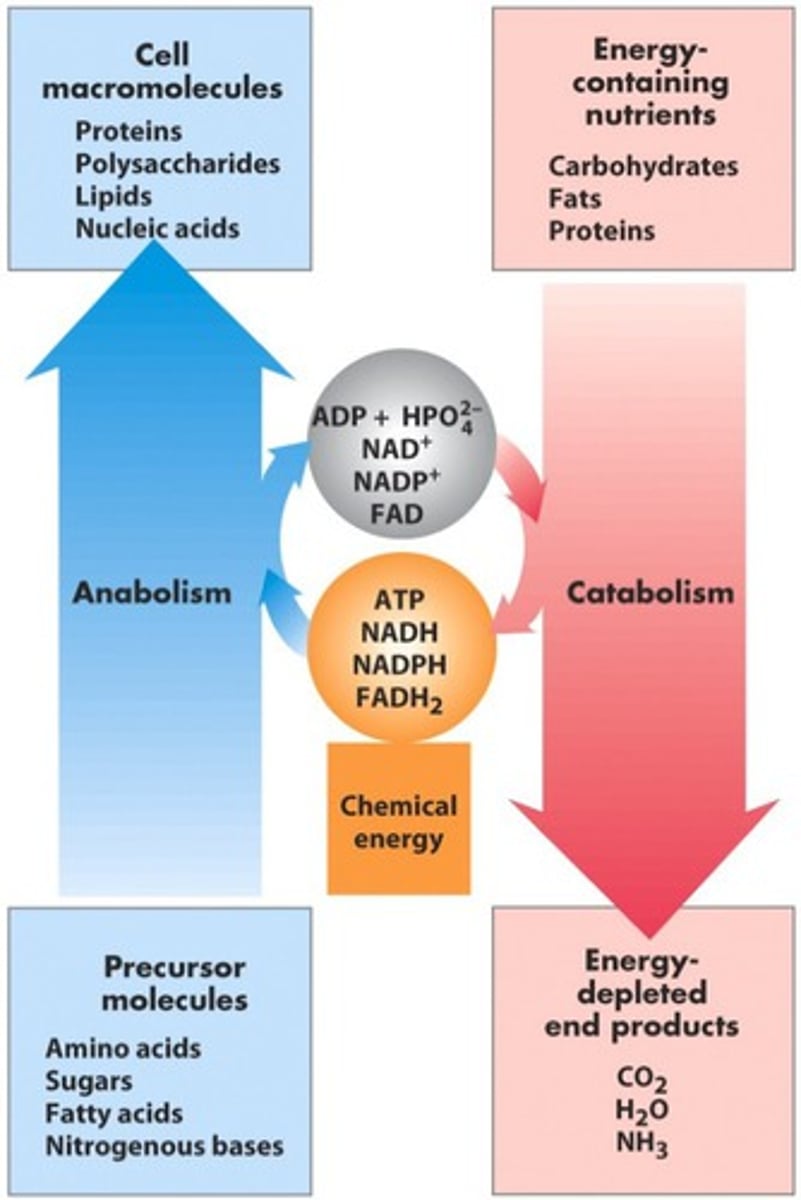
Define anabolic and catabolic
Anabolic: builds complexity (ex: dehydration synthesis)
Catabolic: reduces complexity (ex: hydrolysis)
Define exergonic and endergonic
Exergonic: releases energy (ex: hydrolysis)
Endergonic: requires energy (takes in energy) (ex: dehydration synthesis)
What atoms make up carbohydrates?
carbon, hydrogen, oxygen (1:2:1 ratio)
What do carbohydrates do?
they store short term energy and structure

What are the 3 "main" monosaccharides
glucose, fructose, galactose
How are disaccharides formed?
When two monosaccharides are linked together through dehydration synthesis
What are the three "main" disaccharides? What monomers make them up?
Glucose + Glucose = Maltose
Glucose + Fructose = Sucrose
Glucose + Galactose = Lactose
What are polysaccharides?
massive polymers of sugars
What are the 2 main functions of glucose polymers
energy storage and structural support
What are the polysaccharides used for storage in plants and animals?
plants: starch
animals: glycogen
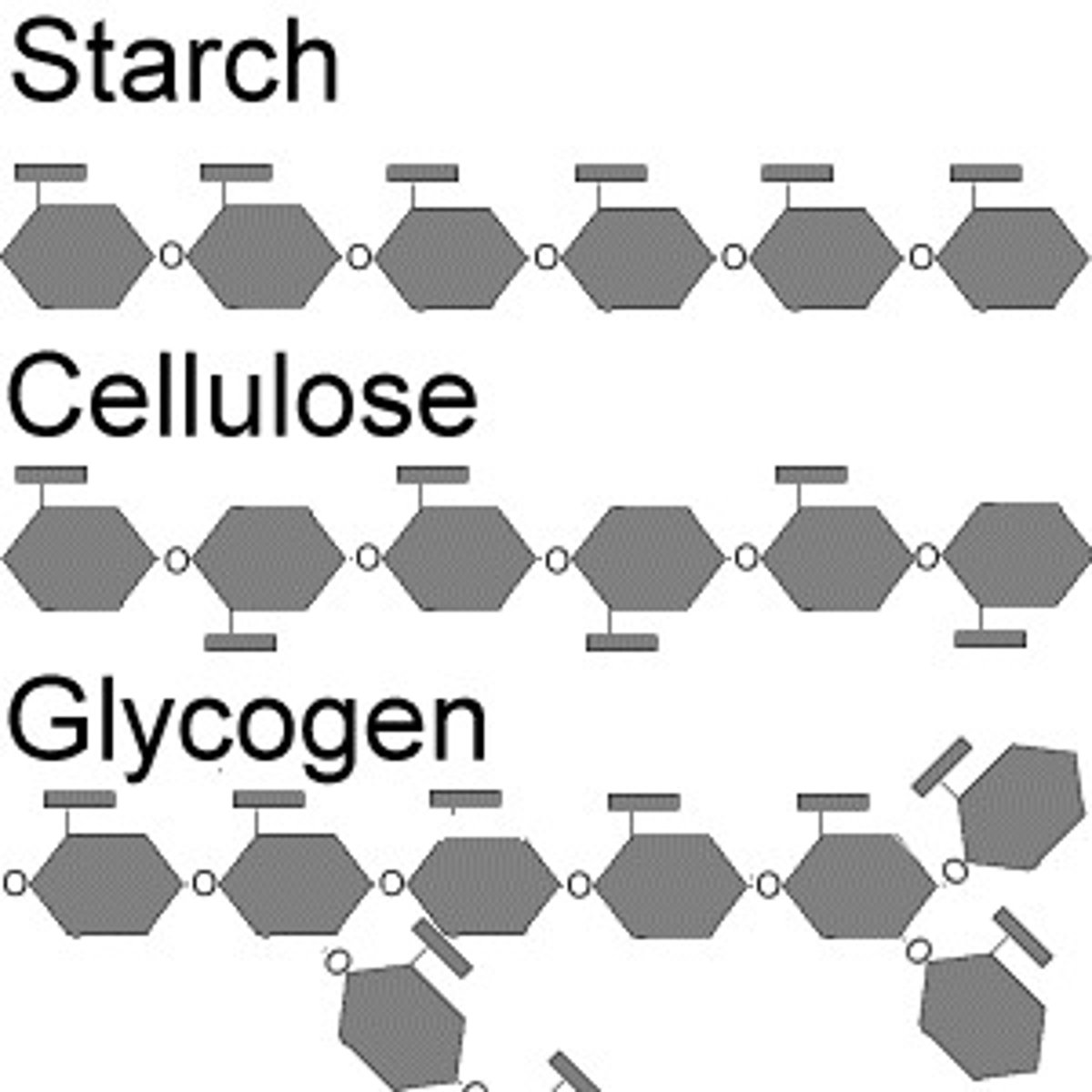
What is the major component of plant-like cell walls?
cellulose
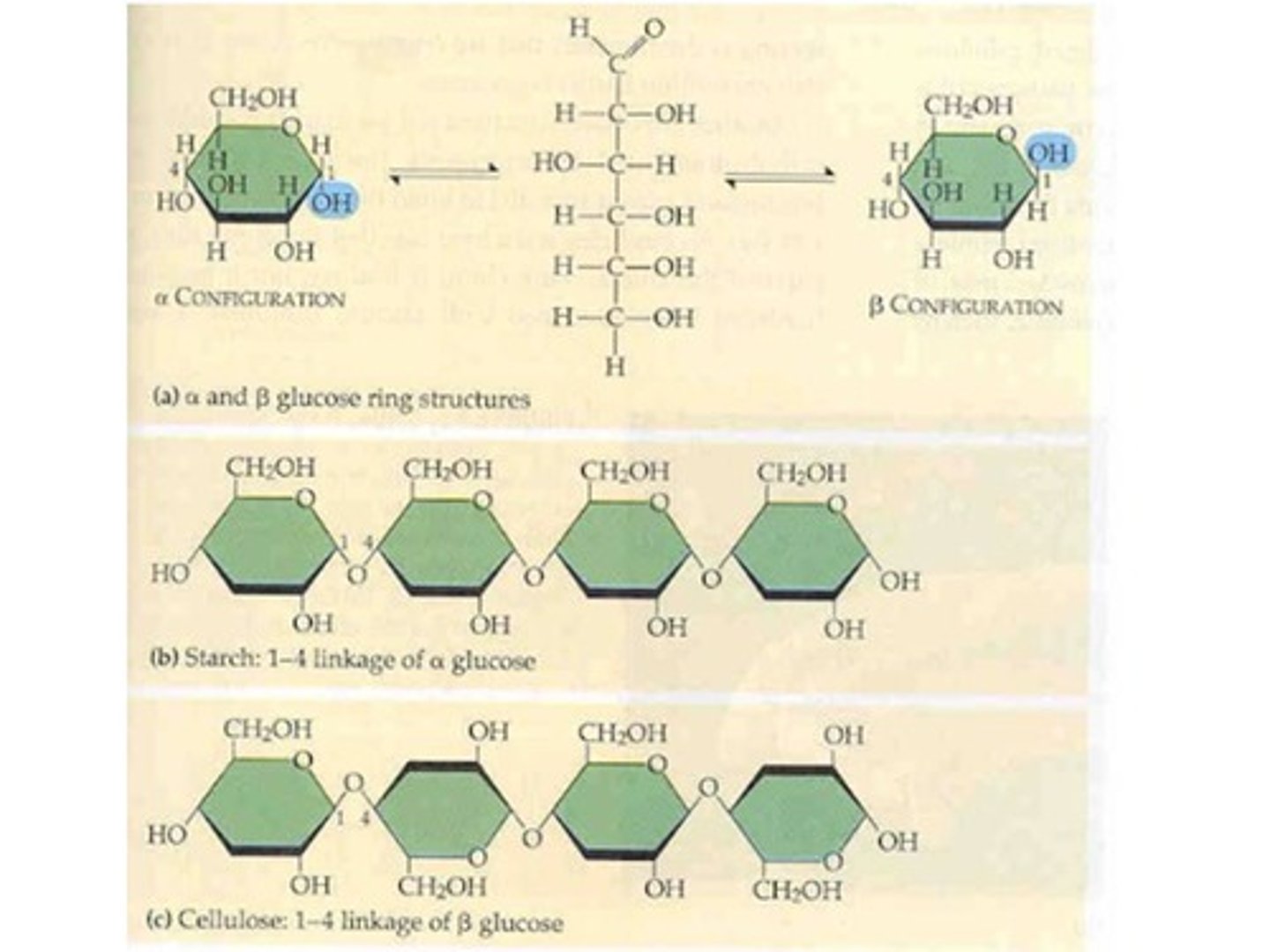
How do the structures of starch and cellulose differ?
Starch has alpha linkages between it's glucoses
Cellulose has beta linkages between it's glucoses
What are lipids?
fats, oils, and waxes

What atoms make up lipids?
carbon, hydrogen, oxygen (very little O compared to C)
What do lipids do?
they are used for long term energy storage and insulation
T or F: lipids are built of polymers
false: they're made of monomers
What are the three major groups of lipids?
triglycerides, phospholipids, steroids
What makes up a triglyceride?
1 glycerol and 3 fatty acids
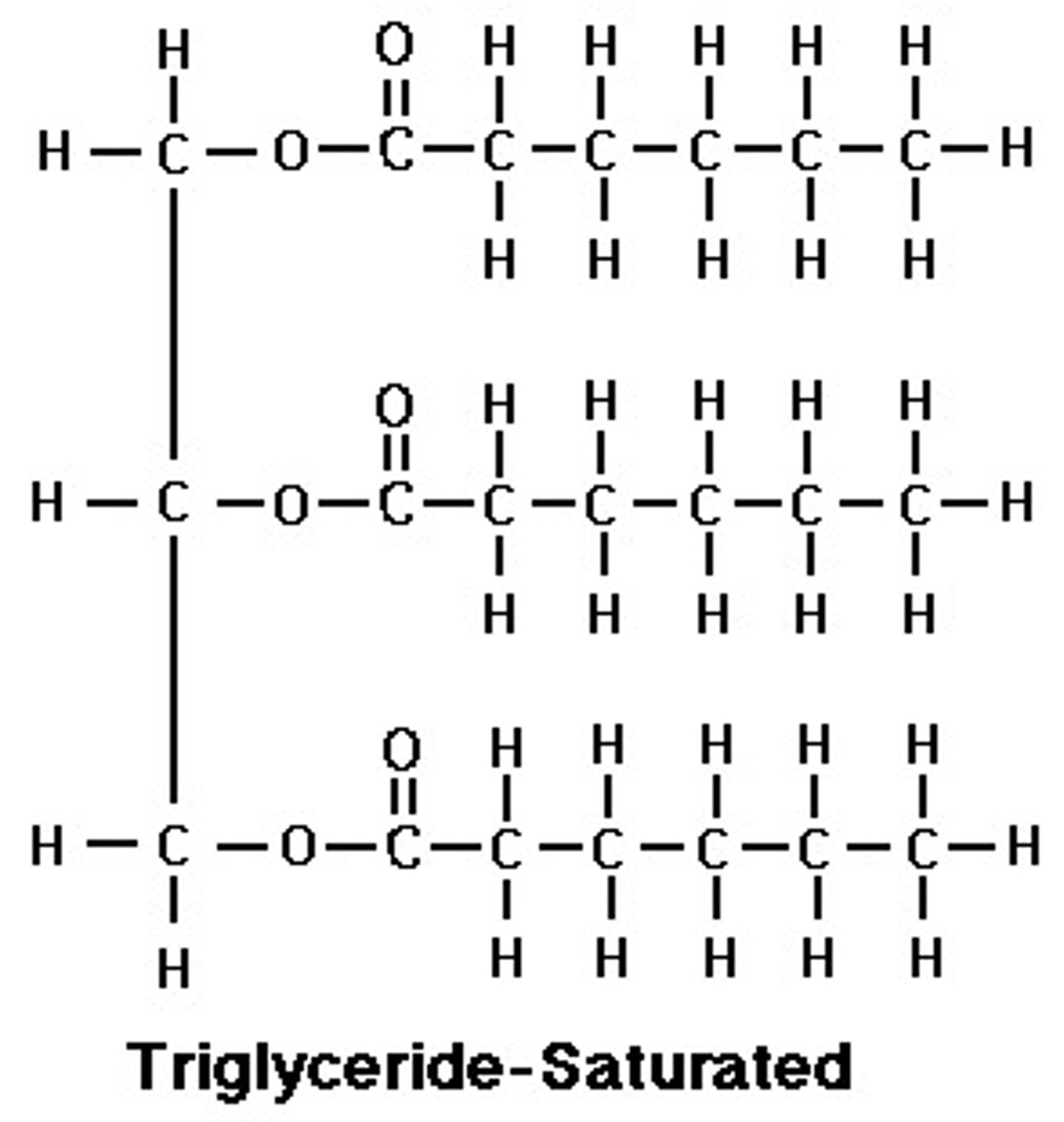
How do unsaturated and saturated fats differ?
Saturated fats have no double bonds, they are unhealthier
Unsaturated fats have at least one double bond, and are healthier; the double bond causes a bend in the structure
What makes a phospholipid?
1 glycerol, 2 fatty acids and 1 phosphate
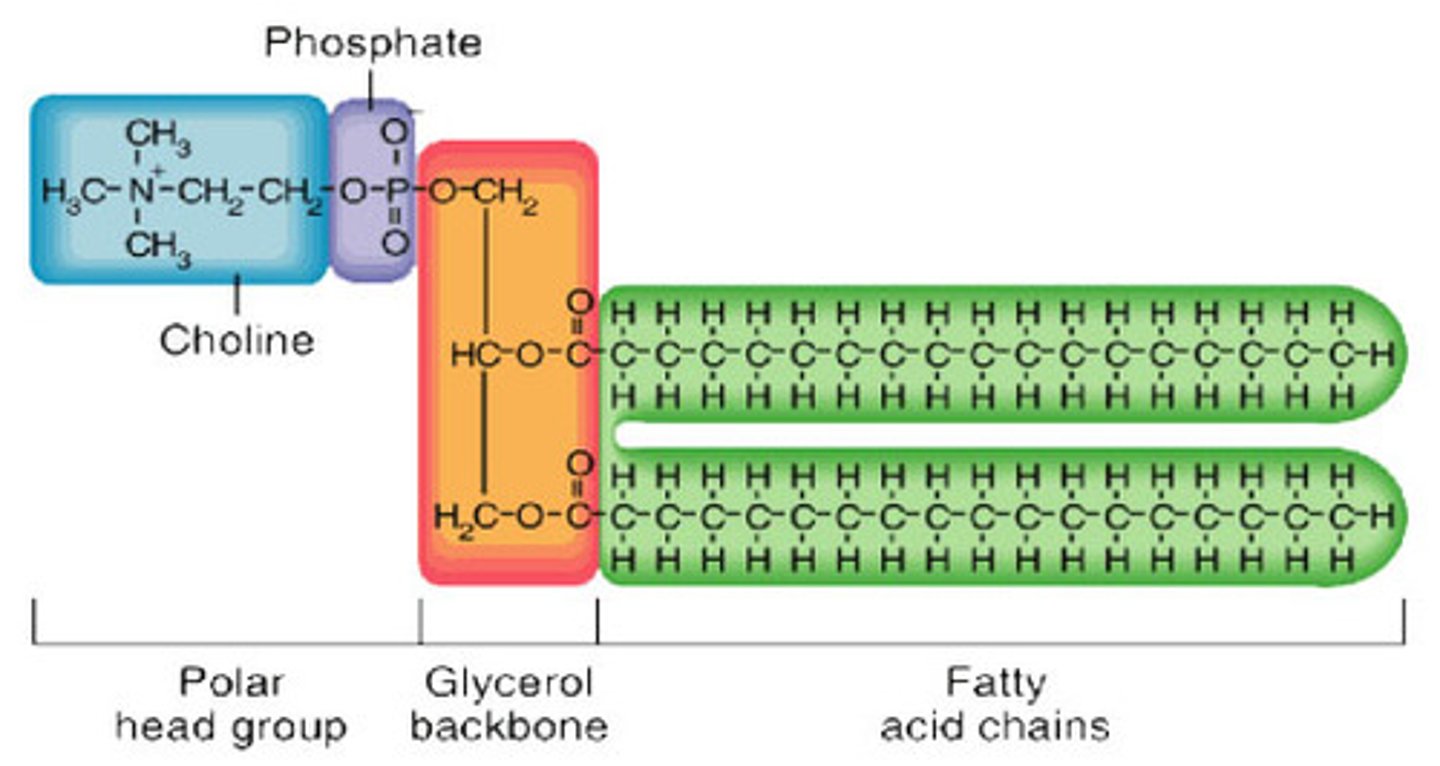
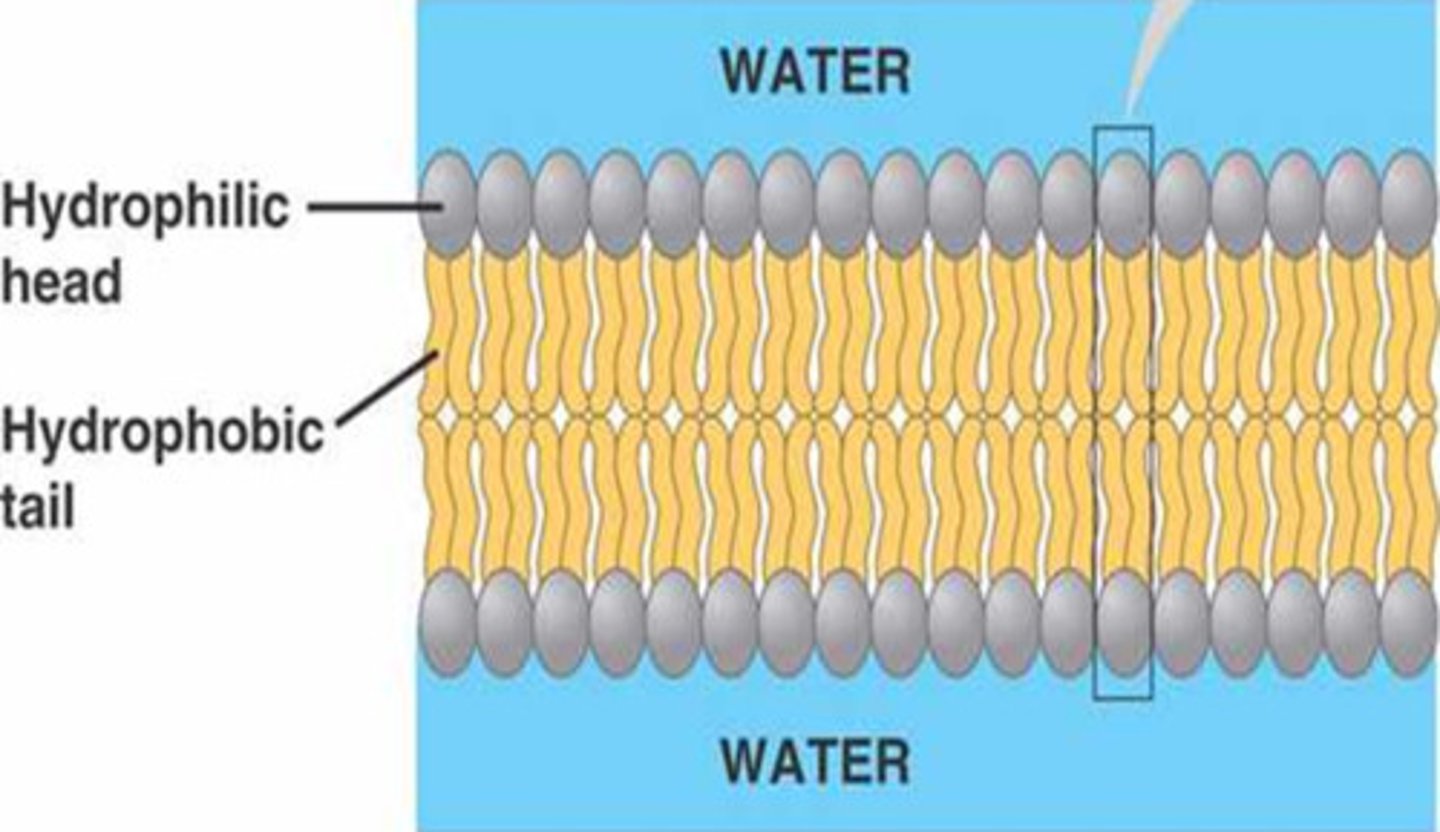
What are the properties of a phospholipid?
The molecule has a polar region (the head which inhabits the phosphate group), and a non polar region (the fatty acid legs). When combined with other phospholipids, they form a bilayer (hydrophobic tails away from water; hydrophilic heads near water) that is a major component of cell membranes
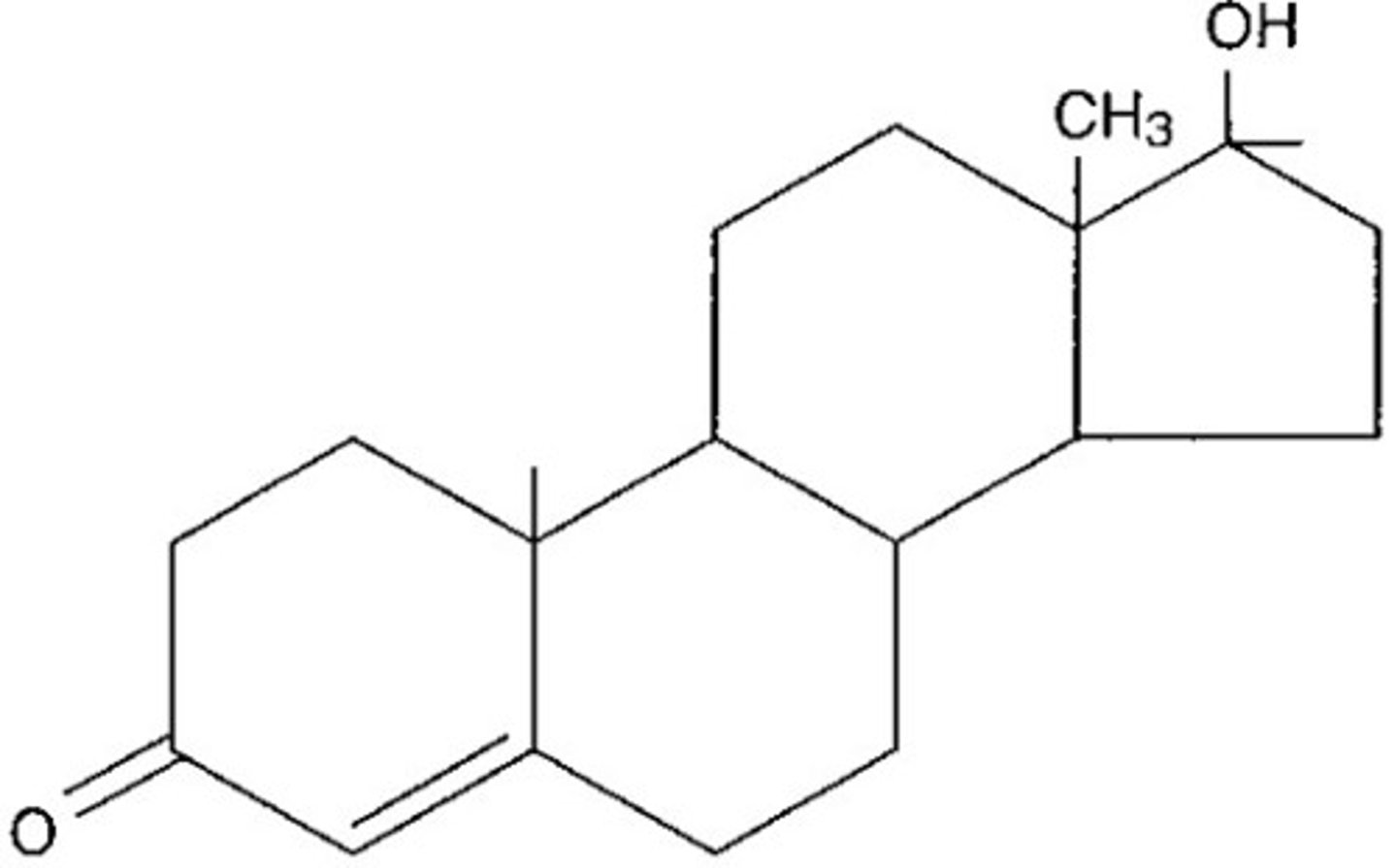
What are steroids?
lipids characterized by a carbon skeleton consisting of four FUSED rings. The presence of different functional groups leads to different functions. Ex: Cholesterol, testosterone, estrogen
What atoms make up proteins?
carbon, hydrogen, oxygen, nitrogen and a little sulfur
What are proteins made of?
polymers made by amino acids joined by peptide bonds (a bond formed by dehydration synthesis)
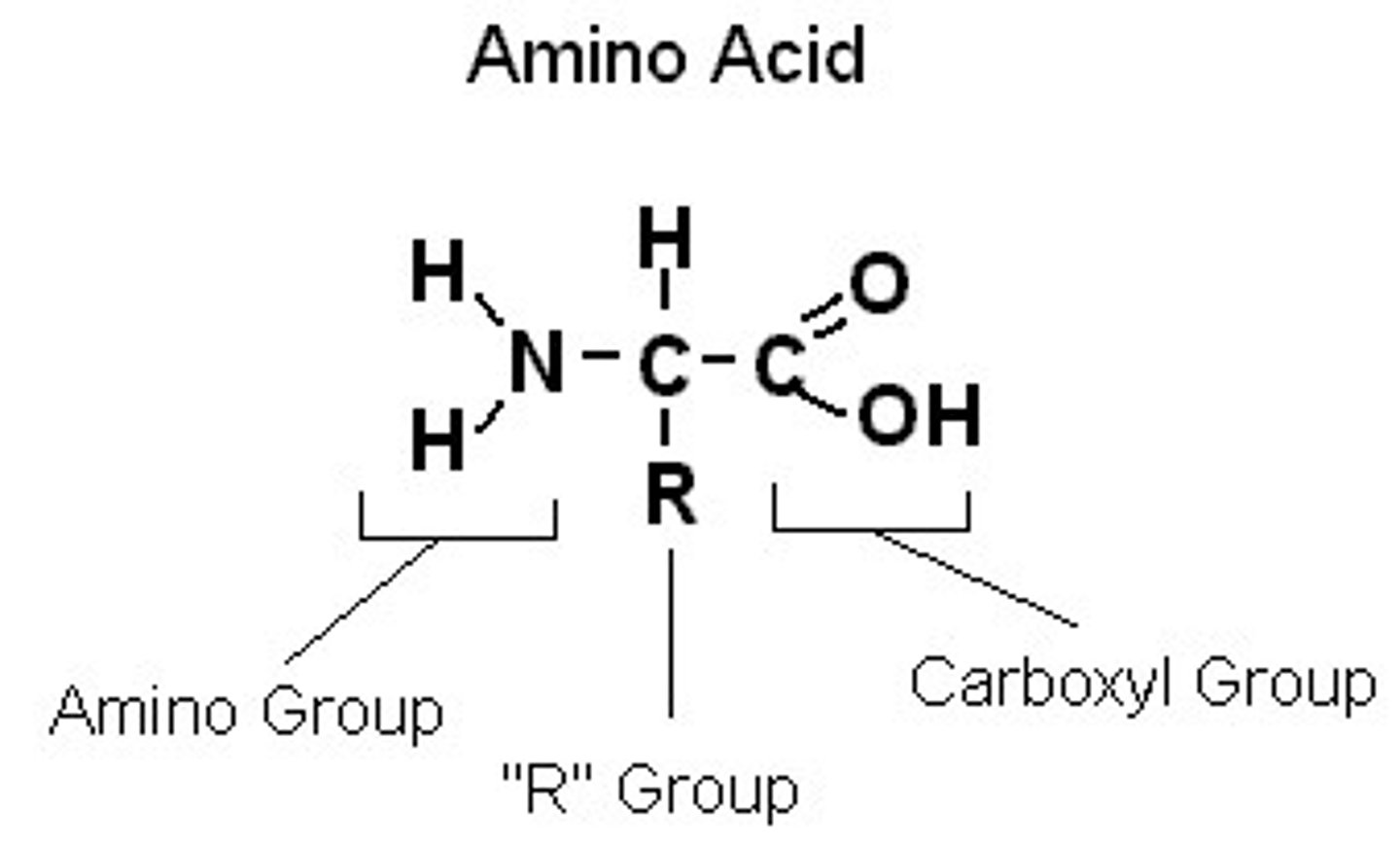
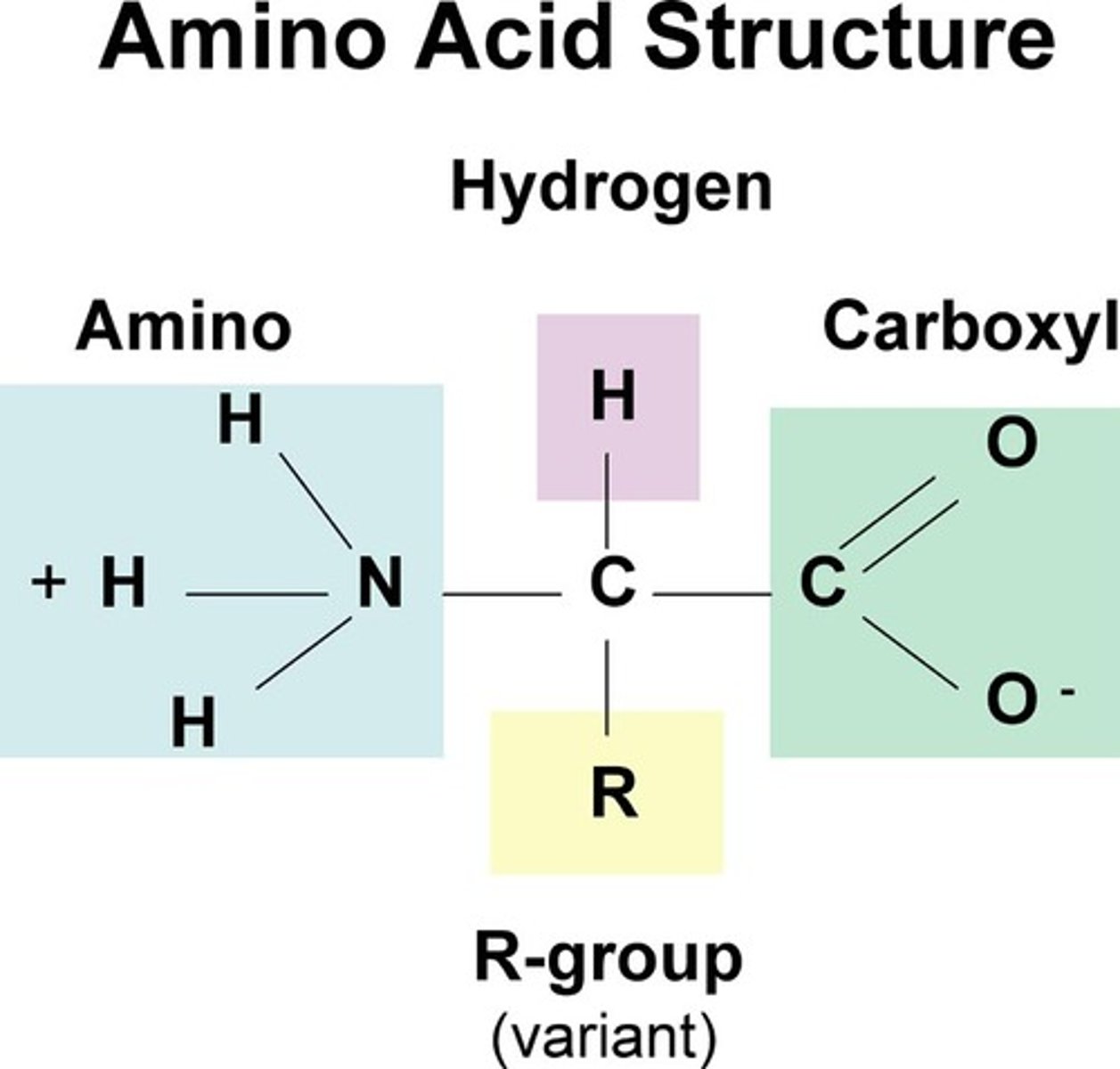
what 2 functional groups are found in amino acids? What are they bonded to?
carboxyl group and amino group. They bond to a central "alpha" carbon
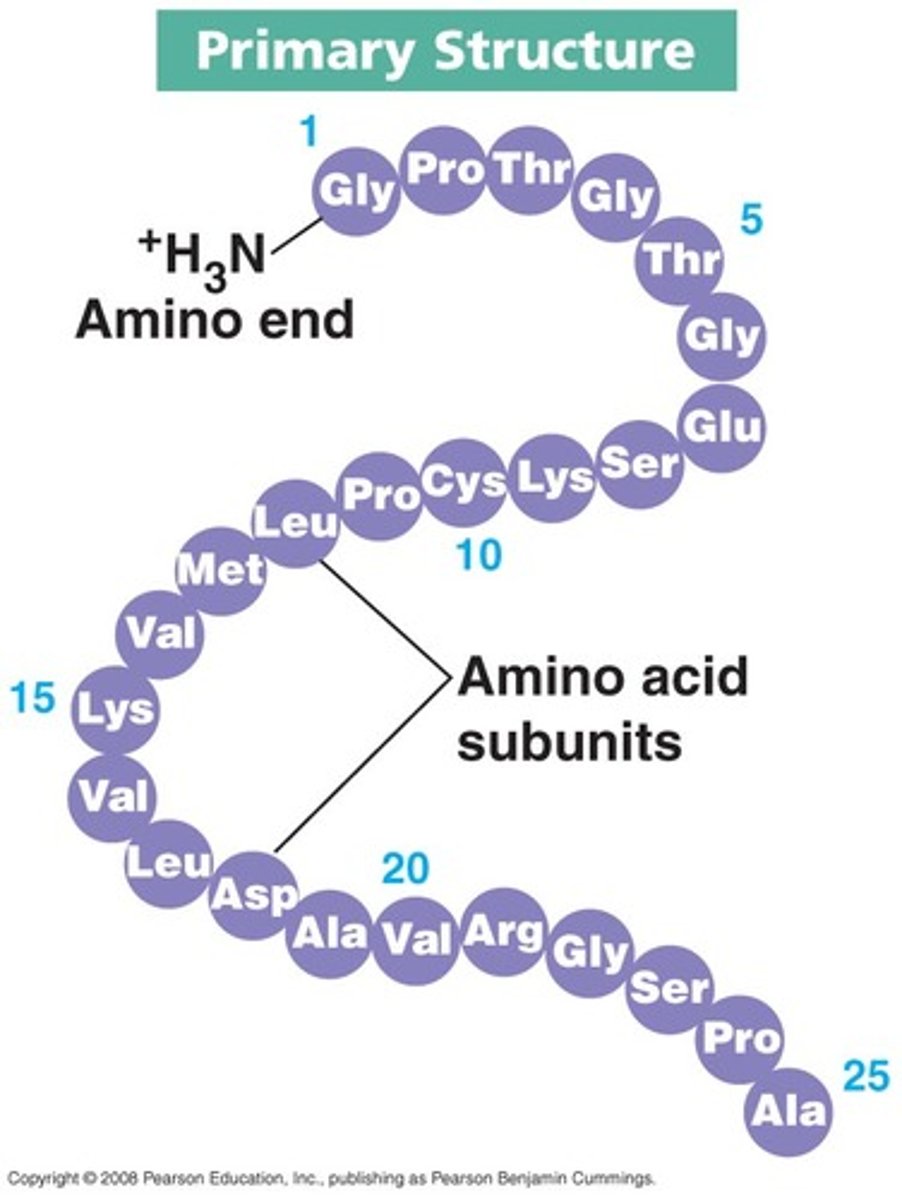
Describe the primary structure of a protein
sequence of amino acids in a polypeptide chain
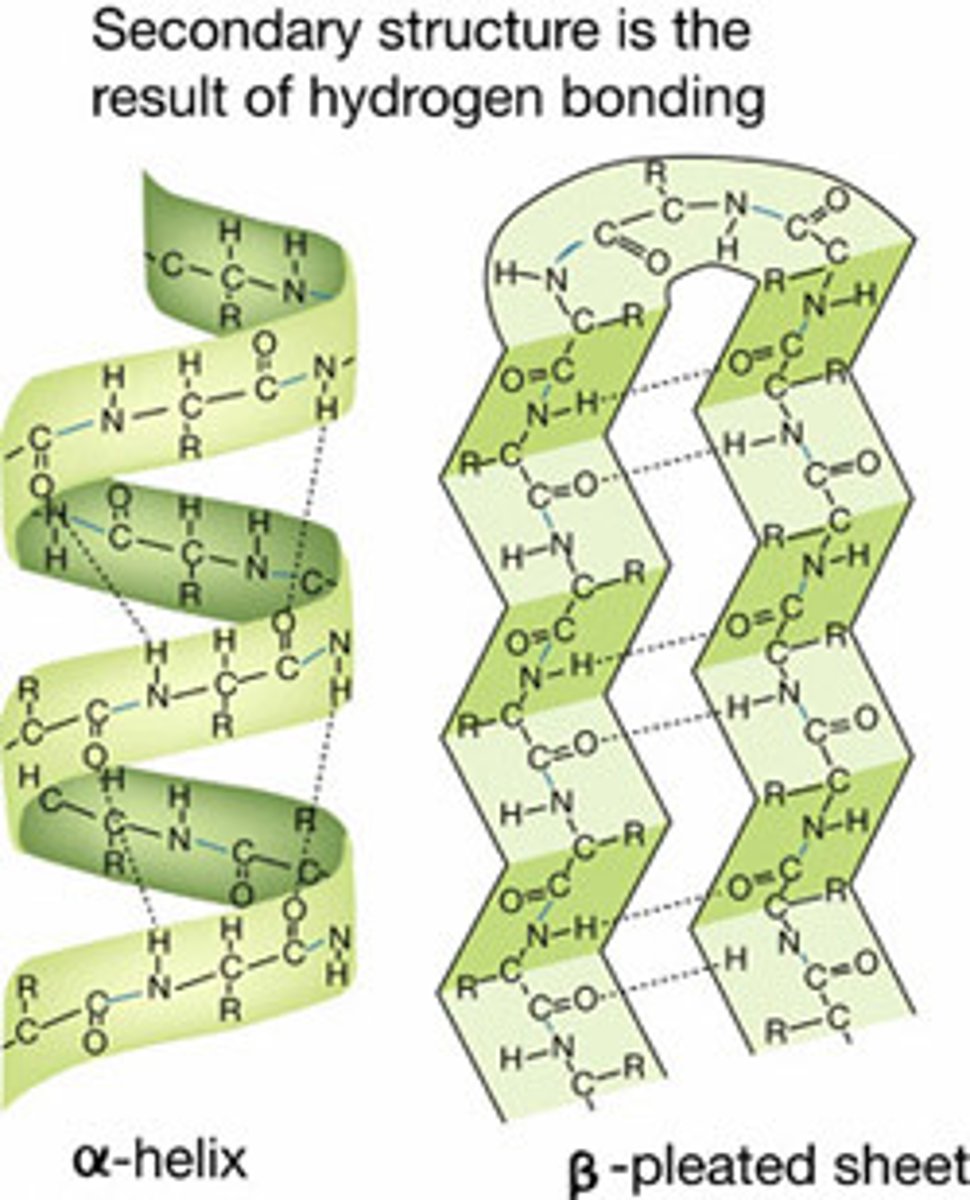
Describe the secondary structure of a protein
3D structures formed by hydrogen bonding between atoms in the carbon nitrogen backbone. (no R groups are involved). Either an "Alpha Helix" or "Beta Pleated Sheet" can be formed
Describe the tertiary structure of a protein
The specific 3D shape of a particular polypeptide chain ("conformation"). It's determined by the various interactions between the R-Groups
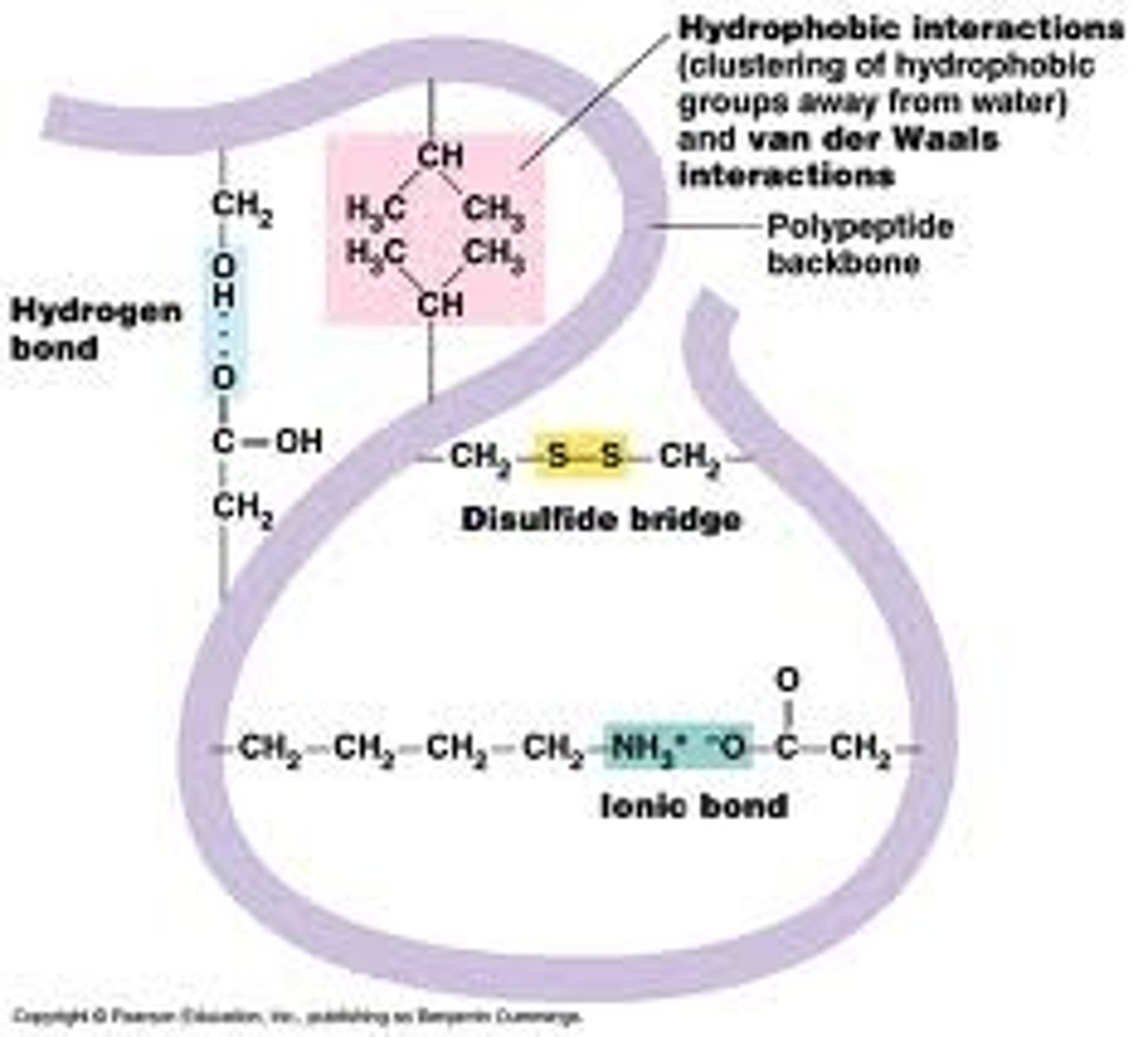
What kind of interactions occur to determine the tertiary structure?
Hydrogen bonds, hydrophobic interactions/van Der Waals interactions, disulfide bridge, ionic bonds
Describe the quaternary structure of a protein
Two or more polypeptide chains, each with its own tertiary structure, combine to form a functional protein
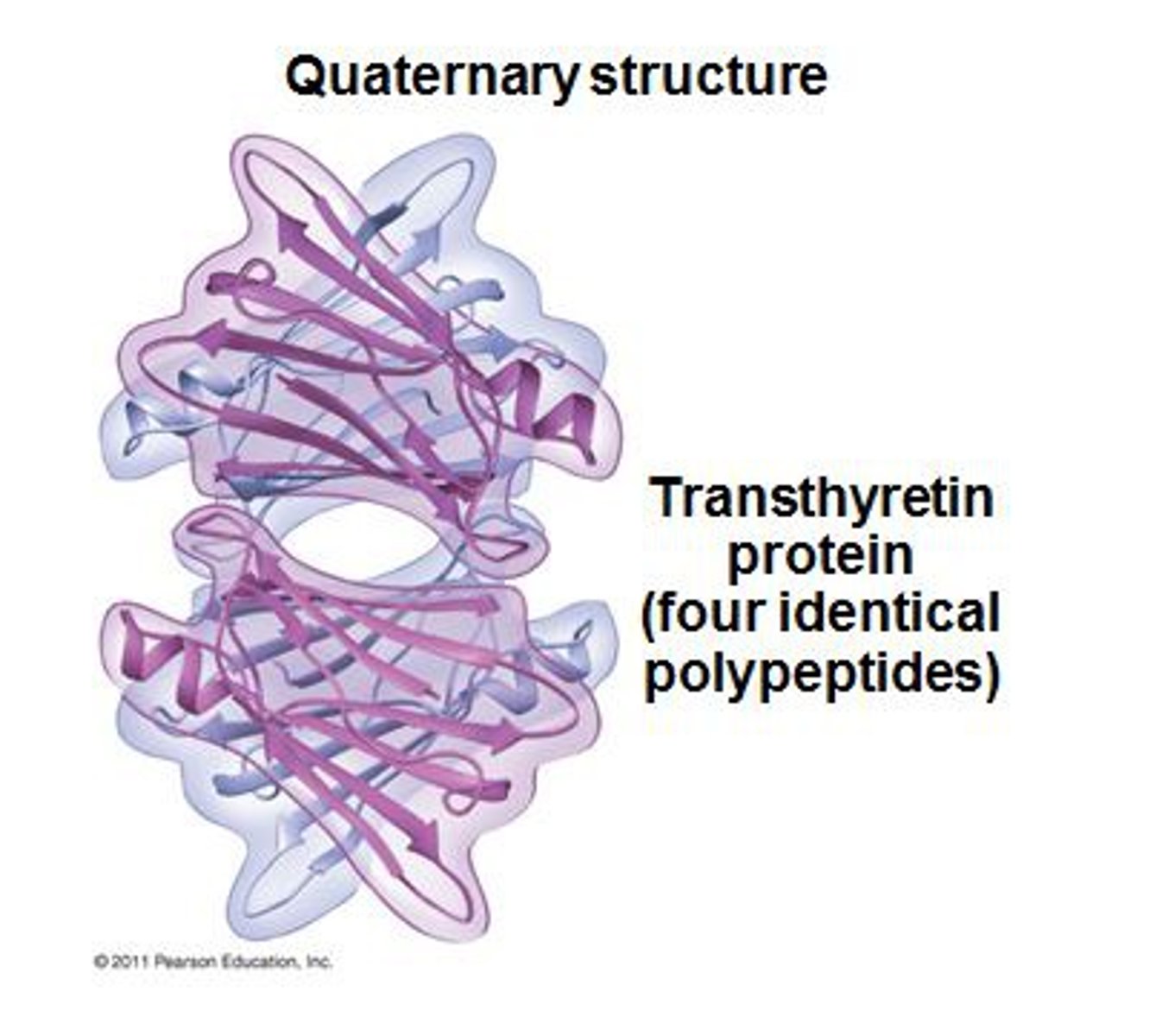
What do proteins do?
they're responsible for all life activities of the cell (Ex: enzymes and transport proteins)
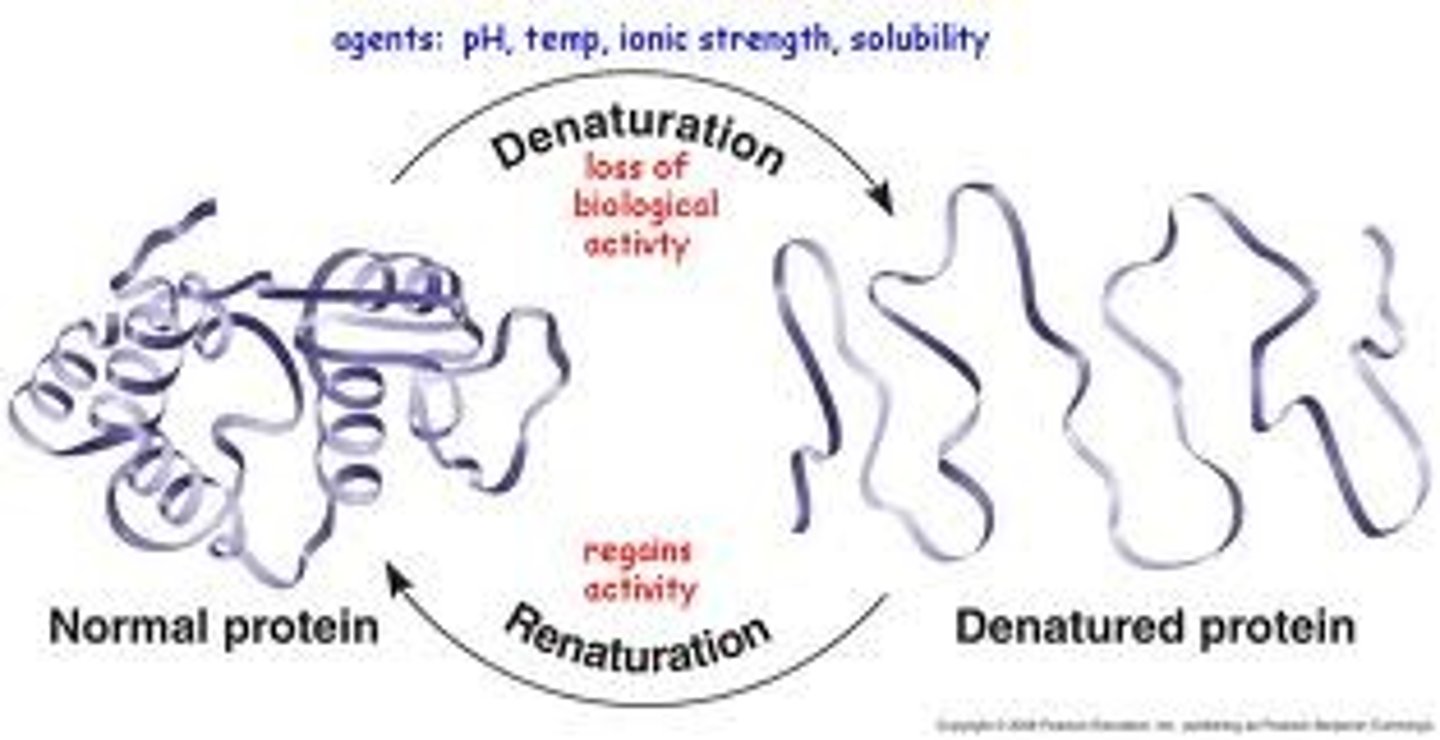
Define denaturation
a change in the structure of a protein. Denatured proteins do not work well (if it all)
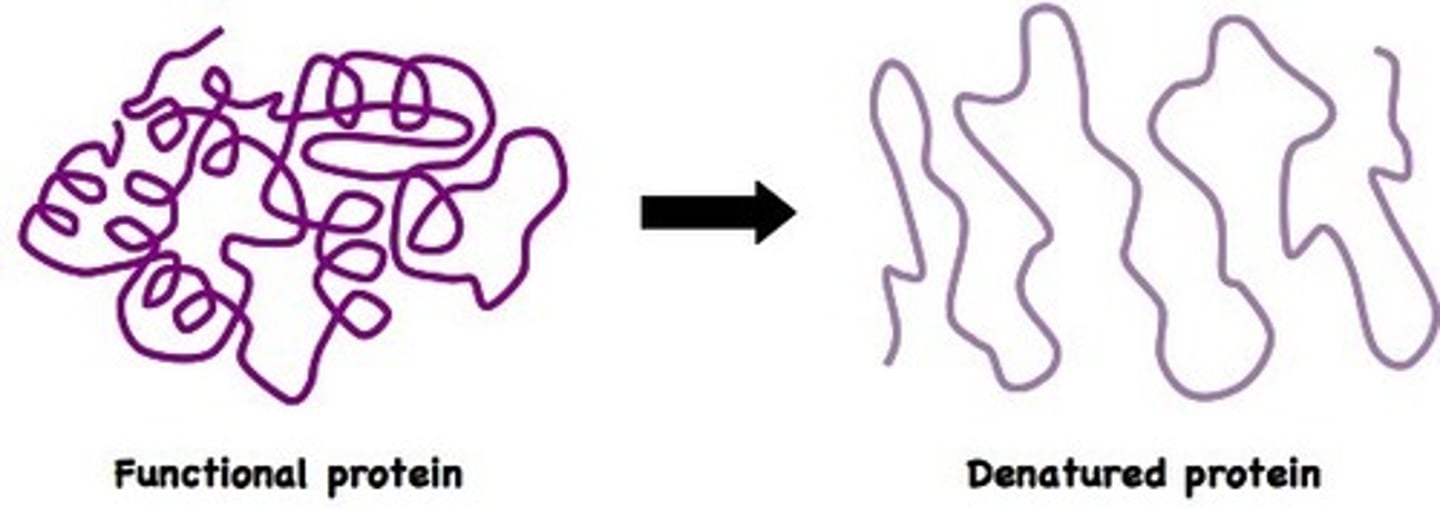
what causes a protein to denature?
change in temp, pH, salinity (amount of salt), polarity of solvent.
What atoms make up Nucleic Acids?
carbon, hydrogen, oxygen, nitrogen, phosphorus (no sulfur!)
What do nucleic acids do?
store and transmit genetic information
What are the two kinds of nucleic acids?
DNA and RNA
What are nucleotides?
monomers that make up nucleic acids
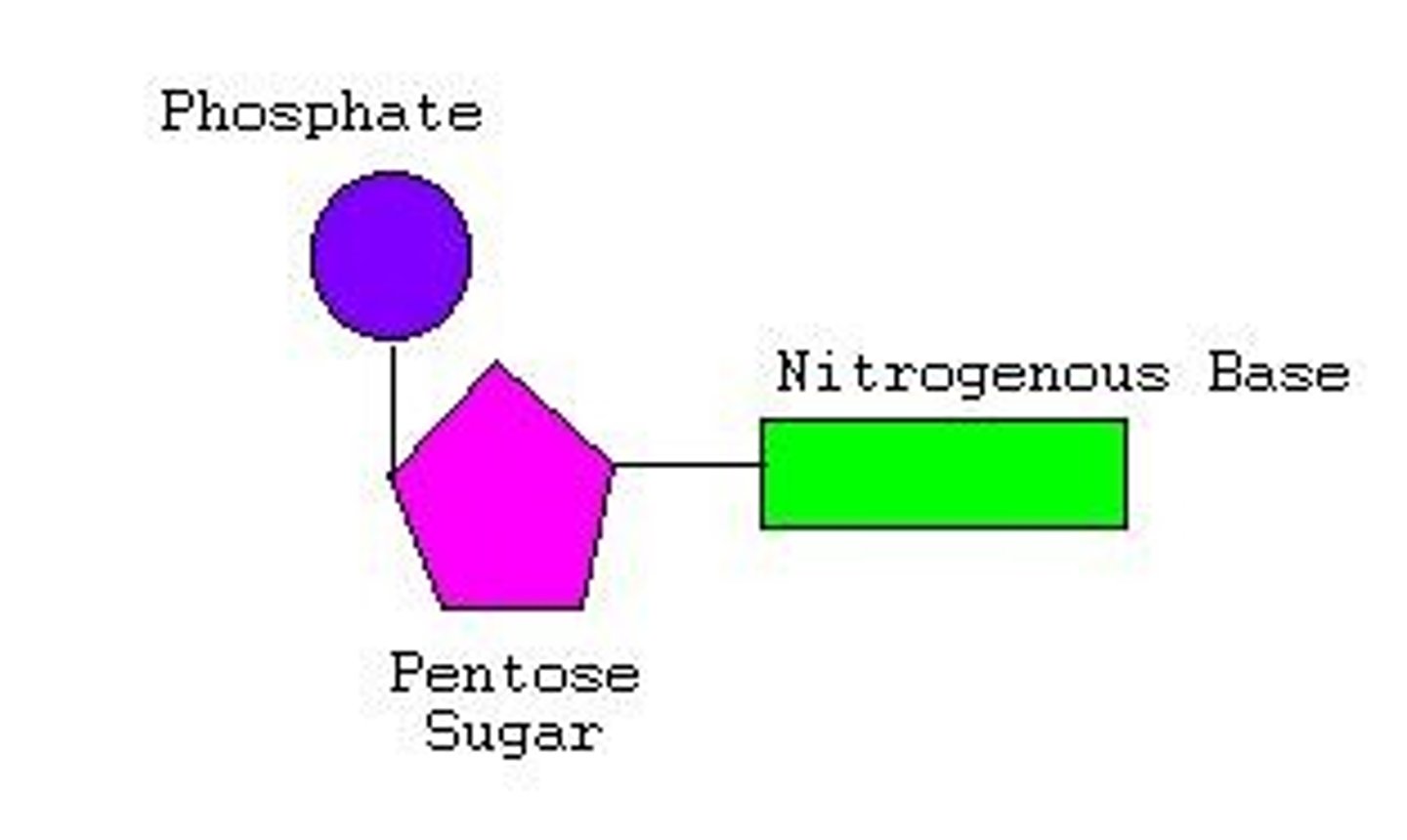
What do nucleotides consist of?
nitrogenous base ("ring" shape(s)), pentose sugar, phosphate group
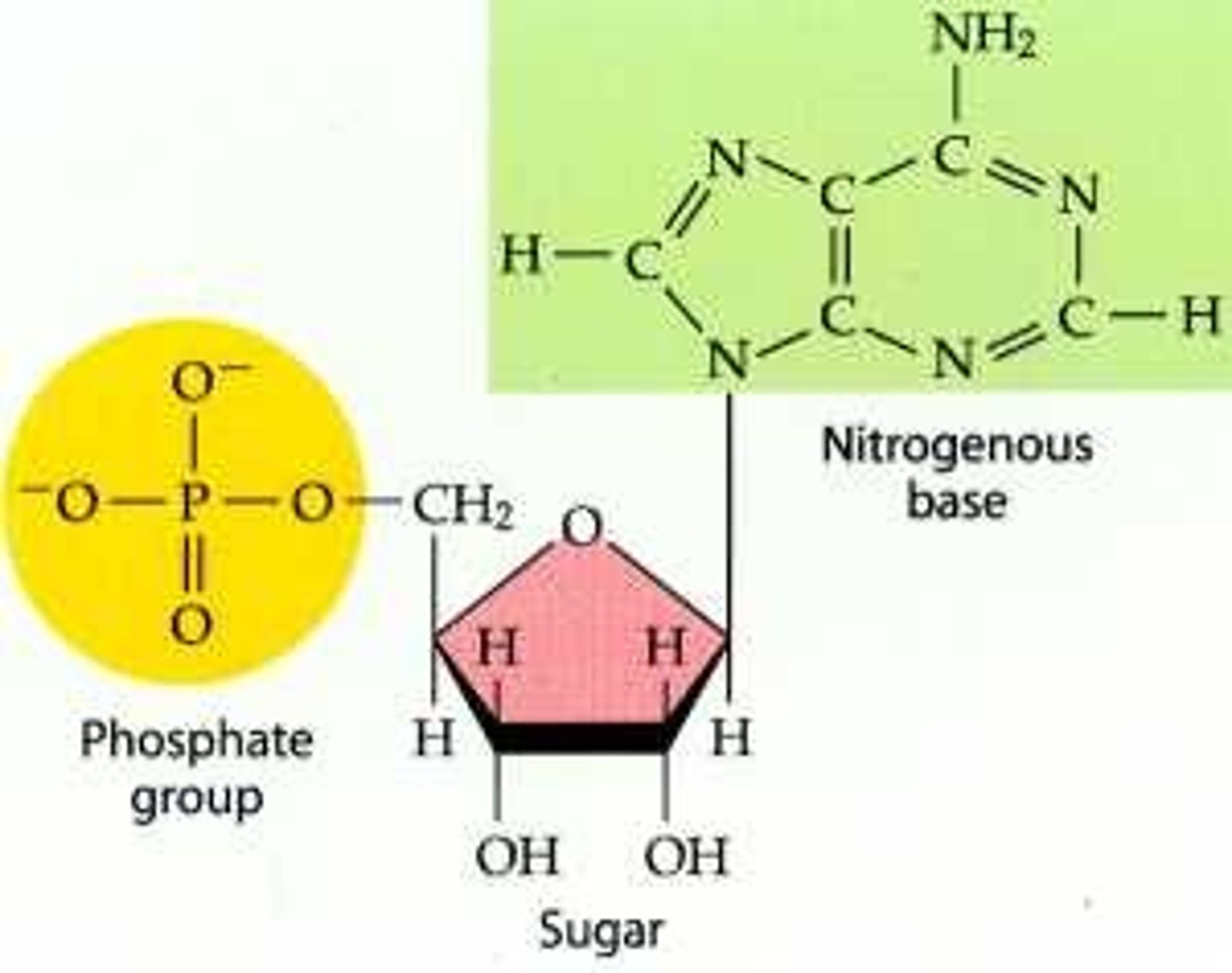
Difference between DNA and RNA Pentose
DNA: deoxyribose
RNA: ribose
Difference between DNA and RNA Bases
DNA: Adenine, Thymine, Guanine, Cytosine
RNA: Adenine, Uracil, Guanine, Cytosine
Difference between DNA and RNA Strands
DNA: 2 strands
RNA: 1 strand
What functions do DNA serve?
it stores information about the primary structure of proteins (It tells the cell what order to put amino acids in to build each protein), and the sequence of RNA molecules
It's heritable (passed down through genes)
Describe the structure of DNA
There are 2 chains of covalently bonded nucleotides from sugar to phosphate ("phosphodiester bonds"). The chains are bonded to each other by weak hydrogen bonds between Nitrogenous bases.
- A bonds to T, and G bonds to C
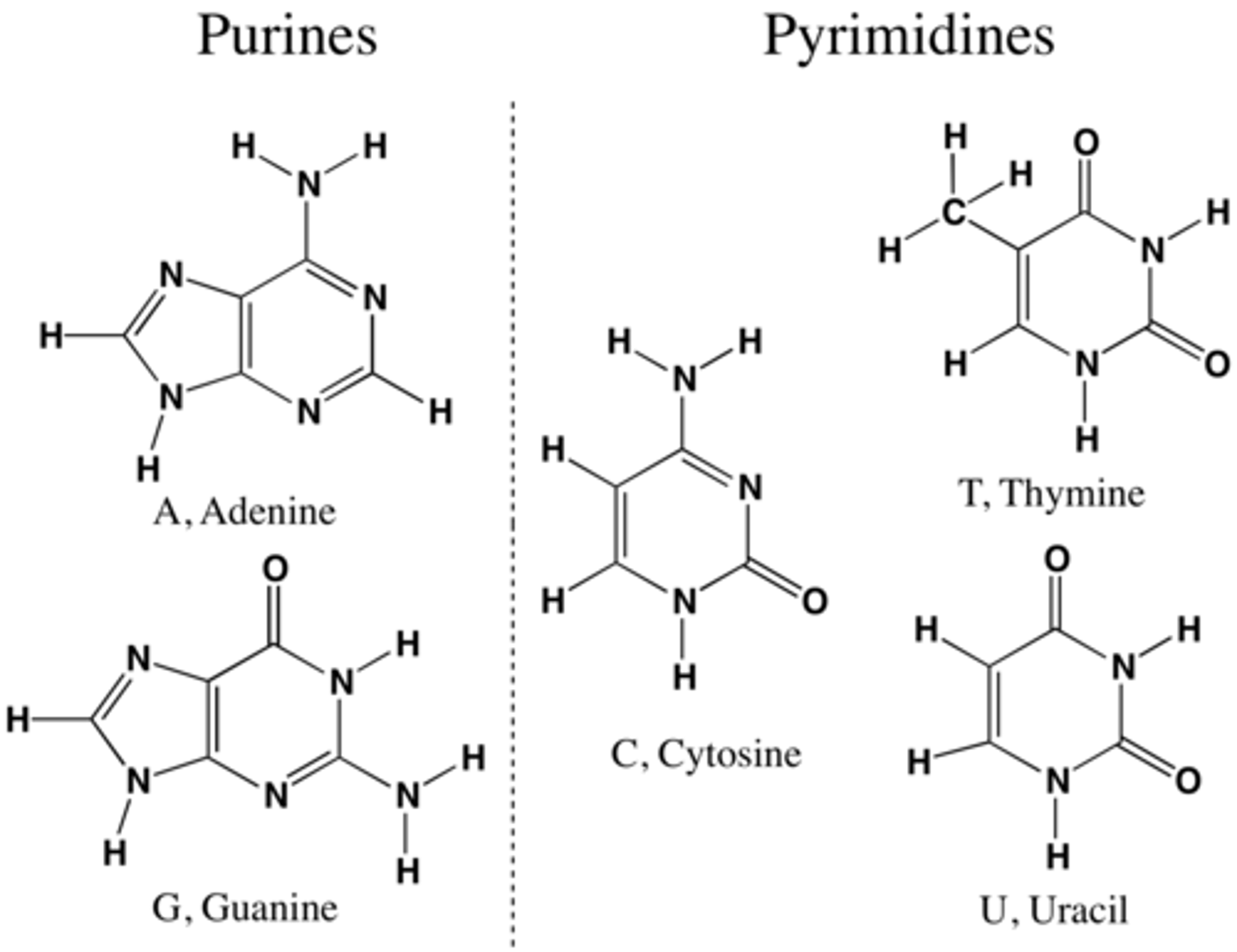
what are purine and pyrimidine bases?
nitrogen bases that hold DNA strands together through hydrogen bonds.
Purines are Adenine and Guanine (A, G)(double rings)
Pyrimidines are Cytosine, Thymine, and Uracil (C, T, U) (single rings)
What functions do RNA serve?
they transmit and translate DNA information into proteins primary structures. They also have many enzymatic and regulatory functions
Describe the structure of RNA
less stable than DNA. 1 strand, but base pairing can still occur (Adenine bonds to Uracil)
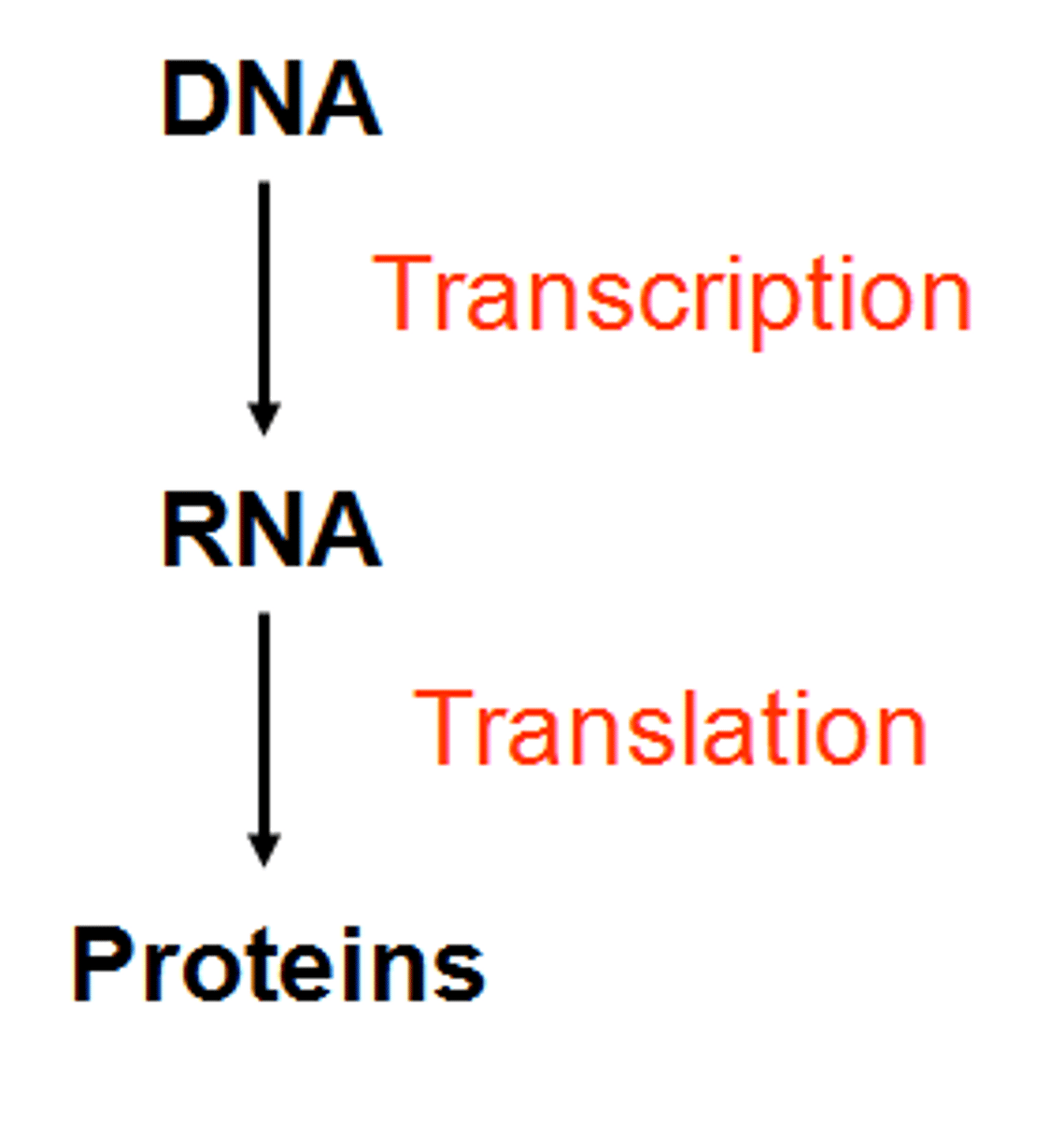
What is the central dogma of molecular biology?
information stored in DNA moves to RNA before some of that information finally directs the construction of proteins:
DNA--> RNA --> Proteins