15. Food standards
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
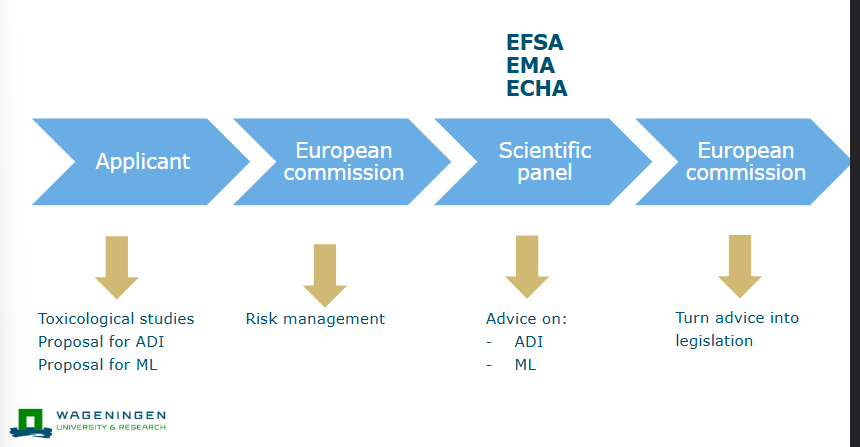
Common market authorisation procedure of chemicals
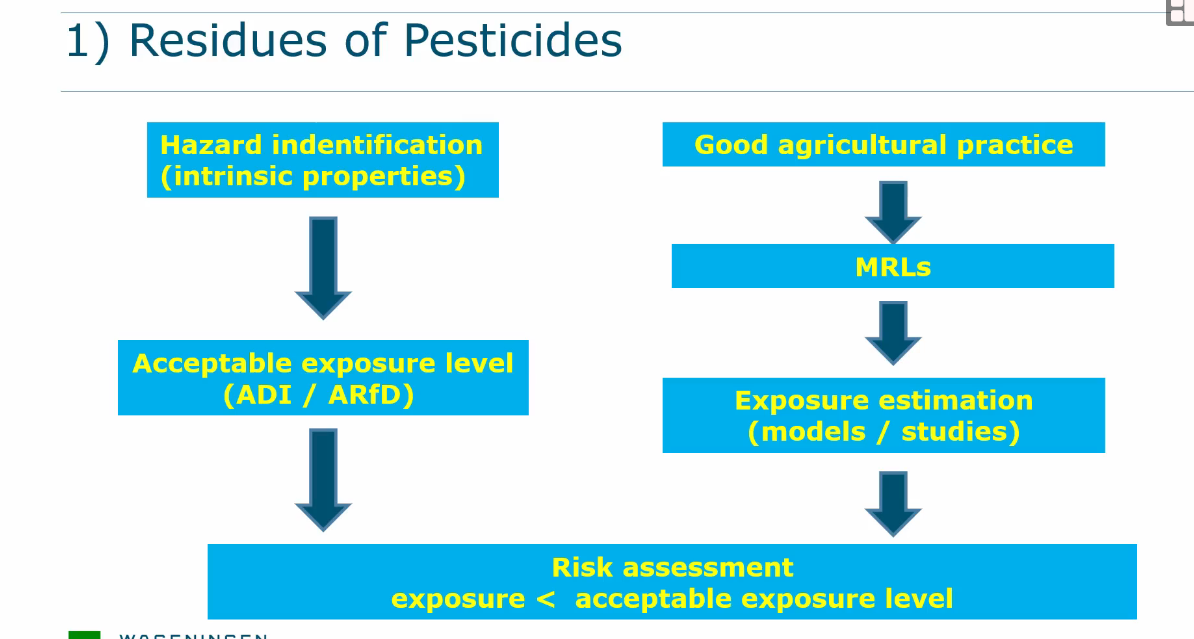
Residues of pesticides
Very tightly regulated
Pesticide products contain at least one active substance to kill insects/weeds
No active chemical = it is not pesticide
Active substance must be approved
ADI must be set
Residues
Traces of active ingredients in treated products
Maximum residue levels: highest level of a residue that is legally tolerated in or on food
Good agricultural practices
Exposed populations to residues of pesticides
Operators: performs activities relation to the applicaiton:
At factory
Farmers
Amateur users
Bystanders:
Presence is quite incidental and unrelated to work
Exposed during a short period of time (Acute exposure)
Take no action to avoid or control exposure
Residents:
Lives, works or attends school clsoe to an area
Presence is unrelated to work involving PPPs
Take no action to avoid or control exposure
Might be in the location for up to 24 hours per day
Workers:
as part of employment entering an area
Handling a crop that has been treated
Essential information to known regarding residues of pesticides?
Toxicological profile of the active substance
ADI or BMDL must be determined
Toxicological information of the plant protection product
Population(s) exposed
Different based on ages?
Exposure scenario
outdoor/indoor?
dose level?
route of exposure?
etc
What are the two streams of assessment in pesticide regulation?
Stream one:
Looks at substance or product that substance is in and toxicological profile
Stream two:
A certain concentration is necessary to be effective - good agricultural practice
Maximum residue level (MRL) - is determined to see what the maximum amount is that is necessary for the pesticide to be effective.
This has NOTHING to do with safety.
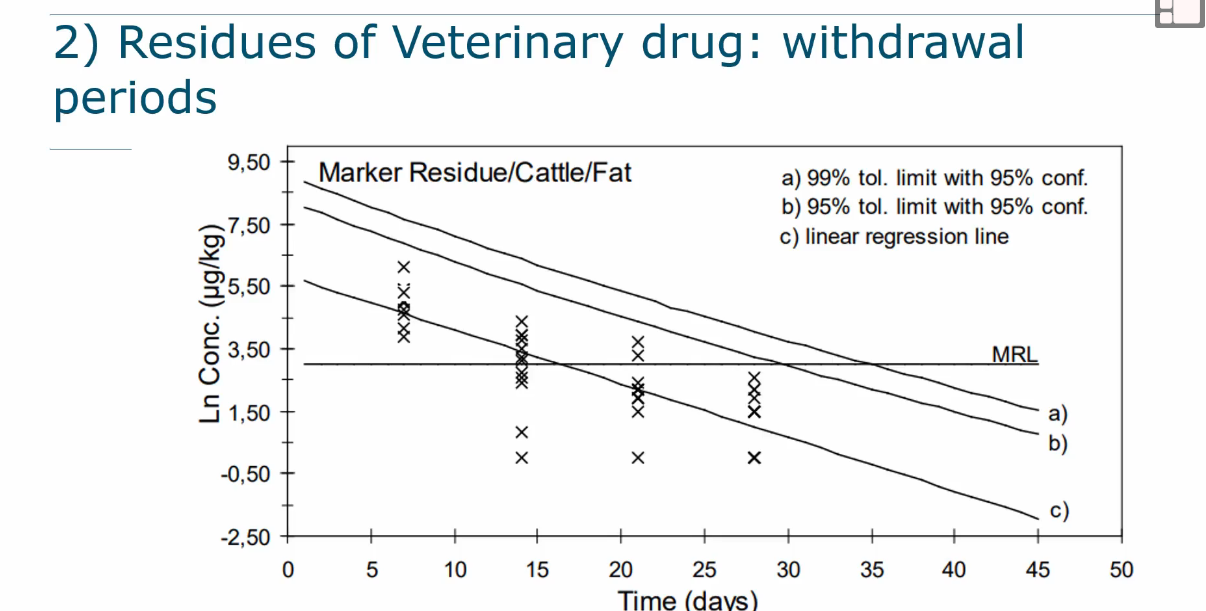
Residues of veterinary medicines
Scientific advice European medicines Agency
Set ADI based on toxicological profile of active ingredient
Study efficacy and toxicity of product with active ingredient
Set MRL in tissues
Medicine are also applied in different ways (injection, pill, etc) these all affect how medicine affects body
Residues of veterinary drugs “Food Basket”
This food basket is an assumption of how much meat we eat
Based on that assumption they can determine how many veterinary drugs you consume and what level of veterinary drugs are safe
Which chemicals end up in the meat? (veterinary drugs)
During metabolism the drug is changed by the body (especially the liver)
Therefore the question is if the metabolite that is left is dangerous
Also concentrations change over time and after a certain amount of time the metabolite (marker residue) has decreased below the MRL and is safe (see picture)
Therefore an ADI is set for the active ingredient but an MRLs for the species and tissue.
Withdrawal period is sometimes set if needed to remove marker residue.
Maximum levels of contaminants in food
Maximum levels should be set at a strict level which is reasonably achievable by following good agricultural, fishery and mnaufacturing practices and taking into account the risk related to the consumption of the food.
To ensure an efficient protection of public health, products containing contaminants exceeding the maximum levels should not be placed on the market either as such, after mixture with other foodstruffs or used as an ingredient in other foods
How are maximum levels of contaminants in food determined for non-genotoxic contaminants?
Non-genotoxic contaminants
No producer (industry) involved
Data from scientic literature
Specific EU funded research projects
Set a TDI
MRL based on:
TDI or,
Sensitivity or, sensitivity of analytical methods
Contaminants in food for genotoxic contaminants
MRL is based on:
ALARA principle
Margin of exposure
Sensitivity of analytical methods
Industrial chemicals legislation
Europe: REACH Registration Evaluation and Authorization (and Restriction) of CHemicals
Producer has to provide toxicity data on their chemical in order to allow the chemical on the market
Many ‘existing’ chemicals need to be tested
Combination toxicology
The idea that we know for a lot chemicals how they are dangerous by themselves but we do not know their risks when combined
The three ways in which chemicals exist with each other:
Similar action
Dissimilar actions
Interactions
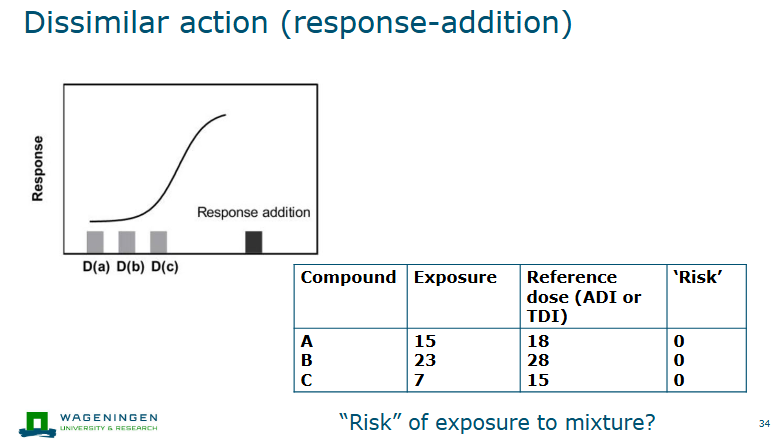
Dissimilar action (response-addition)
Separate mechanisms/modes of action
Chemicals do not influence each other’s action
Effects of combination: sum of the effects of the compounds (response addition)
If each chemical is present at doses below ADI/TDI, no adverse effects of mixture expected
Similar action (dose-addition)
Same mechanism/modes of action, possible differences in potencies
Effect of combination: effects of the sum of the compounds (dose addition)
Even if each chemical is present at doses below ADI/TDI, adverse effects may be induced
Dioxins/PCBs → activate aryl hydrocarbon receptor
Organophosphates → inhibit acetylcholinesterase (AChE)
Relative potency factor (RPF)
Potency of chemical congener as a relative potency compared to the index chemical
Index chemical
A potent congener of a chemical group for which adequate data are available for the risk assessment (hazard characterization)
Interactions
Interaction effects may be expected when a chemical affects:
Uptake of another
Biotransformation of another
Competition for enzymes
Enzyme induction
Excretion/clearance of another