FDA Regulation
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
How are dietary supplements regulated compared to drugs?
Regulated as food even though they can have therapeutic benefit, toxicity, or unwanted interactions with other medications (like drugs)
Describe the historical events and legislation leading to the development of the modern FDA
1906 Pure Food and Drug act - standards for purity and drug strength, The Jungle
1938 Food Drug and Cosmetic Act - elixir sulfanilamide tragedy » mandated safety testing
1962 FD&C Amendments - stronger safety and efficacy testing, thalidomide disaster (birth defects)
2007 FDA Amendments - COX2 inhibitors, greater FDA control
Describe phase I-IV clinical trials, including the approximate number of participants, whether the participants are healthy or have the condition the drug is intended to treat, and whether the phase is conducted pre or post approval
Phase 1 - healthy volunteers dosing, safety, and toxicity, pre approval
Phase 2 - small group of volunteers with disease, test efficacy of treatment, pre approval
Phase 3 - randomized control trial (new drug vs placebo or standard care), larger sample, people with disease, pre approval
Phase 4 - continued monitoring of safety and efficacy, after approval
Describe the off-label use of prescription drugs
Prescribing outside the scope of FDA-approved indication label
FDA permits healthcare providers to do this based on personal judgement, but not pharma companies
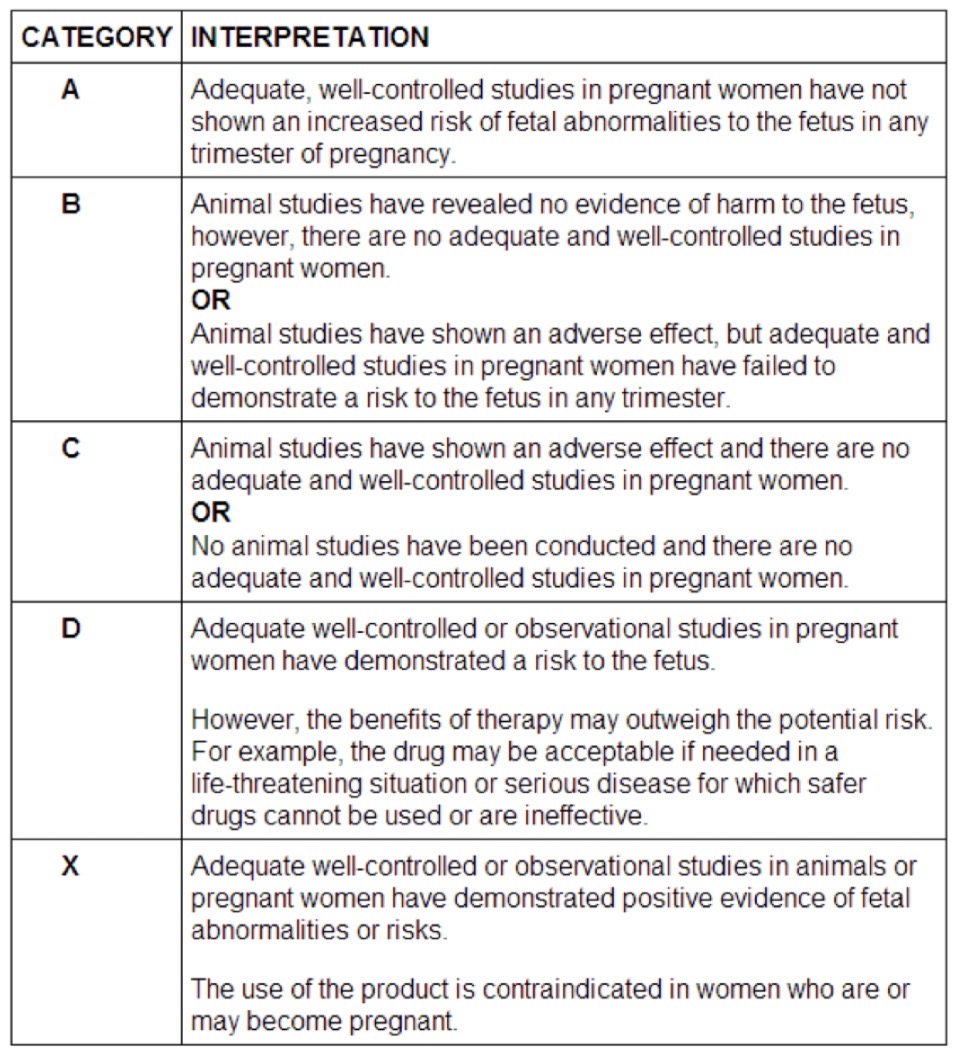
Define the term teratogen, and be familiar with the FDA categories for safety in pregnancy
teratogen - any substance that will harm a developing fetus or embryo
A = no risk, proven by studies in pregnant women
B = animal studies show risk but no controlled studies in pregnant women show risk OR animal studies do not show risk but no studies in pregnant women exist
C = animal studies show risk but no controlled studies in pregnant women OR no animal or pregnant women studies
D = studies in pregnant women show risk, but risk may outweigh benefit
X = guaranteed risk and contraindication
Describe the nomenclature of drugs in terms of their chemical, code, generic, official, and trade names
Chemical - formal drug name based on chemical structure
Code - short name provided by pharma company for use during drug evaluation
Generic - non-proprietary name for drug or class of drugs
Brand/trade - proprietary name provided by manufacturer
List how generic drugs can and cannot differ from original brand name drugs
CANNOT DIFFER - active ingredients/admin route/potency (pharmaceutically equivalent), delivered into blood at same time period with no more than 20% variation (bioequivalent), indications and safety profiles (therapeutically equivalent), FDA standards
CAN DIFFER - flavors
MUST DIFFER - appearance (size, shape, color)
Understand why some drugs require prescriptions
deemed safe for unsupervised use because they meet one of the following criteria:
habit forming
toxic
are for medical conditions that are not easily self-diagnosed
Know differences between prescription and OTC drugs
Rx requires federally and state licensed healthcare professional authorization for purchase
OTC is available without Rx, same FDA regulation, easy to understand labeling, can be same as Rx but lower dose than Rx formulas, generic rules apply, can be restricted due to abuse liability or meth ingredient capability
Understand and describe the scheduling of controlled drugs and recognize that this is overseen by the DEA
Schedule 1 - high abuse with no legal accepted medical use in the USA (LSD, heroin, marijuana)
Schedule 2 - high abuse with accepted medical use (morphine, oxycodone, cocaine, ritalin, amphetamine)
Schedule 3 - intermediate abuse potential (ketamine, suboxone)
Schedule 4 - drugs with lower abuse potential (benzodiazepines)
Schedule 5 - low abuse potential, may be dispensed by a pharmacist without Rx under specific circumstances (opiate controlling cough and antidiarrheal drugs)