Barbier Reaction
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
What is the Barbier Reaction?
Organometallic + Aldehyde → Alcohol
How are organometallic reagents normally prepared?
Normally prepared with a C=O added to it
Why is the reaction referred to as an “in situ” reaction?
→In situ: The organometallic precursor and carbonyl are added to generate the organometallic compound in the solution.
What are Organolithiums
→Its pyrophoric (ignites on exposure to air) thus extremely moisture sensitive
→Its super basic (pKb -35) and requires hydrocarbon solvents.
What are Organomagnesium halides (Grignard Reagent)
→NOT pyrophoric but still extremely moisture sensitive.
→Extremely basic and requires ether solvents
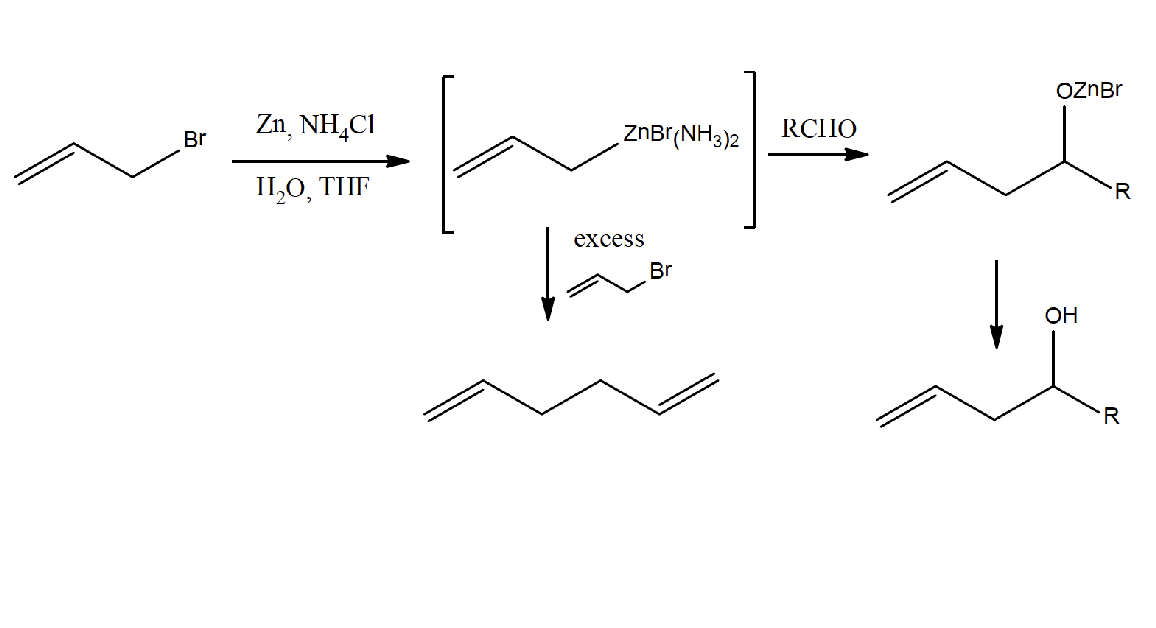
Describe the Activated (Stabilized) Organozinc Reaction
An organozinc reagent will be generated, in situ, under Barbier conditions.
→Activated organozinc complexes generated in the presence of ammonium has a much slower reaction kinetics for water than for addition to an aldehyde or ketone.
→No need for inert atmosphere or dry conditions.
→Reaction is very fast and high yield.
Draw the Barbier Reaction
What are some cautions for Allyl bromide?
-Its a lachymator thats highly irritating on the skin. Keep in the fume-hood!
What are some cautions for Saturated NH4Cl
-It’s acidic and irritating (wash skin w/ cold water for ~15min)
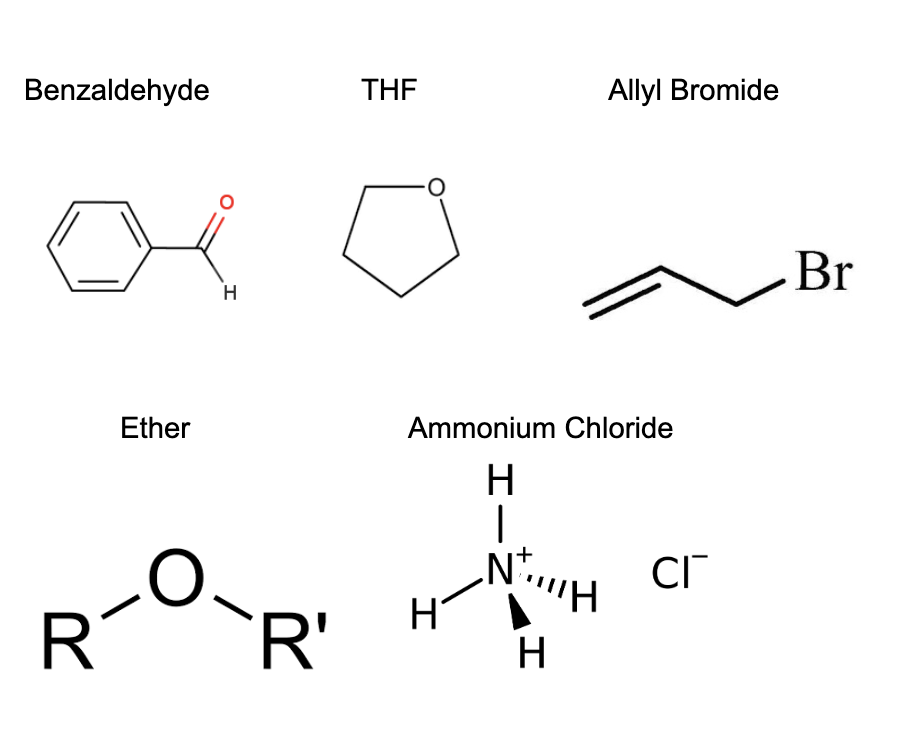
What are the 6 reagents? Draw them
Benzaldehyde
THF
Allyl Bromide
Ammonium Chloride
Zinc
Ether
Describe the procedure (4 steps)
Add 20 mL NH4Cl, 5 mL THF, 20mmol benzaldehyde, and 34mmol allyl bromide to flask. Then add 24mmol zinc to flask and swirth for 45 min.
Transfer mix to separatory funnel, wash with 10mL ether and then add 15mL more ether.
Extract organics, wash ether with water and 5% NaHCO3. Extract aqueous layer with 5mL of ether.
Dry ether with Na2SO4 then filter to evaporate on hot plate, then cool temp and weight.